Abstract
Background and Objective
Direct oral anticoagulants (DOACs) have been proven to be cost-effective for treating various conditions, including venous thromboembolism (VTE). Nevertheless, there are no studies assessing the cost-effectiveness of DOACs for VTE treatment in Saudi Arabia using real-world data. Hence, this study seeks to examine the costs and medical consequences of apixaban compared to rivaroxaban in treating VTE patients in Saudi Arabia.
Methods
A retrospective cohort study was carried out in three tertiary care hospitals spanning from January 2016 to December 2020. The measure of effectiveness is defined as the likelihood of preventing the composite of VTE recurrence (rVTE), major bleeding (MB), or clinically relevant non-major bleeding (CRNMB) within 90 days of the indexed VTE event, and rehospitalization due to rVTE, MB, or CRNMB. The effectiveness was determined by calculating 1 minus the probability of experiencing the composite outcome. The incremental cost-effectiveness ratio (ICER) was computed from the perspective of the Saudi National Health System, therefore only direct costs were considered. The 95% confidence interval surrounding mean costs and effectiveness rates was calculated using the bootstrapping method. Sensitivity analyses were also carried out.
Results
In the analysis, 367 patients were included, with 176 on apixaban and 191 on rivaroxaban. The average annual medication costs for apixaban and rivaroxaban were $547.05 and $577.77, respectively. The mean annual direct medical costs for apixaban and rivaroxaban were $6496.83 (95%CI 5748.86-7457.97) and $5528.58 (95%CI 4836.21-6024.52), respectively. Apixaban's and rivaroxaban's mean effectiveness rates were 0.91 (95%CI 0.87-0.96) and 0.77 (95%CI 0.71-0.83), respectively. This resulted in an ICER of $6916.07 for the prevention of an additional composite outcome when using apixaban instead of rivaroxaban. The use of apixaban for preventing composite outcomes has been found to be more effective but costlier in 94.81% of the bootstrap cost-effectiveness distributions compared to rivaroxaban. It was also found to be more effective and less costly in 5.19% of the bootstrap cost-effectiveness distributions.
Conclusion
Apixaban demonstrated superiority over rivaroxaban in preventing composite outcomes, encompassing rVTE, MB, CRNMB, and rehospitalization. Nevertheless, this was correlated with increased direct medical expenses. These findings emphasize the necessity of a well-rounded approach when choosing anticoagulants, considering both clinical effectiveness and economic considerations. This study's results are impactful for improving patient care and resource allocation, underscoring the pivotal role of cost-effectiveness in healthcare decision-making.
Introduction
Venous thromboembolism (VTE) is highly preventable, yet it poses a significant burden on healthcare systems. 1 It is estimated to impact around one million people in Europe, up to 900,000 people in the United States, and approximately 10 million people globally each year.2−4 In Saudi Arabia, an estimated 25,000 patients are affected by VTE annually. 5 Furthermore, the risk of VTE recurrence can be as high as 40% within ten years after the initial diagnosis. VTE can present as deep vein thrombosis (DVT) or pulmonary embolism (PE), with surgery, older age, immobility, and pregnancy being the most common risk factors for VTE. 1
Before the introduction of direct oral anticoagulants (DOACs) such as dabigatran, rivaroxaban, apixaban, and edoxaban, warfarin was the primary therapy for VTE. However, warfarin necessitates frequent international normalized ratio (INR) monitoring and interacts with various medications and foods.6,7 As a result, there was a need for alternatives with less frequent monitoring and fewer drug-drug and drug-food interactions in clinical practice. Clinical trials and real-world studies have demonstrated that DOACs are more effective than warfarin in preventing VTE recurrence and have similar or lower risks of bleeding events. Consequently, clinical practice guidelines now recommend DOACs over warfarin for treating patients with VTE.6,7
Limited data exists on the efficacy and safety of DOACs in the Saudi population. A real-world observational study that included 492 patients with VTE revealed that apixaban was as effective as warfarin in reducing VTE recurrence, but with fewer major bleeding events. 8 Economic evaluations conducted in high-income countries have demonstrated the cost-effectiveness of DOACs in treating VTE.9−11 In Saudi Arabia, some economic evaluations have been conducted to assess the cost-effectiveness of DOACs compared to warfarin in patients with atrial fibrillation (AFib), but none of them included patients with VTE. One study indicated that using rivaroxaban for stroke prevention in patients with AFib led to lower direct medical costs, resulting in a mean annual cost saving of $13,260.79. 12 Another study found that apixaban was a cost-saving option for patients with AFib. 13 As a result, this study aims to conduct a cost-effectiveness analysis of using apixaban versus rivaroxaban in the treatment of VTE in Saudi Arabia.
