Abstract
There is little data regarding the diagnostic ability of neutrophil–lymphocyte ratio (NLR) in acute coronary syndrome. Therefore, we aimed to assess the predictive value of NLR in the discrimination of troponin-positive patients. We enrolled 244 patients (mean age 61.3 ± 11.7 years, 70.5% male) who presented to the emergency service with acute chest pain. Patients were divided into 2 groups based upon the troponin positivity in the 12- to 24-hour follow-up. The admission NLR was significantly higher in the troponin-positive group (5.49 ± 4.01 vs 2.40 ± 1.36, P < .001). A cutoff point of 2.80 for NLR measured on admission had 79% sensitivity and 73% specificity in predicting follow-up troponin positivity. After multivariate analysis, admission NLR and glucose levels remained significant predictors of follow-up troponin positivity. In conclusion, NLR may be an early diagnostic marker in emergency service for discriminating patients who will have a positive troponin test in the follow-up.
Introduction
Total white blood cell (WBC), neutrophil, and lymphocyte counts, as well as the neutrophil–lymphocyte ratio (NLR; a derivative of the latter 2), are markers of systemic inflammation. 1,2 In previous reports, the NLR was especially shown to be a predictor of cardiac events and mortality in patients with stable coronary artery disease (CAD) and severity of coronary atherosclerosis. 3,4 In addition, several studies showed that total WBC count, its differential, and the NLR have prognostic value in patients with acute coronary syndromes (ACSs). 5 –7
Acute stress-induced lymphopenia and neutrophilia reflect the acute changes in the immune system in the setting of ACS. Lymphopenia was linked to the release of stress-induced cortisol, and it was shown to be one of the earliest findings after acute myocardial injury in previous studies. 8
In this study, we aimed to determine the early diagnostic ability of a recently emerged marker of inflammation, the NLR, which is an index reflecting both the elevated neutrophils in acute inflammatory conditions and the lymphopenia occurring after acute physiological stress, in patients presenting to the emergency department with anginal complaints.
Methods
Study Population
The present study is a single-center, observational study that was conducted between June 2012 and July 2013. The study population consisted of 244 consecutive patients who presented to the emergency department with an angina or anginal equivalent complaint within the first 12 hours of symptom onset (working diagnosis of ACS). The resting 12-lead electrocardiogram (ECG) was obtained within 5 minutes after first medical contact. If the initial ECG was normal or inconclusive, additional recordings were obtained in 30 minutes. The ECG recordings were repeated at 3-hour intervals and immediately in the case of aggravation of symptoms. The same cardiology specialist evaluated all patients. All patients underwent detailed cardiovascular examination and were questioned about smoking, family history of CAD, previous myocardial infarction, hypertension, diabetes mellitus, and noncardiac diseases. Risk factors for CAD were defined as previously described. 4 Exclusion criteria were clinically significant valvular heart disease, significant congestive heart failure, hematological disease, cancer, severe renal or liver disease, ongoing infection or systemic inflammatory conditions, autoimmune disease, or ST-segment elevation myocardial infarction (STEMI). In addition, patients with admission troponin values above the upper reference were excluded, in which the reference values are below 0.010. Acute coronary syndrome diagnosis was made according to the presence of angina in association with electrocardiographic changes or typical elevation of cardiac enzymes (troponin). Patients with troponin positivity formed the study group, while patients with negative troponin levels formed the control group. Patients diagnosed as ACS were referred for coronary angiography. All participants gave informed consent, and the local ethics committee approved the study.
Laboratory Analysis
Peripheral venous blood samples were drawn at admission and every 4 to 6 hours for 12 to 24 hours follow-up. Total and differential leukocyte counts were measured by an automated hematology analyzer (Beckman Coulter LH 750; Brea, California). Absolute cell counts were used in the analyses. Six hours apart, at least 2 troponin tests were performed using the BioMérieux VIDAS troponin I ultra assay (BioMérieux SA, Marcy l'Etoile, France). Biochemical parameters were measured by standard methods.
Statistical Analysis
Data were analyzed with the SPSS software version 17.0 for Windows (SPSS Inc, Chicago, Illinois). The Kolmogorov-Smirnov test was used to verify the normality of the distribution of continuous variables. Continuous variables are presented as mean ± standard deviation, and categorical variables are given as counts and percentages. The independent sample t test or the Mann-Whitney U test was used for continuous variables and the chi-square test for categorical variables. Spearman test was used for correlation analysis. The effects of different variables on troponin positivity were calculated in a univariate analysis for each. Variables with a P value of <.10 were further analyzed by multivariate logistic regression analysis, and results are shown as odds ratio (OR) with 95% confidence intervals (CIs). Receiver–operating characteristic (ROC) curve analysis was used to determine the optimum cutoff level of the NLR in association with positive troponin levels. Statistical significance was defined as P < .05.
