Abstract
This study compared the incidence of M2/ANXA5 haplotype carriage, a documented repeated miscarriage risk factor, in patient groups with normal and elevated lipoprotein(a) (Lp(a)) levels. A total of 138 women with ≥2 consecutive, idiopathic recurrent miscarriages, categorized in patients with elevated (≥30 mg/dL, n = 44) and normal Lp(a) level (<30 mg/dL, n = 94) were recruited at the recurrent pregnancy loss (RPL) clinic of Munich Großhadern University Hospital. A total of 500 fertile women served as controls. All patients were genotyped for ANXA5 promoter haplotypes, genetic frequencies were compared, and odds ratios (ORs) and relative risks of M2 carriers were calculated. Women with M2 haplotype had an almost 2 times higher relative risk of RPL (OR 2.6, 95% confidence interval 1.5-4.6, P = .001) than fertile controls. Furthermore, risk rises to 2.47 in patients having normal Lp(a) levels (OR 3.2, 95% confidence interval 1.7-5.9, P = .001), whereas women with high Lp(a) levels exhibit notably lower apparent RPL risk of 1.39 (OR 1.4, 95% confidence interval 0.5-4.1, P = .659).
Introduction
Thrombophilia predispositions are a recognized factor for miscarriages and placental obstetric complications. Significant share of these conditions are hereditary and occur due to genetic alterations in the availability or affinity of blood coagulation factors and auxiliary proteins involved in the thromboplastin complex. 1 Reduced expression of the ANXA5 gene product, potent placental anticoagulant, has been documented in series of obstetric pathologies, antiphospholipid syndrome, 2 preeclampsia (PE), 3,4 fetal growth restriction (FGR), 4,5 and nonpregnant women with previous recurrent pregnancy loss (RPL). 6 A promoter haplotype of ANXA5 identified in 2007 was shown to drive down gene expression. 7,8 This haplotype-designated M2 has been documented as RPL predisposition factor in populations of Central Europe 7,9 and recently in the Japanese population. 10 The M2 consists of four consecutive nucleotide exchanges in the proximal core promoter region of ANXA5 gene and a subset of this haplotype harboring only the 2 “central” SNPs, SNP2 and SNP3, and defined as M1 haplotype, 7 does not appear to be associated with any obstetric complications from the previous studies. A recent study demonstrated fetal loss, FGR, and placental thrombosis in homo- and heterozygous animals of an AnxA5 loss of function murine model. 11
A direct interaction of ANXA5 with lipoprotein (a) (Lp(a)) leads to attenuated membrane binding of the former, upon physiologically relevant high Lp(a) concentrations of 10 to 40 mg/dL. 12 High Lp(a) is a well-established risk factor for coronary disease and stroke. 13,14 Pathologically elevated Lp(a) has been associated with PE, 15 FGR, 16 and RPL. 17,18
Since both proteins, ANXA5 and Lp(a), are involved in the formation of the thromboplastin complex and high circulating concentrations of Lp(a) would effectively reduce the anticoagulation potential of ANXA5, we aimed to evaluate the distributions of M2/ANXA5 carriers in patients with RPL having normal and elevated Lp(a) levels.
Patients and Methods
Study Population
The present study complied with the ethical guidelines of all institutions involved. Furthermore, it was approved by the review board of the Ludwig-Maximilians University of Munich (institutional review board), and informed consent was obtained from all patients examined.
A total of 138 women were prospectively recruited between June 2011 and July 2012 at the Recurrent Pregnancy Loss Clinic of the Division of Gynecological Endocrinology and Reproductive Medicine. All women were screened negative for any potential cause of their recurrent miscarriage as described previously. 19,20 Lipoprotein(a) levels of women were screened at least 6 weeks after the last pregnancy had ended. According to these measured levels, study patients were grouped in patients with elevated Lp(a) levels ≥30 mg/dL (subgroup 1) or normal Lp(a) levels <30 mg/dL (subgroup 2) according to the general definition. 21
DNA was extracted from white blood cells, using QIAmp DNA blood mini kit (Qiagen Hilden, Germany) and stored in 100 µL aliquots at −20°C for further analyses.
