Abstract
We aimed to investigate whether prolonged treatment with dalteparin could inhibit plaque progression. With C57BL/6J mice as the control, genetically deficient apolipoprotein E (apo E) male mice of C57BL/6J strain (apo E−/−) were randomly divided into 3 groups. The model group received no dalteparin, while the other 2 groups received dalteparin at 100 and 200 U/kg d, respectively. The aorta was harvested for hematoxylin and eosin staining to observe plaque formation and for immunohistochemical staining to detect the expression of oxidized low-density lipoprotein receptor 1 (LOX-1). The expression of LOX-1 messenger RNA was detected by reverse transcription polymerase chain reaction, while the expression of LOX-1 protein was detected by Western blotting. Dalteparin decreased aortic plaque volume and inhibited aortic LOX-1 protein expression in apo E−/− mice. The effect persisted 4 weeks after dalteparin treatment was discontinued. Dalteparin may inhibit atherosclerotic lesions by downregulating the expression of LOX-1 protein.
Introduction
Lectin-like oxidized low-density lipoprotein receptor 1 (LOX-1), a cell surface endocytosis receptor for atherogenic Ox-LDL, appears to play key roles in the pathogenesis of atherosclerosis. 1,2 The LOX-1 is involved in foam cell transformation of macrophages, apoptosis and necrosis of vascular endothelium, and migration and proliferation of smooth muscle cells. The LOX-1 is also correlated with plaque instability in human coronary atherosclerotic lesions. 3,4
Thrombin and factor Xa are 2 key factors in the formation of arterial thrombi and venous thrombosis. Our preliminary study of vascular smooth muscle cells indicated that thrombin and factor Xa could induce the expression of LOX-1 protein in a dose-dependent manner. 5 Low-molecular-weight heparins, which have anti-Xa and antithrombin activity, are important therapeutic agents in the treatment of patients with acute coronary syndromes and deep venous thrombosis.
Whether low-molecular-weight heparin causes inhibition of the LOX-1 receptor activity is unknown. We speculate that low-molecular heparin, by inhibiting the expression of factor Xa and thrombin, may decrease the expression of LOX-1 induced by thrombin and factor Xa, reduce the lipid deposition within plaques, and inhibit the plaque formation.
We selected dalteparin for this trial on the basis of the preparatory research. 6 In the Fragmin in Unstable Coronary Artery Disease (FRIC) study, prolonged treatment with dalteparin led to a trend toward reduced recurrence of angina, even though the dose of dalteparin was reduced to half. 7 In the Fast Revascularization During Instability in Coronary Artery Disease II (FRISC-II) trial, after continuous injection of dalteparin for 1 month, patients with unstable coronary artery disease had decreased mortality, myocardial infarction (MI), and vascular reconstruction rates. 8
The purpose of this study was to investigate whether prolonged treatment with dalteparin could inhibit atherosclerotic lesion formation by downregulating the expression of LOX-1 protein and explore the potential antiatherosclerotic mechanism of heparin apart from anticoagulation in apolipoprotein E-deficient (apo E−/−) mice, a murine model of atherosclerosis. 9
Materials and Methods
Animals
The C57BL/6J mice and apo E−/− C57BL/6J mice were purchased from Peking University Animal Laboratory (license SYXK 2006-0009; Beijing, china). All mice were male and 6 weeks old. The mice were kept in microisolator cages on a 12-hour day/night cycle. All experimental procedures and protocols involving animals were approved by the Institutional Animal Care Committee of Peking University People’s Hospital (Beijing, China).
The C57BL/6J mice were randomly divided into 2 groups, a normal diet control group and a high-fat diet control group. The high-fat diet consisted of a standard diet plus 21% lard plus 0.15% cholesterol. 10 The apo E−/− mice were all fed a high-fat diet and were randomly divided into 3 groups, model group, dalteparin 100 U/kg d group, and dalteparin 200 U/kg d group. There were 12 mice in each group. After 12 weeks of ingesting a high-fat diet, different dose of dalteparin were alternately injected subcutaneously in the right or left groin region for 4 weeks. The model group received no dalteparin, while the other 2 groups received dalteparin 100 U/kg d and dalteparin 200 U/kg d, respectively. Then, 6 mice from each group were randomly sacrificed to examine the effects of dalteparin. The remaining mice were fed without dalteparin treatment for another 4 weeks to study the delayed effects of dalteparin on atherosclerosis.
