Abstract
This study presents sample size considerations derived from the Efficacy of Thromboprophylaxis as an Intervention during Gravidity (EThIGII) trial (ClinicalTrials.gov: NCT00400387) to address the question of low-molecular-weight heparin (LMWH) treatment in women with recurrent pregnancy loss (RPL) depending on the M2/ANXA5 haplotype. To evaluate the possible influence of such treatment on miscarriage rates of trial participants, a post hoc analysis of ANXA5 promoter genotypes in the light of M2/ANXA5 (RPRGL3) distribution was performed using logistic models. DNA for genotyping was available from 129 LMWH and 95 control patients, 44 (19.6%) of whom were M2/ANXA5 carriers. Miscarriages occurred in 1 (4.0%) of 25 M2/ANXA5 carriers from the LMWH group compared to 4 (21.1%) of 19 in the control group, resulting in an odds ratio (95% confidence interval) for miscarriage of 0.16 (0.016-1.5) for women treated with LMWH. In noncarriers, miscarriage rates were 6 (5.8%) of 104 versus 7 (9.2%) of 76 for the LMWH and the control groups, respectively, corresponding to an odds ratio for miscarriage of 0.60 (0.19-1.9). The apparent beneficial effects of miscarriage rate reduction in M2/ANXA5 carriers with RPL concur with biological considerations about improvement in reduced ANXA5 function through LMWH treatment in an adequate murine model. The data obtained were instrumental to design proper assessment of the existence and magnitude of this effect.
Introduction
Hereditary thrombophilia may well be one etiology for recurrent pregnancy loss (RPL), 1 and evidence on impeded placental perfusion shows an increased risk for adverse pregnancy outcome. 2 –5 The influence of genetic thrombophilia has been reviewed in several clinical studies 6 and initially concentrated mostly on the common, low thrombotic risk factor V Leiden (G1691A, R506Q, and rs6025) and prothrombin (PTm) G20210A (rs1799963) variants. 7 In 2007, another potential hereditary factor for thrombophilia-related RPL was found, termed as “M2,” a haplotype in the proximal core promoter region of the annexin A5 (ANXA5) gene, defined as a constellation of 4 single-nucleotide polymorphisms (SNPs), c.−467G>A, rs112782763; c.−448A>C, rs28717001; c.−422T>C, rs28651243; and c.−373G>A, rs113588187. 8 The haplotype, a risk factor for RPL susceptibility, RPRGL3, OMIM entry 614391, was confirmed through molecular cloning and direct sequencing of the relevant amplicon clones, so the minor alleles of all 4 SNPs comprising it were proven to reduce the expression of a reporter gene in a functionally representative cell line compared to a background construct harboring all the major (normal) alleles. 8 Reduction in ANXA5 messenger RNA abundance was demonstrated in chorion-carrying M2, 9 which was confirmed to be haplotype specific, 10 and concomitant decrease in ANXA5 protein levels has been detected in placental tissue of M2 carriers with a thrombophilic placental complication. 11 Differential enrichment of the haplotype has been observed in RPL patient-groups, contributing to relative risks for carriers from 1.5 to 2, compared to random population controls of German, 5,8 Bulgarian, 5 and Malaysian 12 ethnic backgrounds. Estimated relative risks in M2 carriers in relation to healthy control patients with negative history for infertility or miscarriage are between 1.8 and 3, depending on the number of miscarriages and categories of recurrent spontaneous abortion and embryonic development in patient cohorts of German, 5,8 Italian, 4 Bulgarian, 5 and Japanese 13 extraction. In Germany alone, the RPL risk attributed to M2 carriers of retrospective and prospective clinical cohorts has been estimated in altogether 600 recurrent miscarriage patients and 1123 control individuals. 5,8,14 –17 In 2012, another haplotype classification system H1 to Hn was introduced to characterize nucleotide variations in the 5′ nontranslated region of ANXA5, 18 including altogether 7 SNPs, rs62319820, rs112782763, rs28717001, rs28651243, rs113588187, rs1050606, and rs1131239, resulting in 4 to 7 constructed haplotype combinations, depending on genotyping scope and depth in Dutch patients. 18 –20 According to this classification, 18 haplotype H3 would represent an expansion of M2, including the minor alleles of rs62319820 upstream and of rs1050606 and rs1131239 downstream. The association of M2 with the downstream SNPs rs1050606 and rs1131239 of uncertain functional relevance is apparently not perfect, as evident from previous 8 and more recent studies. 15,20 Since M2/ANXA5 is linked with reduced expression of ANXA5 in chorionic placenta, 10,11 the associated comparable risks of male carriers in RPL couples may indicate impaired embryonic anticoagulant function. 5,12,14 This is in accordance with the well-studied role of ANXA5 as placental anticoagulant, enriched on the surface of chorionic syncytiotrophoblasts. 1