A previously recruited control group consisted of 500 fertile women from the registry of the Institute of Human Genetics, UKM Muenster, and genetic backgrounds of patients and control groups were confirmed comparable, as previously described. 7
Genotyping and Statistical Analysis
Extracted DNA was genotyped by direct amplicon Sanger sequencing as previously described. 7 Differences of M2 carriage in women with elevated or normal Lp(a) levels were assessed using the 2-tailed Fisher exact test. Statistical significance was interpreted at odds ratios (ORs) of 2 (±0.25) and above, characteristic for the RPL populations genotyped so far. 7,9,10 The significance level of the analysis was set at P < .05.
Results
In all, 44 (32%) women had elevated (median: 60 mg/dL, min–max: 30-141 mg/dL) and 94 (68%) had normal Lp(a) levels (median 12 mg/dL, min-max: 1-29 mg/dL). Patient subgroups did not differ in terms of common clinical or specific obstetric history (Table 1).
Relevant Clinical Data of the Study Population.a
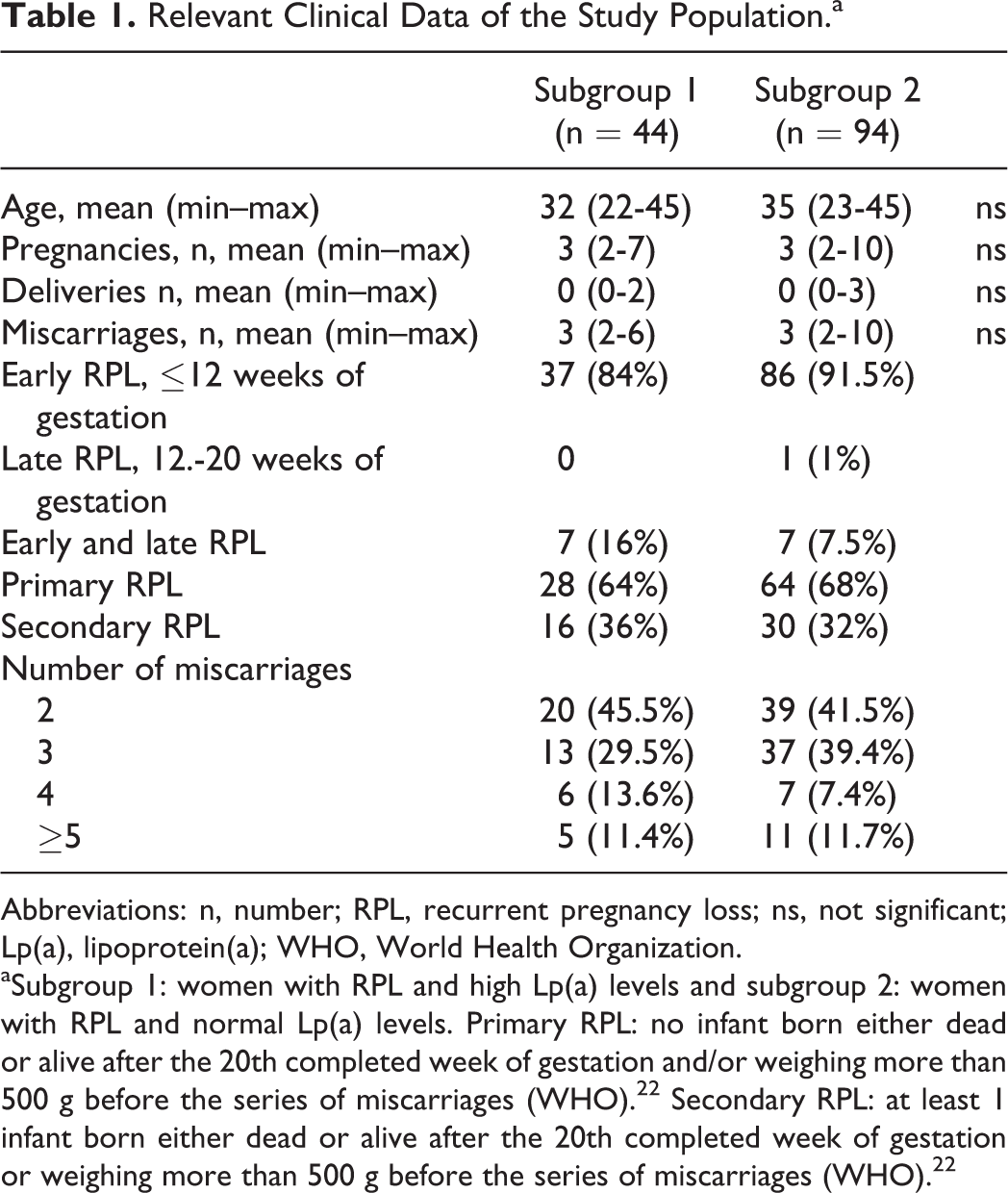
Abbreviations: n, number; RPL, recurrent pregnancy loss; ns, not significant; Lp(a), lipoprotein(a); WHO, World Health Organization.
