Abstract
Background:
Periprocedural anticoagulation is primarily used in endovascular procedures to prevent acute reocclusion of the target vessel, but periprocedural anticoagulation might also have an impact on long-term outcome. Consecutive bleeding events are feared complications. Despite changes in peripheral endovascular revascularizations (EVRs), the periprocedural management has remained unchanged for years. Unfractionated heparin is still the treatment of choice during and immediately after EVR.
Materials and Methods:
We performed a prospective, single-center, open-label phase III study comparing 2 different regimes of enoxaparin peri-interventional to peripheral EVR stratified into low- and high-risk groups according to the acute and long-term reocclusion risk due to their vessel morphology. In both groups, 0.5 mg/kg of enoxaparin as a bolus was administered intravenously 10 to 15 minutes before the start of the procedure. In the low-risk group, 40 mg of enoxaparin were administered once daily for 7 days; whereas in the high-risk group, 1 mg/kg of enoxaparin was administered subcutaneously (sc) 2 times a day for 48 hours after the procedure and afterward 40 mg of enoxaparin was administered sc once daily for 5 days.
Results:
For the analysis of the per protocol population, 44 patients remained in the low-risk group and 140 in the high-risk group. Concerning the primary safety end point, a total of 25 (13.59%) bleeding events occurred until day 30; 5 (11.36%) of them in the low-risk group and 20 (14.29%) in the high-risk group (P = .809 for low vs high risk). None of the bleeding events observed were major according to Thrombolysis In Myocardial Infarction criteria. Concerning our primary efficacy end point, none of the patients showed an acute reocclusion classified as a significant decrease in ankle–brachial index (ABI) or elevated peak systolic velocity ratio confirmed by duplex sonography until day 30. Concerning the second end point of prevention of chronic reobstruction, at day 180 ABI has decreased in the low-risk group from mean 0.94 at day 30 to mean 0.89 and from 1.28 at day 30 to 0.85 after 6 months in the high-risk group. No significant reobstruction was found in the low-risk group, whereas 5 significant reobstruction events were objectified in the high-risk group, all of them in the femoropopliteal arterial segment at day 180.
Conclusion:
We conclude that low-molecular-weight heparin either in a low-dose or high-dose regime during a peripheral EVR is safe concerning bleeding complications and acute reobstructions. The long-term follow-up showed no significant difference between our high- and low-risk groups concerning reobstruction. The periprocedural anticoagulation seems to have no influence on the long-term patency rate after peripheral EVR.
Keywords
Introduction
Despite the dramatic change in daily practice of peripheral endovascular revascularization (EVR) procedures, the periprocedural management has remained unchanged for years. In contrast to coronary interventions, unfractionated heparin (UFH) is still used during and also often in the after course of peripheral interventions. 1 The UFH is an effective antithrombotic agent, but it produces a highly variable anticoagulant response, 2 therefore resulting in either over- or under-anticoagulation in patients and hence potentially inducing hemorrhage or thromboembolic episodes in the course of endovascular interventions. The reason for this is the unpredictable course of action by plasma proteins. Low-molecular-weight heparin (LMWH) on the contrary exhibits considerably less nonspecific binding to plasma proteins than UFH when their inhibitory activities against activated factor X (anti-Xa) activities are compared in vitro or ex vivo. These observations may explain why the anticoagulant response, as measured by anti-Xa levels, is more predictable for LMWH than for UFH, with a highly defined bioavailability of 80%. 3
The LMWHs have proven to be a preferable alternative to UFH in the setting of percutaneous coronary interventions as the use of LMWHs is associated with a significant reduction in major bleeding events compared with UFH, without compromising outcomes on hard ischemic end points. 4,5 Although the superiority of LMWH has been proven for many indications in several cardiovascular trials, there is a lack of data for the use of LMWH in the field of peripheral endovascular interventions. If peripheral interventionists use LMWH, they extrapolate data from cardiology interventions. 6,7
The purpose of our study was to evaluate the efficacy and safety of 2 different regimes of LMWH (enoxaparin), during and immediately after endovascular treatment in patients with peripheral arterial disease (PAD), and their relevance for target vessel reobstruction in the long-term follow-up. Anti-Xa serum levels were also determined in order to assess the predictability of the anticoagulatory potential of the 2 regimens.