Main Reagents and Instruments
Polymerase chain reaction (PCR) primers were provided by CyberSyn (California, USA). Lowry protein reagent determiner was from BioRad (Pennsylvania, USA). Reverse transcription reagent kit was from Promega (Wisconsin, USA). The nitrocellulose membranes (Protran) were purchased from Protran and Schleicher and Schuell. The enhanced chemiluminescence (ECL) Western immunoblotting reagents were from Amersham (Uppsala, Sweden). Horse anti-mouse immunoglobulin G (IgG) was purchased from Amersham, and rabbit anti-LOX-1 polyclonal IgG was purchased from Abcam (item: ab31238; London, Britain). Dalteparin sodium (Fragmin) 5000 IU (anti-Xa)/0.2 mL was provided by Pfizer Limited (New York, USA).
Experimental Method
After sacrifice of the mice, the upper segment of the aorta was separated for hematoxylin and eosin (HE) and immunohistochemical staining. The lower segment of the aorta was used to detect the expression of LOX-1 messenger RNA (mRNA) by reverse transcription PCR (RT-PCR) and the expression of LOX-1 protein by Western blotting.
Hematoxylin and Eosin Staining and Pathological Morphology
The upper segment of the aorta was conventionally fixed, ethanol gradient dehydrated, and paraffin embedded. Slices were cut intermittently and uniformly with a slice thickness of 5 μm. One slice was taken from an interval of 5 slices and stained with HE. Three sections from each mouse sample were randomly chosen. Three fields in each slice were randomly selected for atherosclerotic lesion measurement using Image J software (NIH Company, Japan). The ratio of atherosclerotic lesion to wall area was calculated as was the ratio of lipid core area to plaque area.
Immunohistochemistry Detection
Paraffin slices were dewaxed, dehydrated, and incubated in 0.3% H2O2 in methanol. The antigen was repaired by high pressure. Slices were probed with rabbit anti-LOX-1 polyclonal antibody IgG (Abcam). Brown-stained particles were regarded as positive staining. Densitometric analysis was performed using Image J software.
Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted by trizol reagent, and the absorbency at the 260/280 nm was measured by an ultraviolet spectrophotometer. The complementary DNA was synthesized using a reverse transcript reagent kit. The PCR system was 25 μL, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal reference. The LOX-1 upstream primer was 5′-TGTCCACAAGACTGGCTCTG-3′, and the downstream primer was 5′-TGCACAGTTGCCTGATGAAT-3′. The LOX-1 PCR product was 300 bp. The PCR conditions for LOX-1 determination were, first denaturation at 94°C for 3 minutes, followed by 35 cycling reactions (94°C 30 s, 60°C 30 s, and 72°C 30 s), then extension at 72°C for 7 minutes. The GAPDH upstream primer was 5′-ACGCATTTGGTCGTATTGGG-3′, and the downstream primer was 5′-TGATTTTGGAGGGATCTCGC-3′. The GAPDH PCR product was 231 bp. The PCR conditions for GAPDH were, first denaturation at 94°C for 3 minutes, followed by 35 cycling reactions (94°C 30 s, 58°C 30 s, and 72°C 30 s), and then extension at 72°C for 7 minutes. After the amplification cycles, PCR products were subjected to agarose gel for electrophoresis. Densitometric analysis was performed using a gel-analyzing system.
Western Blotting Analysis
Briefly, the aorta was cut into pieces on ice, then the protein extracts were collected after centrifugation at 8000 rpm at 4°C. Protein concentrations were measured by the Lowry protein assay (BioRad). Equal concentrations of the protein extracts were subjected to sodium dodecyl sulfate-polyacrylamide (12%) gel electrophoresis and transferred onto nitrocellulose membranes (Protran; Schleicher and Schuell) by electroblotting. Membranes were probed with monoclonal antibody against LOX-1 (Abcam). Bands were visualized by ECL Western blotting detection reagents (Amersham). Densitometric analysis was performed using Image J software.
