Abstract
Retinal vein occlusion (RVO) and retinal artery occlusion (RAO) cause significant visual impairment. The role of thrombophilia and cardiovascular testing is uncertain, and optimal treatment strategies have not been determined. We reviewed medical records of 39 patients with RVO and RAO (23 women and 16 men). Thrombophilia and cardiovascular evaluations were performed and outcomes were reviewed. In all, 24 (61.5%) patients had at least 1 thrombophilia. Elevated factor VIII levels were found in RVO (n = 5) but not in RAO. There are no other significant differences in thrombophilias in RVO compared to those in RAO. Most patients had hypertension(41.2% RAO and 55% RVO) and hyperlipidemia (35.5% RAO and 81.8% RVO). In all, 4 women were using oral contraceptives, 2 were pregnant or postpartum. Follow-up data was available for 28 patients (13 RAO, 15 RVO). Nineteen were treated with aspirin, four with warfarin, and one with low molecular weight heparin. Eight patients reported improvement in vision at time of follow-up (5 RAO, 3 RVO). Multiple risk factors are associated with RVO and RAO, and a complete assessment should include thrombophilia and cardiovascular studies.
Introduction
Retinal vascular occlusions represent a significant source of morbidity in the United States and may cause permanent visual impairment. Retinal vascular occlusions are frequently linked to systemic risk factors including hypertension, hyperlipidemia, diabetes, smoking, use of oral contraceptives, 1 and the pregnancy-puerperal state. 2 Retinal artery occlusions (RAOs) are more commonly associated with vascular inflammation, cholesterol emboli, and enhanced platelet aggregation, while retinal vein occlusions (RVOs) are associated with traditional venous thrombosis risk factors such as thrombophilias, venous stasis, and endothelial injury. Both types of occlusions are associated with ischemia, neovascularization, and macular edema during progression. 3 Macular edema is associated with the acute phase of RAO but may last months or years after RVO. Thrombophilias such as factor V Leiden (FVL), the prothrombin gene G20210A mutation (PGM), deficiencies of the natural anticoagulants protein C (PC), protein S (PS), or antithrombin (AT), elevated levels of coagulation factors VIII (FVIII), IX (FIX), and XI (FXI), and antiphospholipid antibodies (aPLs) have all been investigated as potential risk factors. 4,5 However, the data have been conflicting. An increased prevalence of FVL and PGM has been reported in patients with RVOs. 6 –8 Other reports have found significant associations with elevated homocysteine and aPLs but not with other thrombophilias. 9 Resistance to PC has also been reported in younger patients. 10
Cardiovascular risk factors are also important, especially in older patients. 11,12 However, if cardiovascular risk factors are not present, thrombophilias may be more relevant to pathology. 13 Elevated homocysteine is an established cardiovascular risk factor and is associated with both retinal arterial and retinal venous occlusions. 14 –17 Detection of thrombophilias is important in patients with retinal vascular occlusions, because they are at higher risk of future systemic venous and arterial thrombosis. 18
The purpose of our study was to establish the frequency of thrombophilias and cardiovascular risk factors in our referral population of patients with RAO and RVO. We sought to determine whether thrombophilic and cardiovascular risk factors were different between these two types of occlusions. We also evaluated treatment strategies and visual outcomes in our population to determine whether any treatment strategy is preferential.
Materials and Methods
Medical records were retrospectively reviewed from all patients with documented retinal vascular occlusions who were referred between January 2001 and December 2011 for thrombophilia evaluation at the hematology clinics of two academic institutions (Mount Sinai Medical Center and New York Presbyterian Hospital). Thirty-nine consecutive patients with retinal vascular occlusions were identified. Baseline demographic variables recorded were age and gender. Patients were classified as having RVO, RAO, or both based on the ophthalmological examinations at the time of diagnosis. Thrombophilia evaluation included FVL, PGM, AT, PC, PS, aPL, serum homocysteine, and activity of FVIII, FIX, and FXI. 19 Cardiovascular risk factors were identified from a chart review of previously diagnosed conditions. The risk factors were hypertension, hyperlipidemia, diabetes, and smoking. Additional thrombotic risk factors evaluated for women were the use of female hormones including second- or third-generation combined oral contraceptives, progesterone-only contraceptives, fertility medications, hormonal therapy, and selective estrogen receptor modulators or concomitant pregnancy or puerperal state.