Method
Study Design, Setting, and Patients
A retrospective cohort study was conducted using data collected from January 2016 to December 2020 at three major hospitals in Riyadh, Saudi Arabia: King Abdulaziz Medical City (KAMC), King Saud University Medical City (KSUMC), and King Abdullah bin Abdulaziz University Hospital (KAAUH). The study focused on adult patients (aged 18 and above) who were treated with either apixaban or rivaroxaban for a new episode of VTE and were not on therapeutic anticoagulants when the first VTE incident occurred. The study aimed to compare the direct medical costs and effectiveness of apixaban with rivaroxaban. Effectiveness was measured as the probability of preventing a combination of VTE recurrence (rVTE), major bleeding (MB), or clinically relevant non-major bleeding (CRNMB) within 90 days of the initial VTE event, as well as re-hospitalization due to rVTE, MB, or CRNMB. Effectiveness was calculated as 1 minus the probability of experiencing the composite outcome. The study was approved by the Institutional Review Boards (IRBs) of the three sites: KAMC (ref. No. NRC21R/400/09), KSUMC (ref. No. E-21-6295), and KAAUH (Ref.# 22-0139).
Data Collection
We collected data on age, gender, body mass index (BMI), underlying health conditions (such as diabetes, hypertension, dyslipidemia, etc), history of major bleeding (MB) or clinically relevant non-major bleeding (CRNMB) within 12 months before the index date, as well as other medical history including hypertension, diabetes, chronic kidney disease, Afib, coronary artery disease, valvular disease, stroke, transient ischemic attack, active smoking, and active cancer. The analysis considered the occurrence of rVTE, MB, or CRNMB within 90 days of the index date, as well as rehospitalization due to these events, to assess the effectiveness measure. The definitions of MB and CRNMB were in line with the criteria set by the International Society on Thrombosis and Haemostasis (ISTH). 14
The direct cost data covers the expenses related to prescription drugs such as anticoagulants (eg, rivaroxaban, apixaban, unfractionated heparin, enoxaparin, and fondaparinux), doctor office visits, hospitalizations, emergency room visits, lab tests (eg, CBC, lipid profile, liver function tests, renal function tests, etc), imaging studies (eg, CT scans, Doppler ultrasounds, Holter monitor tests, ECGs, etc), and packed red blood cells. The prices for medical services were sourced from the Saudi Ministry of Health cost center, and the acquisition costs for medications were obtained from the Saudi Food and Drug Authority (SFDA).
Statistical Analysis
The baseline characteristics and costs of included patients with VTE who were treated with apixaban or rivaroxaban were presented using descriptive statistics, including frequencies with percentages or means with standard deviations. To estimate the odds ratio for having the composite outcomes, we conducted a multivariable logistic regression controlling for the effect of all baseline characteristics and risk factors for having rVTE. Then, the effectiveness estimate was reported as adjusted odds ratio (OR) with 95% confidence interval (95%CI).
To generate the mean rate of effectiveness (ie, probability of not experiencing the composite outcome), a covariate-adjusted linear mixed model was conducted controlling for the effect of all baseline characteristics and risk factors for having rVTE. Then, to adjust the generated mean rate of effectiveness for the baseline characteristics of patients on apixaban or rivaroxaban, inverse probability of treatment weighting (IPTW) was conducted. Bias-corrected confidence intervals for the mean difference in effectiveness rates and total annual direct medical costs were calculated using bootstrapping with 10,000 replications to generate the bootstrap cost-effectiveness distributions.
Furthermore, univariate sensitivity analysis was conducted using the SFDA prices for apixaban and rivaroxaban as well as the discounted prices for the public healthcare institutions. The cost data were presented in United States dollar (USD) (1 USD = 3.75 Saudi Riyal (SAR)). All these statistical analyses were performed using the widely recognized SAS® version 9.4 (SAS Institute, Cary, NC, USA.), ensuring the validity of the results.
