Abstract
Objective:
The aim of this study was to evaluate the relationship between vitamin D levels and hemostatic factors like tissue factor pathway inhibitor (TFPI).
Methods:
Patients who had 25-hydroxyvitamin D3 (25(OH)D3) levels measured were included. Coagulation and hemostatic parameters were evaluated. Patients were divided into 3 groups based on 25(OH)D3 levels as group 1 (25(OH)D3 < 10 ng/mL, n = 25), group 2 (25(OH)D3 = 10-19.9 ng/mL, n = 22), and group 3 (25(OH)D3 ≥ 20 ng/mL, n = 28).
Results:
A total of 75 patients with a mean age of 39 (range 18-57) years were included in the study. Prothrombin time was longer in group 3 than in group 2 (P = .043). The TFPI levels were higher in group 3 than in the other groups (P < .001). There was a strong positive correlation between 25(OH)D3 and TFPI levels (r = .47, P < .001).
Conclusion:
Further studies are needed for evaluation of the role of TFPI in hemostasis and thrombotic process in patients with vitamin D deficiency.
Introduction
Vitamin D is not a pure vitamin, because it is not only taken by food but it is also mainly synthesized by the body with the help of sun exposure. 1,2 Vitamin D deficiency is a highly prevalent condition worldwide, reportedly present in 30% to 60% of the general population. 3 –5 Clinically, vitamin D3 has a key role in calcium homeostasis and bone mineralization. In recent years, there has been an accumulating data from studies that support an association between inadequate vitamin D3 status and increased risk of cardiovascular disease (CVD) and death. 6
Receptors of vitamin D have been found in various tissues, including the vascular endothelium. The role of vitamin D in arterial disease is controversial; it has been found that both high 7,8 and low 9 levels were associated with myocardial infarction.
The function of vitamin D in the hemostatic process is uncertain. The thrombus formation is triggered by a damaged vessel wall that exposes tissue factor (TF) together with activated factor VIIa (FVIIa) to initiate the blood clotting cascade. 10 In addition, reduced fibrinolysis 11 and increased platelet adhesion 12 may contribute to thrombus formation. Increasing experimental evidence from cell culture studies suggests that vitamin D may exert anticoagulant effects, thus supporting the data that vitamin D derivatives may develop as new types of antithrombotic agents. 13
Laboratory tests, such as the prothrombin time (PT) and activated partial thromboplastin time (aPTT), are frequently used in clinical setting to assess the activity of the coagulation system. However, these tests do not provide much information about global coagulation, and they are not useful in evaluating the levels of TF or TF pathway inhibitor (TFPI). The TF/FVIIa complex is the primary initiator of coagulation in vivo. The TFPI is a protease inhibitor and is the primary physiological inhibitor of the TF/FVIIa complex. 14 Under normal conditions, vascular endothelial cells mostly express TFPI. The vast majority of TFPI in the body is bound to the endothelium. Approximately 20% of TFPI is present in the plasma where the majority is bound to the low-density lipoproteins and only almost 2% is unbound. 14 Both TF and TFPI have essential roles in hemostasis, and the alteration in the balance between the 2 proteins can lead to development of coagulation problems such as thrombosis or bleeding.
Inflammatory markers in the circulation are also potential predictors of the CVD. High-sensitivity C-reactive protein (hs-CRP) is accepted as a marker of low-grade inflammation and a predictor of CVD. 15 –17 Hyperhomocysteinemia is also an independent risk factor for atherosclerosis. 18,19 Elevated homocysteine levels are associated with coronary artery, cerebrovascular, and peripheral vascular diseases. 20
The aim of this study was to evaluate the vitamin D status and changes in levels of a hemostatic regulator such as TFPI. In addition, hs-CRP and homocysteine were measured for evaluation of predisposition to CVD.