As RPL constitutes huge psychological stress to the couples, different therapeutic options have been evaluated and discussed. A number of observational studies suggested that low-molecular-weight heparin (LWMH) increases live birth rates. 21 –25 Results of 2 earlier randomized trials demonstrated the efficacy of LMWH for women with unexplained RPL. 26,27 In contrast, 3 later studies did not confirm these findings. 28 –30 Conclusions of a study comparing LMWH to aspirin drew no significant advantage of LWMH versus aspirin. 31 However, these studies differ considerably in their selection of potential genetic conditions associated with RPL. A recent observational study in factor V Leiden (FVL)- and PTm-carrying RPL women demonstrated benefits of LMWH treatment in terms of fewer fetal deaths, less preeclampsia cases, and lower rate of small for gestational age babies. 32 The last Cochrane review on anticoagulant treatment in women with unexplained recurrent miscarriage with or without inherited thrombophilia concluded that the effect of such treatment in women with hereditary thrombophilia “needs to be assessed in further randomized controlled trials.” 33 To meet the need for properly powered, multicenter, randomized assessment, the Efficacy of Thromboprophylaxis as an Intervention during Gravidity (EThIGII) trial planned to enroll 486 patients (expecting a 10% dropout rate), and it took >5 years and much effort to attain this goal. This then permitted the rare opportunity of performing a post hoc analysis of genetic RPL predisposition factors from retrospective DNA samples. The EThIGII trial compared LMWH and multivitamins versus multivitamins alone in unexplained RPL. 33 The authors concluded that LMWH had no influence on ongoing pregnancies or live birth rates. Additionally, in this trial, a subgroup analysis evaluating RPL patients (n = 63) with inherited thrombophilia (FVL or PTm mutations, antithrombin deficiency, protein C deficiency, and protein S deficiency) was performed. Low-molecular-weight heparin and multivitamins showed no significant advantage versus multivitamins alone in this particular patient-group. However, the number of women with each of the above thrombotic risk factors or their combinations was fairly small, so that further stratification did not appear meaningful. When the trial was designed, M2/ANXA5 had not been identified as a possible RPL risk factor. Given the uncertain effect of LMWH on RPL carriers of genetic thrombophilia, it was of interest to examine a possible therapeutic influence on women carrying the M2 haplotype of the ANXA5 gene as a predisposition factor with a prevalence of 10% to 15% in European parous controls without miscarriages. 4,5,8,14 With experimental evidence coming from a heterozygous knockout murine model of AnxA5 function, LMWH could substitute for reduced abundance of ANXA5 as anticoagulant on relevant molecular targets. 34
Materials and Methods
Trial Design
The EThIGII was a multicenter, randomized, controlled, and open-label trial. 35 The trial protocol and subsequent genetic analysis for “possible predisposing hereditary factors” approval was obtained from the federal authorities in Germany and Austria and the ethics boards of all attending trial sites. All patients signed written informed consent for trial participation. The study randomized 449 women without indication for LMWH use, such as homozygous FVL or factor II mutations, and observed a further 112 women, of whom, in total, 228 provided informed consent for the use of their genomic DNA to study potential inherited RPL factors. Four women were lost to follow-up, and from the remaining 224 patients, 95 were randomly assigned to the control and 94 to the intervention arm. A further 35 received LMWH but were not randomized, 10 of whom had antiphospholipid syndrome, 18 of whom had had problems related to venous thromboembolisms, and 5 who would have been eligible but chose to use LMWH.
