Abstract
Postthrombotic syndrome (PTS) is a common complication of deep vein thrombosis. This study aims to assess the role of recurrent venous thrombosis and inherited thrombophilia in the pathogenesis of PTS. A series of 206 patients diagnosed with lower extremity venous thrombosis were retrospectively reviewed. The PTS was observed in 30.58% of the patients. Recurrent venous thrombosis was identified in 3.4% of the patients without PTS and in 33.3% of patients with PTS (P < .001). Inherited thrombophilia alone or in association with recurrent venous thrombosis was more commonly detected when PTS was moderate to severe (P = .04 and <.001) or severe (P < .001). Recurrent venous thrombosis increases the incidence of PTS significantly. The severity of PTS raises when an underlying thrombophilia is present either alone or in association with recurrent venous thrombosis.
Introduction
Post-thrombotic syndrome (PTS) is a costly and morbid long-term complication of deep vein thrombosis (DVT). Although less dramatic than pulmonary embolism, postthrombotic syndrome is responsible for the greater degree of chronic socio-economic morbidity. After an episode of acute DVT, long-term manifestations of PTS have been reported in 29% to 79% of the patients, 1 and severe forms including skin ulcers have been identified in 4% to 23% of the patients. 2 In the United States, skin changes and ulceration are present in 6 to 7 million and 400 000 to 5 00 000 people, respectively. 3 Estimates of the overall cost of chronic venous insufficiency in the United States reach 3 billion US dollars annually. 4
Pathophysiology of PTS is complex and not entirely understood. The PTS is seldom studied in long-term clinical trials, mainly because its evaluation requires very long-term follow-up. Treatment modalities of PTS are limited, and their results are often disappointing. Recognition of risk factors involved in the development of PTS is essential for the prevention and management of this syndrome. The purpose of this study is to evaluate the contribution of venous thrombosis recurrence and inherited thrombophilia to the constitution of PTS.
Materials and Methods
From January 2002 to January 2012, 206 consecutive patients (89 males and 117 females, diagnosed in an academic tertiary care center with lower extremity DVT by color flow duplex scan examination [Prosound alpha 7 ALOKA, Zug, Switzerland]) were retrospectively reviewed. Age varied between 25 and 96 years (mean: 60.6 years). Patients were followed for a period ranging from 3 to 120 months (mean: 16 months). A clinical research form was filled by every patient by a retrospective evaluation of the incidence and the severity of PTS; data were entered and analyzed by Statistical Package for the Social Sciences statistics (IBM Corporation, Somers, New York) software, version 13.0. A chi-square test was used to correlate between dichotomous variables, and a Fischer exact test was used in case of calculated values that were lower than 5.
The severity of PTS was evaluated according to clinical, etiological, anatomical, and pathophysiological classification recommended by the International Consensus Committee on chronic venous disease 5 and revised by the American Venous Forum. 6 Patients without visible or palpable signs of venous disease (C0), patients with telangiectasia or reticular veins (C1), with varicose veins (C2), with moderate edema (C3), with or without moderate symptoms such as aching, pain, tightness, skin irritation, heaviness, muscle cramps, and patients with moderate venous reflux or obstruction involving localized venous segments, identified on duplex scan, were considered having moderate PTS. Patients with severe edema >3 cm (C3), skin pigmentation and/or eczema (C4a), lipodermatosclerosis and/or atrophie blanche (C4b), healed venous ulcer (C5), active venous ulcer (C6) with severe clinical symptoms, and patients with severe venous obstruction and/or severe venous reflux involving extensive and multiple venous sites, essentially iliac, popliteal, and posterior tibial veins were classified as having severe forms of PTS. The presence of recurrent venous thrombosis and inherited thrombophilia was determined among patients with and without PTS. Patients were considered carriers for inherited thrombophilia when one or more of these tests was positive, antithrombin III deficiency, protein C deficiency, protein S deficiency, prothrombin G 20210 A mutation, factor V Leiden mutation, factor V H 1299 R R2 mutation, and methylenetetrahydrofolate reductase (MTHFR) C 677T or A 1298 C mutation leading to hyperhomocysteinemia.