Materials and methods
Study Design and Patient Selection
This cross-sectional study was done in the Department of Endocrinology and Metabolism. Patients presenting to the checkup outpatient clinic and whose 25-hydroxyvitamin D3 (25(OH)D3) levels were measured between December 2011 and February 2012 were included in the study. Patients with a history of diabetes mellitus, impaired glucose tolerance, laboratory findings of renal or hepatic dysfunction, a coagulation disorder, atherosclerotic disease, and those of any form of medication effect on coagulation and hemostatic parameters were excluded. Patients with a history of endocrine disorders that affect coagulation system like thyroid dysfunction, hyperprolactinemia, Cushing’s disease, or those with a previous clinical diagnosis of osteomalacia were also excluded. Patients were divided into 3 groups based on the levels of 25(OH)D3 as group 1 (25(OH)D3 < 10 ng/mL, n = 25), group 2 (25(OH)D3 = 10-19.9 ng/mL, n = 22), and group 3 (25(OH)D3 ≥ 20 ng/mL, n = 28). All individuals underwent physical examination, anthropometric measurement, and biochemical screening.
Height and weight were measured in light clothing wearing no shoes, and the body mass index (BMI) was calculated as weight divided by the squared height.
The local ethics committee approved the study protocol, and all participants gave written informed consent.
Blood Sampling and Laboratory Assay
Blood samples were collected and centrifuged after 30 minutes. We measured serum high-density lipoprotein cholesterol (elimination/catalase method), low-density lipoprotein cholesterol (LDL-C, elimination/catalase method), total cholesterol (enzymatic method), triglycerides (GPO, trinder without serum blank method), and glucose (glucose oxidase method) levels by spectrophotometric assay (Advia 2400, Siemens Healthcare Diagnostics Inc., Tarrytown). Fasting serum insulin was measured using the chemiluminescent immunoassay method (Advia Centaur XP, Siemens Healthcare Diagnostics Inc).
Plasma PT (Thromborel S reagent), aPTT (Dade Actin FSL aPTT reagent), fibrinogen, and
The 25(OH)D3 levels in serum samples were studied by direct competitive chemiluminescence immunoassay method (Diasorin Liaison, Italy).
Plasminogen activator inhibitor 1 (PAI-1) and thrombomodulin concentrations were quantitatively determined by commercial enzyme-linked immunosorbent assay (ELISA) kits (eBioscience, Austria; Uscn Life Science Inc, China), according to the manufacturers’ instructions. The intraassay and interassay coefficients of variation for thrombomodulin were <10% and <12%, respectively. The plasma TFPI level was measured using ELISA kit (Uscn Life Science Inc).
Statistical Analysis
Normal distributions of the quantitative variables were analyzed by Kolmogorov-Smirnov test. Parametric tests were applied when variables distributed normally, and nonparametric tests were applied when variables did not distribute normally. Parametric test results were given as means ± standard deviations. Nonparametric test results were given as means, minimum–maximum. Qualitative variables were presented as counts and proportions. For normally distributed variables, analysis of variance was applied to evaluate the differences among groups, and Bonferonni test was used for post hoc analysis. Comparison of variables, which did not distribute normally, was performed using Kruskal-Wallis test. To evaluate the correlation between the 25(OH)D3 levels and other variables, we used the Pearson/Spearman correlation. A P value <.05 was considered as statistically significant. Statistical analysis was performed using the SPSS version 17.0 software (SPSS Inc, Chicago, Illinois).
Results
A total of 75 patients attended the examination, and their basal characteristics are shown in Table 1. Distribution of patients based on 25(OH)D3 was as follows: group 1, 25 patients with a mean age of 32.4 years; group 2, 22 patients with a mean age of 34.0 years; and group 3, 28 patients with a mean age of 33.9 years. The difference between groups was not statistically significant (P > .05). Gender distribution in groups was also comparable (P = .097; Table 1).
Basal Characteristics and Laboratory Findings of Study Population.
