Abstract
Objective:
To examine the efficacy and adverse events of recombinant human thrombopoietin (rhTPO) in the treatment of infection-associated thrombocytopenia in senile patients.
Methods:
The current study is a retrospective analysis of the patients receiving rhTPO for infection-associated thrombocytopenia in our hospital.
Results:
Forty-nine cases were included in the analysis as rhTPO group. The absolute platelet count after treatment, increase in platelet count, and the overall response rate were considerably higher in the rhTPO group than that in the control group. Improvement in bleeding score was higher in the rhTPO treatment group than that in the control group (2.1 ± 5.4 vs 0.4 ± 1.7). Bleeding event was stopped in 68.2% of the patients after rhTPO treatment and in 35% of the patients in the control group (P = .032). A stratified analysis indicated that the therapeutic efficacy is much better in patients without organ failure.
Conclusion:
Recombinant human TPO is effective in alleviating infection-associated thrombocytopenia and hemorrhage in senile patients, particularly if given prior to the emergence of organ failure.
Introduction
Thrombocytopenia is a common complication in severe infection, 1 particularly in elderly population due to decreased hemotopoiesis. Thrombocytopenia could lead to internal hemorrhage and is an independent prognostic factor in patients with severe infection. 2 –4 Several investigations show that dysregulation of the thrombopoietin (TPO)/Mpl receptor system is present in sepsis. 5 –7 Thrombopoietin TPO, known as c-mpl ligand or megakaryocyte growth and development factor (MGDF), promotes proliferation of commited megakaryocytic precursors and differentiation of immature megakaryoblasts in vitro and in vivo. 8,9 Thrombopoietin is constitutively produced by the liver and kidneys and is then cleared from circulation upon binding with its receptor, c-mpl, and expressed mainly on platelets and megakaryocytes. 10,11 Many evidences indicate that TPO acting through c-mpl is essential for normal thrombopoiesis. 12 Recombinant human TPO (rhTPO) was approved in China in 2006. The efficacy of rhTPO has been established for chemotherapy-induced thrombocytopenia and idiopathic thrombocytopenic purpura (ITP). 13 –16 The current study is a retrospective analysis of the potential therapeutic efficacy and adverse events of rhTPO in senile patients with infection-associated thrombocytopenia.
Methods
Patients
The data from the first 90 consecutive patients admitted in the Southern ward of PLA General Hospital and treated with rhTPO, from October 2007 to October 2010, for retrospective analysis.
All the following criteria must be met for inclusion: (1) >60 years of age; (2) presence of thrombocytopenia (platelet count <100 × 109/L); (3) infection confirmed with microbiological evidence; (4) fever >38°C; and (5) complete medical record that could be used to evaluate the therapeutic effects. Exclusion criteria were thrombocytopenia induced by other reasons such as chemotherapy, hypersplenism, drug-induced thrombocytopenia, disseminated intravascular coagulation, ITP, and other hematological disorders. A group of patients not receiving rhTPO for infection-associated thrombocytopenia admitted in our hospital in the same time were included as a control. The 2 groups were matched on a 1:1 ratio for gender and age.
Treatment
Recombinant human TPO was injected subcutaneously at a dose of 15 000 units/day. Recombinant human TPO was stopped when the platelet count became normal (platelet > 100 × 109/L) or there was no response after 14 days of treatment. Its usage was similar to that used for chemotherapy-induced thrombocytopenia. Antibiotic treatment and other supportive care (eg, platelet transfusion when platelet count <20 × 109/L or active bleeding exists) were available based on physicians’ discretion. Patients in the control group received identical treatment options other than rhTPO.
Study Measures
The following characteristics were recorded on a standardized form: age, sex, past history, days of rhTPO application, blood routine test, liver and kidney function (alanine transaminase [ALT], gamma-glutamyl transpeptidase [GGT], total bilirubin [TBIL], direct bilirubin [DBIL], blood urea nitrogen [BUN], creatinine [Cr]), coagulation function (prothrombin time [PT], activated partial thromboplastin time [APTT], and fibrinogen [Fbg]), platelet transfusion, hemorrhage events, and adverse reactions. Bleeding severity was evaluated at inclusion and during follow-up, with a previously reported standardized bleeding score. 17 Blood routine test was recorded once every other day. In the rhTPO group, the baseline platelet count was defined as the last measure prior to the treatment, and in the control group, it is the third consecutive platelet count at less than the lower limit of the normal range. In the rhTPO group, the platelet count after the treatment was defined as the first measure after the discontinuation of the treatment, and in the control group, it is the measure at the closest date of that in the matched patient.
Efficacy Assessment
Treatment efficacy was evaluated according to the criteria from an international working group, in 2009. 18 “Complete response” (CR) is defined as any platelet count of at least 100 × 109/L. “Response” (R) is defined as any platelet count between 30 and 100 × 109/L and at least doubling of the baseline count. “No response” (NR) is defined as any platelet count lower than 30 × 109/L or less than doubling of the baseline count. Total response rate is defined as CR rate plus the R rate. The definition of R requires concurrent resolution of bleeding symptoms.
