Abstract
Background: Dalteparin and enoxaparin are recommended as thromboprophylaxis for at least 10 days in patients undergoing abdominal surgery (AS) or hospitalized patients with acute medical illnesses. Even though both agents have proven clinical effectiveness through randomized trials, there have been no head-to-head studies. In this evaluation, indirect statistical techniques were used to compare safety and efficacy between dalteparin and enoxaparin in these 2 high-risk patient populations. Methods: A literature search was conducted from January 1980 to November 2010 for randomized trials evaluating dalteparin or enoxaparin prophylaxis following AS or in hospitalized patients. Binary outcomes for safety and efficacy were statistically pooled using fixed or random effects models in cases of significant heterogeneity. In trials where a common control was used (eg, unfractionated heparin [UH]), indirect statistical comparisons between dalteparin and enoxaparin were performed using meta-regression analysis with active drug as the primary independent variable. Results: The meta-analysis in AS patients showed that enoxaparin or dalteparin had comparable efficacy to UH in terms of venous thromboembolic events (VTEs; relative risk reduction [RR] = 0.87, P = .46). The indirect statistical comparison was unable to find significant differences between enoxaparin and dalteparin in terms of risk for VTE (P = .84), major bleeding (P = .38), heparin-induced thrombocytopenia ([HIT]; P = .084), or death (P = .97). In acutely ill medical patients, treatment with enoxaparin or dalteparin had a 52% VTE risk reduction compared to placebo (RR = 0.48, P < .001). The indirect comparison was also unable to find significant differences between enoxaparin and dalteparin in terms of VTEs (P = .15), major bleeds (P = .39), HIT (P = .48), and death (P = .41). Conclusions: The findings suggest comparable safety and efficacy between dalteparin and enoxaparin in AS and in acutely ill medical patients.
Introduction
Randomized comparative trials of adequate statistical power remain the gold standard for establishing superiority in terms of safety and efficacy between the 2 drugs indicated for the same condition. However, it is not uncommon in medical decision making to find no direct head-to-head trials of competing agents. In contrast, the drugs of interest may have been evaluated in separate trials relative to a common control group such as placebo. In such situations, clinicians have to rely on indirect comparisons of individual arms of randomized trials which hopefully had used similar patient inclusion criteria. Such an approach is crude, but it does give an indication of differential safety and efficacy between treatments of interest. A preferred approach is an indirect statistical comparison of different treatments that performs an adjustment based on the results of their direct comparison on a common control group. 1 The advantage of such an approach is that the internal validity associated with randomization is maintained. 1,2
Given the challenges associated with conducting multiple large randomized trials, the importance of indirect statistical comparisons of alternative therapies has increased. The advantages of using these techniques to conduct comparative effectiveness evaluations are that they utilize the best available evidence to provide answers to questions that have not been addressed through a randomized trial. Examples in the literature where indirect statistical analyses have been successfully used to conduct comparative effectiveness research include renal cell carcinoma and pulmonary arterial hypertension. 3,4 As an extension, one area that would benefit from indirect statistical evaluations is in the prevention of venous thromboembolic events (VTEs) in high-risk patient populations.
Two such populations consist of patients undergoing abdominal surgery (AS) or those who have been hospitalized with acute medical illnesses. The American College of Chest Physicians (ACCP) guidelines recommend that a low-molecular-weight heparin (LMWH) is a reasonable option for thromboprophylaxis in these 2 high-risk groups. 5 There are several LMWHs available, but 2 of the most commonly used are enoxaparin and dalteparin. Despite being available for clinical use since the early 1990s, there are no large head-to-head randomized studies between dalteparin and enoxaparin in AS or in hospitalized patients with acute medical illnesses. As a result, the application of indirect statistical techniques to support a comparative safety and efficacy assessment between dalteparin and enoxaparin in these 2 high-risk populations is warranted. The results from such analysis can then provide the foundation for further comparative effectiveness research between the newer oral agents rivaroxaban and dabigatran. 6,7
Methods
Literature Review and Meta-Analysis of Randomized Trials
A computer literature search of PubMed, Embase, the Cochrane Database, and Google Scholar was conducted from January 1980 to November 2010 for randomized trials evaluating dalteparin and enoxaparin for the prevention of VTEs in patients undergoing AS and in hospitalized patients with acute medical illnesses. Search terms consisted of “{dalteparin or enoxaparin}, AND {prophylaxis} AND {abdominal surgery}, OR {medical patients}, AND {randomized clinical trial}.”
The inclusion criteria for trial acceptance consisted of the following: The trial must have utilized a parallel group design. Cross-over trials were excluded given the inherent contamination from latent treatment effects in their overall design. The trial population must have consisted of patients undergoing AS or hospitalized patients with acute medical illnesses. Must have enrolled patients greater than 18 years of age. The control arm in accepted trials must have been placebo, unfractionated heparin (UH), or warfarin. Accepted trials must have evaluated the clinically appropriate doses of dalteparin or enoxaparin in at least 1 of the trial arms. Trials must have been a randomized comparison with at least 25 patients enrolled into each arm.
Once the trials meeting the inclusion criteria were identified, the following data were extracted: baseline patient information, indication, sample size in each group, drug and dosage, duration of therapy, study duration, definition of primary and secondary end points, and primary and secondary results. Other data extracted from the accepted trials consisted of the number of deep venous thromboses ([DVTs] both venographically detected and symptomatic) and pulmonary embolism (PE) in each group. Safety data consisted of bleeding events (minor and major), heparin-induced thrombocytopenia (HIT), number of withdrawals caused by adverse drug reaction, and all-cause mortality in each of the study groups.