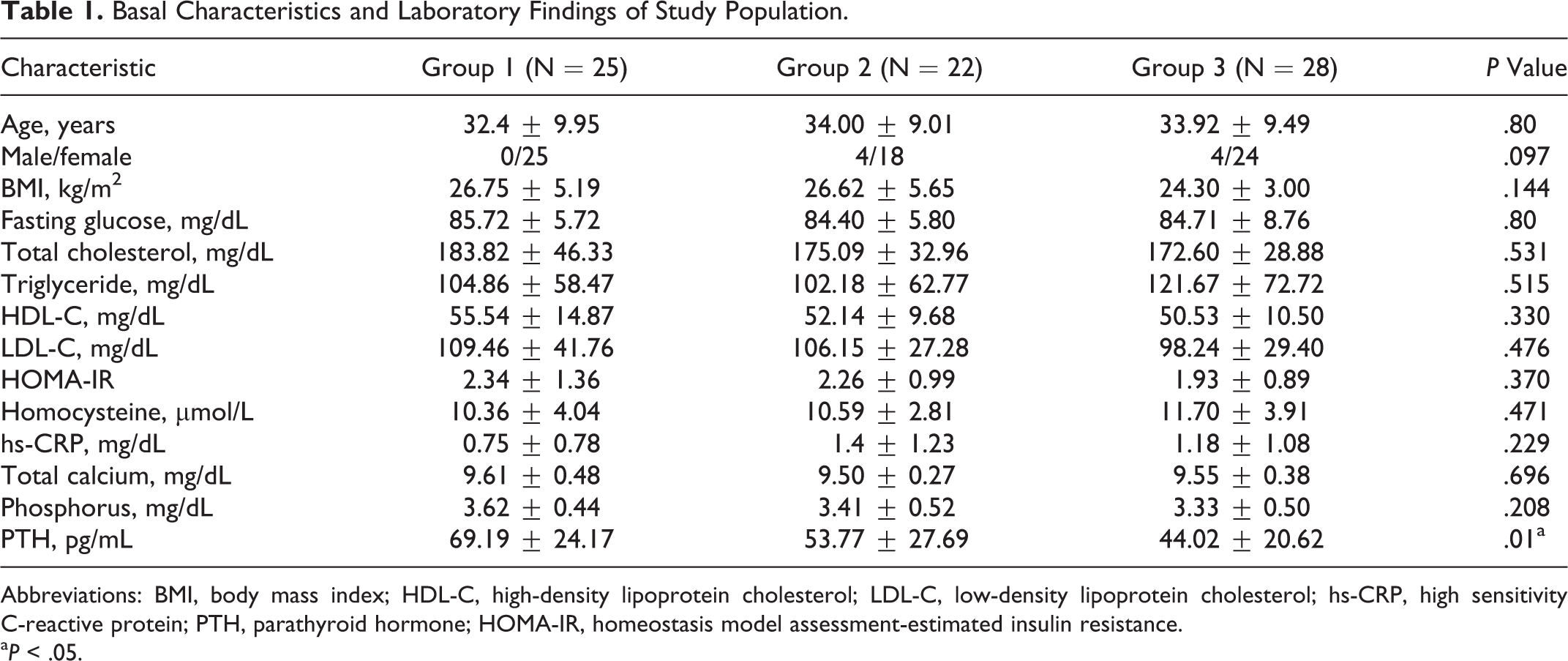
Abbreviations: BMI, body mass index; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; hs-CRP, high sensitivity C-reactive protein; PTH, parathyroid hormone; HOMA-IR, homeostasis model assessment-estimated insulin resistance.
a P < .05.
The mean TFPI levels of groups 1 and 2 were each significantly lower than that of group 3 (P < .001; Figure 1). The difference between group 1 and group 2 was evaluated as statistically insignificant (P = .938). The PT of group 3 was found as prolonged when compared to the other 2 groups, but all 3 groups had PT levels within normal ranges. This difference was evaluated as statistically significant (P < .05). Comparisons of other hemostatic and coagulation parameters of groups are shown in Table 2.