Factor V Leiden and PGM polymorphisms were tested using the polymerase chain reaction. Protein C and AT activity were measured using chromogenic assays. Protein C activity <70% and AT activity <80% were considered deficient. Protein S activity was measured with a STACLOT LA assay according to the manufacturer’s instructions and <60% was considered low. Free and total PS antigen levels were measured by enzyme-linked immunosorbent assay (ELISA) and were considered deficient if <60% and <70%, respectively. Functional levels of FVIII, FIX, and FXI were measured using the Behring Sysmex CA 1500 Coagulation analyzer (Dade Behring: Siemens, Germany) with factor VIII-, IX-, and XI-deficient plasmas. Levels of these factors were considered increased if the levels were greater than 150%. 19 –21 Lupus anticoagulant testing used the dilute Russell viper venom time (dRVVT) with the CRYOcheck dRVVT screening reagent and dRVVT confirmatory reagent followed by the STACLOT LA 20 (Diagnostico Stago, France) as a confirmatory test. For the purposes of this study, a lupus anticoagulant test result was considered positive if the results of either the aPTT or the dRVVT mixing studies or the confirmatory lupus anticoagulant tests was positive. The anticardiolipin (aCL) and anti-β 2 glycoprotein I immunoglobulin (Ig) G and IgM testing were performed on serum samples using a commercial ELISA technique. Positive results were IgG greater than 20 aCL IgG/dL and IgM greater than 20 aCL IgM/dL. To be classified as aPLA positive, patients had to test positive on 2 different occasions at least 12 weeks apart for any of the aPLs, lupus anticoagulant alone, or both aPLs and lupus anticoagulant. Total homocysteine concentration was measured by a standard enzymatic method using the Beckman-Coulter-DXC and was considered elevated if the level was >12 µmol/L.
Treatments with antiplatelet agents, oral or parenteral anticoagulation, and intravitreal injections were assessed. Patients were followed at serial visits by both hematologists and ophthalmologists. Visual acuity was measured at initial presentation and then at the time of follow-up, approximately 6 to 12 months from initial presentation. Visual symptoms were assessed at each visit. Outcomes were classified as “complete resolution” if the patient reported that visual symptoms had resolved, “partial resolution” if the patient reported some improvement in visual defect but did not improve to baseline, “unchanged” if the patient did not report any change in vision, and “progression” if the patient reported interval worsening of vision.
The institutional review board approved the study, and the requirement for informed consent was waived.
Statistical Analysis
Descriptive statistics (including frequency and percentage) were calculated to characterize the RAO and RVO patient groups. Fisher exact test was used to compare thrombophilia risk factors, cardiovascular risk factors, and clinical outcomes between the RAO and the RVO groups. All P values are 2 sided with statistical significance evaluated at the .05 α level. All analyses were performed in SPSS Version 21.0 (SPSS Inc, Chicago, Illinois).
Results
A total of 39 patients (25 women and 14 men) with retinal vascular occlusions were identified. The mean age was 51 years (range 22-86). In all, 17 patients (10 women and 7 men) had RAO, 20 patients (13 woman and 7 men) had RVO, and 2 patients had combined RAO–RVO. Both patients with RAO–RVO were female. Thrombophilia testing and cardiovascular risk factors for RAOs and RVOs are listed in Table 1.
Risk Factors Associated With Retinal Artery Occlusions (RAO) and Retinal Vein Occlusions (RVO).