Materials and Methods
Study Design
Our study is a prospective, single-center, open-label phase III study comparing 2 different regimes of enoxaparin during and immediately after peripheral EVR stratified into a low- and a high-risk group for reobstruction (acute and chronic) as defined below. The trial started in April 2006 and ended in August 2008 and was approved by the local ethics committee. All patients gave written informed consent.
The risk group was defined based on the reobstruction risk as follows.
The low-risk group was defined as patients with an obstruction <4 cm in the pelvic or <7 cm in the femoropopliteal arterial region and <100% stenosis of the target vessel undergoing balloon angioplasty or bailout stenting and with >2 vessels leading to the forefoot.
Patients assigned to the high-risk group were patients presenting with 100% stenosis (occlusion) of the vessel undergoing balloon angioplasty or bailout stenting and strong decrease in distal flow defined as <2 vessels leading to the forefoot, or patients presenting with an obstruction >4 cm in the pelvic or >7 cm in the femoropopliteal arterial region.
The differentiation between low- and high-risk groups was made according to the probability of acute and reobstruction after the procedure, according to lesions morphology (length and total occlusion).
All patients scheduled for EVR of the lower limbs due to PAD at our institution were screened for participation in the study. The study participants had to meet the following inclusion criteria: lower extremity PAD Rutherford category 2 to 6, age >18 years, and capable of giving informed consent to participate in the study. They had to be scheduled to undergo balloon angioplasty with or without stent insertion of the lower limb arteries (pelvic, superficial femoral, or popliteal arteries) with at least 60% stenosis grade. Exclusion criteria were as follows: pregnant or breast-feeding females, females of childbearing potential not using effective contraception, patients with the evidence of an active bleeding disorder, acquired or inherited coagulopathy, prior history of major bleeding disorder (eg, intracranial hemorrhage), thrombolytic therapy prior to balloon angioplasty or patients treated with oral anticoagulation within 72 hours prior to inclusion, as well as treatment with LMWH or UFH within 72 hours prior to inclusion, major surgery, or recent major trauma within the previous 3 months, uncontrolled hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure > 110 mm Hg), and participation in another clinical study with other investigational agents or devices within the previous 30 days.
Dosage Schedule
In the low-risk group, 0.5 mg/kg of enoxaparin as a bolus was administered intravenously (iv) 10 to 15 minutes before the start of the procedure. The first enoxaparin injection of 40 mg was administered subcutaneously (sc) the following day and repeated daily for 7 days (a total of 8 days of investigational treatment: 1 day of iv and 7 days of enoxaparin low-dose sc).
In the high-risk group, 0.5 mg/kg of enoxaparin as a bolus was administered iv 10 to 15 minutes before the start of the procedure. enoxaparin, 1 mg/kg, was administered sc 2 times a day in the following manner: first dose 8 (+2) hours after the iv bolus injection on day 1; second dose 12 (±1) hours after the first dose, third dose 12 (±1) hours after the second dose, and fourth dose 12 (±1) hours after the third dose. Administration of 40 mg of enoxaparin sc once daily for 5 days was started 24 ±2 hours after the last 1 mg/kg sc dose (a total of 8 days of investigational treatment: 1 day of 0.5 mg/kg iv bolus plus first dose of 1 mg/kg enoxaparin sc, followed by 3 × 1 mg/kg enoxaparin sc [48 hours] and 5 days of 40 mg of enoxaparin sc).
Study End points
The primary study end point was the safety of an anticoagulant regime with enoxaparin during and after balloon angioplasty with or without stent insertion in patients with PAD as measured by Thrombolysis In Myocardial Infarction (TIMI) major and minor bleeding criteria combined until day 30.
Secondary study end points were efficacy and safety end points at different time lines: patency rate of the treated arterial vessel segment after balloon angioplasty with or without stent insertion assessed by color-coded duplex sonography and ankle–brachial index (ABI) once at day 30 (still considered to be an acute reobstruction) and second after 6 months, as well as the percentage of patients with adequate anticoagulation during balloon angioplasty defined as a peak anti-Xa level between 0.5 and 1.5 IU/mL and furthermore the safety of the regimen regarding TIMI major and minor bleedings after 6 months.