Statistical Analysis
Experimental data were reported as mean ± standard deviations of 3 repeated experiments. Single-factor (1-way) analysis of variance, after confirmation with the Kolgomorov-Smirnov test, was performed for quantitative analysis. The SPSS 13.0 software was used for statistical analysis (SPSS Inc, Chicago). A value of P < .05 was considered to indicate statistical significance.
Results
Skin and Hair
All mice survived until sacrifice. All C57BL/6J mice had generally good skin and hair condition. All apo E−/− mice showed hair loss (Table 1) and inactivity to outside stimulation. Compared with the apo E−/− model group, the dalteparin 200 U/kg d group showed less hair loss. During the whole process of heparin injection, there were no subcutaneous hemorrhages or bleeding.
Hair Loss in apo E−/− Mice.a

Abbreviation: apo E−/−, apolipoprotein E deficient.
a There was no statistical difference in the 3 groups (P = .21).
Effect of Dalteparin on Atherosclerotic Lesions in the Aorta
Hematoxylin and eosin staining showed that in C57BL/6J mice fed with a normal diet, the intima was smooth without atherosclerotic plaque formation, while in C57BL/6J mice fed with a high-fat diet, the intima was slightly thickened but without atherosclerotic plaque formation. In the model group of apo E−/− mice, the intima was thickened, and there were irregularly distributed plaques in which cholesterol crystals were found sometimes leading to