Results
A total of 244 consecutive patients (mean age 61.3 ± 11.7 years, 70.5% male) were included in the study. Patients were divided into 2 groups based upon troponin positivity in the 12- to 24-hour follow-up period. Baseline demographic, biochemical, and hematological data of the patients by group are summarized in Table 1. Groups were similar with respect to cardiovascular risk factors, CAD, previous percutaneous coronary intervention (PCI), and coronary artery bypass graft (CABG) operation.
Clinical and Hematological Characteristics of Patients According to the Follow-Up Troponin Levels.
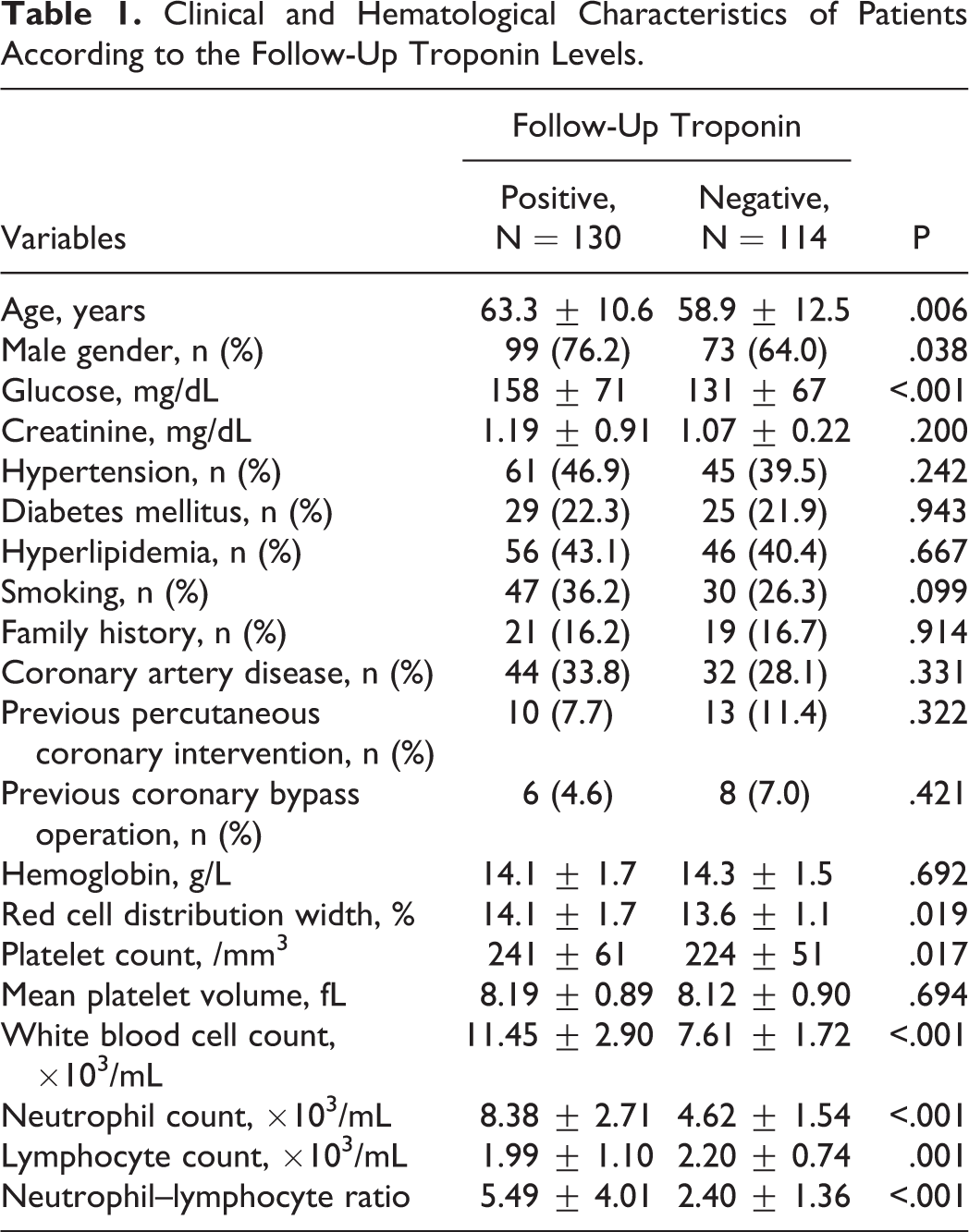
The patients in the troponin-positive group were more likely to be older and male and have higher glucose, platelet, WBC, and neutrophil counts than the troponin-negative group. However, lymphocyte count was lower in the troponin-positive group. In addition, the NLR was significantly