Abstract
Due to the pronounced hypercoagulable state in heparin-induced thrombocytopenia (HIT), alternatives to heparin that do not interact with HIT antibodies are needed for anticoagulation management. This study was designed to determine whether the oral factor Xa inhibitor apixaban could be used. Functional platelet activation with apixaban in the presence of HIT antibodies was evaluated by the 14C-serotonin release assay (SRA; washed platelets) and the heparin-induced platelet aggregation assay (PA-HIT; platelet-rich plasma). A consistent absence of platelet activation by apixaban (0.05-50 μg/mL) was observed: SRA (n = 35) 11 ± 4% and PA-HIT (n = 37) 8 ± 3% (mean ± standard error of the mean; positive is >20%) versus heparin (0.1 U/mL) 82 ± 3% SRA and 78 ± 6% PA-HIT (P < 0.01) versus enoxaparin (10 μg/mL) 73 ± 5% SRA and 62 ± 7% PA-HIT. Apixaban may provide an option for oral anticoagulation in patients with HIT, particularly for extended management and prevention.
Introduction
Apixaban (Eliquis) is an orally bioavailable, small-molecule (459 Da molecular weight), direct factor Xa (FXa) inhibitor. Apixaban has been recently approved by the US Food and Drug Administration for the prevention of stroke and systemic embolism in patients with atrial fibrillation. Apixaban is also approved in the European Community and in Japan for stroke prevention in patients with atrial fibrillation, and it is approved in several countries (excluding the United States) for preventing blood clots following knee or hip replacement surgery. Apixaban is under investigation for the treatment of venous thromboembolism in the Apixaban after the initial Management of Pulmonary embolism and deep vein thrombosis with First-line therapy (AMPLIFY) and Apixaban after the initial Management of Pulmonary embolism and deep vein thrombosis with First-line therapy-Extended Treatment (AMPLIFY-EXT) trials, results of which will be reported in 2013.
At present, unfractionated heparin (UFH) and low-molecular-weight (LMW) heparin anticoagulation are widely used in the management of these and other patient populations. However, their use is restricted by the heparin-induced thrombocytopenia (HIT) syndrome caused by the heparin-based drugs. The pathophysiological mechanism of HIT involves the generation of antibodies targeted to neoepitopes on the platelet factor 4 (PF4) molecule which are exposed when PF4 is complexed with heparin or other highly charged materials. 1-4 Polyclonal antibodies are generated which include immunoglobulins (Ig) G, A, and M. Complexes of IgG/PF4/heparin bind to the FcγIIa receptor on platelets causing platelet activation (with more PF4 release). 5 This immune-mediated disorder also causes thrombin generation, endothelial cell damage, upregulation of the inflammatory response, and a pronounced hypercoagulable state. 6,7 Untreated HIT results in amputation or death in more than 35% of the affected patients. 8,9
Patients with HIT need anticoagulation but require drug alternatives to the heparins. Direct thrombin inhibitors (DTIs) such as argatroban and lepirudin administered intravenously do not cross-react with HIT antibodies, 10-13 and thus became the first effective nonheparin/nonglycosaminoglycan anticoagulant drug treatments for patients diagnosed with HIT. The current consensus guideline for patients who have HIT is to immediately remove the heparin and provide anticoagulation with an alternative anticoagulant to prevent or treat associated thrombosis. 9,14
Although intravenous DTIs are effective in patients with HIT, 15- 18 they are associated with a bleeding risk and drug-specific limitations. 19- 23 These include anaphylactic reactions and antibody production with lepirudin and interfering effects in clinical laboratory assays with all DTIs, particularly argatroban. For long-term anticoagulation, the patients are switched from the intravenous DTI to an oral vitamin K antagonist (VKA) in a bridging manner akin to switching from UFH to a VKA. This presents its own set of problems that cause concern for the clinicians from the standpoint of patient safety. 23 -26 Apart from the unpredictable pharmacokinetics and the need for routine monitoring, the VKAs have a slow onset of action and the potential to precipitate venous limb gangrene or skin necrosis due to its inhibition of protein C. 27 The latter 2 properties of VKAs are of particular concern when treating patients with HIT.
The anticoagulation management in patients with HIT has not yet been optimized. In particular, anticoagulant options other than a drug administered by intravenous infusion in a hospital setting and drugs with a lesser bleeding risk are needed for prevention of thrombosis in patients with mild symptoms of HIT and also where the diagnosis of HIT is not clear. Further, oral anticoagulant options other than VKAs are needed for patients requiring long-term anticoagulation.