stenosis. The size of the atherosclerosis was reduced after treatment with dalteparin (Figure 1). The plaque area–lumen area ratio and the lipid composition–plaque area ratio of the dalteparin 100 U/kg d group and the dalteparin 200 U/kg d group were both smaller than that of the model group (P < .05). Dalteparin 200 U/kg d inhibited the formation of atherosclerosis more than dalteparin 100 U/kg d (Table 2). The HE staining showed these differences still existed 4 weeks after cessation of dalteparin therapy.
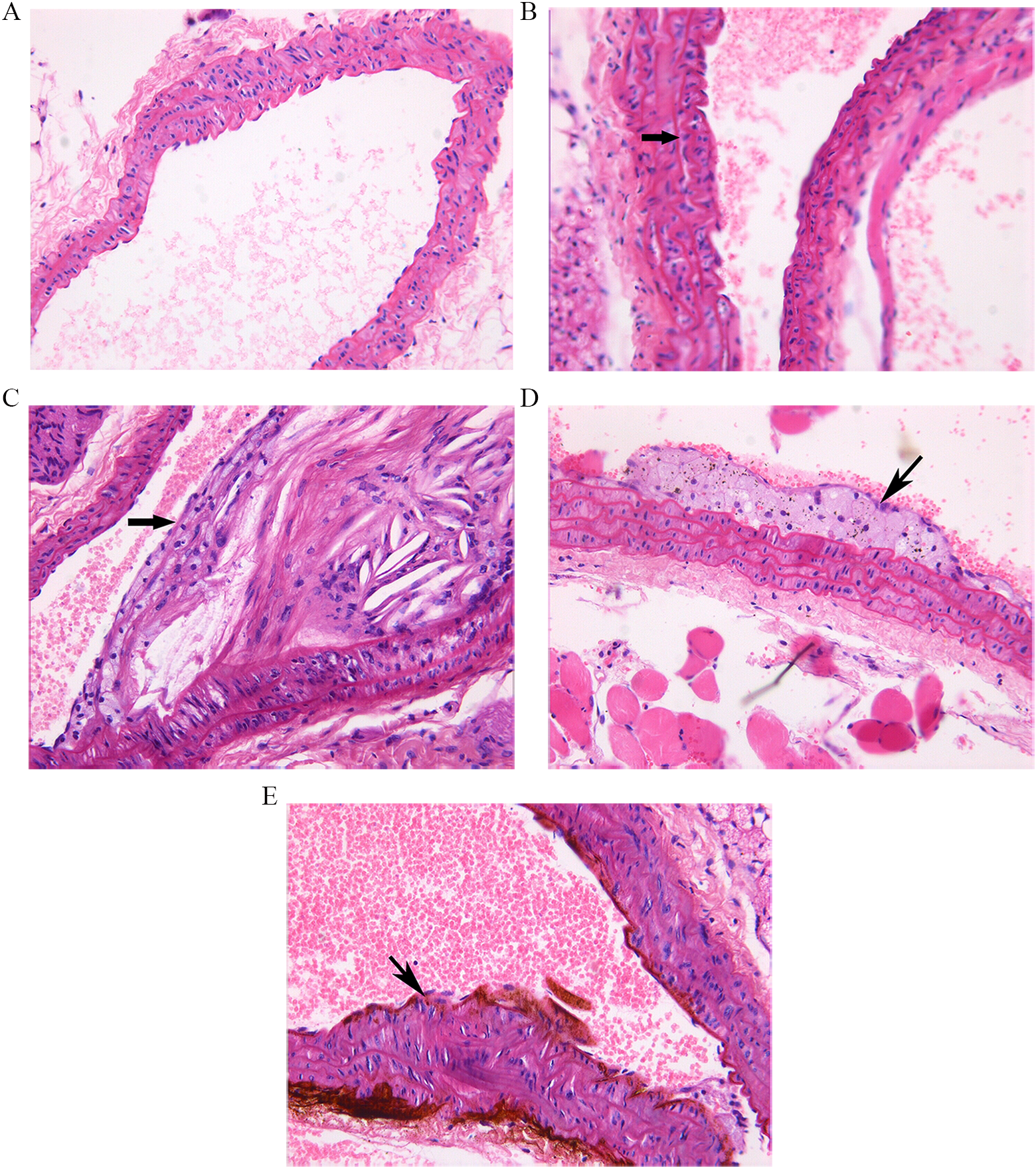
The 16-week atherosclerotic lesions in mice aortic root (hematoxylin and eosin [HE] staining ×200). A, The C57BL/6J mice of normal diet group showing no atherosclerosis. B, The C57BL/6J mice of high-fat diet group showing slightly thickened membrane and intima (arrow). C, Model group of apo E−/− mice showing apparent plaque formation (arrow), leading to lumen stenosis. D, Dalteparin 100 U/kg d group of apo E−/− mice showing moderate atherosclerotic plaques (arrow). E, Dalteparin 200 U/kg d group of apo E−/− mice showing slight atherosclerotic plaques (arrow). apo E−/− indicates apolipoprotein E deficient.
The Impact on apo E−/− Mice Aortic Atherosclerosis Lesions by Low-Molecular Heparin.
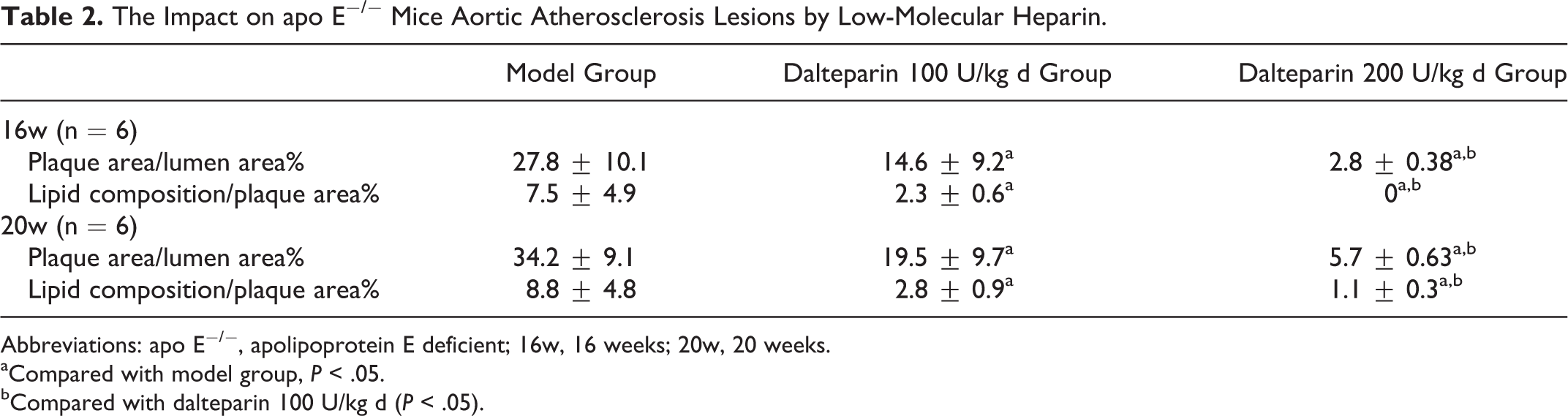
Abbreviations: apo E−/−, apolipoprotein E deficient; 16w, 16 weeks; 20w, 20 weeks.
aCompared with model group, P < .05.
bCompared with dalteparin 100 U/kg d (P < .05).