higher in the troponin-positive group (P < .001; Figure 1). The NLR was positively correlated with age, admission glucose level, and red cell distribution width (RDW) and negatively correlated with hemoglobin level (Table 2). Using multivariate logistic regression analysis, higher admission NLR values were found to independently predict the patients in which troponin became positive in the follow-up period (OR: 1.859, 95% CI: 1.507-2.293, P < .001; Table 3). In ROC analysis, a cutoff level of NLR > 2.80 had 79% sensitivity and 73% specificity in predicting troponin positivity (ROC area under curve: 0.829, 95% CI: 0.777-0.881, P < .001; Figure 2). According to the coronary angiography reports, 15 (11%), 63 (48%), and 52 (40%) of the patients were treated with medication, PCI, and CABG, respectively.
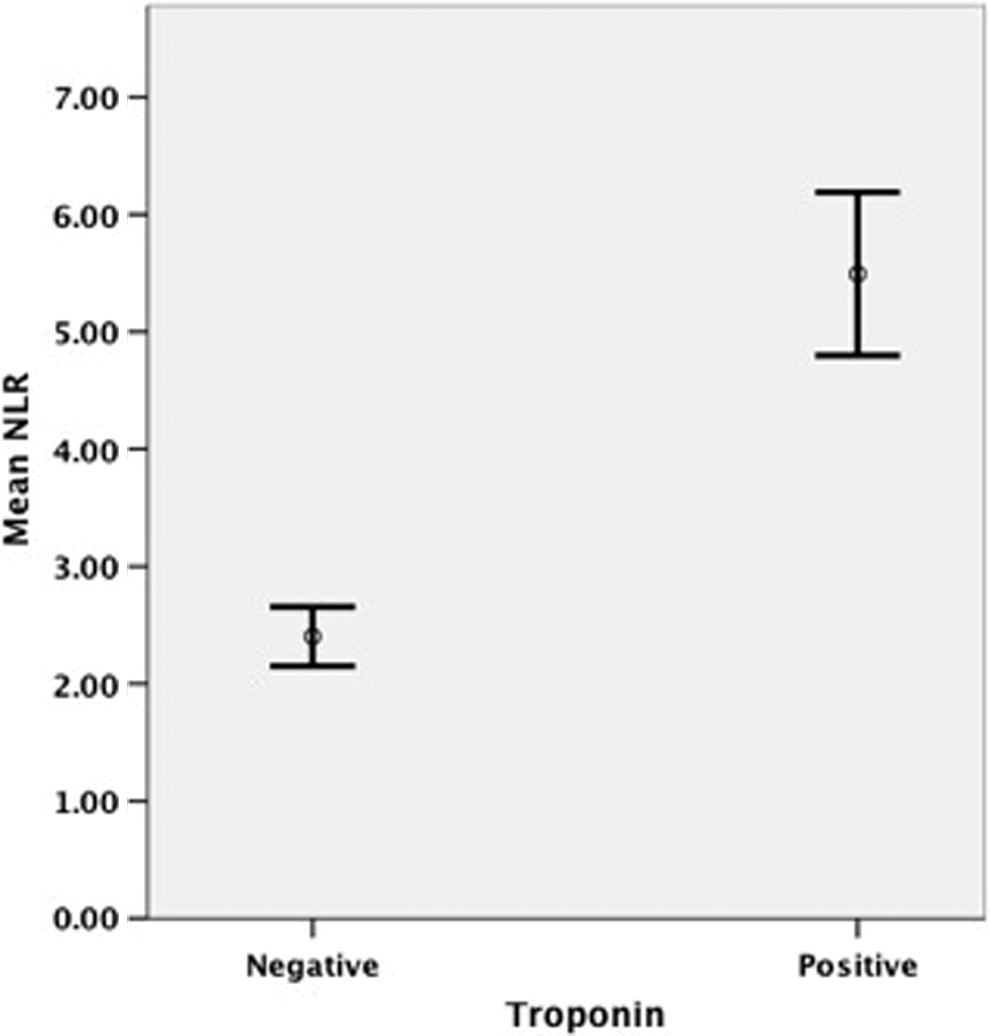
Admission neutrophil–lymphocyte ratios according to the follow-up troponin levels.