The heparin-derived pentasaccharide, fondaparinux (Arixtra), is an indirect acting FXa inhibitor, requiring binding to antithrombin and administered subcutaneously. Because it does not have a strong in vitro cross-reactivity with HIT antibodies due to its small molecular size, 28 fondaparinux has been used with success in the management of patients with HIT 20 ,29 ,30 (and other case studies). Clinicians are beginning to view fondaparinux as a promising prevention/treatment option for patients with HIT. Yet, questions remain as fondaparinux is not inert as an immunogen. 31 ,32 Moreover, fondaparinux is not an orally administered drug.
The FXa inhibitor apixaban is structurally different from UFH, LMW heparins, and fondaparinux. It is therefore expected that apixaban will not interact and complex with PF4 in a manner that would generate neoepitopes of PF4 inducing HIT antibody formation. It is also expected that apixaban will not be recognized by preexisting HIT antibodies and thus will not contribute to the propagation of an existing HIT syndrome.
The present study was undertaken to validate the hypothesis that apixaban lacks the ability to interact with HIT antibodies causing activation of platelet functional activities as a first step to establish the potential for apixaban to be used for the anticoagulation management of HIT.
Materials and Methods
Laboratory
Apixaban was evaluated in 2 platelet function assays, the 14C-serotonin release assay (14C-SRA) and the platelet aggregation assay (PA-HIT). Tests were performed by highly skilled and experienced staff. 33
Study Drugs
Apixaban (Eliquis; Bristol-Myers Squibb, Princeton, New Jersey): a range of concentrations for apixaban (0.05 to 50 μg/mL) including the clinical dosing range were evaluated for a comprehensive analysis.
Unfractionated sodium heparin (APP, Schaumburg, Illinois): classical concentrations were used (0.1 and 100 U/mL) in the 14C-SRA which depict the bell-shaped curve response of UFH to HIT antibodies. In the PA-HIT, the final concentrations of 0.1 to 10 μg/mL of UFH were used. This served as the positive control. Saline was used as a negative control.
The LMW heparin enoxaparin (Lovenox; Sanofi US, Bridgewater, New Jersey) was purchased from the Loyola hospital pharmacy. Dilutions of drug in saline were made such that the final concentrations from 0.1 to 100 µg/mL were studied.
Fondaparinux (Arixtra; GlaxoSmithKline, Philadelphia, Pennsylvania) was purchased from the Loyola hospital pharmacy. Dilutions of drug in saline were made such that the final concentrations from 0.1 to 100 µg/mL were studied.
Heparin-Induced Thrombocytopenia Antibodies
Patient sera were obtained from the Serotonin Release Assay Laboratory of the Loyola University Medical Center (Maywood, Illinois). Patient specimens were collected under ethical board approval. Only sera testing positive with heparin in the 14C-SRA were used. Prior to use, the patient serum was heat inactivated at 56°C for 45 minutes to remove residual enzymatic activity.
Platelets
Platelets were prepared from fresh normal blood collected by venipuncture after obtaining written informed consent from the donor. Studies were performed immediately upon collection of the blood. Only prescreened donors whose platelets were known to be reactive in the 14C-SRA and the PA-HIT assays were used.
14C-Serotonin Release Assay
In brief, the 14C-SRA method was performed as follows. Washed platelets were prepared immediately after collection of whole blood into acid–citrate–dextrose anticoagulant then loaded with radiolabeled 14C-serotonin. Washed, labeled platelets, serum, and drug were mixed together and incubated for 60 minutes at 22°C to 24°C. The reaction was stopped with EDTA, and platelets were pelleted by centrifugation. Supernatant radioactivity was measured in a scintillation counter. Each run included counts for background activity and total 14C-serotonin release. The radioactivity counts were used to calculate a positive (≥ 20% serotonin release) or negative response for each specimen. Internal assay serum controls for positive and negative responses were included in each run.
Heparin-Induced Platelet Aggregation Assay
In brief, the PA-HIT method was performed as follows. Platelet-rich plasma (PRP) was prepared immediately after whole blood collection into tubes containing sodium citrate. The PRP, serum, and drug were mixed together and immediately assayed for platelet aggregation over a 20-minute period on the PAP-4 Platelet Aggregometer (BioData, Horsham, Pennsylvania). The results were reported as maximum percentage of platelet aggregation and slope (rate) of the reaction using the light transmission of platelet-poor plasma to identify 100% aggregation. A positive response was defined as aggregation ≥20%.