Effect of Dalteparin on Aortic LOX-1 mRNA Expression
Aortic LOX-1 mRNA expression was detected by RT-PCR, and the level was represented by relative coefficient. The relative coefficient = absorbance of LOX-1 strips/absorbance of GAPDH strips. The relative coefficient of the apo E−/− model group was 0.882 ± 0.135, which was significantly greater than that of the C57BL/6J normal diet group (relative coefficient 0.602 ± 0.079; P < .05). After dalteparin treatment, LOX-1 mRNA expression exhibited a downward trend, but the difference did not reach statistical significance (Figure 2A and B).
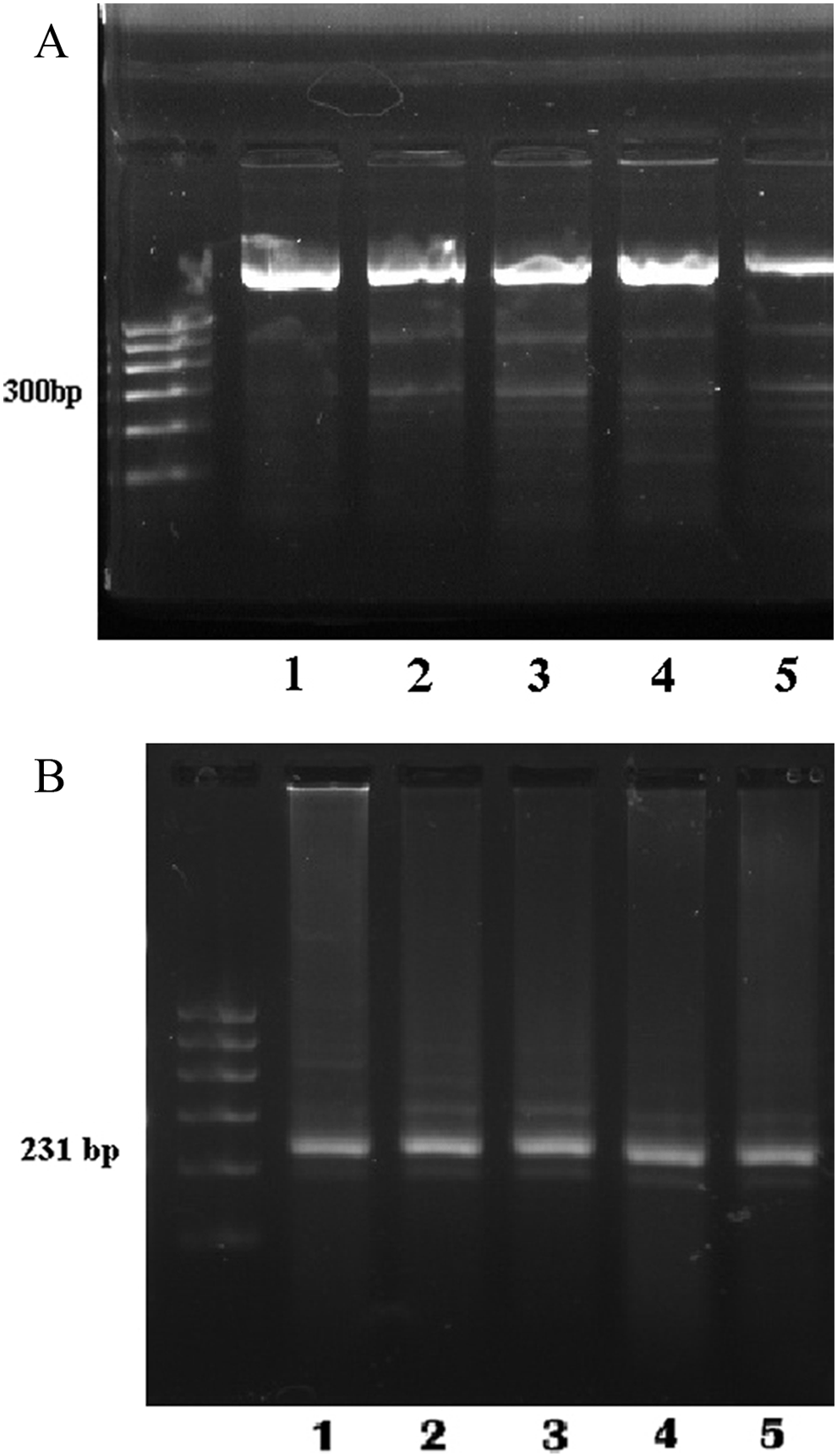
A, Oxidized low-density lipoprotein receptor 1 (LOX-1) mRNA expression in the mice aorta (RT-PCR). Strip 1: C57BL/6J mice of normal diet group. Strip 2: C57BL/6J mice of high-fat diet group. Strip 3: the model group of apo E−/− mice. Strip 4: dalteparin 100 U/kg d group of apo E−/− mice. Strip 5: dalteparin 200 U/kg d group of apo E−/− mice. B, The glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA expression in the mice aorta (RT-PCR). Strip 1: C57BL/6J mice of normal diet group. Strip 2: C57BL/6J mice of high-fat diet group. Strip3: the model group of apo E−/− mice. Strip 4: Dalteparin 100 U/kg d group