Results
Baseline Characteristics
Overall, 367 patients were included in the analysis; 176 were on apixaban, while 191 were on rivaroxaban. The mean age for the whole population was 53.1 ± 19.6 years, and patients in the apixaban group were significantly older than patients in the rivaroxaban group (55.7 ± 19.6 vs 50.7 ± 19.4 years; p-value = 0.0141). Most of the patients were female (64.3%), and there was no significant difference in their distribution between the two groups. Various underlying medical comorbidities existed in our patients, and obesity was the most prevalent condition, affecting nearly half of the population (49.3%). The number of patients with hypertension (46.0% vs 28.8%; p-value = 0.0006), diabetes (41.5% vs 26.7%; p-value = 0.0028), or stroke (12.5% vs 5.8%; p-value = 0.0241) in the apixaban group was significantly higher compared to the rivaroxaban group. The number of patients with cancer in the rivaroxaban group was higher than that of the apixaban group (0.6% vs 5.8%; p-value = 0.0052). In terms of other risk factors for VTE, smoking and history of surgery were more prevalent in the rivaroxaban group compared to the apixaban (for smoking: 19.3% vs 38.2%; p-value=<0.0001 and surgery: 13.1% vs 24.1%; p-value = 0.0070). These data were summarized and depicted in Table 1.
Patient Baseline Characteristics for Patients on Apixaban (n = 176) Rivaroxaban (n = 191).

The numbers in the table are presented as means ± Standard deviation or frequency (%).
Abbreviations: BMI: body mass index; VTE: venous thromboembolism.
Adjusted Association Between the use of Apixaban or Rivaroxaban and Composite Outcome
Patients who received apixaban had significantly lower odds of experiencing composite outcomes (OR = 0.33; 95%CI 0.166-0.649) compared to those who received rivaroxaban. However, it is important to note that this association was notably influenced by the presence of two VTE risk factors: being bedridden (OR = 2.44; 95%CI 1.299-4.564) or smoking (OR = 2.43; 95%CI 1.150-5.143). These findings were detailed in Table 2 of the multiple logistic regression analysis assessing the relationship between apixaban use and the composite outcome.
Multiple Logistic Regression for the Association Between the use of Apixaban and the Composite Outcome.

The composite outcome was calculated as dichotomous variable (0-1), where “1” denotes full effectiveness in preventing the composite outcome (ie, rVTE, MB, or CRNMB within 90 days of the index date and the rehospitalization resulting from these rVTE, MB, or CRNMB). Abbreviations: BMI: body mass index; VTE: venous thromboembolism; rVTE: recurrent venous thromboembolism; MB: major bleeding; CRNMB: clinically relevant non-major bleeding.
Cost-Effectiveness
The mean annual cost of medications for apixaban and rivaroxaban, based on SFDA prices, was $547.05 and $577.77, respectively as presented in Figure S1. In contrast, the mean annual direct medical costs for apixaban and rivaroxaban were $6496.83 (95%CI 5748.86-7457.97) and $5528.58 (95% CI4,836.21-6024.52), respectively. The mean effectiveness rates for patients on apixaban and rivaroxaban, with regards to preventing rVTE, MB, or CRNMB and associated rehospitalization within 90 days of the index date, were 0.91 (95%CI 0.87-0.96) and 0.77 (95%CI 0.71-0.83), respectively. Based on this data, the incremental cost-effectiveness ratio (ICER) to prevent an additional composite outcome with the use of apixaban as opposed to rivaroxaban was calculated at $6916.07, as reflected in Table 3. The analysis of the cost-effectiveness distributions revealed that the use of apixaban for VTE prevention was more effective but costlier in 94.81% of the bootstrap distributions, and more effective and less costly in 5.19% of the distributions, as indicated in Figure S2.
the Mean Effectiveness Rate and Treatment Cost for Patients on Apixaban (n = 176) Versus Rivaroxaban (n = 191) Using the SFDA Prices (in 2021).