All of the study outcomes were presented as binary end points (eg, VTE rates, major bleeding events) and were combined using a fixed or random effects model in cases of significant heterogeneity. 8 Random effects meta-analysis assumes that the effect of the intervention varies across studies. When significant between-study variation is present, the 95% confidence interval (CI) for the summary measure tends to be larger with a random effects model. Treatment effects from individual trials were also presented as Forest Plots.
Statistical heterogeneity between studies was assessed by both the Q statistic and the I 2 test statistic. 9 Briefly, the I 2 statistic measures the proportion of variance across studies due to heterogeneity. It is considered to be a superior measure of study heterogeneity than the Q statistic because the latter is often underpowered when evaluating homogeneity in meta-analyses. The P values associated with the Q statistic (chi-square with k − 1 degrees of freedom, where k is the number of studies) were also reported. In situations where the Q statistic was statistically significant or the I 2 statistic was greater than 25%, a random effects meta-analysis model was used as described earlier. Publication bias was assessed through an evaluation of funnel plots and by the method proposed by Egger, which provides a significant P value when the publication bias may be present. 10,11
Indirect Statistical Comparisons Between Drugs
For each patient population, an indirect statistical assessment between dalteparin and enoxaparin was performed using 2 approaches. The first indirect method was a meta-regression analysis on the primary clinical outcomes and on adverse event rates reported in the trials. A meta-regression analysis is an appropriate method for conducting an indirect comparison in cases where trials evaluating the drugs of interest used a common comparator. 4,12 Therefore, indirect comparisons between dalteparin and enoxaparin were performed with those trials that had the same control group. Separate analyses were performed for each patient population. Unfractionated heparin was the common control in the case of AS. In hospitalized patients with acute medical illnesses, placebo was the common control that allowed for the indirect comparison.
Active drug (dalteparin and enoxaparin) was the independent variable in the regression model.
12
Other independent variables considered in each model included duration of therapy, treatment schedule (pre- vs post-surgery initiation), geographic region where the study was conducted (ie, North America vs Europe vs global), and year of publication. Therefore, a typical meta-regression model would be as follows: Meta-regression model: log[VTE_Relative_Risk] = drug + dosing + duration + treatment schedule + geographic region+ year of publication. All of these analyses were performed using Stata V 9.0 (Stata Corp, College Station, Texas). Indirect treatment comparisons were also performed using the method of Bucher and colleagues, which partly maintains the benefits of randomization on the effect size.
1,13
Briefly, this is a simple method for an adjusted analysis, in which the indirect comparison of A and B is adjusted according to the results of the direct comparisons with a common intervention—C. Let lnORAC
denote log odds ratio of A
1 versus C
1 in trial 1, and lnORBC
denote log odds ratio of B
2 versus C
2 in trial 2. The log odds ratio of the adjusted indirect comparison of A and B (lnOR’AB
) can then be estimated by: lnOR’AB = lnORAC − lnORBC
The standard error would be SE(lnOR’AB) = √ [SE(lnORAC)2 + SE(lnORBC)2].
Although an odds ratio is used in the above equations, this adjusted method may also be used when the relative efficacy is measured as a relative risk (RR), risk difference, or mean difference. Empirical evidence indicates that results of adjusted indirect comparison are usually, but not always, similar to those of direct head-to-head trials.
2
It is important to make a distinction between the 2 methods for indirectly comparing dalteparin to enoxaparin. In the meta-regression approach, the evaluation is between a LWMH (dalteparin and enoxaparin) versus a common control (ie, placebo or UH) and the effect measure is expressed as a RR difference between drugs. In contrast, the method of Bucher et al uses a common comparator (eg, placebo) to statistically link the 2 treatments. As a result, the generated outcome is an effect measure (RR in this case) comparing enoxaparin to dalteparin with an associated P value for statistical significance. 13
Results
From the 2 patient populations, 249 citations were reviewed for eligibility. A total of 14 randomized trials meeting the inclusion criteria were appropriate for the statistical pooling exercise. By indication and drug, the trials were broken down as follows: AS—dalteparin = 7, enoxaparin = 3; and acute medically ill patients—dalteparin = 1 and enoxaparin = 3. The common comparator to the LMWHs was UH in the AS trials and placebo in studies that evaluated acute medically ill patients. There were no studies where warfarin was a common control for both drugs. Detailed information on each of the trials in presented in Tables 1 and 2 .