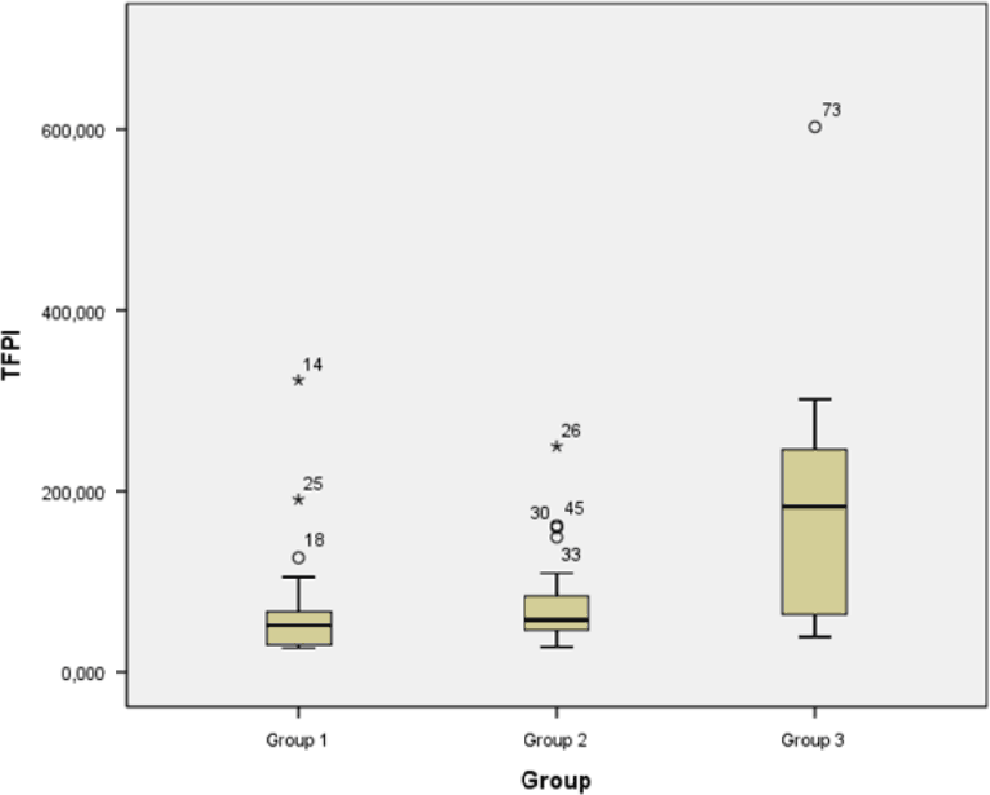
Comparison of tissue factor pathway inhibitor (TFPI) levels between groups.
Distribution of Hemostatic and Coagulation Parameters According to Groups.
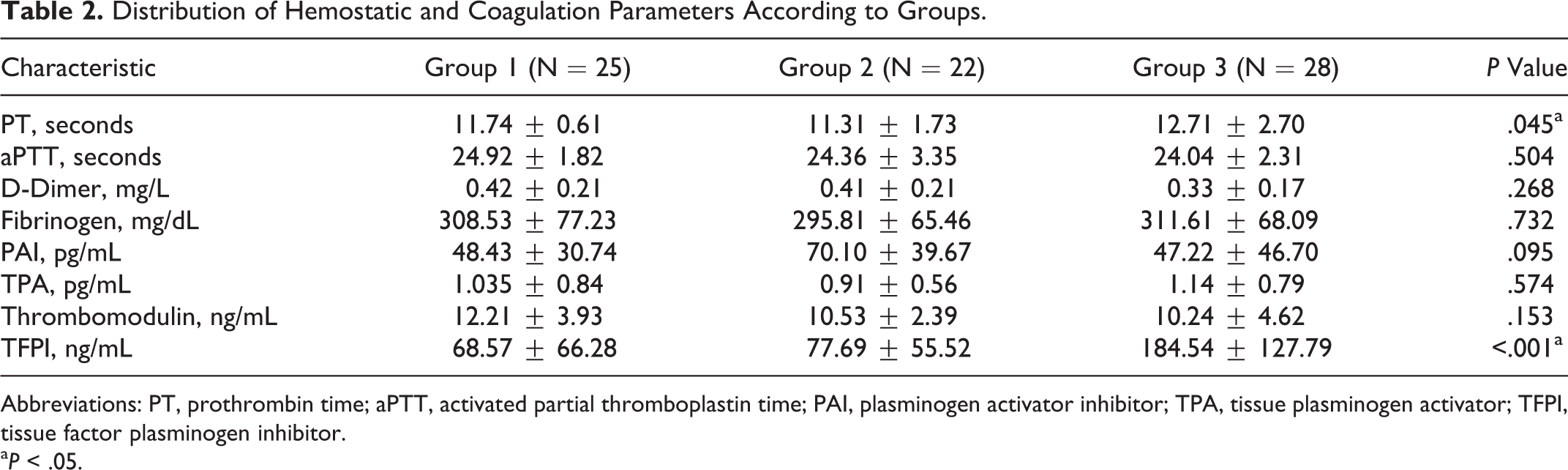
Abbreviations: PT, prothrombin time; aPTT, activated partial thromboplastin time; PAI, plasminogen activator inhibitor; TPA, tissue plasminogen activator; TFPI, tissue factor plasminogen inhibitor.