The numbers in the table are presented as means with 95% confidence intervals (95%CI) * The mean effectiveness rate was calculated as dichotomous variable (0–1), where “1” denotes full effectiveness in preventing the composite outcome (ie, rVTE, MB, or CRNMB within 90 days of the index date and the rehospitalization resulting from these rVTE, MB, or CRNMB). Abbreviations: ICER: incremental cost-effectiveness ratio; rVTE: recurrent venous thromboembolism; MB: major bleeding; CRNMB: clinically relevant non-major bleeding; 95%CI: 95% confidence intervals.
Sensitivity Analysis
Based on the discounted tender prices for apixaban and rivaroxaban, the average annual medication costs were $481.56 and $417.47, respectively, as depicted in Figure S3. This resulted in mean annual direct medical costs of $6079.12 (95% CI 5338.31-7031.26) for apixaban and $5061.27 (95% CI 4378.24-5549.59) for rivaroxaban. In light of these findings, the incremental cost-effectiveness ratio (ICER) increased to $7270.36, as illustrated in Table 4. In the cost-effectiveness distributions, apixaban was deemed more effective but costlier in 95.88% of cases, and more effective and less costly in 4.12% of cases, when compared to rivaroxaban (as shown in Figure 1).
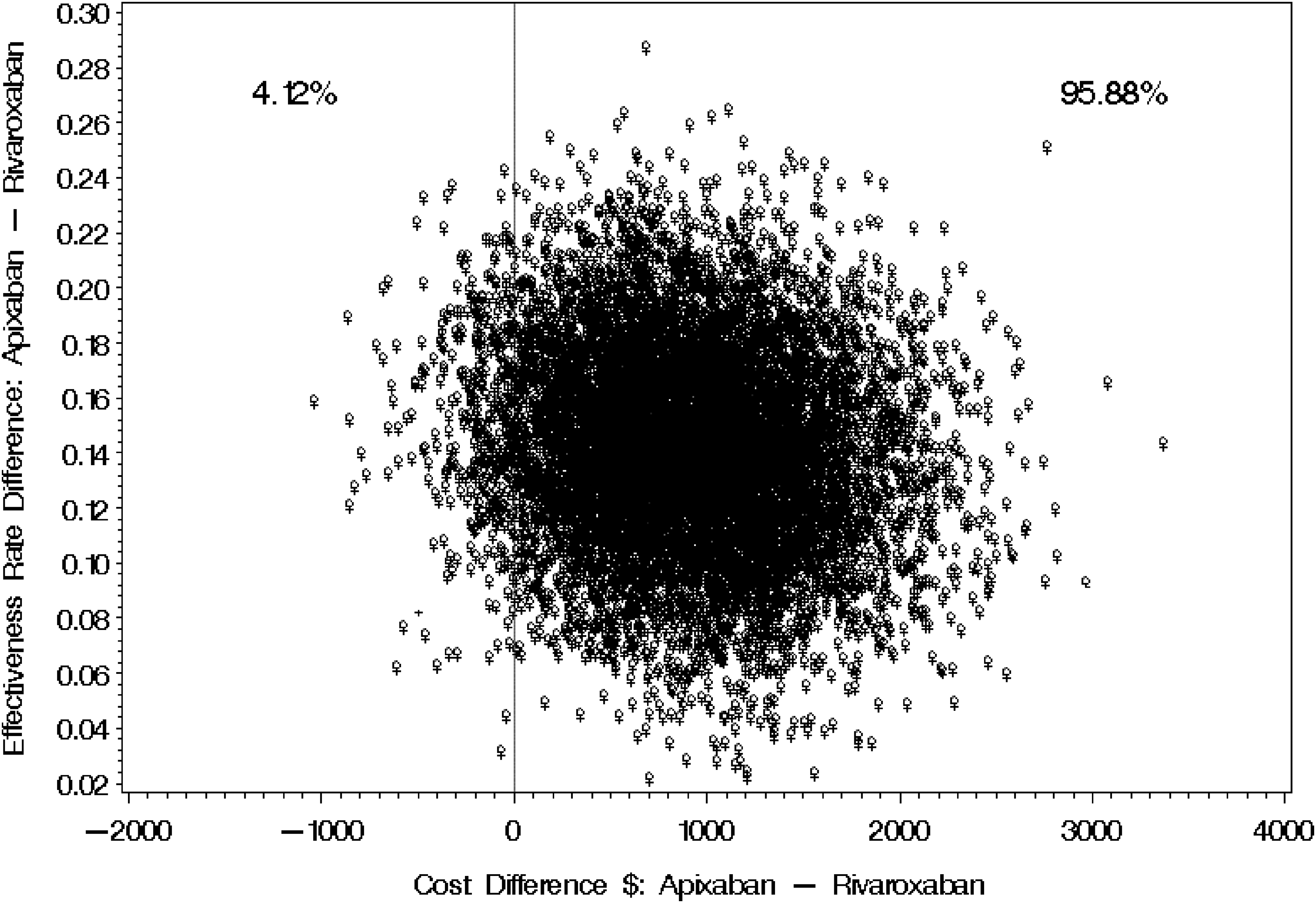
Bootstrap distribution of cost-effectiveness for Apixaban versus Rivaroxaban (Tender Price).