aSubgroup 1: women with RPL and high Lp(a) levels and subgroup 2: women with RPL and normal Lp(a) levels. Primary RPL: no infant born either dead or alive after the 20th completed week of gestation and/or weighing more than 500 g before the series of miscarriages (WHO). 22 Secondary RPL: at least 1 infant born either dead or alive after the 20th completed week of gestation or weighing more than 500 g before the series of miscarriages (WHO). 22
The role of M2/ANXA5 as a risk factor for idiopathic RPL was verified, comparing the 138 women with recurrent miscarriages to the control group (Table 2). The study group slightly deviated from Hardy-Weinberg equilibrium (Markov chain Monte Carlo [MCMC] P = .1951), mainly due to an excess of homozygous carriers of both haplotypes. Allelic frequencies (AFs) of M2 were found twice as high in the study group (0.105) compared to the control group (0.051). Consequently, women with M2/ANXA5 appeared to have a 1.98 higher risk of RPL than noncarriers, compared to fertile controls (OR 2.6, 95% confidence interval 1.5-4.6, P = .001).
Genotype Frequencies of ANXA5 Gene Promoter Haplotypes in the Patient Group With Subgroups and in the Fertile Controls Group.
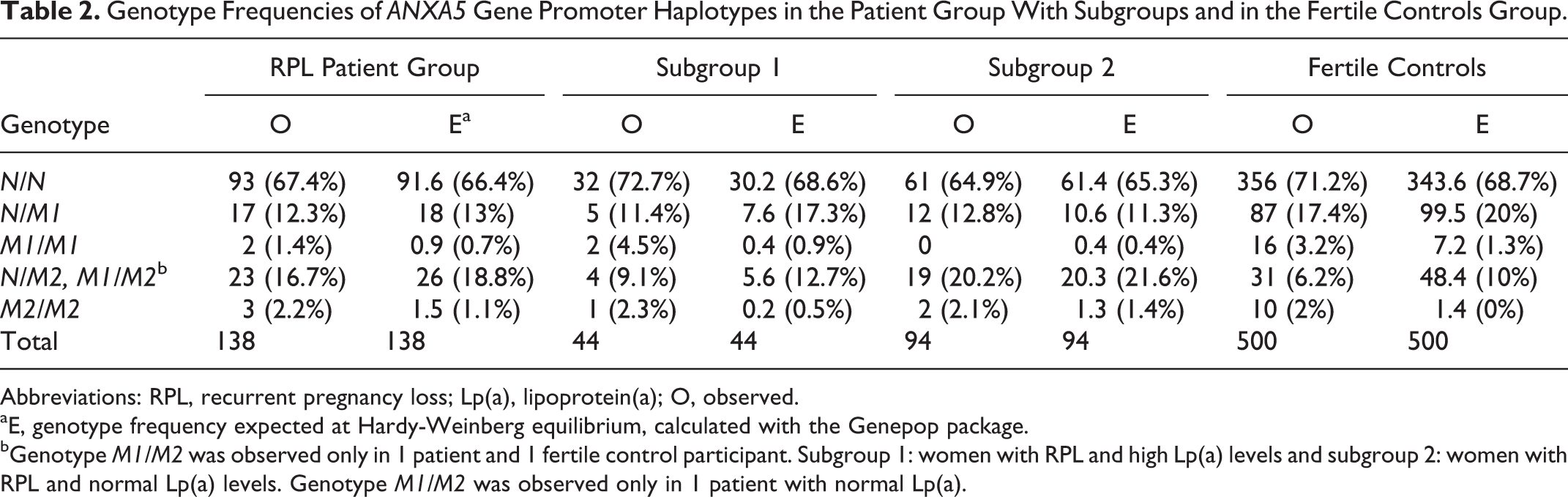
Abbreviations: RPL, recurrent pregnancy loss; Lp(a), lipoprotein(a); O, observed.