Randomized Trials Comparing Dalteparin or Enoxaparin in Abdominal Surgery Patients
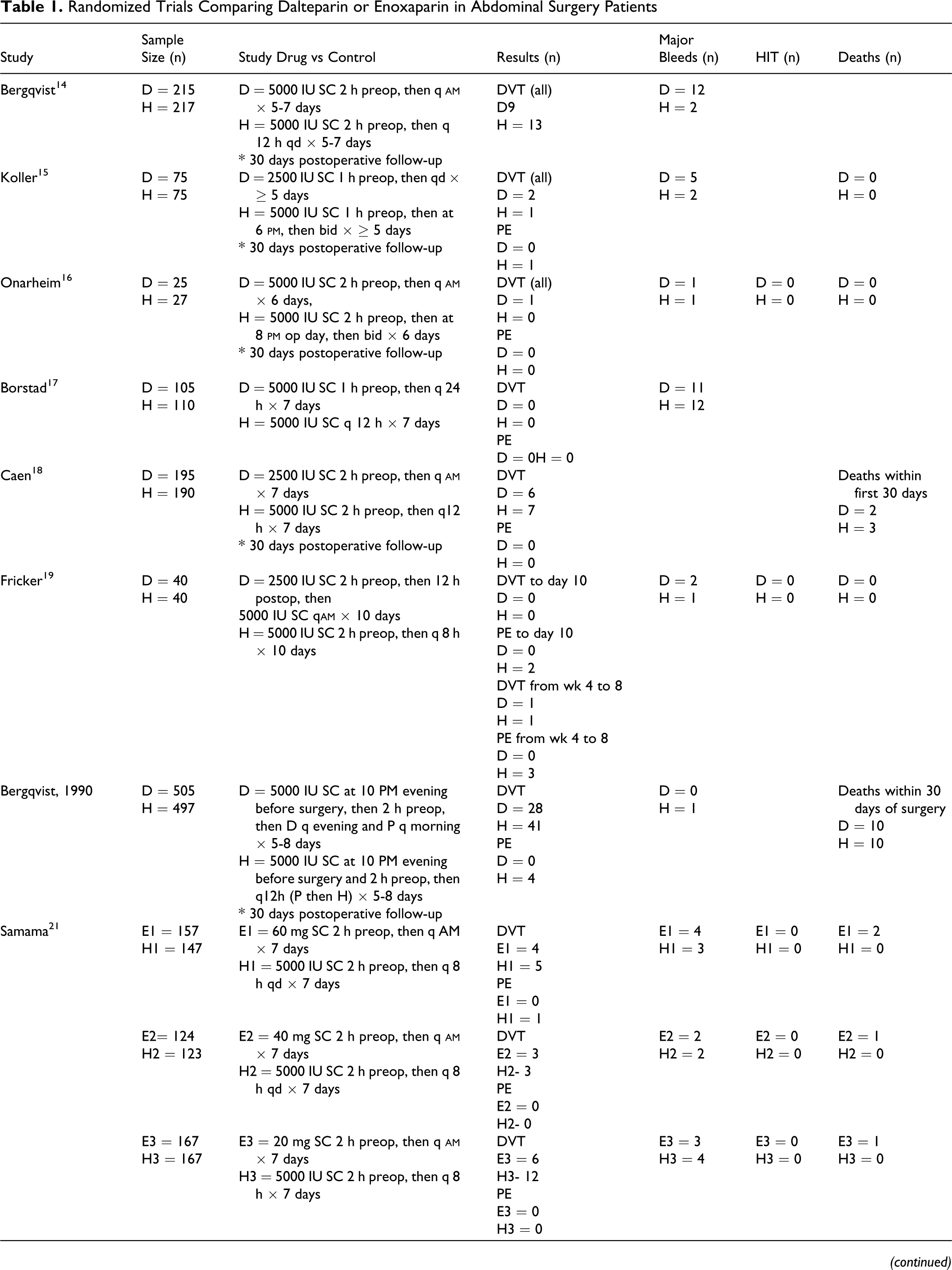
Abbreviations: D, dalteparin, E, enoxaparin, P, placebo, H, unfractionated heparin, SC, subcutaneously, IV, intravenously, THR, total hip replacement, HIT, heparin-induced thrombocytopenia, VTE, Venous thromboembolism, DVT, deep vein thrombosis, PE, pulmonary embolism; SC, subcutaneously; bid, twice daily, q, every; tid, thrice daily; op, operative.
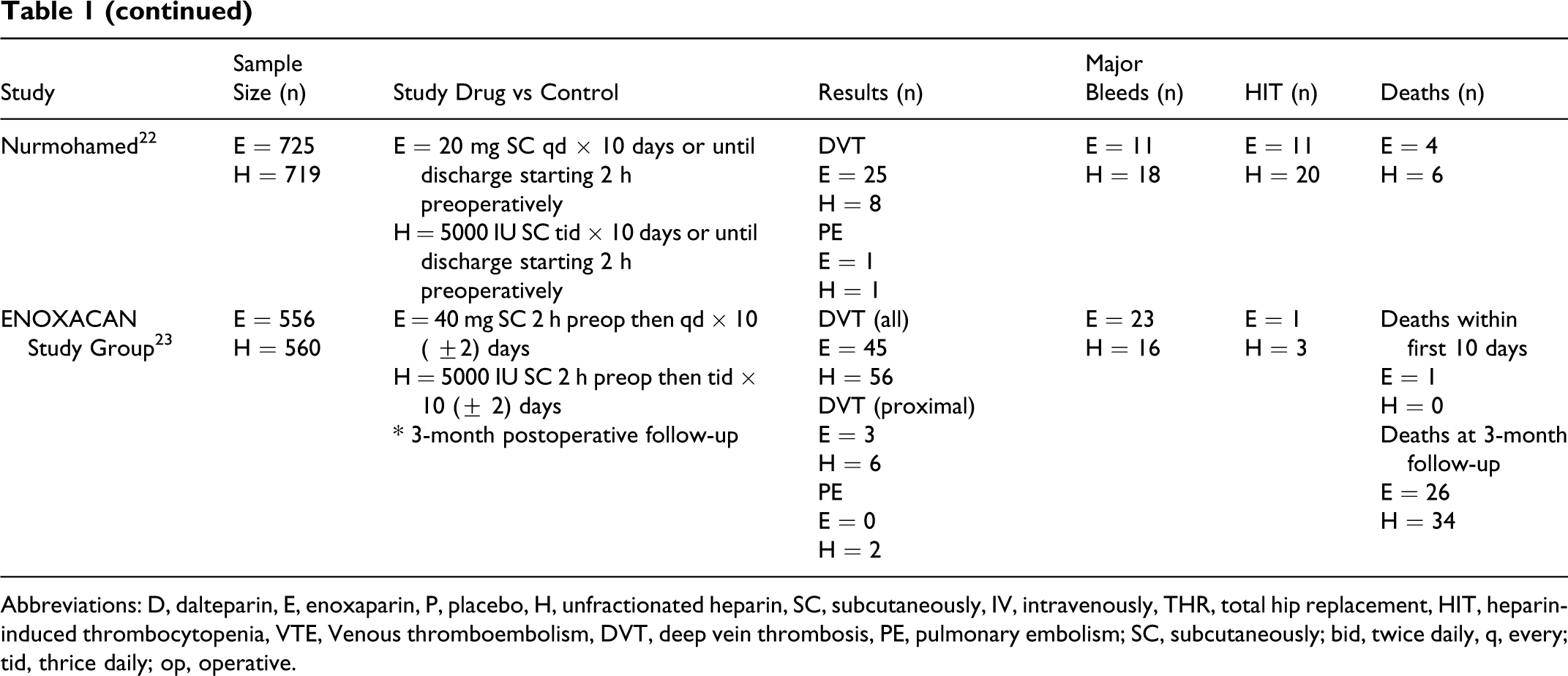
Randomized Trials Comparing Dalteparin or Enoxaparin in Hospitalized Patients With Acute Medical Illnesses

Abbreviations: D, dalteparin, E, enoxaparin, P, placebo, H, unfractionated heparin, SC, subcutaneously, IV, intravenously, THR, total hip replacement, HIT, heparin-induced thrombocytopenia, VTE, venous thromboembolic events, DVT, deep vein thrombosis, PE, pulmonary embolism; q, every.