Results
14C-Serotonin Release Assay
The 14C-SRA evaluated functional platelet activation. A total of 35 specimens from patients with HIT/donor platelet combinations were used to evaluate apixaban for cross-reactivity to HIT antibodies (Figure 1A). Apixaban did not interact with preformed HIT antibodies to cause platelet activation as determined by the 14C-SRA for any specimen.
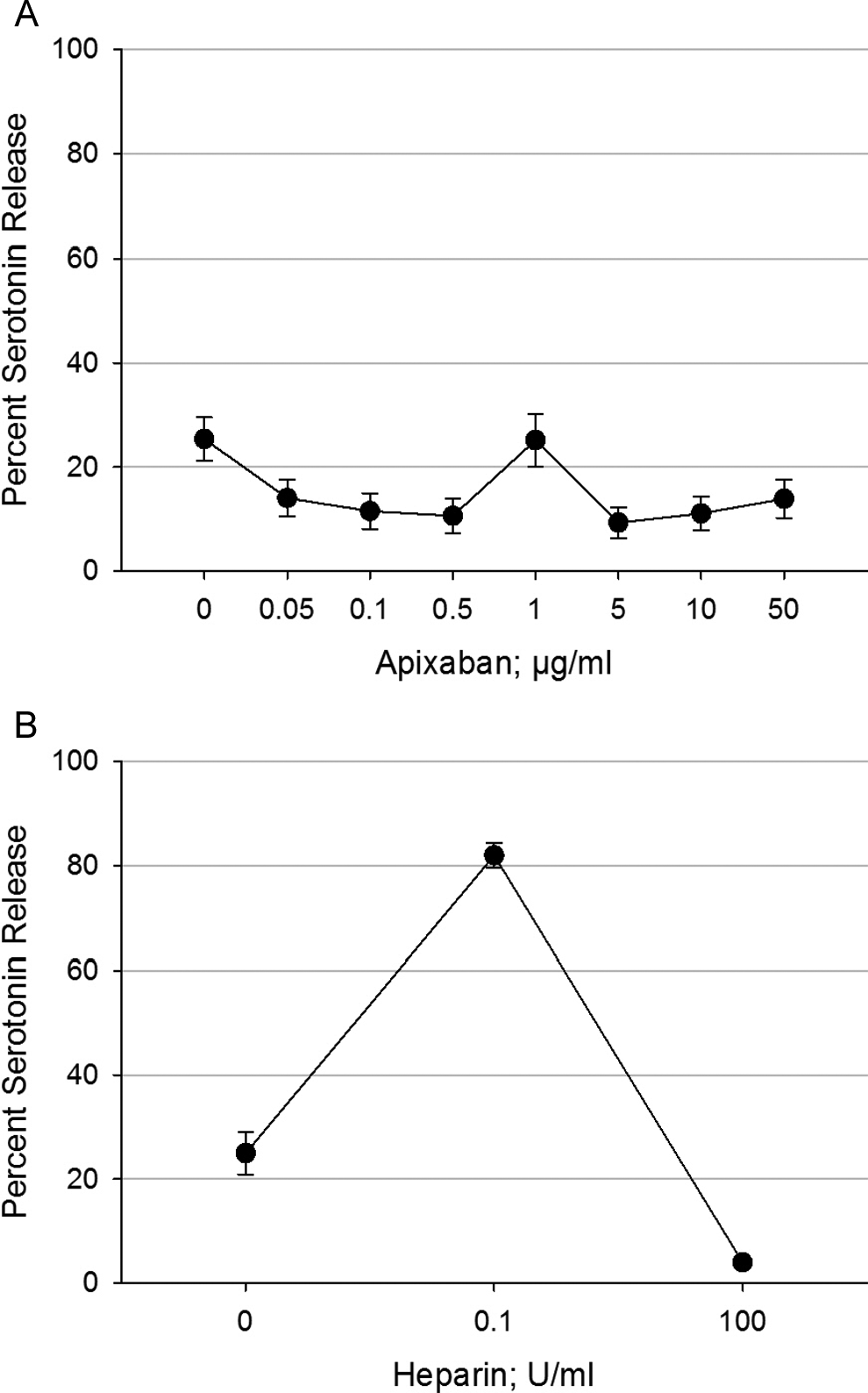
Cross-reactivity of apixaban and heparin with HIT antibodies. A, Apixaban (0.05-50 µg/mL) was tested in vitro in the 14C-SRA using HIT serum (antibodies) from different patients and platelets from different donors (n = 35). Saline was used as a negative control. Negative 14C-SRA responses are <20% serotonin release. Apixaban did not cross-react with any HIT antibody. B, In the 14C-SRA, unfractionated heparin was tested at 0.1 and 100 U/mL (n = 35). The positive control is represented by the 0.1 U/mL heparin concentration (>80% serotonin release) and a negative platelet activation response for the supratherapeutic concentration of 100 U/mL heparin, as shown here. Data in (A) and (B) are from the same assay runs with apixaban and heparin tested simultaneously on the same HIT antibody/platelet combinations. Results are shown as mean ± standard error of the mean. 14C-SRA indicates 14C-serotonin release assay; HIT, heparin-induced thrombocytopenia.