aE, genotype frequency expected at Hardy-Weinberg equilibrium, calculated with the Genepop package.
bGenotype M1/M2 was observed only in 1 patient and 1 fertile control participant. Subgroup 1: women with RPL and high Lp(a) levels and subgroup 2: women with RPL and normal Lp(a) levels. Genotype M1/M2 was observed only in 1 patient with normal Lp(a).
Analyzing the distribution of M2 carriers in women with elevated (1) versus normal (2) Lp(a) levels, we found subgroup 1 was not in Hardy-Weinberg equilibrium (MCMC P = .0231), whereas patients with normal Lp(a) of subgroup 2 were in Hardy-Weinberg equilibrium for ANXA5 haplotypes (MCMC P = .8504).
Allelic frequency of M2 in elevated Lp(a) level women was 0.068, being close to the AF determined in controls (0.051) with resulting relative risk of 1.39 (OR 1.4, 95% confidence interval 0.5-4.1, P = .659) compared to fertile women. In contrast, those with normal Lp(a) levels had a relative risk of 2.47 for RPL (OR 3.2, 95% confidence interval 1.7-5.9, P = .000) with an M2 AF of 0.122 (Table 2). There is a greater apparent risk of 1.24 for M2 carriers of the second subgroup when performing intragroup comparison of patients with RPL (OR 2.2, 95% confidence interval 0.8-6.4, P = .059, Fisher's exact test). The subgroup analysis of women with RPL and elevated versus normal Lp(a) levels indicates that M2 carriers are substantially rarer in the subgroup of patients with higher Lp(a).
Discussion
There was an enrichment trend of M2 carriers in the patient subgroup with normal Lp(a) levels. In contrast, among patients with RPL having elevated Lp(a), M2 carriers seemed underrepresented, with genetic frequency very close to this of the fertile controls and of the general population. 7 This underrepresentation could be hardly evaluated as statistical trend because of the low number of patients with RPL having elevated Lp(a) levels. On the other hand, the distribution of M2 carriers in this particular subgroup is in strong deviation of Hardy-Weinberg equilibrium that reflects upon the whole patient group. In contrast, the M2 distribution in the second subgroup, with normal Lp(a), is in almost perfect Hardy-Weinberg equilibrium. The observed underrepresentation of M2 heterozygotes and overrepresentation of M2 homozygotes in subgroup 1 would lead to the conclusion of a negative ascertainment bias if generally the homozygotes count in both subgroups was not that small. For a pilot study, no statistically justified conclusion can be made on such effect at present because of the small sample size, although the genetic distribution even at this scale is evidently correct. In a study patient population, one would expect distribution of M2 carriers that is similar to the fertile controls (positive ascertainment bias under the “fertility” criterion) if a competitive genetic interaction model is in play for this particular group, meaning negative ascertainment bias due to a second risk factor (sic, high Lp(a)). We believe this to be indeed the case for this particular subgroup, and our hypothesis is supported by biochemical evidence of Lp(a)/ANXA5 direct interaction in physiologically relevant concentrations, 12 which effectively reduces the ANXA5 anticoagulation potential. This would in consequence yield a prothrombotic phenotype. This initial finding has to be confirmed in larger patient groups, in order to gain on statistical significance and possibly be instrumental in the diagnostic workup of patients with RPL.
Footnotes
Acknowledgments
The authors would like to thank Ursula Antkowiak for the competent technical assistance. The authors acknowledge the continuous support of IZKF Muenster.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