Venous Thromboembolic Event Prophylaxis Following AS
A total of 10 randomized trials met the study inclusion criteria, providing a total of 11 treatment arms that were suitable for meta-regression analysis. There were 3 enoxaparin and 7 dalteparin studies using UH as the common control. All of the trials started active drug therapy a few hours before surgery and the duration of treatment did not exceed 10 (±2) days (Table 1). The initial meta-analysis suggested that regardless of the drug (enoxaparin or dalteparin), there was no significant difference in the RR of VTEs between the LMWHs and UH (RR = 0.87; [0.62-1.25], P = .46; Figure 1 ). Similar results were also observed in terms of safety. There were no significant differences between the LMWHs and UH with respect to major bleeds (RR = 1.17, P = .38; Figure 2 ), HIT (RR = 0.57, P = .084) and post surgical death (RR = 0.99, P = .97; Table 3 ). An indirect statistical comparison between enoxaparin and dalteparin in this patient population was then conducted using meta-regression analysis. There was no significant difference in VTE risk between drugs (P = .46). In addition, geographic region where the trial was conducted (global vs North America) and year of publication were not significantly associated with the pooled RR. Similarly, the analysis also indicated that the 2 drugs were also comparable in terms of risk for major bleeds, HIT, and post surgical death (Table 3).
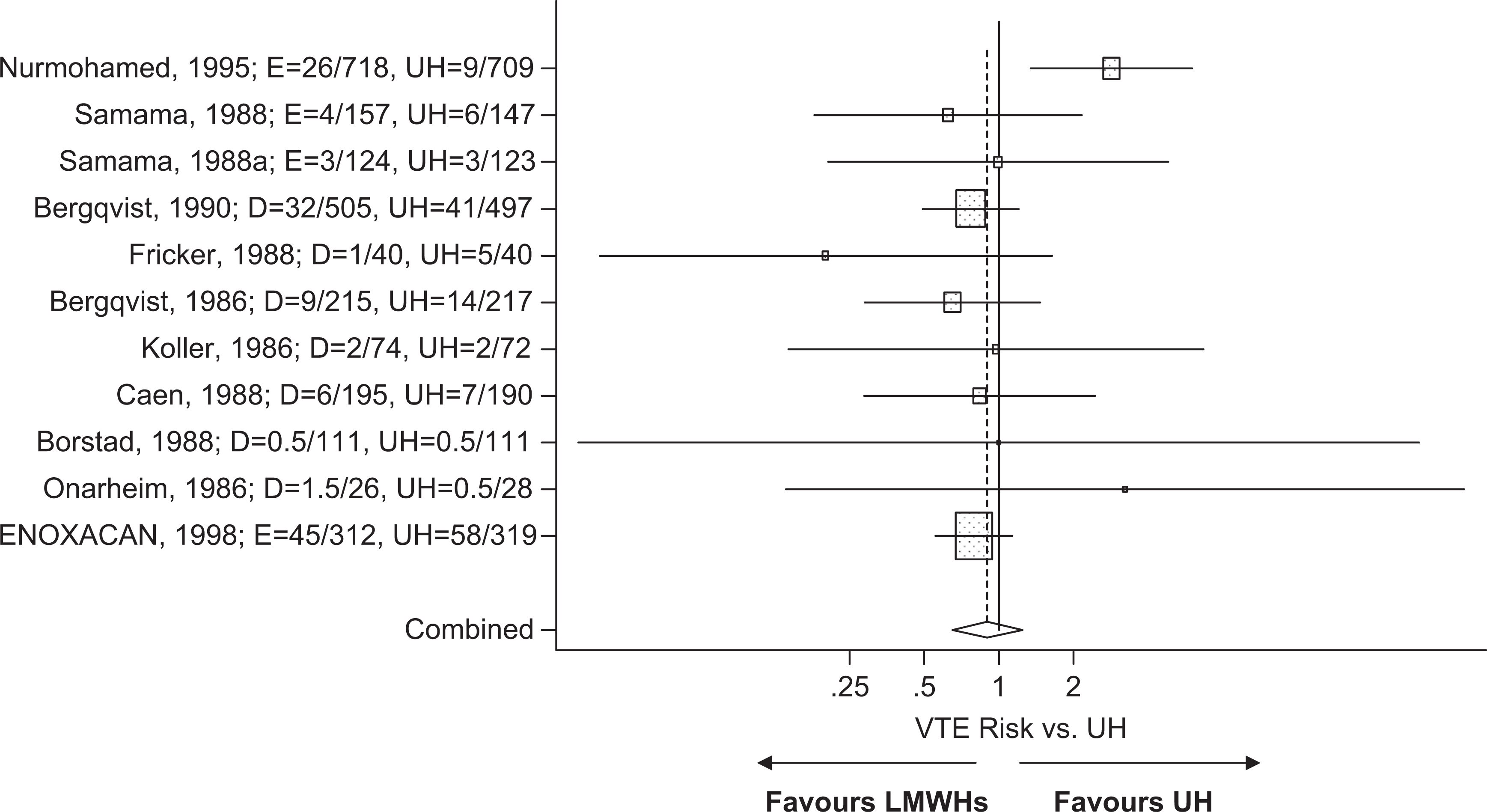
Meta-analysis on the relative risk for the prevention of VTEs in unfractionated heparin–controlled trials evaluating enoxaparin or dalteparin in abdominal surgery. D indicates dalteparin; E, enoxaparin; UH, unfractionated heparin.