a P < .05.
Levels of TFPI did not correlate with serum calcium, phosphorus, or parathyroid hormone (Table 3). On the other hand, a significant positive correlation was established between 25(OH)D3 and TFPI (r = .47, P < .001; Figure 2). However, a similar correlation could not be found for the other hemostatic and coagulation parameters.
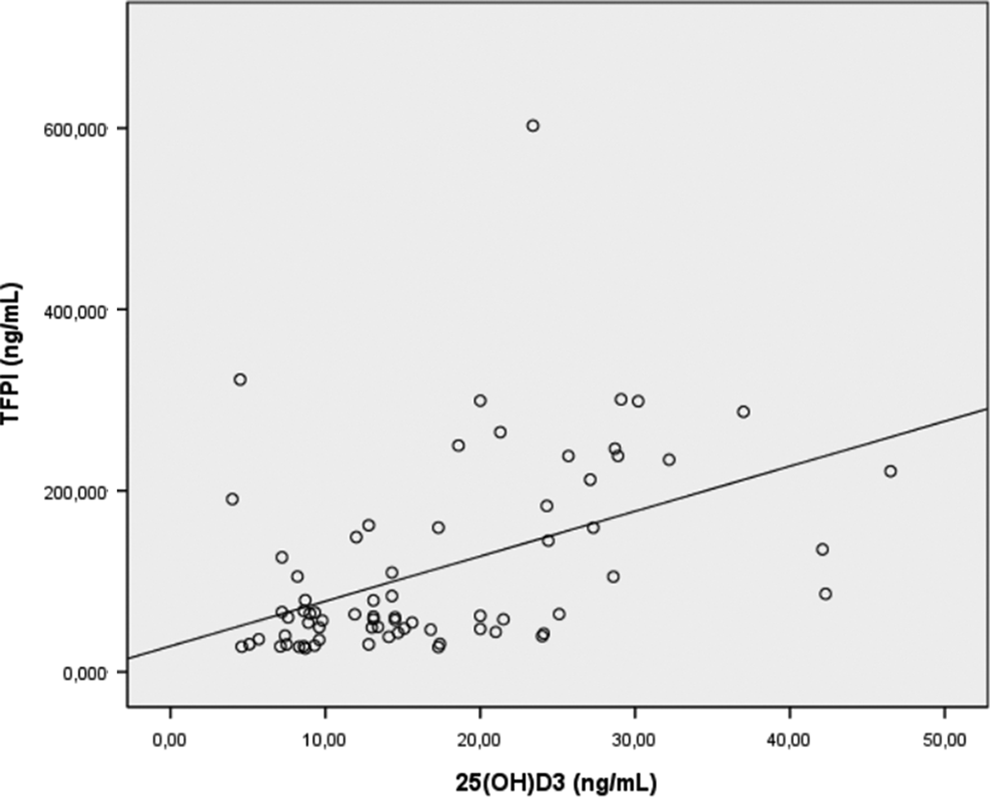
Correlation between 25(OH) vitamin D3 and tissue factor pathway inhibitor levels.
Results of Correlation Analysis Between TFPI and Vitamin D, Hemostatic and Coagulation Parameters, hs-CRP, and Homocysteine.