In comparison, UFH gave a positive response to each of the same HIT antibody specimen/platelet donor combinations in the 14C-SRA (Figure 1B).
Another experiment was performed to compare the apixaban response to that of the LMW heparin enoxaparin and to fondaparinux in the 14C-SRA. Like apixaban, fondaparinux did not cause platelet activation with HIT antibodies (n = 10; Figure 2). Enoxaparin, on the other hand, had a strong activation response similar to that of UFH but shifted to the right (ie, beginning at higher concentrations) with a somewhat lower maximum activation response.
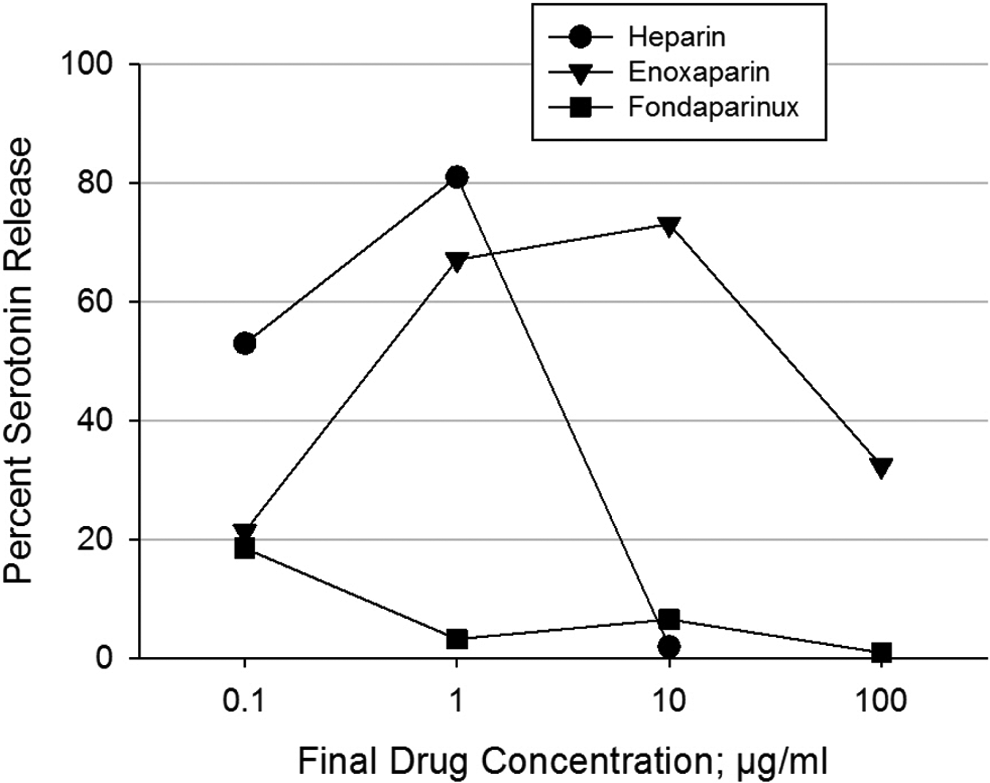
Cross-reactivity of LMW heparin, fondaparinux, and heparin with HIT antibodies. The LMW heparin enoxaparin, the FXa inhibitor fondaparinux, and unfractionated heparin were tested in vitro in the 14C-SRA at 0.1, 1.0, 10, and 100 µg/mL using HIT serum (antibodies) from different patients and platelets from different donors (n = 10) chosen from the same combinations used in Figure 1A and B. Negative 14C-SRA responses are <20% serotonin release. Fondaparinux did not show cross-reactivity with any HIT antibody. Enoxaparin had a strong cross-reactivity similar in strength to that of heparin. Results are shown as mean ± standard error of the mean. 14C-SRA indicates 14C-serotonin release assay; FXa, factor Xa; HIT, heparin-induced thrombocytopenia; LMW, low molecular weight.