Definitions of Parameters and Outcomes
“Early RPL” was defined as at least 2 consecutive miscarriages before 12 weeks’ gestation, and women with 1 or more “late” miscarriages (after 12 weeks’ gestation) were also included. Main outcomes were intact pregnancy at 24 weeks’ gestation and live birth according to the birth records. These records were also used to assess the secondary outcomes preeclampsia, placental abruption, preterm delivery <37th gestational week, or intrauterine growth restriction <5th percentile according to Voigt et al. 36 “Miscarriage” refers herein to spontaneous pregnancy loss before 24 weeks’ gestation.
Genotyping of ANXA5 Proximal Promoter Region
Presence of the common M2 haplotype in the annexin A5 gene (M2/ANXA5) was assessed in genomic DNA of 224 enrolled patients upon trial completion. DNA was extracted from 1 mL peripheral blood, and genotyping of ANXA5 promoter haplotypes was performed by amplicon sequencing as described by Bogdanova et al. 8 Accuracy of genotyping was verified by randomly blinded inclusion of 2% repeated samples. Genotypes were scored in table format and 4 digits coded for further processing.
Statistical Methods
The analyses presented here were all decided upon at the outset of this add-on project to the EThIGII trial and before any genotyping data were collected. All assessments were carried out using the R statistical software package version 3.1.0, 37 and all tests were considered significant for P ≤ .05. Tests for contingency tables used the χ2 test without continuity correction or the Fisher test if expected counts were below 5. The significance for differences between subgroups was tested based on the interaction term in a logistic model. Odds ratios (ORs) and confidence intervals (CIs) were estimated from the logistic model if a category had 3 or more levels or using a Wald estimate and normal approximation using the “odds ratio” command from the “epitools” package. 38
Departures from the Hardy-Weinberg equilibrium were assessed using the “genetics” package. 39 Statistical power calculations were performed with the “pwr” package. 40
Results
A total of 224 patients agreed to DNA genotyping for potential hereditary RPL factors, 129 of whom received LMWH and 95 who did not (see Table 1). The observed prevalence of M2 carriers was 19.6% and did not differ markedly between the groups, with 20.0% versus 19.4% in control and LMWH, respectively. M1 carriers were also similar between the groups (13.7% vs 13.2%). M2/ANXA5 carrier rates were also examined in the clinical subgroups with 3 or more pregnancy losses and 3 or more early pregnancy losses and were essentially the same at 20.7% and 23.5% (see Table 2). When compared to 533 controls from the German population, 8 the observed OR for being an M2 carrier in the EThIGII sample was a modest 1.3 (95% CI, 0.89-2.0) in contrast to a comparison with 500 German parous controls without miscarriages, 8 where the OR was 2.7 (1.7-4.3; see Figure 1). Miscarriage rates of M2 carriers and noncarriers in both intervention groups and clinically relevant subgroups were compared (Table 2). M2 carriers in the control groups exhibited higher miscarriage rates compared to the M2 noncarriers with an OR of 2.6 (95% CI, 0.6-10), with similar results for the subgroups, although all differences are not significant.
Genotype Frequencies of ANXA5 Gene Promoter Haplotypes in RPL Patients of the EThIGII Trial Are Provided as Count (Percentages).