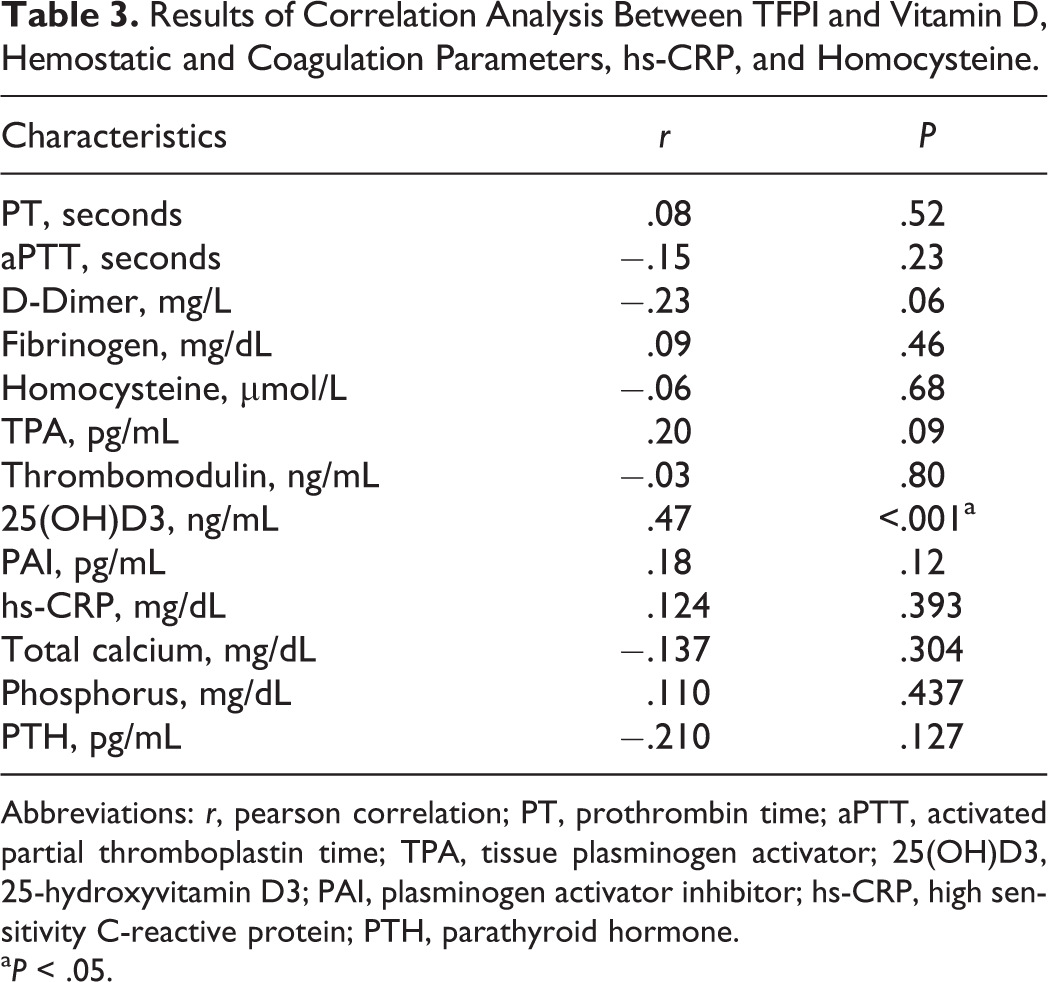
Abbreviations: r, pearson correlation; PT, prothrombin time; aPTT, activated partial thromboplastin time; TPA, tissue plasminogen activator; 25(OH)D3, 25-hydroxyvitamin D3; PAI, plasminogen activator inhibitor; hs-CRP, high sensitivity C-reactive protein; PTH, parathyroid hormone.
a P < .05.
Discussion
In the present study, we found a significant positive association between serum 25(OH)D3 and TFPI levels. We also demonstrated higher PT values in patients with vitamin D levels ≥20 ng/mL. According to our knowledge, this is the first report evaluating the TFPI, which is known as major inhibitor of coagulation, in patients with vitamin D deficiency.