the Mean Effectiveness Rate and Treatment Cost for Patients on Apixaban (n = 176) Versus Rivaroxaban (n = 191) Using the Mean Tender Prices (2021–2023).

The numbers in the table are presented as means with 95% confidence intervals (95%CI).
The mean effectiveness rate was calculated as dichotomous variable (0–1), where “1” denotes full effectiveness in preventing the composite outcome (ie, rVTE, MB, or CRNMB within 90 days of the index date and the rehospitalization resulting from these rVTE, MB, or CRNMB).
Abbreviations: ICER: incremental cost-effectiveness ratio; rVTE: recurrent venous thromboembolism; MB: major bleeding; CRNMB: clinically relevant non-major bleeding; 95%CI: 95% confidence intervals.
Discussion
The use of DOACs for the treatment of VTE is well-established and recommended by various clinical guidelines. They have been shown to be more effective than vitamin K antagonists (VKA) with low-molecular-weight heparin (LMWH), and have a favorable safety profile in terms of bleeding risks.6,7,15,16 This study further expands on the existing evidence by conducting a thorough real-world head-to-head cost-consequence analysis comparing apixaban to rivaroxaban. The results of the study indicate that apixaban is more effective than rivaroxaban in reducing the composite outcome of rVTE, MB, and CRNMB within 90 days of the initial VTE event. However, it is associated with higher costs and an ICER of US$ 6916.1. This advantage persisted even after sensitivity analysis using tender prices, which raised the ICER to US$ 7270.4. It is worth noting that apixaban had an OR of 0.33 for the composite outcomes, which may have been significantly influenced by the presence of two risk factors: smoking and being bedridden.
In 2016, a study by Lanitis et al compared the cost-effectiveness of apixaban with other oral anticoagulants for VTE treatment over six months utilizing the Markov model and data from phase 3 trials. This analysis found apixaban to be a dominant alternative to rivaroxaban and LMWH/dabigatran in terms of efficacy and cost, regardless of the type of VTE, DVT or PE. The study also reported that apixaban was a dominant option for lifelong treatment in an exploratory analysis of treatment duration. 10 However, this study relied solely on data from phase 3 trials and did not use real-world data, like our case. A systematic review incorporating the Lanitis et al study and six additional studies concluded that apixaban demonstrated superior efficacy compared to other DOACs in preventing and treating VTE, followed by rivaroxaban, edoxaban, and dabigatran. 17 However, it's important to note that the data from these studies primarily relied on phase 3 clinical trials instead of direct comparisons between DOACs. Additionally, the studies were conducted in various countries (United States, United Kingdom, and Canada) using different economic models, which suggests that the findings should be interpreted with caution. Our current study provides unique real-world evidence supporting the superior effectiveness of apixaban over rivaroxaban, albeit with higher associated costs. Nevertheless, previous research has also demonstrated apixaban's superiority over other DOACs in terms of both cost and effectiveness when it comes to stroke prevention in patients with Afib.12,18,19
In contrast, when assessing the cost-effectiveness of apixaban versus rivaroxaban from the perspective of the United Kingdom National Health Service (NHS) and Personal Social Services (PSS), rivaroxaban was found to have marginally lower total costs and increased quality-adjusted life years (QALYs) compared to apixaban. 20 Furthermore, a study conducted by Sun et al examined the cost-effectiveness of DOACs versus VKA for VTE treatment in China and concluded that rivaroxaban was the most cost-effective option when compared to VKA + LMWH or apixaban. 21