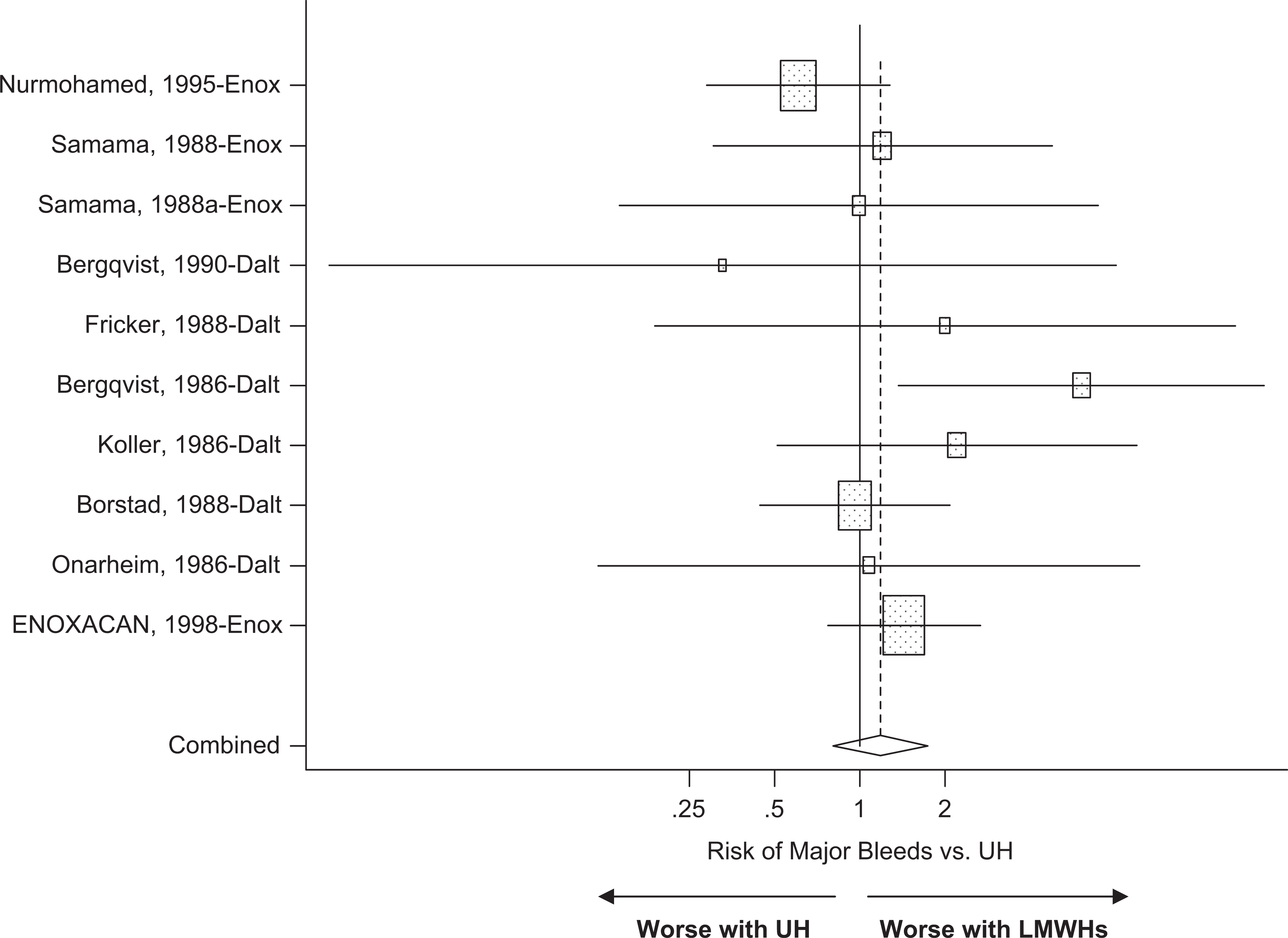
Meta-analysis on the relative risk for major bleeds in unfractionated heparin–controlled trials evaluating enoxaparin or dalteparin in abdominal surgery. D indicates dalteparin; E, enoxaparin; UH, unfractionated heparin.