Platelet Aggregation Assay
The PA-HIT evaluated functional platelet aggregation. A total of 37 specimens from patients with HIT/platelet donor combinations were used to evaluate apixaban for cross-reactivity to HIT antibodies (Figure 3A). Apixaban did not interact with preformed HIT antibodies to cause platelet aggregation as determined by the PA-HIT for any specimen.
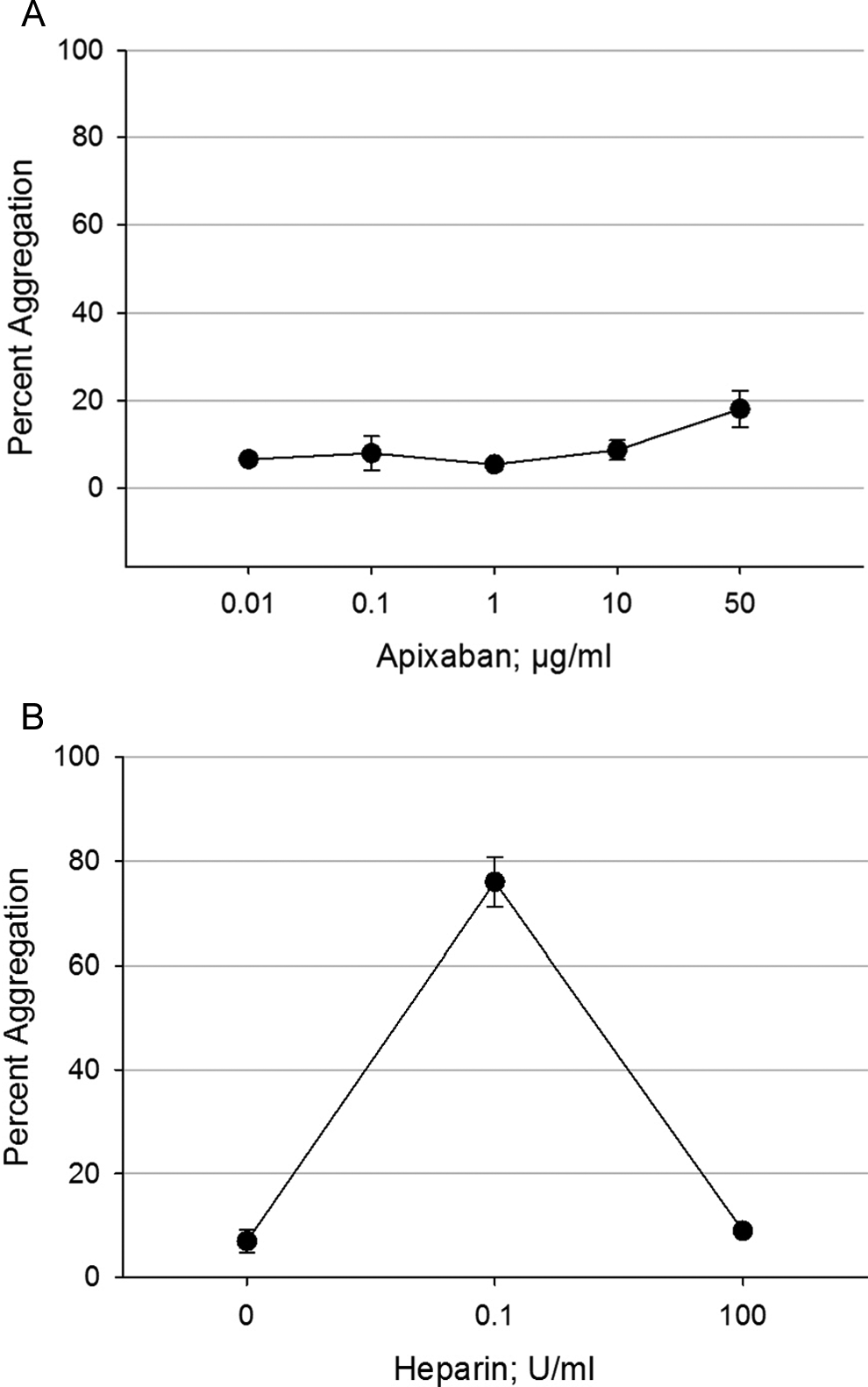
Cross-reactivity of apixaban and heparin with HIT antibodies. A, Apixaban was tested in vitro in the PA-HIT from 0.01 to 50 µg/mL using HIT serum (antibodies) from different patients and platelets from different donors (n = 37). A negative PA-HIT response is <20% aggregation. Apixaban did not cross-react with any HIT antibody. B, In the PA-HIT, unfractionated heparin was tested at 0.1 and 100 U/mL (n = 37); saline was used as the negative control. The positive control is represented by the 0.1 U/mL concentration (>70% aggregation). For a positive PA-HIT result, heparin at the supratherapeutic concentration of 100 U/mL will give a negative platelet aggregation response, as shown here. The data in (A) and (B) are from the same assay runs with apixaban and heparin tested simultaneously on the same HIT antibody/platelet combinations. Results are shown as mean ± standard error of the mean. HIT indicates heparin-induced thrombocytopenia; PA-HIT, heparin-induced platelet aggregation assay.