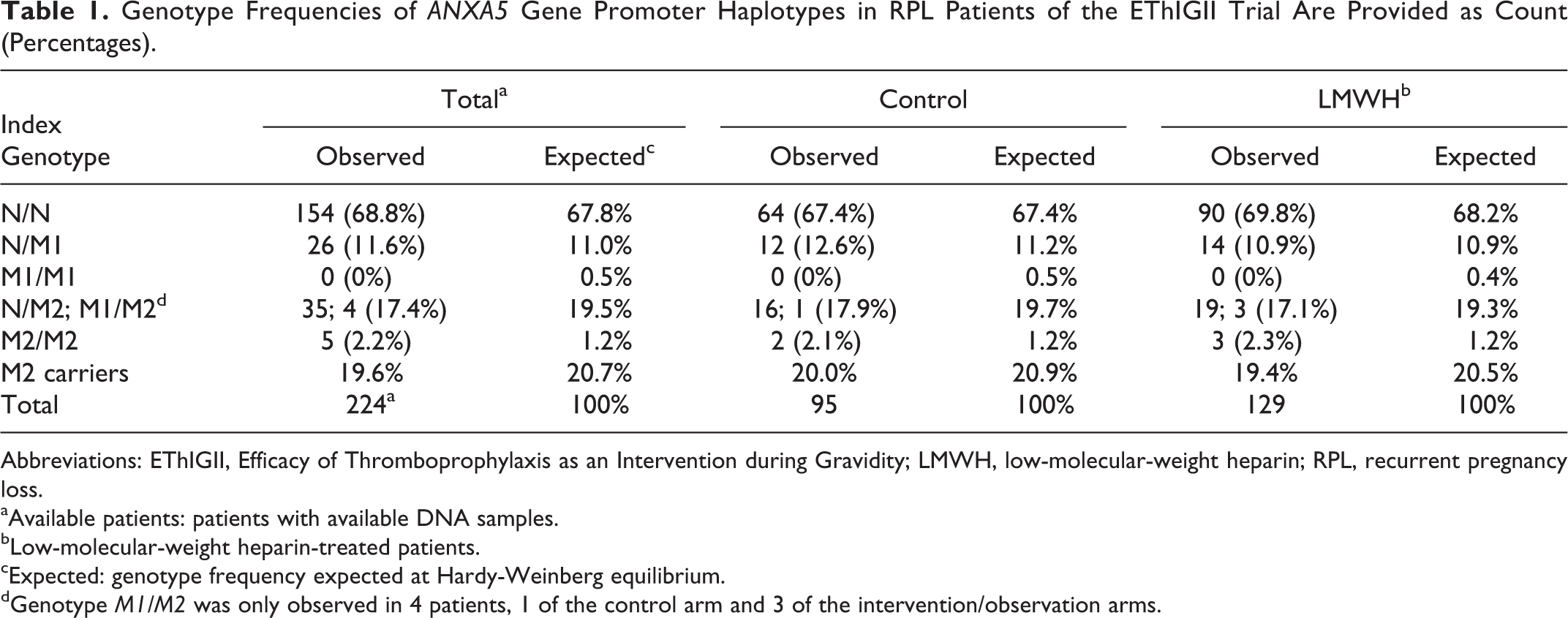
Abbreviations: EThIGII, Efficacy of Thromboprophylaxis as an Intervention during Gravidity; LMWH, low-molecular-weight heparin; RPL, recurrent pregnancy loss.
aAvailable patients: patients with available DNA samples.
bLow-molecular-weight heparin-treated patients.
cExpected: genotype frequency expected at Hardy-Weinberg equilibrium.
dGenotype M1/M2 was only observed in 4 patients, 1 of the control arm and 3 of the intervention/observation arms.
Miscarriages at 24 Weeks’ Gestation Are Provided as Counts (Percentages) Depending on M2 Carrier Status in the Control and Intervention Groups.a
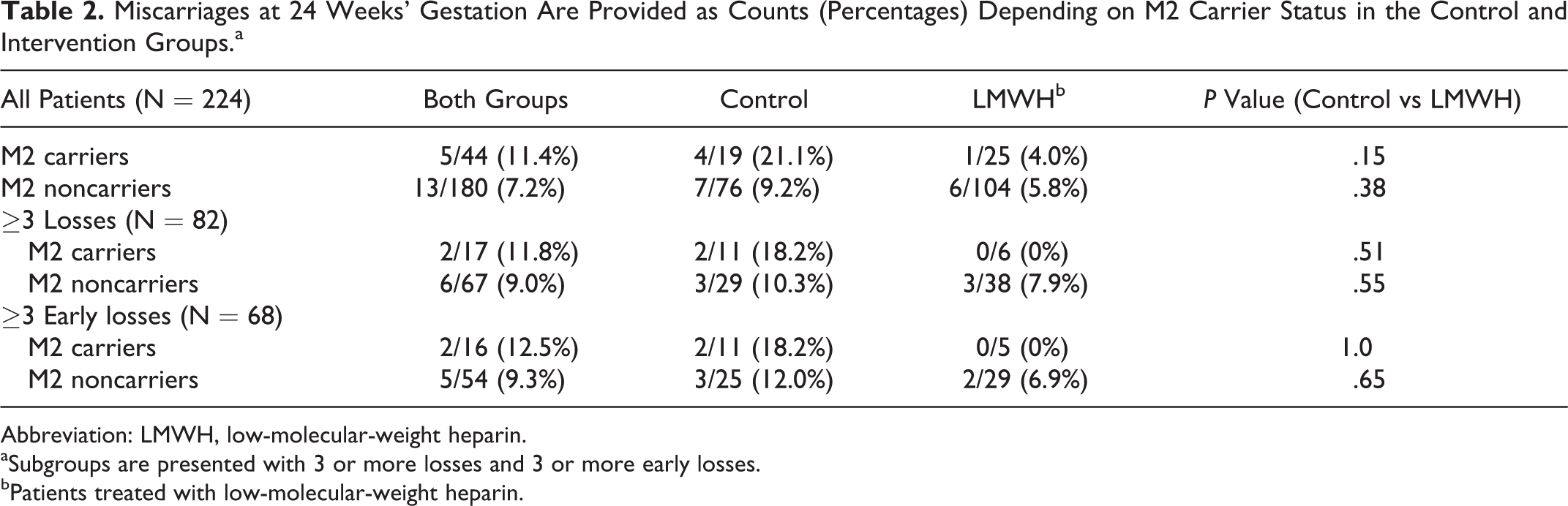
Abbreviation: LMWH, low-molecular-weight heparin.