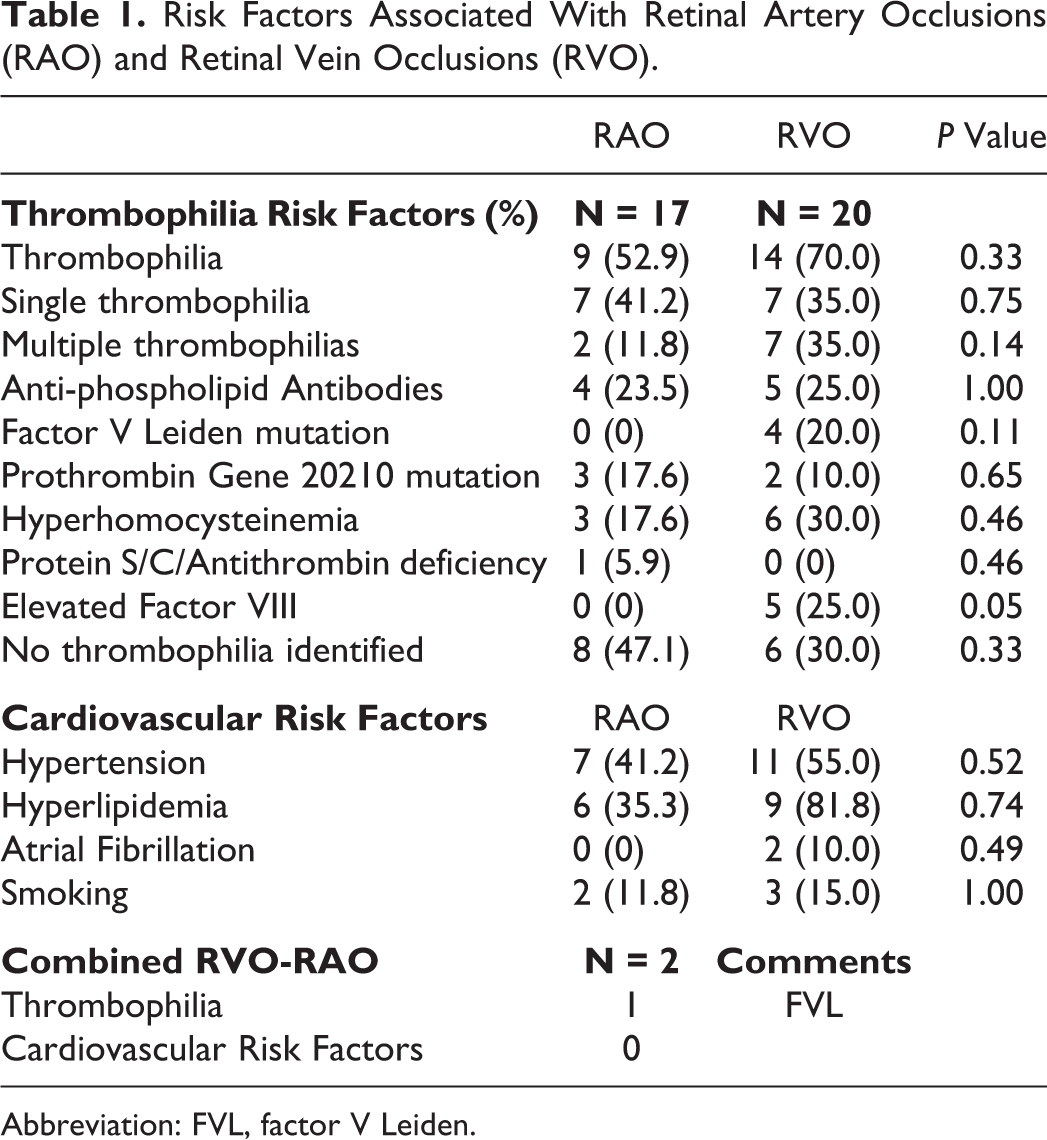
Abbreviation: FVL, factor V Leiden.
Thrombophilias were detected in most patients with RAO. Of the 17 patients, 9 had at least 1 identified thrombophilia. Of these, 4 patients had aPLs, 3 patients had PGM, 3 patients had elevated homocysteine, and 1 patient had PS deficiency. No patients with RAOs had FVL or elevated coagulation factor levels. Similarly, cardiovascular risk factors were identified in most patients. Hypertension was observed in 7 patients, hyperlipidemia in 6, and 2 were smokers. None of the patients had diabetes. One female patient was using oral contraceptive pills (OCPs), and 2 patients had RAOs during pregnancy or the puerperium (Table 2).