Study Procedures
Patients assigned to the study after giving informed consent had to undergo the following visits. At the baseline visit, the following parameters were assessed for all patients: patient demography, clinical examination, medical history, concomitant medication, electrocardiography, and laboratory parameters including hemostatic parameters, color-coded duplex sonography, and ABI. If the patients were randomized after successful EVR, these parameters were also assessed at day 30 and day 180 during the follow-up period. Adverse events or serious adverse events were assessed and reported at any time during the study. All study-relevant procedures and measurements were done by 2 experienced study technicians who were also familiar with ultrasound procedures and acquirement of relevant reobstructions in the treated vessel segment confirmed by duplex ultrasound. Reobstruction was defined as restenosis in the case of a peak systolic velocity of >2.4 m/s or total occlusion objectified with duplex sonography. Revascularization or bypass surgery of the target vessel was also assumed to be related to a reobstruction.
The endovascular interventional procedures were performed referring to the standard of EVRs in peripheral arteries at our institution. All patients were treated either by an antegrade or by a retrograde access, either to perform a plain retrograde or to crossover the endovascular procedure, and a diagnostic angiogram was obtained. Due to the morphological findings of the angiography, the patients were randomized to the 2 different treatment groups, either low- or high-risk group as defined above.
In the case of unsuccessful EVR, by means of an unsuccessful passage of the target lesions, the patients were withdrawn from the study. For plain old balloon angioplasty (POBA) or stent-assisted balloon angioplasty, all commercial products available at that time were used. An EVR was defined as successful if there was no residual stenosis >50%, flow limitation dissection, or peripheral embolization requiring acute treatment verified by angiography.
After successful EVR, manual compression at the puncture site was applied for 15 minutes. No closure devices were used at that time as a standard at our institution. The peri-interventional anticoagulant treatment was applied as described above. Concomitant therapy for the procedure was as follows: if patients were already on acetylsalicylic acid (ASA), this regime was continued after the procedure. Dual antiplatelet therapy or switching to clopidogrel after stent implantation was not a standard at our institution at the time the trial was conducted. If patients were ASA naive, 500 mg of ASA were administered iv the day before the procedure and secondary prophylaxis was started as already described.
Statistical Analysis and Sample Size Justification
Generally, a level of significance α = .05 corresponding with a level of confidence 1 α = .95 was used. The 2-sided 95% confidence interval (CI) was calculated for the primary objective as well as for all secondary objectives.
Summary tables are provided for the baseline, demographic, and vital signs data of all patients included in the study. Data were summarized to render median, mean, minimum, and maximum values. Data were analyzed in a descriptive manner including the necessary and suitable measures of location (means, medians, quartiles, minimum, and maximum values), measures of spread (standard deviations, ranges), and some explorative diagrams. Some explorative tests were also done.
The primary goal of the study was to estimate the frequency of major and minor bleeding assessed by TIMI criteria until day 14. The frequency of major and minor bleeding assessed by TIMI criteria until day 14 has been chosen as the primary safety parameter and was the only decisive for sample size justification in this study.
Bruno et al showed that 48 patients (10.7%) from a total of 448 patients receiving enoxaparin as a single 30-mg iv bolus followed by 1.0 or 1.25 mg/kg sc twice a day experienced 38 minor (8.5%) and 10 major (2.2%) bleeding events during the study, within 2 weeks of enrollment. Therefore, the anticipated rate of TIMI minor and major bleeding events could be estimated by 10%.
To confirm this estimation (2-sided 95% CI of ±4.3%), a total number of nevaluable = 190 evaluable patients were required.
Assuming a dropout rate during the therapy phase of the trial of 5%, a total number of ntotal = 200 patients were required. The sample size was calculated by nQuery Advisor 6.1.
Results
A total of 218 patients were included in our study. In 24 patients, the EVR was unsuccessful and therefore these patients were not randomized to either of the treatment groups. Finally, 194 patients were randomized, 44 into the low-risk group and 150 into the high-risk group. For the final analysis, only 184 patients were available. These patients have been defined as patients available for analysis per protocol, which means that they did not show any major protocol deviations (Figure 1).