aSubgroups are presented with 3 or more losses and 3 or more early losses.
bPatients treated with low-molecular-weight heparin.
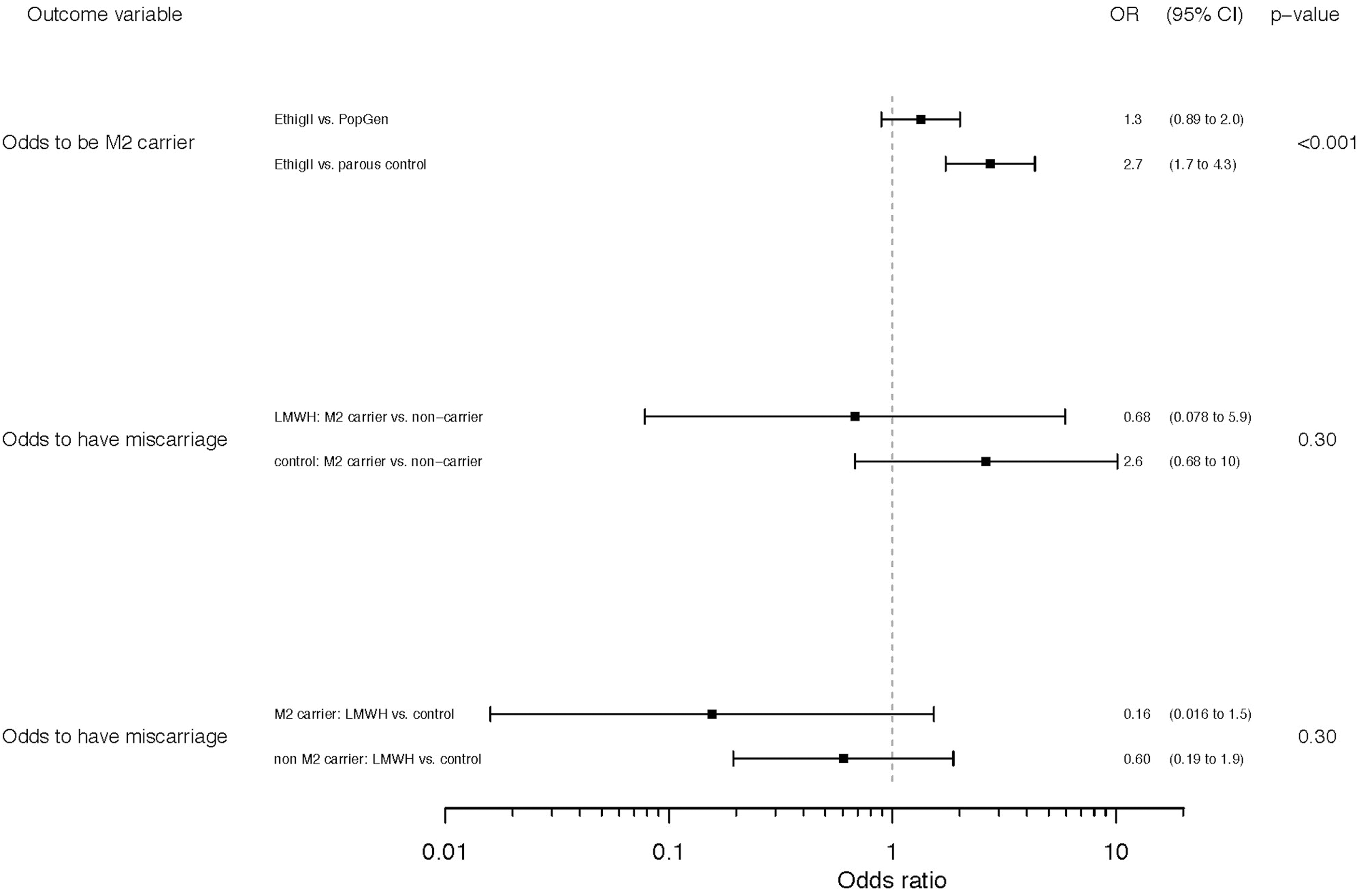
Forest plot to the odds ratios of the outcomes “M2 carrier status” in EThIGII patients versus population and parous controls, and “miscarriage” in the LMWH-treated versus vitamins-treated (control) groups, respectively, among M2 carriers versus noncarriers.