Summary of Meta-Regression Analysis on the Risk of VTE and Adverse Events in Abdominal Surgery and in Hospitalized Patients With Acute Medical Illnesses
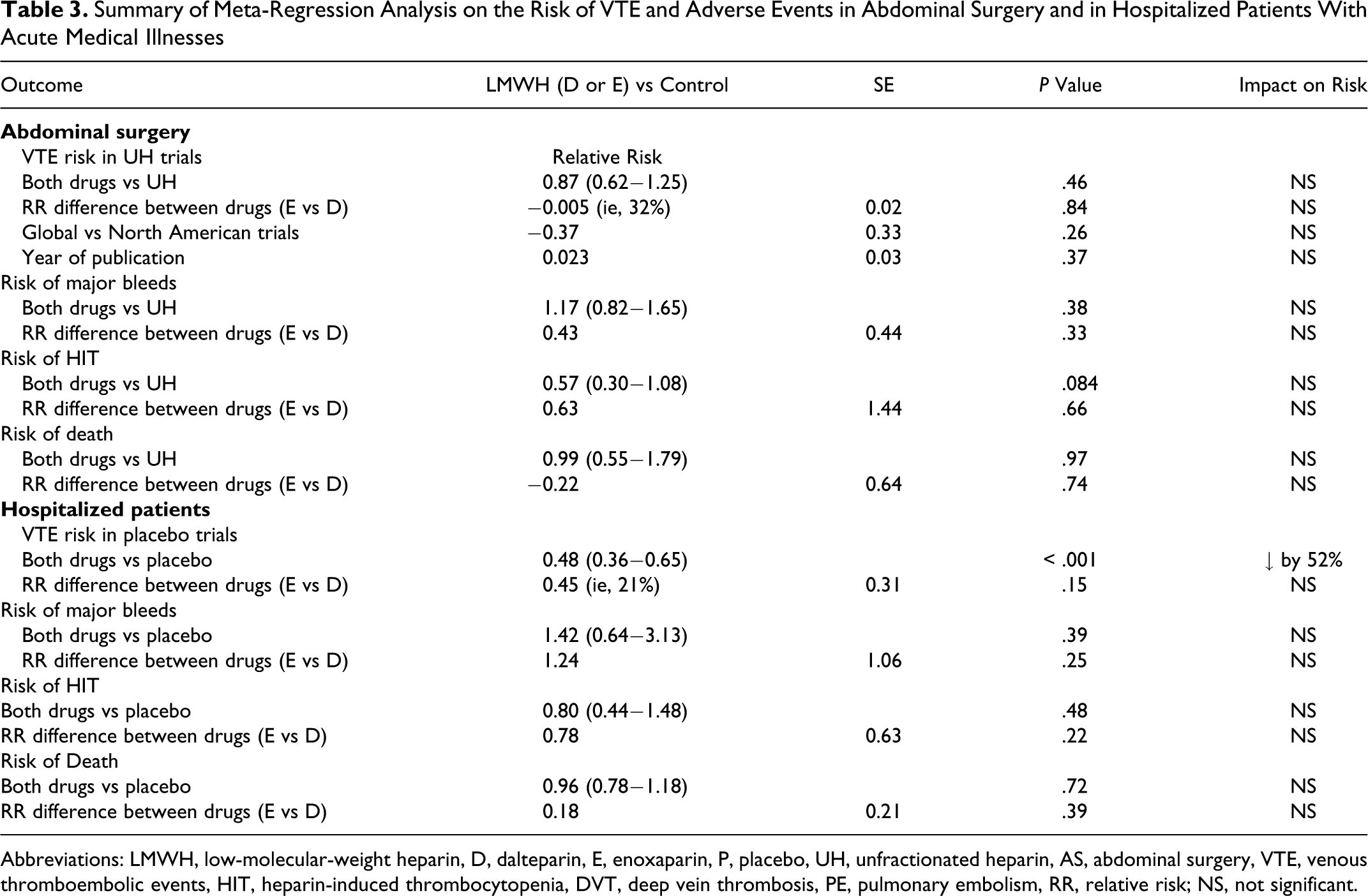
Abbreviations: LMWH, low-molecular-weight heparin, D, dalteparin, E, enoxaparin, P, placebo, UH, unfractionated heparin, AS, abdominal surgery, VTE, venous thromboembolic events, HIT, heparin-induced thrombocytopenia, DVT, deep vein thrombosis, PE, pulmonary embolism, RR, relative risk; NS, not significant.
Hospitalized Patients With Acute Medical Illnesses
There were a total of 3 enoxaparin trials and a single study with dalteparin which met the inclusion criteria in this patient population. All of the trials used a placebo control and the duration of therapy was up to 14 days or until hospital discharge for 3 studies and up to 90 days or until discharge in 1 trial (Table 2). The trial by Lederle et al could not be included in the VTE analysis because it only provided symptomatic DVT rates. 27 The meta-regression analysis of all trial data suggested that hospitalized patients treated with an LMWH had a 52% VTE RR reduction compared to the placebo control (RR = 0.48, P < .001; Figure 3 ). This benefit was achieved without a significant increase in the risk for major bleeds (RR = 1.42, P = .39), HIT (RR = 0.80, P = .48), or death (RR = 0.96, P = .72).