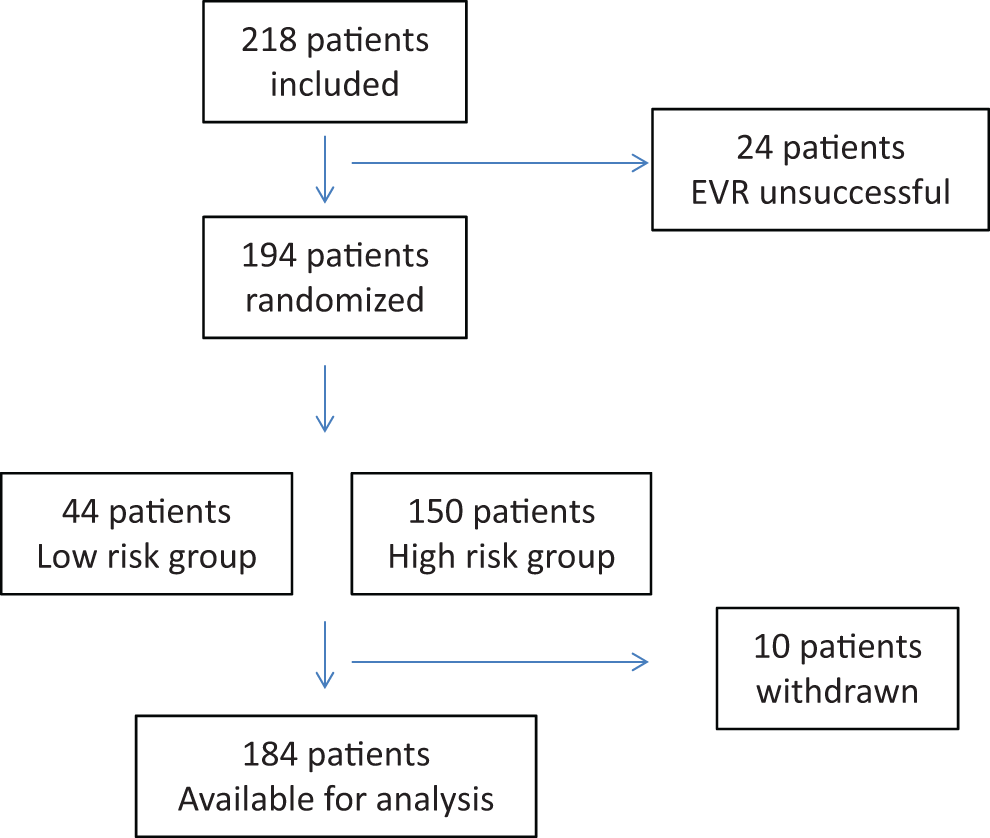
Study profile.
For the analysis of the per protocol population (PP), 44 patients remained in the low and 140 in the high-risk group. Demographic data are shown in Table 1.
Baseline Characteristics of 184 Patients Available for Per Protocol Population (PP) Analysis Per Risk Group.
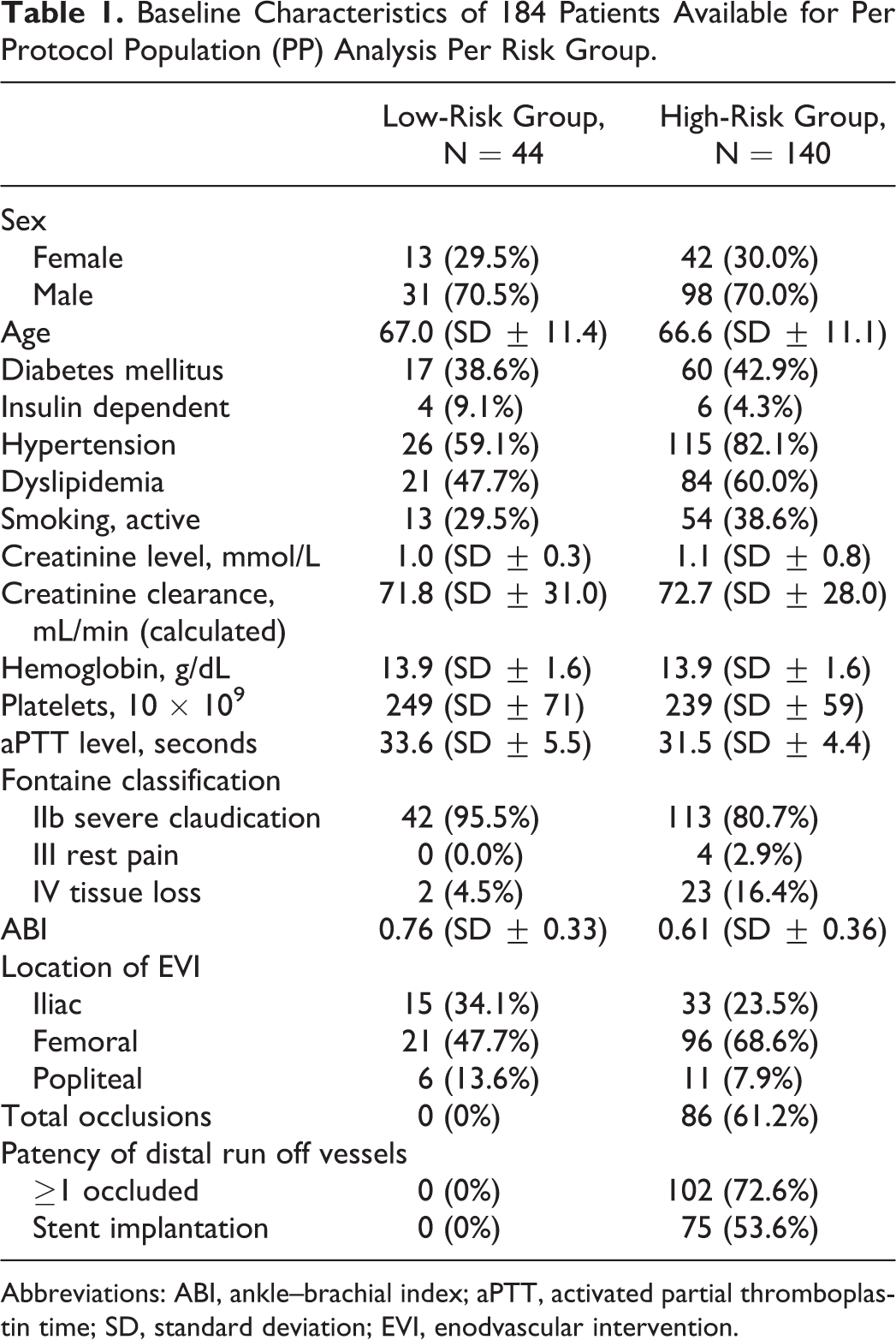
Abbreviations: ABI, ankle–brachial index; aPTT, activated partial thromboplastin time; SD, standard deviation; EVI, enodvascular intervention.
Concerning the primary safety end point, a total of 25 (13.59%) bleeding events occurred until day 30; 5 (11.36%) of them in the low-risk group and 20 (14.29%) in the high-risk group (P= .809 for low- vs high-risk). At day 180, 5 additional (2.72%) bleeding events occurred in the whole PP, all of them in the high-risk group (3.57%; P = .459 for low vs high risk). None of the bleeding events were classified as major bleeding events according to TIMI criteria. All bleeding events were minor bleeding events. A detailed description of the bleeding events is shown in Table 2.