Risk Factors in Female Patients With RVO and RAO.
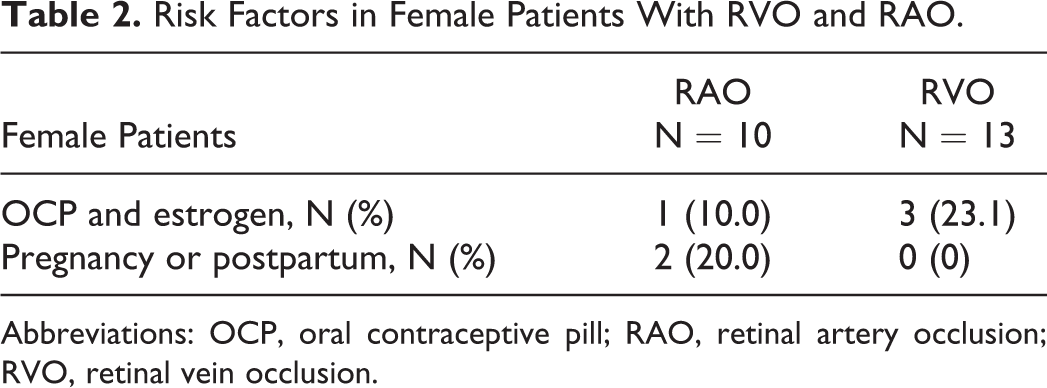
Abbreviations: OCP, oral contraceptive pill; RAO, retinal artery occlusion; RVO, retinal vein occlusion.
More thrombophilias were detected in the RVO group compared to the RAO group. However, only elevated FVIII levels that were seen in the RVO group but not in the RAO group were statistically significant. Of the 20 patients, 14 had at least 1 identified thrombophilia and 7 patients had more than 1 thrombophilia (Table 1). Antiphospholipid antibodies were detected in 5 patients, FVL in 4, PGM in 2, elevated homocysteine in 6, and elevated FVIII in 5. We did not identify any individuals with PC, PS, or AT in this group. Cardiovascular risk factors in patients with RVO included hypertension in 11, hyperlipidemia in 9, and smoking in 3. Two patients had atrial fibrillation and were on anticoagulation treatment at time of presentation. No patients had diabetes. Three female patients used OCPs or estrogen-containing products (Table 2).
Aspirin treatment was used by a total of 22 patients. In all, 13 patients were taking 81 mg daily and 8 were taking >81 mg. One patient used aspirin plus clopidogrel. Other treatments were also prescribed at the physician’s discretion. In all, 5 patients were on warfarin, 2 were 2 with low-molecular-weight heparin (LMWH) and 3 were treated with folic acid. One patient received intraocular bevacizumab and laser therapy only.
After initial presentation, 28 of the 39 patients were available for interval follow-up at 6 to 12 months with the hematologist and the ophthalmologist. Patients were also queried as to the change in their vision after the initiation of treatment. Treatment outcomes varied significantly in our patients with either type of retinal vascular occlusion (Table 3). Of the 7 patients with RAO taking aspirin, 5 reported at least some improvement in vision. Of the 3 patients with RAO on warfarin, 2 had improvement in vision. One patient had spontaneous resolution without treatment. The patient treated with LMWH and the patient treated with folic acid did not improve (Table 3). Of the 15 patients with RVO who had follow-up data, 12 were on aspirin. Of these 12 patients, 8 had no improvement in vision while 3 had improvement. One patient was treated with warfarin with some improvement in vision. Two patients had spontaneous complete vision recovery without treatment. One asymptomatic patient was treated with aspirin without improvement in fundoscopic examination. There were no patients treated only with clopidogrel or folic acid in this group who were available for follow-up. One patient with combined RAO–RVO was available for follow-up. This patient was treated with bevacizumab and photocoagulation but did not have improvement in vision.
Treatment and Follow-Up.