Vitamin D deficiency has been suggested as a cardiovascular risk factor. There has been increasing data about vitamin D3 status and thrombolytic profile before progression to CVD. Tissue-type plasminogen activator (tPA) is predominantly an endothelial cell enzyme. It is released from the vascular endothelium and acts on plasminogen to produce plasmin. In plasma, it circulates as a complex with its natural inhibitor PAI. Both tPA and fibrinogen bind to fibrin in the fibrin clot. Measurement of tPA may be useful as an indicator of endothelial dysfunction.
21
The tPA is also thought as a risk marker for atherothrombotic disease.
22
The PAI-1 level was accepted marker for fibrinolytic activity.
23
There have been a few studies evaluating the association between these parameters and vitamin D status in the literature. Hyppönen et al reported that serum 25(OH)D3 level was inversely associated with tPA, fibrinogen, and
An upregulated expression of thrombomodulin by 1,25(OH)2D3 has been reported in osteoblasts. 28 Koyama et al demonstrated that activated vitamin D3 exerts anticoagulant effects by upregulating the expression of an anticoagulant protein, thrombomodulin, and downregulating the expression of TF as a coagulation factor in monocytic cells. 29 In a further study, the same group reported that 1,25(OH)2D downregulates TF and upregulates thrombomodulin expression in monocytic cells, counteracting the effect of oxidized LDL-C. 30
The hs-CRP is accepted as a marker of endothelial dysfunction, 15 and it is also a risk factor for coronary heart disease. 17 Ngo et al reported that lower levels of 25(OH)D were associated with increased plasma hs-CRP in a normal population cohort of 253 individuals aged 51 to 77 years. 31 Jorde et al also found a significant and negative association between 1,25(OH)2D and hs-CRP that disappeared after adjustment for BMI. 25 Cigolini et al demonstrated that type 2 diabetic patients with vitamin D3 deficiency had significantly higher plasma hs-CRP levels independent of anthropometric parameters, glycemic control, and traditional CVD risk factors. 32 Conversely, Shea et al did not find any significant associations between 25(OH)D, plasma hs-CRP and fibrinogen including 1381 participants from the Framingham Offspring Study. 33 We also did not find such an association between hs-CRP and vitamin D status. This can be also explained with the small study population size when compared with the previous studies.
The TFPI is primarily synthesized by the microvascular endothelium. It circulates in plasma but at very low concentrations. 34 About 20% of TFPI circulates in plasma in association with lipoproteins. 35 A low level of circulating TFPI has been shown to be a risk factor for developing venous thromboembolism. Dahm et al demonstrated that individuals with TFPI levels <10th percentile has a slightly increased odds ratio (OR: 1.7; confidence interval [CI], 1.1-2.6) for developing deep vein thrombosis (DVT). 36 In a case–control study of 122 patients with DVT, the total TFPI level was found as significantly lower in patients with DVT than in 126 controls. 37 Hoke et al demonstrated that low levels of TFPI, <second percentile, were associated with an increased risk of recurrent venous thromboembolism in a prospective multicenter study (relative risk: 2.7; CI, 1-7.4). 38 In addition, reduced levels of TFPI have been found in women taking oral contraceptives or treated with hormone replacement therapy, which may be one of the mechanisms for the increased thrombotic risk. 39 According to our knowledge, there are no studies evaluating the association between TFPI levels and vitamin D status in the literature. We demonstrated a significant positive correlation between 25(OH)D3 and TFPI levels.
The present study has some limitations. There is a wide range of TFPI among normal individuals. Our study population is too small to decide an association between low levels of TFPI and thrombotic event. Furthermore, this is a cross-sectional study, so changes in TFPI levels with vitamin D replacement therapy must be confirmed with prospective studies.
In conclusion, there was a positive association between serum vitamin D levels and TFPI. However, no statistically significant relation was found between 25(OH)D3 and other hemostatic and inflammatory markers. In the future, with the further studies, TFPI can be used as a predictor of thrombotic process like well-known other parameters such as PAI and TPA in patients with vitamin D deficiency.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