Variations in reported cost-effectiveness results for DOACs in the literature may arise from several factors, such as differences in the definition of bleeding (major vs non-major), the duration of anticoagulation treatment, and reliance on phase 3 trials using Markov models rather than real-world data. Additionally, variations in patient baseline characteristics, types of VTE diagnoses, and the use of different economic models across countries contribute to the diversity of reported results. Notably, apixaban presents specific characteristics that make it a potentially cost-effective option for VTE treatment compared to rivaroxaban, particularly if the medication acquisition cost is reduced. These characteristics include a shorter lead-in dosing of 10 mg twice daily for seven days 22 when compared to rivaroxaban's 15 mg twice daily for 21 days, 23 potentially leading to a reduced duration of hospitalization for the index event management. Furthermore, recent systematic reviews and meta-analyses have highlighted apixaban's superior safety and efficacy, with reported reductions in rVTE, MB, and CRNMB compared to rivaroxaban.24−26 On the other hand, rivaroxaban offers the advantage of once-daily maintenance dosing, potentially enhancing patient adherence to the treatment regimen. 23
This study is the first to evaluate the cost-effectiveness of apixaban versus rivaroxaban for VTE management in Saudi Arabia using real-world data. Despite its strengths, the study has several limitations that should be taken into account. The retrospective design and data sourced from only three hospitals in Riyadh may restrict the generalizability of the findings to other regions or healthcare settings. Moreover, the treatment duration and follow-up period were confined to 90 days, limiting the applicability of the results to longer treatment periods. There is also a potential for information bias, given that the data were retrospectively extracted from electronic medical records. Additionally, considering the retrospective design and the availability of sufficient data, the analysis did not encompass patient-centered outcomes such as quality of life, treatment adherence, dietary factors, health access and socioeconomic status which are vital for a comprehensive assessment of the cost-effectiveness of these anticoagulants. Future research should address these factors, utilizing larger sample sizes, more diverse populations, and prospective study designs to validate our findings and offer deeper insights into the long-term cost-effectiveness of apixaban and rivaroxaban.
Conclusion
In summary, while apixaban demonstrates greater effectiveness in preventing combined outcomes such as rVTE, MB, CRNMB, and rehospitalization compared to rivaroxaban, it is tied to higher direct medical expenses. These findings highlight the need for a well-rounded approach when choosing anticoagulants, taking into account both clinical effectiveness and economic considerations to enhance patient care and resource management.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251318705 - Supplemental material for Cost-Consequence Analysis of Apixaban Versus Rivaroxaban for Managing Patients with Venous Thromboembolism in Saudi Arabia
Supplemental material, sj-docx-1-cat-10.1177_10760296251318705 for Cost-Consequence Analysis of Apixaban Versus Rivaroxaban for Managing Patients with Venous Thromboembolism in Saudi Arabia by Abdulaali R. Almutairi, Yazed Alruthia, Majed S. Alyami, Omar A. Alshaya, Taif Z. Alanazi, Sarah I. Al Daghreer, Ghazwa B. Korayem, Sarah A. Alrasheed, Reema A. Alorf and Omar A. Almohammed in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors would like to extend their appreciation to King Saud University for funding this work through the Researcher Supporting Project (RSP2025R77), King Saud University, Riyadh, Saudi Arabia.
Authors’ Contributions
All listed authors have significantly contributed to the research, providing direct and intellectual input throughout the process, and have given their approval for the publication of this work.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was approved by the supervising institutional review boards (IRB) at the three study sites; KAMC (ref. No. NRC21R/400/09), KSUMC (ref. No. E-21-6295), and KAAUH (Ref.# 22-0139), with the need for written consent waived by the ethical committee due to the retrospective nature of the study.
Funding
The author (OAA) received funding from the Researcher Supporting Project (RSP2025R77), King Saud University, Riyadh, Saudi Arabia to support the publication of this article. The funding agency played no role in designing the study, analyzing and interpreting the data, or writing the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