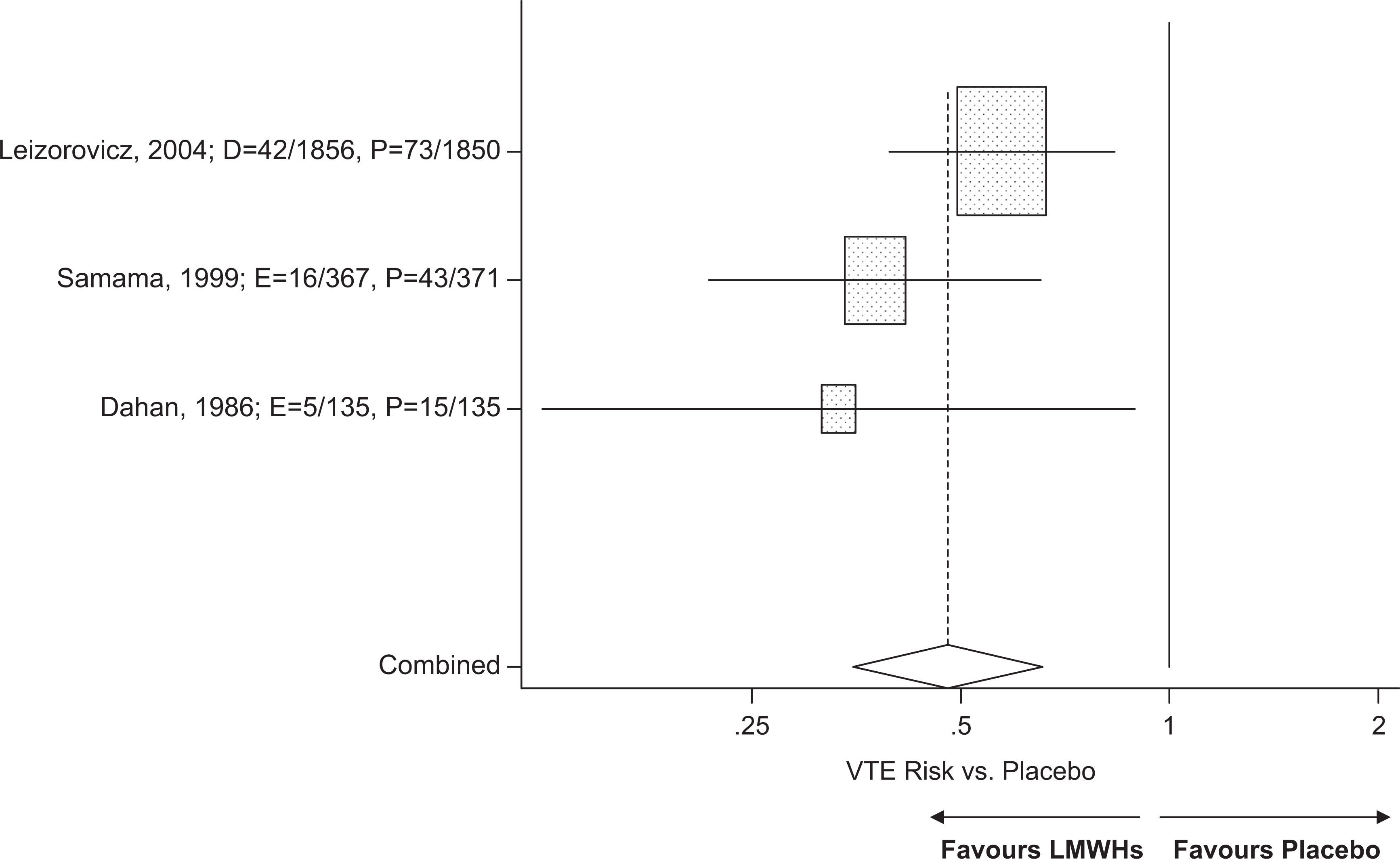
Meta analysis on the relative risk for the prevention of VTEs in placebo-controlled trials evaluating enoxaparin or dalteparin in hospitalized patients.
In the second part of the evaluation, the indirect statistical comparison between enoxaparin and dalteparin was not able to find significant differences between drugs in terms of VTE risk (Table 3). In addition, the safety assessment between drugs also indicated comparable risks for major bleeds, HIT, and death (Table 3). Consistent with the findings in the previous patient population, the results implied that dalteparin was comparable to enoxaparin in terms of efficacy and adverse events when used in hospitalized patients with acute medical illnesses.
Indirect Comparison of Safety and Efficacy Using the Method of Bucher (1997)
The indirect method developed by Bucher and colleagues, one of the most cited approaches for performing indirect comparisons of randomized trials, 13 was applied for the indirect assessment of enoxaparin versus dalteparin with respect to VTE prevention and adverse events such as major bleeds, HIT, and death in each of the 2 patient populations. As was previously revealed by the meta-regression analysis, there was no evidence to suggest that one agent was superior to another with respect to VTE prevention following AS or in hospitalized patients with acute medical illnesses (Table 4 ). Similarly, there was no significant difference in the risk of major bleeds, HIT, and death between drugs as indicated by the 95% CIs for the RR which crossed the 1.0 threshold. Hence, these data imply that enoxaparin and dalteparin have comparable safety and efficacy when used to prevent VTEs in these high-risk patient groups.