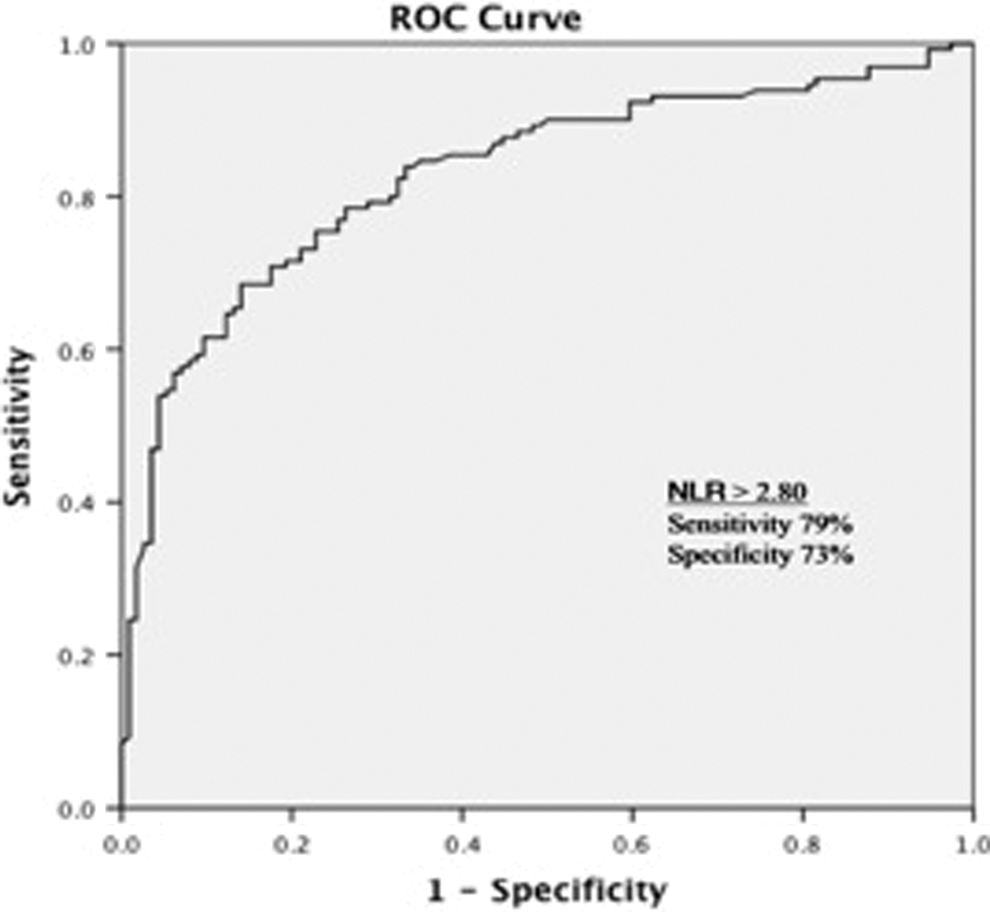
The receiver–operating characteristic curve analysis of neutrophil–lymphocyte ratio for predicting troponin positivity.
Spearman Correlation Analysis Between the NLR and Other Blood Parameters.