of apo E−/− mice. Strip 5: dalteparin 200 U/kg d group of apo E−/− mice. apo E−/− indicates apolipoprotein E deficient; mRNA messenger RNA; RT-PCR, reverse transcription polymerase chain reaction.
Effect of Dalteparin on Aortic LOX-1 Expression
Immunohistochemistry of the aorta showed that brown positive granules were mainly found in the endothelial cells of aortic intima. All C57BL/6J mice showed no apparent LOX-1 expression. The model group of apo E−/− mice exhibited stronger LOX-1 expression than that found in the 2 C57BL/6J groups (P < .05). Compared with the apo E−/− model group, the expression of LOX-1 was decreased in the dalteparin 200 U/kg d group (P < .05), with only sparse and thin brown granules noted. There was no significant difference in LOX-1 expression between apo E−/− model group and dalteparin 100 U/kg d group (P > .05; Figure 3).
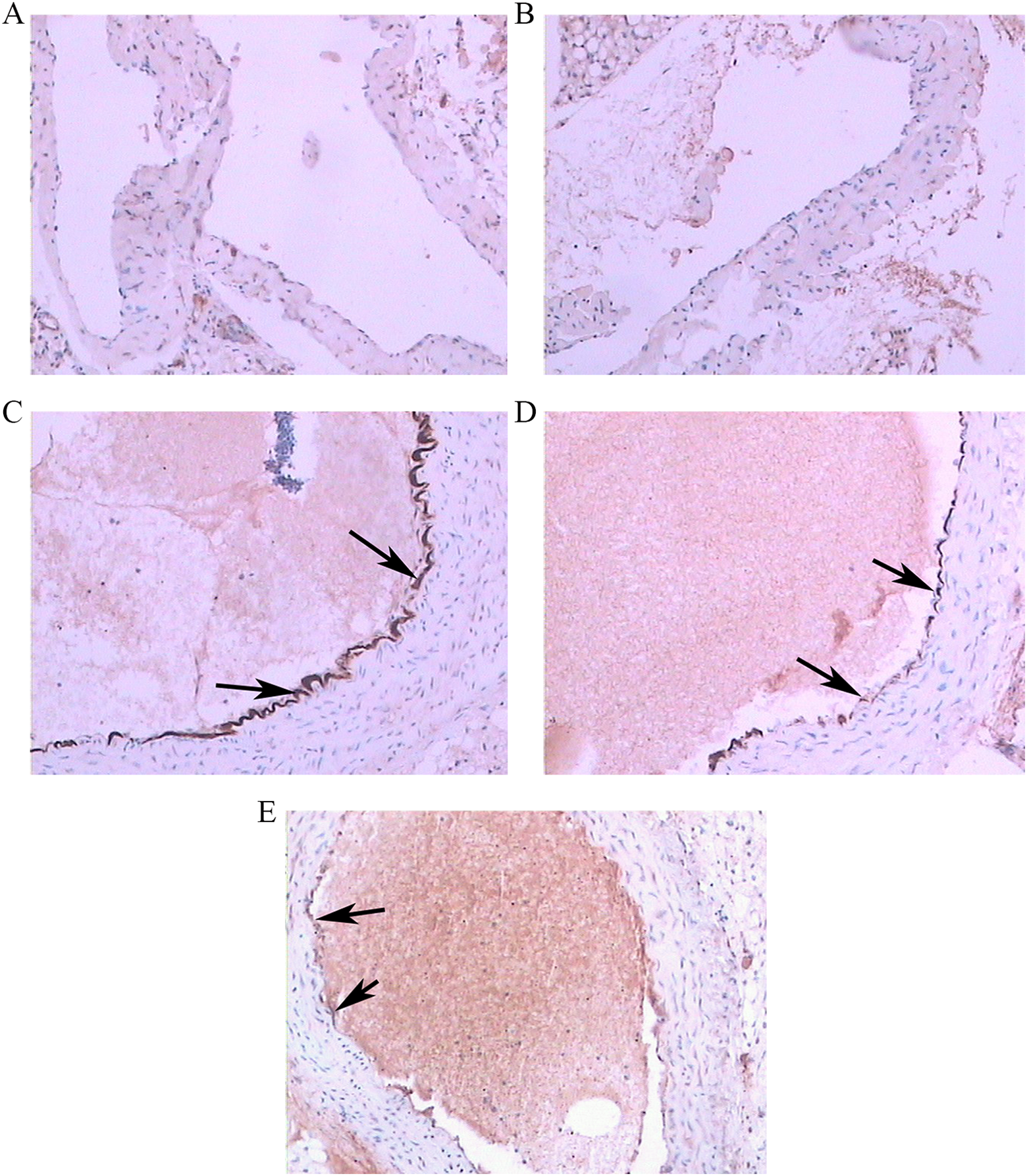
The expression of LOX-1 in the mice aorta root (immunohistochemical staining ×100). A, C57BL/6J mice of normal diet group showing no apparently expression of LOX-1. B, C57BL/6J mice of high-fat diet group showing no apparently expression of LOX-1. C, The model group of apo E−/− mice showing apparently positive expression of LOX-1 (arrow). D, Dalteparin 