Summary of Indirect Statistical Comparisons Between Dalteparin and Enoxaparin Using the Method of Bucher et al 13
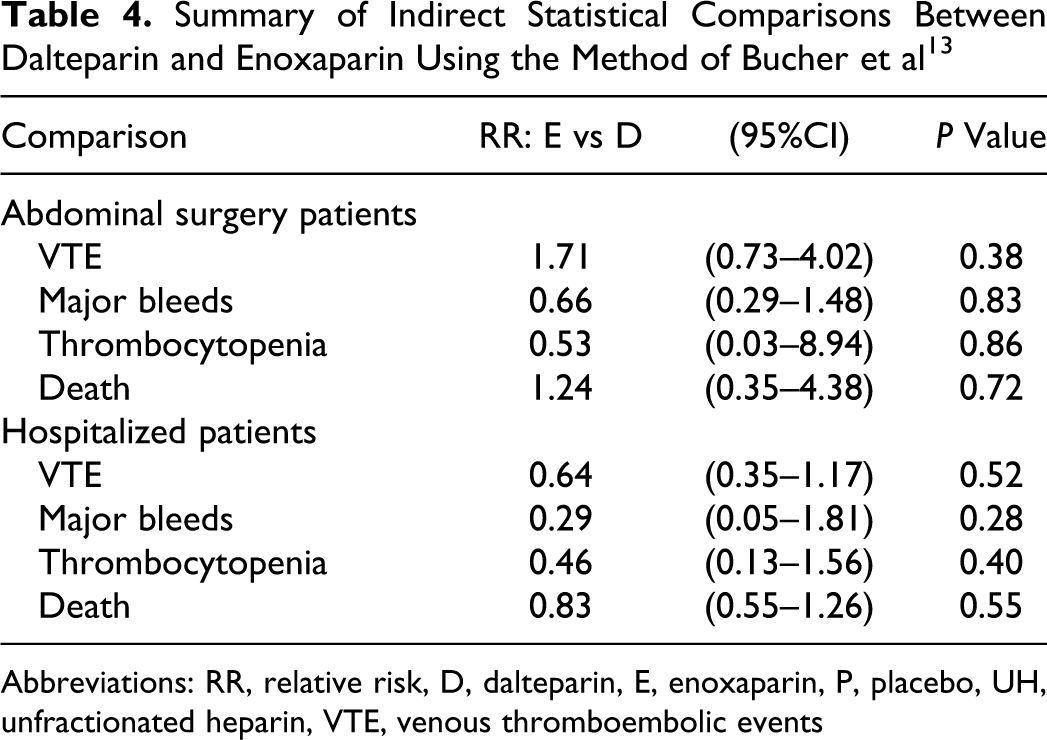
Abbreviations: RR, relative risk, D, dalteparin, E, enoxaparin, P, placebo, UH, unfractionated heparin, VTE, venous thromboembolic events
Testing of Publication Bias
The potential for publication bias was assessed. There was no suggestion of publication bias in trials evaluating AS (Egger test, P = .62) and hospitalized patients with acute medical illnesses (Egger test, P = .35).
Discussion
Patients undergoing AS and those who have been hospitalized with acute medical illnesses represent 2 groups who are at higher than average risk for the development of VTEs. 5 Therefore, the ACCP guidelines state that prophylaxis with an LMWH such as enoxaparin or dalteparin is an acceptable option. 5 However, it is currently unknown whether one agent can be used in place of another without a loss in efficacy or safety. Furthermore, a systematic review of the literature was also unable to conclude therapeutic equivalence due to insufficient comparative data. 28 In such situations, indirect statistical comparisons are a reasonable approach for evaluating 2 similar agents. 2
The current study applied a meta-analysis of randomized trials and indirect statistical techniques to compare dalteparin and enoxaparin in terms of safety and efficacy in the 2 high-risk patient groups. The meta-analysis suggested that enoxaparin or dalteparin had comparable efficacy to UH in terms of VTE RR reduction in patients who had undergone AS. In hospitalized patients with acute medical illnesses, thromboprophylaxis with either dalteparin or enoxaparin was superior to placebo in VTE risk reduction and were not associated with any statistically significant increases in major bleeds or HIT. When the data was used to perform an indirect comparison between dalteparin and enoxaparin in each patient population, the findings were unable to find significant differences in VTE relative risk reduction, major bleeds, HIT or death. Therefore, treatment decisions should be based on other considerations, such as patient or physician preference, ease of administration, and/or cost.
The application of indirect statistical techniques in the current study is an acceptable approach for comparative effectiveness research. However, there are several limitations that need to be acknowledged. All meta-analyses are affected by the quality of the studies analyzed. For that reason, we limited our review to prospective randomized trials with sufficient sample size. Regardless of the data source, our analysis was indirect and does not replace a well-designed noninferiority trial comparing the 2 drugs. Therefore, we must be aware of the potential biases associated with indirect comparisons. There were only 3 and 10 randomized trials suitable for meta-analysis in our 2 patient populations. A larger sample size would have been preferable. As a result, this may have limited our statistical power, so the risk of a type II error (ie, false negative) must be recognized. The current analysis was not a noninferiority study because a prespecified “minimally clinically important difference” in efficacy between the 2 drugs has not been established by regulatory authorities or the academic community. Some of the trials provided multiple treatment arms for statistical analysis via meta-regression. This may violate the independent assumption of regression modeling. Lastly, the failure to find statistically significant differences in the clinical end points between the 2 drugs through indirect methods does not confirm comparable efficacy. The only way to definitively answer this question is through a noninferiority trial. However, it is a reasonable alternative in the absence of such a trial.
Conclusions
The findings of this meta-analysis of prospective randomized trials suggest that dalteparin and enoxaparin are effective for VTE prophylaxis following AS or in hospitalized patients with acute medical illnesses. The indirect statistical evaluation implied comparable safety and efficacy between dalteparin and enoxaparin. Therefore, treatment decision making should be based on other factors such as patient preferences, ease of administration, and cost considerations.
Footnotes
Yoonhee Choe was an employee of the study sponsor at the time of this analysis.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Eisai Inc.