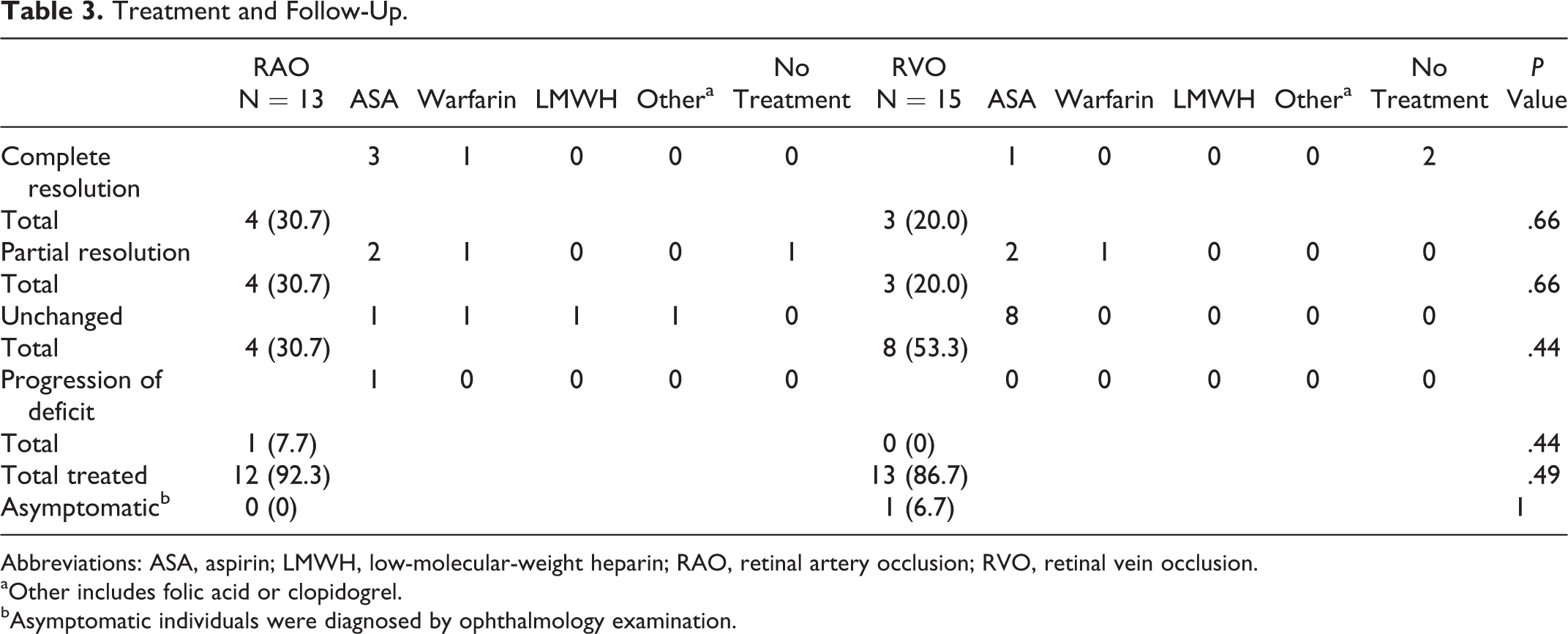
Abbreviations: ASA, aspirin; LMWH, low-molecular-weight heparin; RAO, retinal artery occlusion; RVO, retinal vein occlusion.
aOther includes folic acid or clopidogrel.
bAsymptomatic individuals were diagnosed by ophthalmology examination.
Discussion
In this small retrospective study, a thrombophilia was identified in the majority of a series of 39 consecutive patients presenting with RAO and/or RVO as in many previous reports. 6 –8,13,22 We also observed that hypertension and hyperlipidemia were the 2 most common cardiovascular risk factors in both the groups. With the exception of FVIII elevation, we found a similar incidence of thrombophilias, cardiovascular risk factors, and oral contraceptive use. The most prevalent acquired thrombophilias identified in our study were aPLs and elevated homocysteine, which have been previously reported in patients with RVO and is unrelated to the C677T MTHFR mutation. 14 –17,23 One patient using folic acid did not experience improvement in our study, also in agreement with the cardiovascular literature. 24
The finding of elevated FVIII in RVO but not in RAO is of interest. Elevated FVIII is recognized as a risk factor for systemic thrombosis, and this risk increases incrementally with every 10% increase in FVIII activity, even in the absence of systemic inflammation. 21,25,26 The previous literature is conflicting on this issue in the setting of retinal vascular occlusions. One study did not find an association between unselected RVO and FVIII levels, but another recent report suggested significantly elevated levels of FVIII were present in patients with ischemic RVO. 22,27 This finding deserves further exploration in larger multicenter studies.