100 U/kg d group of apo E−/− mice moderately positive expression of LOX-1 (arrow). E, Dalteparin 200 U/kg d group of apo E−/− mice showing slightly positive expression of LOX-1 (arrow). apo E−/− indicates apolipoprotein E deficient; LOX-1, oxidized low-density lipoprotein receptor 1.
Western blotting showed that the LOX-1 protein strip was 36 kDa. Compared with the level of LOX-1 protein expression of the apo E−/− mice model group, the levels of LOX-1 protein expression in the dalteparin-treated groups was significantly decreased (P < .05), indicating that dalteparin could inhibit aortic LOX-1 protein expression in apo E−/− mice. There was no statistical difference in LOX-1 protein expression between the 2 dalteparin-treated groups (Figure 4; P > .05).

Oxidized low-density lipoprotein receptor 1 (LOX-1) protein expression in the mice aorta (Western blot). Strip 1: C57BL/6J mice of normal diet group. Strip 2 :C57BL/6J mice of high-fat diet group. Strip 3: the model group of apo E−/− mice. Strip 4: dalteparin 100 U/kg d group of apo E−/− mice. Strip 5: dalteparin 200 U/kg d group of apo E−/− mice. apo E−/− indicates apolipoprotein E deficient.
In summary, LOX-1 protein levels were downregulated by dalteparin treatment.