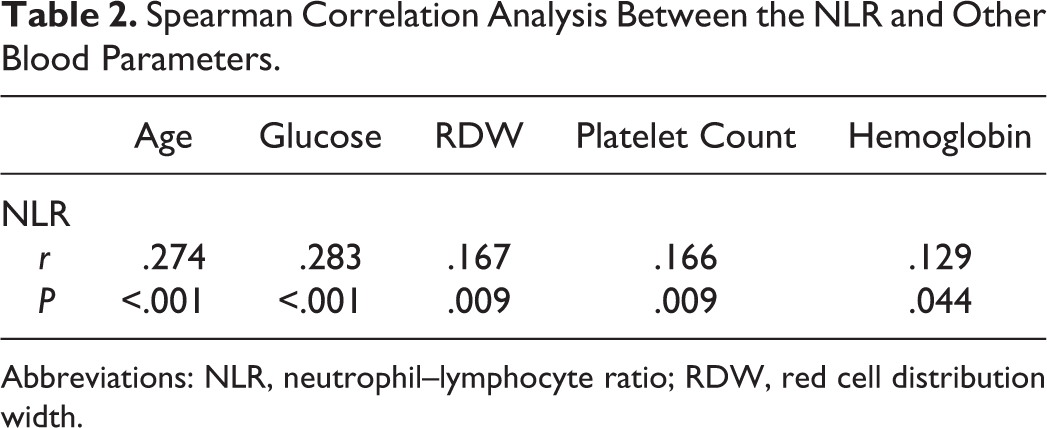
Abbreviations: NLR, neutrophil–lymphocyte ratio; RDW, red cell distribution width.
Multivariate Logistic Regression Analysis to Assess Predictors of Troponin Elevation.
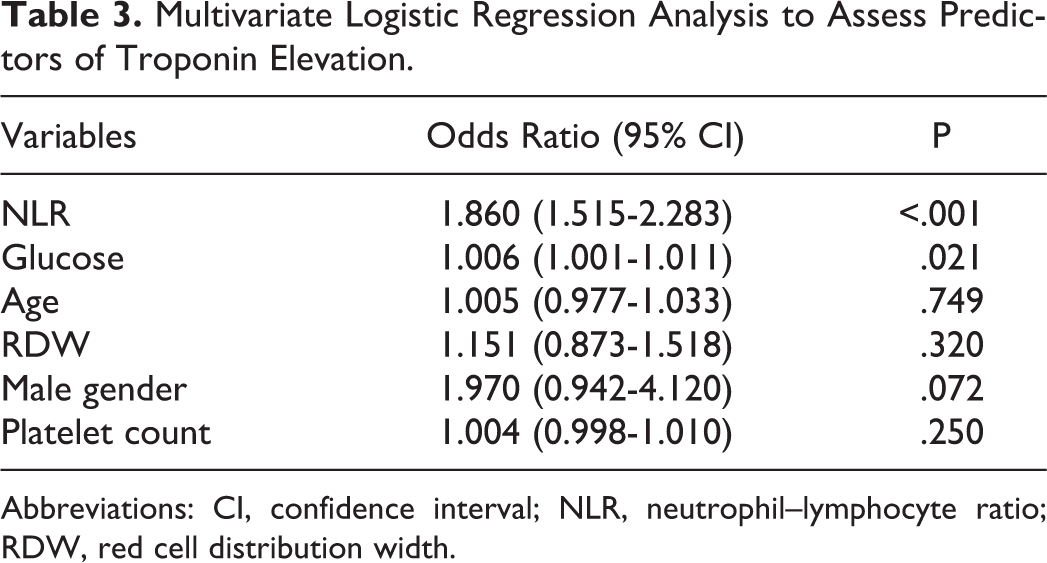
Abbreviations: CI, confidence interval; NLR, neutrophil–lymphocyte ratio; RDW, red cell distribution width.
Discussion
Our study demonstrated that higher admission NLR levels can be useful in discriminating patients in whom troponin will become positive among all patients who were admitted to emergency service with acute chest pain. The NLR levels were higher in patients diagnosed as ACS compared to control group before troponin tests became positive.
Previous studies showed that increased WBC counts are associated with short-term mortality in patients with acute myocardial infarction and frequent hospitalization as well as mortality in patients with heart failure. 9,10 In addition, Giugliano et al demonstrated the predictive value of higher WBC and neutrophil counts for major cardiac events in peripheral artery disease. 11 The NLR has recently emerged as a promising inflammatory marker and has been used in several studies as a predictor of adverse events. Papa et al showed independent relationship between the NLR and the cardiac mortality in patients with stable CAD. 3 The NLR was reported to be a predictor of long-term mortality in patients undergoing PCI. 12 In a study conducted by Akpek et al, the association of NLR with no-reflow development and in-hospital major cardiac adverse events in patients undergoing primary PCI in STEMI setting was demonstrated. 13 Also, the NLR was shown to be a predictor of short- and long-term mortality in patients with STEMI and non-STEMI (NSTEMI). 5,6,14 Turak et al showed that high NLR at admission is an independent predictor of in-hospital mortality and cerebral events in patients with infective endocarditis. 15 Although the prognostic significance of NLR was studied in several cardiovascular diseases, to our knowledge, it has never been evaluated as a diagnostic tool in ACS settings.