In comparison, UFH produced a strong positive platelet aggregation response with each of the same HIT specimen/platelet donor combinations in the PA-HIT (Figure 3B).
Another experiment was performed to compare the apixaban response in the platelet aggregation test to that of the LMW heparin enoxaparin. Enoxaparin had a strong activation response with HIT antibodies similar to that of UFH but with a slightly lower maximum aggregation response (n = 10; Figure 4). As observed in the 14C-SRA, the activation response of the LMW heparin was shifted to the right (ie, beginning at higher concentrations) compared to that of UFH. Apixaban, unlike UFH and LMW heparin, did not induce platelet aggregation in association with HIT antibodies.
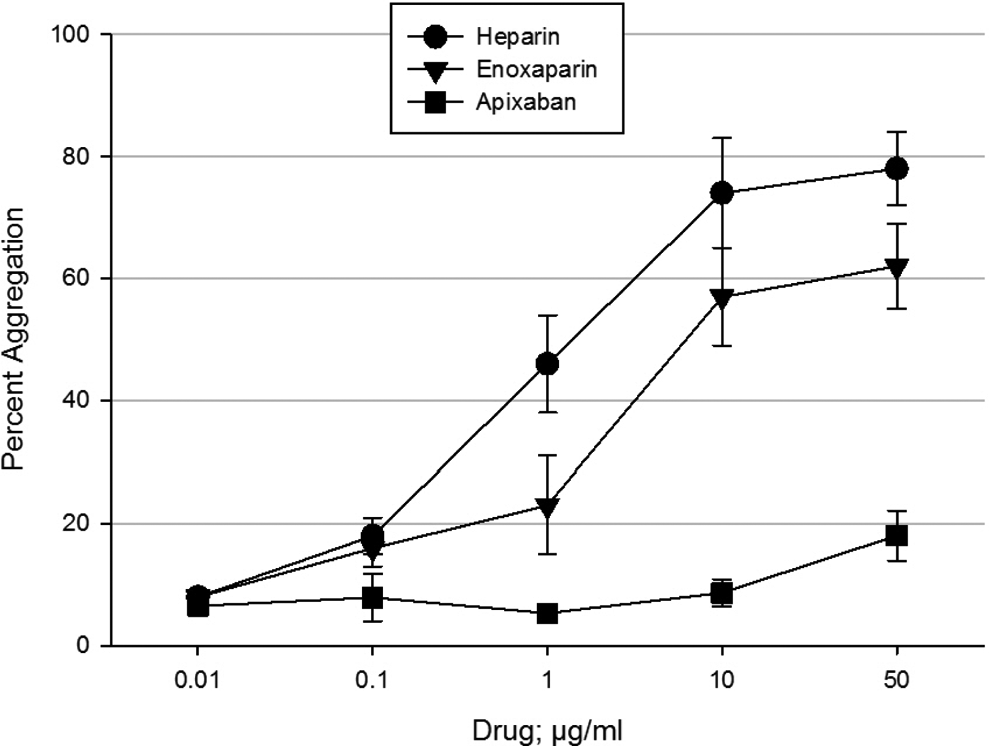
Cross-reactivity of apixaban, LMW heparin, and heparin with HIT antibodies. Apixaban was tested in vitro in comparison to the LMW heparin enoxaparin and unfractionated heparin in the PA-HIT at 0.01, 0.1, 1.0, 10, and 50 µg/mL using HIT serum (antibodies) from different patients and platelets from different donors (n = 10) chosen from the same combinations used in Figure 1A and B. A negative PA-HIT response is <20% aggregation. Apixaban did not show cross-reactivity with any HIT antibody. Enoxaparin had a strong cross-reactivity similar to that of heparin. The results are shown as mean ± standard error of the mean. HIT indicates heparin-induced thrombocytopenia; LMW, low molecular weight; PA-HIT, heparin-induced platelet aggregation assay.