Results
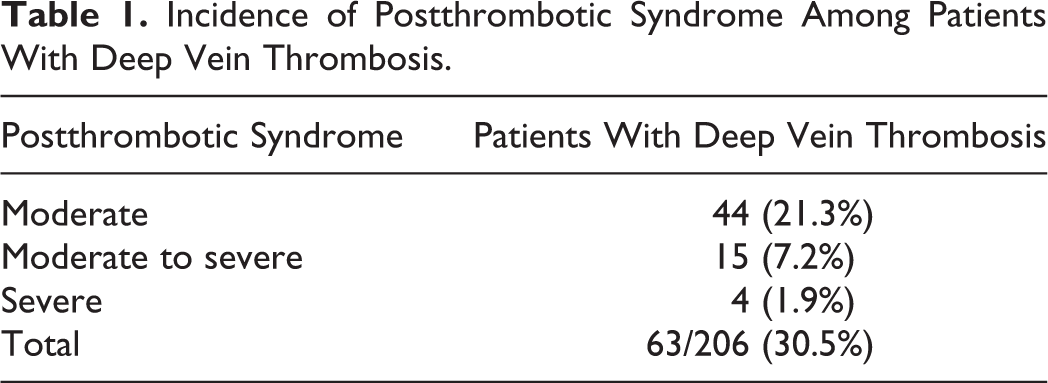
Postthrombotic syndrome occurred in 63 (30.58%) patients. It was moderate in 44 (21.35%) patients, moderate to severe in 15 (7.28%) patients, and severe in 4 (1.94%) patients (Table 1). Recurrent venous thrombosis was reported in 21 (33.33%) of the 63 patients with PTS and in only 5 (3.4%) of the 143 patients without PTS (P < .001). Recurrent venous thrombosis was more commonly observed in the group of patients with PTS, compared to the group of patients without PTS (P < .001; Table 2). A statistical difference in the incidence of inherited thrombophilia between the 2 groups did not reach significant levels (P = .08). However, the presence of thrombophilia increased significantly when PTS was moderate to severe (P = .04) or severe (P < .001; Table 3). The association of recurrent venous thrombosis and thrombophilia was more frequently recorded among patients with PTS (P = .05), essentially when PTS was moderate to severe or severe (P < .001; Table 4).
Incidence of Postthrombotic Syndrome Among Patients With Deep Vein Thrombosis.
Incidence of Recurrent Venous Thrombosis Among Patients With and Without PostThrombotic Syndrome.

Boldface values indicate statistically significant values.
Incidence of Inherited Thrombophilia Among Patients With and Without Postthrombotic Syndrome.

Boldface values indicate statistically significant values.
Incidence of Recurrent Venous Thrombosis and Inherited Thrombophilia Among Patients With and Without Postthrombotic Syndrome.

Boldface values indicate statistically significant values.
Discussion
Pathophysiology of PTS remains incompletely understood. This syndrome is more commonly observed in male gender, obese, and elderly patients. 7 The influence of several risk factors on the incidence of PTS is still controversial. 8 The determinants of postthrombotic manifestations include the rate of recanalization, the global extend of reflux, the anatomic distribution of reflux and obstruction, the recurrent thrombotic event, and thrombophilia.