The ORs for having a miscarriage depending on the intervention (LMWH vs control) do not depend significantly on whether one is an M2 carrier. Nonetheless, it is noteworthy that for M2 carriers, LMWH appears more protective with an OR of 0.16 (95% CI, 0.016-1.5) than for noncarriers with an OR of 0.60 (95% CI, 0.19-1.9; see Figure 1).
M2 carriers treated with LMWH have roughly equal chances of miscarriage compared to treated noncarriers, with an OR of 0.76 (95% CI, 0.03-4.9). For untreated patients, the OR for miscarriage in carriers versus noncarriers is 2.6 (95% CI, 0.6-10). The same direction for the effect was seen in the subgroups of patients with 3 or more abortions and 3 or more early spontaneous abortions, where the LMWH M2 carriers had no miscarriages, but the numbers were extremely small (Table 2).
An evaluation of further subgroups including the LMWH effect on treated M2 carriers versus controls with late pregnancy losses was not performed because all haplotype-carrying patients (n = 4) were in the intervention arm of the trial and had no miscarriages. Likewise, noncarrier groups of late pregnancy loss patients in both trial arms were rather small to allow for a formal comparison. A comparable situation arose when evaluating secondary outcomes because the rates of preeclampsia and intrauterine growth restriction were altogether under 5%, rendering a meaningful analysis impossible. The rate of premature births was similarly low in both trial arms and comparable to that of the general population in Germany and Austria. 41
Genotyping the significant patient sample of the EThIGII trial resulted in 44 M2/ANXA5 carriers distributed about equally in both trial arms, with slight prevalence of the treated group (Tables 1 and 2). These incidence data point at an M2 carrier rate of roughly 20% for similar trials. In order to achieve 80% statistical power in detecting a difference in miscarriage rates of 15% and 25%, about 250 M2 carriers per arm would be required in a prospective study. This result expands the number of required participants to 2500 for RPL women of European descent.
Discussion
Until recently, no study has assessed the efficacy of anticoagulant treatment in carriers of the M2/ANXA5 haplotype. The samples available from the EThIGII trial made it possible to evaluate the influence of LMWH therapy on M2 carriers among RPL patients (n = 44) compared to noncarriers (n = 180). Although the M2 carrier rate of 19.6% in the EThIGII patient sample was higher than the generally estimated 10% to 15% for European parous controls without miscarriages, it was on the low side for RPL patient cohorts, where prevalence of 21% to 29% has been observed depending on the number of consecutive early embryonal losses. 4,5,8,14 This minor deviation could be easily explained by the EThIGII study design, which included patients with single or multiple late fetal losses, since M2/ANXA5 does not appear to be a risk factor for repeated late spontaneous abortions. 4,5 M2 carrier rates were somewhat higher in the subgroups of patients with 3 or more pregnancy losses and 3 or more early pregnancy losses, where they were 20.7% and 23.5%, respectively. Based on the literature, 4,5,8,12,13 one would expect a risk ratio of 1.2 to 1.6 for miscarriage in M2 carriers versus noncarriers, corresponding to an OR of 1.2 to 1.7 for a miscarriage rate of 12%. These previous estimates are slightly lower than the value of 2.6 when comparing miscarriage rates of M2 carriers to noncarriers of the trial’s control group, which however had a wide CI.