Discussion
Patients with HIT develop antibodies which, in the presence of heparin, cause a hallmark measurable platelet activation and aggregation response. To confirm a clinical diagnosis of HIT 1 of the 2 traditional and widely used clinical laboratory tests of platelet function is performed, the gold standard 14C-SRA or the PA-HIT. These 2 test systems are based on different methodologies and thus have different specificities and sensitivities to HIT antibodies. Different yet complimentary information is obtained with each assay. Both tests were employed in this investigation.
Furthermore, the platelet function response to HIT antibodies varies by patient serum (polyclonal HIT antibodies) and by platelet donor. To assure the robustness of the outcome of our data in this study, we utilized HIT antibodies from multiple patients and platelets from different donors. Apixaban concentrations covering the clinical dosing range as well as exceeding the clinical range were compared to the response obtained with clinically relevant concentrations (in units) of heparin.
Since apixaban is structurally unrelated to heparin, HIT antibodies did not target this drug, and platelet activation and aggregation were not observed in the 2 clinical assays for HIT. This response was consistent across all HIT specimens and all platelet donors tested in this study across a range of apixaban concentrations. The in vitro investigations reported herein demonstrate a clear potential for apixaban as an anticoagulant option for the management of thrombosis in patients with HIT. Further assurance of the strength of this finding of a lack of platelet activation in the presence of HIT antibodies rests in the fact that the same finding was obtained with another small molecule FXa inhibitor, rivaroxaban (Xarelto). 34
Apixaban is approved for clinical use in the United States and other countries in atrial fibrillation with additional approvals expected. If utilized in patients with HIT, apixaban would offer several advantages. The most obvious is that apixaban could replace VKAs as an option for oral anticoagulation in patients with HIT in both the inpatient and outpatient settings. This would eliminate the risk of warfarin-induced skin necrosis. Based on the promising results being obtained with the FXa inhibitor fondaparinux for the prevention of thrombosis in HIT 20,29,30 (and other case studies), it is expected that the FXa inhibitory action of apixaban would similarly provide clinical efficacy. Each of the new oral anticoagulants apixaban, rivaroxaban, and dabigatran may potentially fit well into the clinical management protocols for the prevention of thrombosis in patients with HIT. Intravenously administered thrombin inhibitors will likely remain the drug of choice for treatment of acute HIT thrombosis. The general use of nonheparin anticoagulants over heparin and LMW heparin will reduce the occurrence of HIT.
Since anticoagulation in acute episodes of HIT (marked thrombocytopenia and thrombosis) is of critical need, it may be of value to explore the possibility of a parenteral formulation of apixaban in this indication. This option is of increased interest since danaparoid and lepirudin, effective parenteral anticoagulants for the management of patients with HIT, are no longer available. It should also be noted that apixaban could be used not only in patients with HIT but also in all patients who are heparin compromised (eg, antithrombin deficiency).
This study is the first step to assess the potential of apixaban as an alternative anticoagulant for HIT. Patients with HIT can be in a very active hypercoagulable state, accompanied by life- and limb-threatening thrombosis, they can be recovering from thrombosis, or they can be in a nonthrombotic state. Clinical trials to determine dosing regimens that provide safe and effective anticoagulation during the various clinical phases of HIT are necessary. The study reported herein provides the first evidence that apixaban may provide an option for oral anticoagulation in patients with HIT, particularly for extended management and prevention.
Footnotes
Authors’ Note
The sponsors had no role in the study design, data collection, data interpretation, and the writing of this publication. The authors are fully responsible for all content and editorial decisions related to this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research grant from Bristol-Myers Squibb and Pfizer Inc.