Many studies have attempted to clarify what treatments are effective in these patients. Most recently, a study by Napal Lecumberri suggested that aspirin was the preferred option in the treatment of RVO, with traditional anticoagulation useful only in high-risk thrombophilias 28 . Other studies have found improvement in vessel recanalization and visual outcomes in patients on LMWH, warfarin, or aspirin after 6 months. In addition, aspirin maintenance after antithrombotic treatment with LMWH or warfarin decreased the risk of recurrence over a follow-up period of 5 to 10 years. 29
Low-molecular-weight heparin was underutilized in our study. Aspirin and LMWH have been compared as treatments for RVO, and LMWH has consistently resulted in superior composite visual outcomes with minimal bleeding risks. 30,31 In our population, aspirin did not seem highly effective in the initial treatment setting, although 19 patients were placed on aspirin. The formation of small platelet aggregates has been reported in patients with RVO, but interestingly only ticlopidine or beraprost, but not aspirin, decreased these aggregates ex vivo. This may imply that primary platelet responses might be more important than thromboxane synthesis in the development of RVO and explain some of the unresponsiveness to aspirin therapy. 32 In some RVOs, vision generally improves over time without intervention, but a return to baseline visual acuity is not common. 33 Novel treatment options for RVO include laser photocoagulation, local steroids, hemodilution, and systemic angiogenesis inhibition, but more studies are needed,. 3,34 –37 Both RVO and RAO were recently linked to stroke/TIA risk in patients with atrial fibrillation. 38 In our present study, both patients with atrial fibrillation had events while on anticoagulation. This is consistent with other studies where warfarin used for stroke prevention did not result in a protective effect against RVO. 28 Practitioners should be aware that retinal vascular occlusions are still possible in patients on systemic anticoagulation with warfarin.
Our study is limited by our small referral population. We estimate that our center sees a large number of retinal vascular occlusion patients (over 100 per year), and only a small fraction of patients are referred to hematology clinics. Our thrombophilia detection rates of 58.8% in RAO and 70% in RVO are significantly higher than that in the general population or in patients with a diagnosis of deep vein thrombosis, where thrombophilias occur at a frequency of 0.1% to 8% and 2% to 18%, respectively. 39,40 However, these results should be interpreted in the setting of this small retrospective referral population. We cannot make larger inferences as to the background prevalence of these thrombophilias without larger numbers of patients. Similarly, defining risk factors longitudinally is difficult. For example, precise information on the control of cardiovascular risk factors at the time of thrombosis was not available to us. We also did not include patients in our study with prior venous thrombosis, thus the RVOs were all sentinel events that triggered testing for thrombophilias. We are currently collecting information on these patients in terms of long-term risk of cardiovascular disease or thrombosis after presenting with a retinal vascular occlusion.
Overall, the benefit of thrombophilia screening in retinal vascular occlusions is uncertain. However, since patients may be at higher risk of future systemic thrombotic events, screening could help guide treatment. 18 For example, in a patient presenting with a central retinal vein thrombosis in whom a lupus anticoagulant is identified and confirmed, treatment with a vitamin K antagonist may be required. Thrombophilia screening may be particularly effective in younger patients without cardiac risk factors who develop RVOs or RAOs. Further studies should address long-term outcomes of patients with retinal vascular occlusions in the setting of thrombophilias and the role of established and newer anticoagulation agents as treatment options.
Footnotes
Acknowledgments
We would like to acknowledge Dr Babette Weksler for her review and thoughtful comments on the article. We would also like to acknowledge Karen Powell-Boone for her assistance with data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Dr Paul Christos was partially supported by the following grant: Clinical Translational Science Center (CTSC; UL1-TR000457-06).