Discussion
The Ox-LDL appears to play key roles in atherosclerotic progression and atherosclerotic plaque rupture. 11 The LOX-1 is a type II membrane protein belonging to the C-type lectin family, which can act as a cell surface endocytosis receptor for atherogenic oxidized LDL. 4,12 In human atherosclerotic lesions, LOX-1 is expressed in macrophages and smooth muscle cells in advanced plaques as well as endothelial cells covering early lesions. 13 , 14 The LOX-1 activation triggers atherosclerotic plaque formation and progression and has also been shown to lead to plaque vulnerability and potential rupture. 15–17 Acute clinical manifestations of atherosclerosis, including MI, ischemic stroke, and sudden cardiac death, are primarily caused by rupture of advanced atherosclerotic plaques that finally lead to occlusion of the vessel lumen. 3,11 A decrease in LOX-1 expression could partially inhibit the concentration of cholesterol in the plaques and increase plaque stability, so it may be an effective way to reverse atherosclerotic lesions. 18
Thrombin generation is critical to the formation of arterial thrombi. Activated factor X participates in the conversion of a zymogen prothrombin into thrombin, the key enzyme of the coagulation cascade. 19 Factor Xa has been shown to stimulate the proliferation of rat aortic smooth muscle cells and human mesangial cells. 20 Thrombin and factor Xa may contribute to vascular lesion formation. 21
Elevated low-density lipoprotein cholesterol levels increase blood thrombogenicity. 22 In our preliminary study, LOX-1 was induced by thrombin and factor Xa in cultured vascular smooth muscle cells. Thrombin and factor Xa induction of the expression of LOX-1 depends upon epidermal growth factor receptor phosphorylation. The FRISC-II study showed that dalteparin lowers the risk of death, MI, and the need for revascularization during the first month. Initial benefits were not sustained during the longer term if the patients adopted a noninvasive treatment strategy. Reductions in the composite death rate or MI were limited to high-risk patients. Despite dalteparin treatment, the risk of bleeding associated with revascularization was low. 8 A study by Bea et al with elderly apo E−/− mice found that the thrombin inhibitor melagatran can not only promote plaque stability but can also reduce the size of mature plaques, which cannot be achieved by other statins. 23
The percentage of lipid core in a plaque can be used as an indicator of plaque vulnerability. 9,24 This study showed that dalteparin, a thrombin and factor Xa inhibitor, inhibited LOX-1 protein expression and decreased thrombus growth. The inhibition of mRNA expression was less obvious than that of aortic LOX-1 expression in this study. The possible reasons may include the relatively low sensitivity of RT-PCR or limitations of the experiment. Real-time PCR may provide more accurate determination of mRNA expression. The inhibition of LOX-1 protein expression may be a target for antiatherosclerosis therapy by low-molecular heparin, independent of its anticoagulation effects. Therefore, the results of this study can be seen as a promising direction toward a possible advantage of direct thrombin inhibitors of LOX-1 expression.
Among the different strains of apo E−/− mice, the C57BL/6J strain develops the most severe atherosclerosis. With normal feeding, the mice spontaneously develop hypercholesterolemia and atherosclerosis and exhibit a similar plaque distribution as humans. 25 High-cholesterol feeding may accelerate this process. In this study, there was a trend of hair loss and inactivity in the high-fat diet apo E−/− mice, which might indicate aging, although no statistical difference among the apo E−/− groups was found due to the limited number of mice. The most frequent location of plaque formation was the aortic root, followed by the thoracic aorta. Because female mice usually have less severe atherosclerosis than male mice, male mice were chosen in this study. Most animals are likely to have stronger tolerability and require a greater dosage of dalteparin per body unit than humans. The dosage of 200 U/kg d dalteparin in this study is similar to the dosage received by the human body in clinical practices. However, the study could not accurately reflect or represent the complexity of human plaques, so further investigation is required.
This study has some limitations. Further research including using female mice and examination of the possible molecular mechanism are anticipated. The apo E and LOX-1 double knockout mice should be used to validate the effect of dalteparin at the level of LOX-1. Ultrasound imaging of the vessels was not used to show the atherosclerotic plaques noninvasively and consecutively during the experiment. The effects of endogenous antithrombin and endogenous tissue factor pathway inhibitor on the levels of LOX-1 will be explored in our future studies.
Our data showed that dalteparin inhibited atherosclerotic formation by downregulation of the LOX-1 receptor and revealed a novel effect of dalteparin on LOX-1 receptors. The results provide a new theoretical basis for the mechanism of action of low-molecular heparin, paving a way for new therapeutic interventions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This trial was supported by Research and Development Fund of Peking University People's Hospital (RDB2007-19).