Recurrent thrombotic events compete with recanalization early after acute DVT. Failure of recanalization is now recognized as an independent predictor of recurrent DVT. Recurrent thrombotic events are common and have a detrimental effect on valvular competence and on the development of PTS. 9 The risk of recurrent venous thromboembolism (VTE) is 24% at 5 years and 30% at 8 years after initial presentation. 10 –12 It is highly related to the underlying thrombotic risk factors. Assessment of clinical and genetic risk factors for venous thrombosis may provide useful prognostic information for recurrence. The recurrence rate is low in conjunction with a major reversible risk factor (3% in the first year and 10% after 5 years), intermediate in minor reversible risk factor (5% in the first year and 15% after 5 years) and relatively high in idiopathic VTE and elderly patients (10% in the first year and 30% after 5 years). 13 –15 Of the idiopathic DVT,50% to 70% are due to inherited thrombophilia. 16 The risk of recurrence is higher among patients with permanent risk factors including inherited prothrombotic abnormalities than among patients who have trauma or underwent surgery. 17 Watzke suggests a 2-year recurrence rate of 12% in patients with idiopathic DVT or irreversible risk factors and 4.8% in patients with reversible risk factors. 18 Other authors have similarly demonstrated that patients with idiopathic DVT or thrombophilia have 3-fold increased risk of recurrent VTE than those with secondary thrombosis. 10,11 Ongoing hypercoagulopathy is an essential predictor of recurrent VTE risk. 19 Factor V Leiden, prothrombin G 20210 A, and MTHFR mutations leading to hyperhomocysteinemia are the most commonly observed prothrombotic genetic abnormalities associated with venous recurrence. 20,21 Ipsilateral recurrence has been strongly associated with PTS. 15,21,22 Rethrombosis of a partially occluded or recanalized segment further increases the risk of reflux. 16 Reflux has been noted to develop in 36% to 73% of the segments with rethrombosis, a very higher rate compared to segments without rethrombosis. 23 Consistent with these observations, recurrent thrombotic events have been observed in 45% of the patients with PTS in comparison to only 17% of asymptomatic patients. 24 Prandoni et al reported a 6 times greater risk of PTS among patients with recurrent thrombosis. 2
Thrombophilia is increasingly recognized as a risk factor for DVT, particularly among young adults and in countries with high prevalence of prothrombotic genetic mutations. Venous thrombosis is in turn considered a major risk factor for chronic venous insufficiency. However, available information concerning the relation between thrombophilia and PTS is limited and still debatable. Some authors failed to demonstrate any correlation between thrombophilia and PTS. 1,25,26 On the other hand, others reported a high incidence of protein S deficiency and particularly of factor V Leiden mutation among patients with PTS leg ulcers and recommended to screen for this genetic mutation in these patients. 27 –31 Zutt et al observed elevated concentrations of lipoprotein(a) in patients with chronic venous insufficiency and PTS and suggested that lipoprotein(a) may contribute to the pathogenesis of PTS through induction of thrombogenetic microcirculatory dysregulations, impaired extravascular fibrinolysis, or other mechanisms like proinflammatory effects. 29 The PTS is commonly reported among patients with proximal DVT. Thrombophilia, detected among 29% of the patients with proximal DVT, is considered an independent predictor of persistent residual venous thrombosis that has been recognized as an important risk factor for recurrent venous thrombosis and PTS. 31,32 These findings highly suggest that inherited thrombophilia contributes to the constitution of PTS.
The reported study demonstrated that the association of recurrent venous thrombosis and inherited thrombophilia may lead to severe forms of PTS. The effect of these 2 factors on the severity of postthrombotic lesions seems additive. This effect has not yet been evaluated in the literature. In only 1 published study, the authors suggested that severe PTS in young patients may be caused by recurrent venous thrombosis and underlying hereditary disorders of hemostasis, essentially antithrombin III, protein C and S deficiencies, and factor V Leiden mutation and recommended to screen for inherited thrombophilia among young patients with severe PTS and recurrent venous thrombosis. 33
Conclusion
Recurrent venous thrombosis significantly increases the incidence of PTS. The risk of recurrent venous thrombotic events is higher among patients with idiopathic DVT and with permanent risk factors including inherited prothrombotic abnormalities. The presence of inherited thrombophilia either alone or in association with recurrent venous thrombosis correlates with an increased severity of PTS. Thrombophilia seems to interfere in the development of PTS either directly by prolonging residual venous thrombosis or indirectly by increasing the venous thrombotic recurrence rate. Further prospective studies are required to elucidate the pathophysiology of PTS and to confirm the important contribution of recurrence rate and inherited thrombophilia to the pathogeneses of PTS.
Footnotes
Authors’ Notes
This article was presented as a free paper during the XXV World Congress of the International Union of Angiology held on July 1 to 5, 2012 at Prague, Czech Republic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