An OR for miscarriage of 0.16 (95% CI, 0.016-1.5) in M2 carriers treated with LMWH was observed compared to the controls. One might suppose that such a strong effect would be noticeable in trials published so far. However, given a miscarriage rate of, say, 15% in a control population, a carrier prevalence of 20%, and a 5-fold risk reduction for carriers using LMWH, then the intervention arm would have a miscarriage rate of 12.6% and a trial with 450 women would have a power of only 11% for detecting this difference. It is thus conceivable that a strong effect exists, which has not been detected in published trials.
Recurrent pregnancy loss is often defined as a condition with 3 or more consecutive pregnancy losses, but the EThIGII trial design was to include women with 2 or more consecutive miscarriages according to the current definitions used by the American Society for Reproductive Medicine 42 and other comparable trials in the field. This definition takes into account a greater demand of medical attention after 2 miscarriages, on the other hand, a more pronounced effect of LMWH for women at higher risk is conceivable. The comparative analysis of M2 carriers with a history of 3 or more miscarriages and 3 or more early miscarriages supported this notion and is an addition to previous research demonstrating the higher carrier rates of the haplotype in such RPL patients 4,15 and suggesting a biological role in early embryonic anticoagulation. 5,12 In contrast, no effect of LMWH on live birth rates of noncarriers of M2 could be observed.
Genotyping a significant patient sample of the EThIGII trial and comparing the effects of LMWH on M2/ANXA5 carriers versus noncarriers provide data for exploring a potential efficacy of treatment in this particular patient-group, suggesting that a proposed benefit in M2 carriers should be assessed in adequately powered trials. Considering the time, logistics, material, and organizational effort such endeavor would involve, this would require a large multinational coordinated study. Alternatively, or parallel to this, such study could be designed with a different source population. Thus, the results of post hoc analysis of the EThIGII trial provide rare data that can help begin to shed light on this issue and assist in the planning of larger trials.
Altogether, although differences were not significant, it is worth considering that in the relevant subgroups with 3 or more losses/early losses, M2 carriers of the control sample had consistently greater miscarriage risks compared to noncarriers and that treated M2 carriers always had better live birth rates (at 100%) than nontreated carriers (Table 2 and Figure 1). In other words, genetic data of this trial are consistent with a strong protective effect of LMWH for M2/ANXA5 carriers, similar to a response obtained in an AnxA5 maternally deficient murine model, 34 but numbers are too low to be certain.
Considering an incidence of 15% to 17% for the M2 haplotype in ANXA5 among Europeans and a plausible improvement in live birth and thrombophilia-related obstetric complication rates in LMWH-treated pregnant women of M2-carrying couples, it would be an approach of personalized medicine to provide diagnostic care and relevant medication to a population at risk.
In conclusion, there are biological and mechanistic reasons for supposing that M2/ANXA5 carriers may benefit from LMWH treatment. We present evidence that points in this direction, but the existence and magnitude of the effect has yet to be determined.
Footnotes
Authors’ Note
The members of the EThIGII Group are R. Bauersachs, P. Beuter-Winkler, M. K. Bohlmann, O. Brosteanu, A. Brueckmann, J. Dudenhausen, S. Ebner, T. Fischer, S. Gruessner, M. Henes, R. Illing, G. Kamin, B. Kemkes-Matthes, R. Lachmann, B. Lawrenz, P. Neuhaus, E. Neunhoeffer, K. Nitzsche, D. Petroff, N. Rogenhofer, C. Scheler, M. Schenk, E. Schleussner, S. Seeger, C. Seeliger, G. Seliger, C. J. Thaler, and B. Toth.
Acknowledgments
The authors thank the women who allowed their DNA samples to be used for this study. Many thanks go to Thomas Harasim and Alexandra Kotanidou for the excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The EThIGII trial was supported by an unrestricted grant of Pfizer Pharma GmbH. A.M. was funded by a PI grant of the German Research Community, DFG, MA-6288/1-1.