Outcome Parameters, Primary and Secondary Safety, and Efficacy End Points.
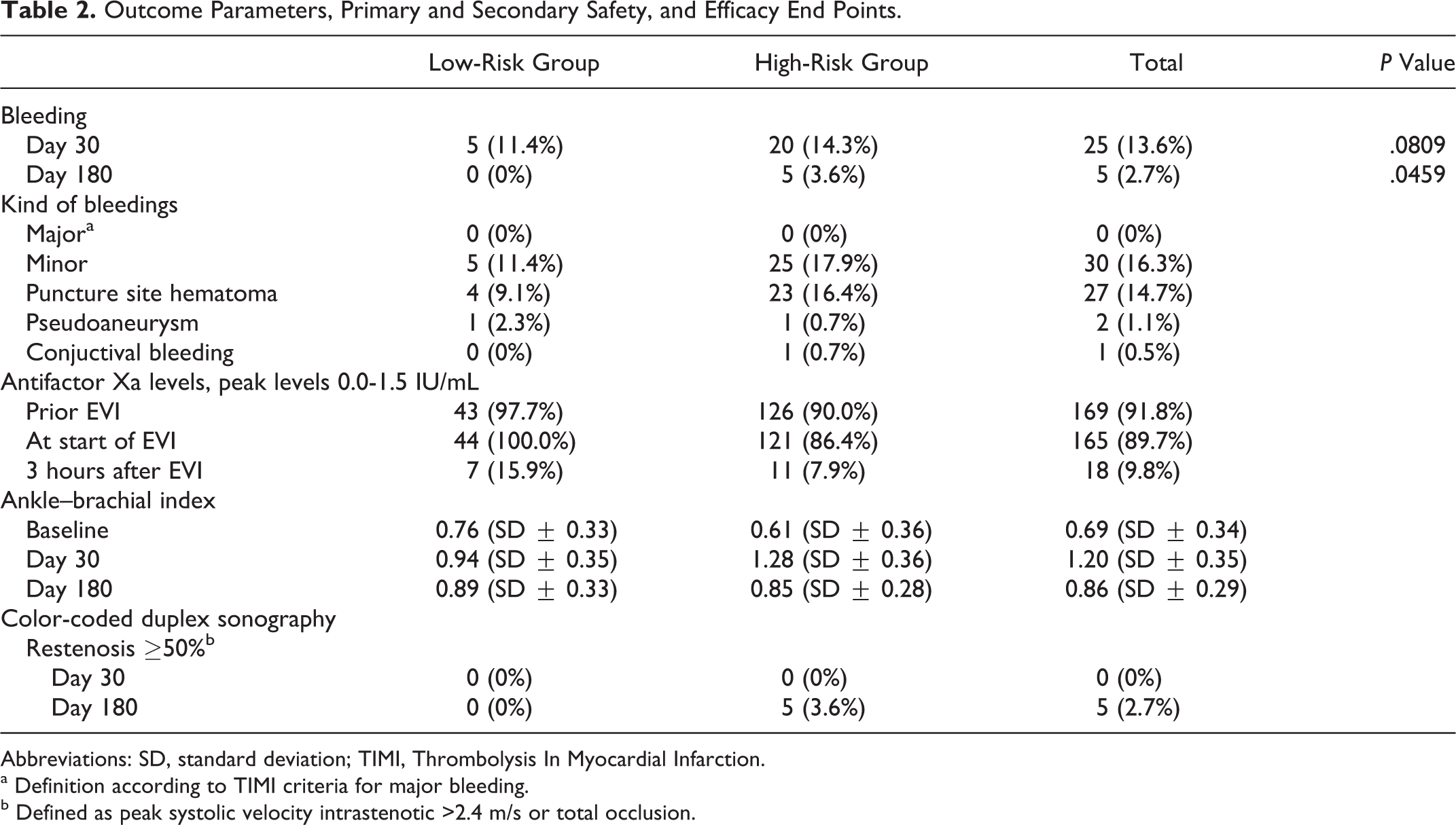
Abbreviations: SD, standard deviation; TIMI, Thrombolysis In Myocardial Infarction.
a Definition according to TIMI criteria for major bleeding.
b Defined as peak systolic velocity intrastenotic >2.4 m/s or total occlusion.
Furthermore, no allergic reaction to enoxaparin at the injection site was reported as well as no occurrence of heparin-induced thrombocytopenia was observed.
In a total of 169 patients, anti-Xa levels were obtained (Table 2). After 3 hours of endovascular intervention (EVI), only 18 patients were found to have peak levels of between 0.5 and 1.5 IU/mL. These findings did not correspond in any way with the occurrence of bleeding events in our patients. All other patients included presented with peak levels below 0.5 IU/mL.
Concerning the efficacy end point, none of the patients showed an acute reobstruction classified by a significant ABI decrease or elevated peak systolic velocity ratio confirmed by duplex sonography, until day 30. At day 180 (month 6) in the low risk group ABI had decreased from 0.94 (day 14) to 0.89, whereas in the high risk group ABI had decreased from 1.28 (day 14) to 0.85 at day 180. No significant reobstruction was found in the low-risk group, whereas 5 patients with significant reobstruction were found in the high-risk group, all of them in the femoropopliteal region. As these patients were completely asymptomatic, no reinterventional procedure was performed. All of these patients had total occlusion at baseline (Table 2).
Discussion
In all kinds of endovascular interventions, anticoagulation is an elementary therapeutic tool for the prevention of reobstructions. For decades, UFH has been the anticoagulant of choice. Due to its pharmacodynamic drawbacks, it has been replaced mainly by LMWH and also by fondaparinux. The major concern when using UFH during endovascular procedures is bleeding complications, 1,8 especially with the additional use of different platelet inhibiting drugs in the after course of endovascular interventions.