It is known that cardiac biomarkers are important in the diagnosis of ACS, as chest pain and ECG findings are usually nonspesific. 16 –18 For the early diagnosis or ruling out of ACS to make an accurate risk stratification, several new biomarkers were developed because classical cardiac markers begin to rise 4 to 6 hours after symptom onset.
Several markers of oxidative stress or vascular inflammatory processes, such as myeloperoxidase, CD40/CD40 ligand, lipoprotein-associated phospholipase A2, heart-type fatty binding protein, and ischemia-modified albumin, have been recently evaluated in acute chest pain based on the underlying pathogenic mechanism. 19 –22 These biomarkers have roles in inflammation, collagen metabolism, plaque erosion, and rupture as well as playing an active role in the development of thrombosis. In our study, as an alternative to these expensive biomarkers, we aimed to establish early diagnostic ability of WBC, their differential counts, and the NLR.
C-reactive protein was thought to be a bystander marker of inflammation until several studies showed that it was an independent risk marker in patients with ACS. 23,24 In some studies conducted on patients with ACS, C-reactive protein (CRP) levels were found to be correlate with peak troponin I levels and short-term adverse events irrespective of the extent of myocardial damage. 24,25 A meta-analysis showed the long-term prognostic value of CRP but indicated controversy regarding the short-term prognostic value. 26 As a matter of fact, controversial results regarding the predictive value of CRP for short-term events do not support the routine use of admission CRP for risk stratification of patients with ACS.
The increase in the number of total leukocytes and the shift in the leukocyte differential in patients with acute myocardial infarction have been known for many years, and these changes were attributed to an increase in cortisol levels. 27 –29 Thomson et al reported that the presence of a relative lymphopenia and rapid increase in creatine kinase MB were early markers of acute myocardial infarction. 8 Additionally, results of a retrospective study conducted by Ozturk et al, showing higher NLR levels in non-ST-segment elevation ACSs than in the control group, seem to support our work. 30
In ACS settings, in the coronary ischemic cascade beginning with atherothrombosis, a certain period of time passes from the onset of symptoms to the elevation in cardiac enzymes. Considering that inflammation is an important factor in acute myocardial ischemia, the leukocytes and the NLR can be regarded as an acute-phase reactant. In the present study, supporting this hypothesis, we found higher NLR values in patients with ACS compared to control patients, all of whom had admission troponin levels in the normal range.
In the study by Kurpesa et al, leukocytosis was independently associated with silent ischemia in postmyocardial infarction patients who were assessed by Holter monitoring for silent ischemia. 31 In patients with CAD, the total leukocyte count and the NLR were shown to be elevated in as little as 15 to 30 minutes as a result of coronary ischemia induced by exercise testing. 32 –34 Our study results support these findings, and admission NLR level may be an early marker for coronary ischemia.
Regardless of the diabetic status of the patients, several studies revealed a relationship between hyperglycemia at admission and increased adverse cardiovascular events in ACS setting. Hyperglycemia in nondiabetic patients treated with reperfusion therapy for STEMI was independently associated with larger infarct size and higher long-term mortality rates. 35 In a study conducted on a broad ACS population, elevated admission glucose level was found to be a strong independent predictor of in-hospital mortality, especially in nondiabetic patients. 36 In summation, hyperglycemia induced by stress after ACS may reflect the severity of myocardial lesion causing troponin elevation.
The major limitations of our study are the cross-sectional design, the lack of in-hospital and long-term follow-up, and the relatively low number of patients. The lack of simultaneous measurement of other inflammatory markers and comparison with the NLR is another limitations. It was shown that hypertension, diabetes mellitus, hyperlipidemia, obesity, and smoking were associated with chronic low-grade inflammation. The NLR and other inflammatory markers such as CRP are prognostic indicators of cardiovascular diseases. For this reason having no information about the basal levels of CRP, WBC, and NLR of the patients before the index event is another limitation.
Conclusion
Neutrophil–lymphocyte ratio is a simple, inexpensive, fast, and easily accessible prognostic parameter; it is an important inflammatory marker that can take a role in the diagnosis of NSTEMI. If supported by further large-scale prospective studies, increased NLR may be useful as a faster diagnostic tool in addition to (or together with) other biomarkers such as troponin.
Footnotes
Acknowledgment
We are grateful to Dicle University DUBAP for their sponsorship of English editing for this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