Numerous interventional trials in the cardiovascular field have shown a better outcome concerning safety and also efficacy for LMWH than UFH. 4,9 It has been proven that body weight–adapted (0.5 mg/kg body weight [BW]) enoxaparin was associated with a lower bleeding rate than UFH,8a without increasing ischemic end points. 5 Evidence for the peripheral field is lacking. So far only data comparing bivalirudin with UFH in the acute setting of peripheral EVR regarding to safety exist. 10,11
Nowadays, the use of UFH during EVR in patients with PAD is very varied. According to the protocols of various randomized multicenter trials with investigational devices, still different regimes are used during the procedure, ranging from 2000 to 5000 IU. The application of UFH is highly appreciated, as in the case of a major bleeding event, and UFH can be antagonised quickly and totally by means of application of protaminsulfat. After the endovascular treatment, further anticoagulant treatment is up to the interventionist. Most sites performing EVR in patients with PAD are administering enoxaparin 40 mg twice daily for 48 hours after the procedure. 6,7
Concerning safety, namely bleeding complications, any anticoagulation regime in patients with PAD must be critically regarded. Patients with PAD are older—usually 1 decade than patients with coronary artery disease (CAD)—and show significantly more comorbidities than patients with CAD. 12,13 Patients with PAD also present with highly calcified vessels, often also at the puncture site at the groin, which may count for local bleeding complications per se. In order to threshold the long-term patency after EVR from the day of the procedure, at least 1 antiplatelet therapy has to be applied additionally.
Referring to bleeding complications at the puncture site and in general, our site found no increase in peripheral EVR with the applied regime. This is an important finding of our study as bleeding, extrapolated from coronary data, is associated with a higher mortality rate in the 30-day follow-up period of EVR. 14 Concerning the primary end point of our study, we could not identify any major bleeding according to the TIMI criteria at any time in our patients, which means that the higher enoxaparin dosage proofed as safe as the lower enoxaparin dosage, independently of the patient’s general condition. 15
Concerning efficacy, the most feared complication is acute reobstruction of the target lesion. In peripheral arteries, mainly POBA procedures are applied and, if necessary, followed by stent insertion. Mainly bare metal stents are used. According to our data, no acute thrombotic occlusion occurred during the applied regime (0.5 mg enoxaparin/kg BW iv right before the procedure); and referring to the measured peak levels of anti-Xa, 3 hours after the procedure, this seems sufficient for the prevention of acute occlusion after an EVR in the peripheral arteries, with no increase in bleeding rate. Furthermore, no peripheral embolization, procedure related, occurred in our study.
Restenosis in the long-term follow-up still remains the main issue for efficacy of EVR in the peripheral arteries. This fact is strongly related to the region of the intervention per se, the lesion length, and the number of outflow arteries. Referring to the occurrence of reobstruction in peripheral EVR in the long-term follow-up period, only limited data exist in accordance with any anticoagulation regime during the intervention. A small single-center prospective study with a small number of patients (N = 42) administered LMWH (reviparin) in additional to UFH with the purpose to prevent instent restenosis in peripheral arteries. The high-dose regime of reviparin 7000 IU sc once daily over 23 days postprocedural proved to be safe and efficient to prevent instent restenosis in the femoropopliteal arterial segment, with a primary patency rate of 88.5% after 12 months. 16
Koppensteiner et al published a study comparing ASA with ASA and LMWH over a period of 3 months as secondary prophylaxis after EVR in the peripheral arteries (femoropopliteal segment), evaluating occurrence of reobstruction. The rationale behind the usage of LMWH in the prevention of reobstruction after EVR is an antiproliferative effect. The study from Koppensteiner et al failed to reduce reobstruction after femoropopliteal percutaneous transluminal angioplasty during a 12-month follow-up period. 17
Till date no data exist, which refer to a differentiation between low- and high-risk groups for the occurrence of reobstruction in reference to their primary vessel obstruction, with a special adoption of periprocedural anticoagulation in relation to different risks. As long lesion length and decreased peripheral run off lead to a higher reobstruction rate in the long-term follow-up, a more differentiated anticoagulation regime in the periprocedural phase of peripheral EVRs could be useful. 18 –20
Our own data suggest that the treatment regimen for high-risk and low-risk patients—defined by the initial vessel morphology—never led to an acute or symptomatic and therefore clinically relevant reobstruction. Reobstructions occurred but only in the high-risk group that was defined as a group with long lesion length. However, patients were asymptomatic concerning their reobstruction.
Conclusion
We conclude that LMWH during a peripheral EVR is considered safe with regard to periprocedural bleeding complications and sufficient to avoid acute reobstruction. For the long-term follow-up, no difference could be found between our high- and low-risk groups and a more intense regime of anticoagulation adjusted to this different risk of reobstruction. Therefore, the periprocedural anticoagulation process may not have any influence on long-term reobstruction rate in peripheral EVR.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