Statistical Analyses
All numerical data showed a normal distribution and were reported as mean ± standard deviation. Paired sample t test was used to compare baseline data between patients who received rhTPO treatment and those who did not receive rhTPO treatment. A stratified analysis of patients with one or more organ failure versus those without was also carried out by using Student t test. Covariance analysis was used to evaluate the changes before and after treatment between rhTPO group and control group and between subgroups with or without organ failure. Event rate was analyzed using chi-square test. Statistical significance was set at P < .05.
Results
General Data
Among the first 90 patients who were to receive rhTPO, 41 patients were excluded from the final analysis because thrombocytopenia was not induced by infection. In the excluded patients, 6 patients had myelodysplastic syndrome, 2 patients had leukemia, 17 patients received chemotherapy, 2 patients had hypersplenism, 3 patients had ITP, 2 patients had disseminated intravascular coagulation, 6 patients had drug-induced thrombocytopenia, and 3 patients had bone marrow metastasis from other cancer. Finally, a total of 49 patients were included in the treatment group. Table 1 summarizes the main characteristics of the 49 patients included. These patients received rhTPO for a period of 14.5 ± 12.7 days. The gender and age did not differ between the 2 groups. The baseline platelet count was 45.9 ± 18.3 × 109/L in the rhTPO group and 48.2 ± 20.5×109/L in the control group. The 2 groups did not differ in their baseline platelet count, white blood cell count, hemoglobin level, and severity of bleeding at inclusion.
Baseline Characteristics.a
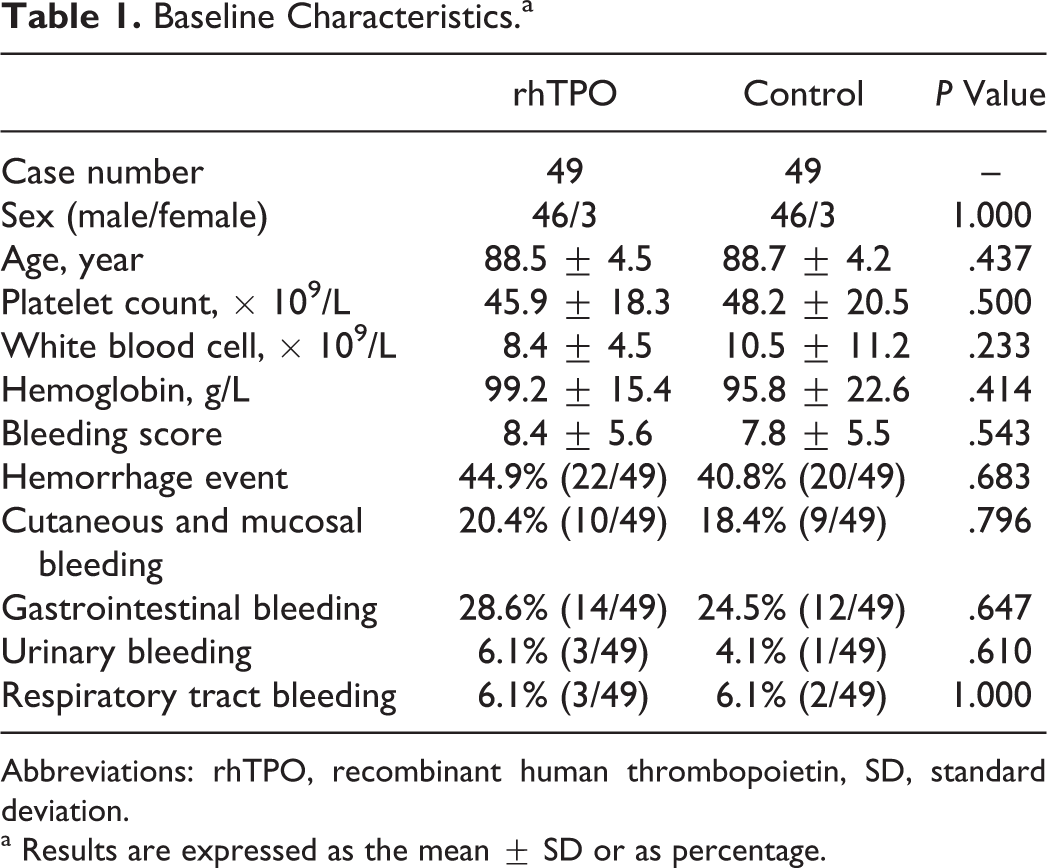
Abbreviations: rhTPO, recombinant human thrombopoietin, SD, standard deviation.
a Results are expressed as the mean ± SD or as percentage.
Therapeutic Efficacy
Analysis of covariance was done using platelet count prior to treatment as the covariance. In the treatment group, platelet count increased from 45.9 ± 18.3 × 109/L at the baseline to 95.4 ± 58.2 × 109/L after rhTPO treatment (Table 2). The rate of CR was 49.0% (24 of 49) and the R rate was 16.3% (8 of 49). In the control group, the platelet count increased from 48.2 ± 20.5 × 109/L at baseline to 71.0 ± 34.1 × 109/L, with a significantly lower CR rate at 26.5%. Improvement in bleeding score was higher in the rhTPO treatment group than that in the control group (2.1 ± 5.4 vs 0.4 ± 1.7, P = .029). After rhTPO treatment, bleeding event was stopped in 68.2% (15 of 22) of the patients and in 35% (7 of 20) of the patients in the control group (P = .032).
Therapeutic Effects in rhTPO and Control Groups.a
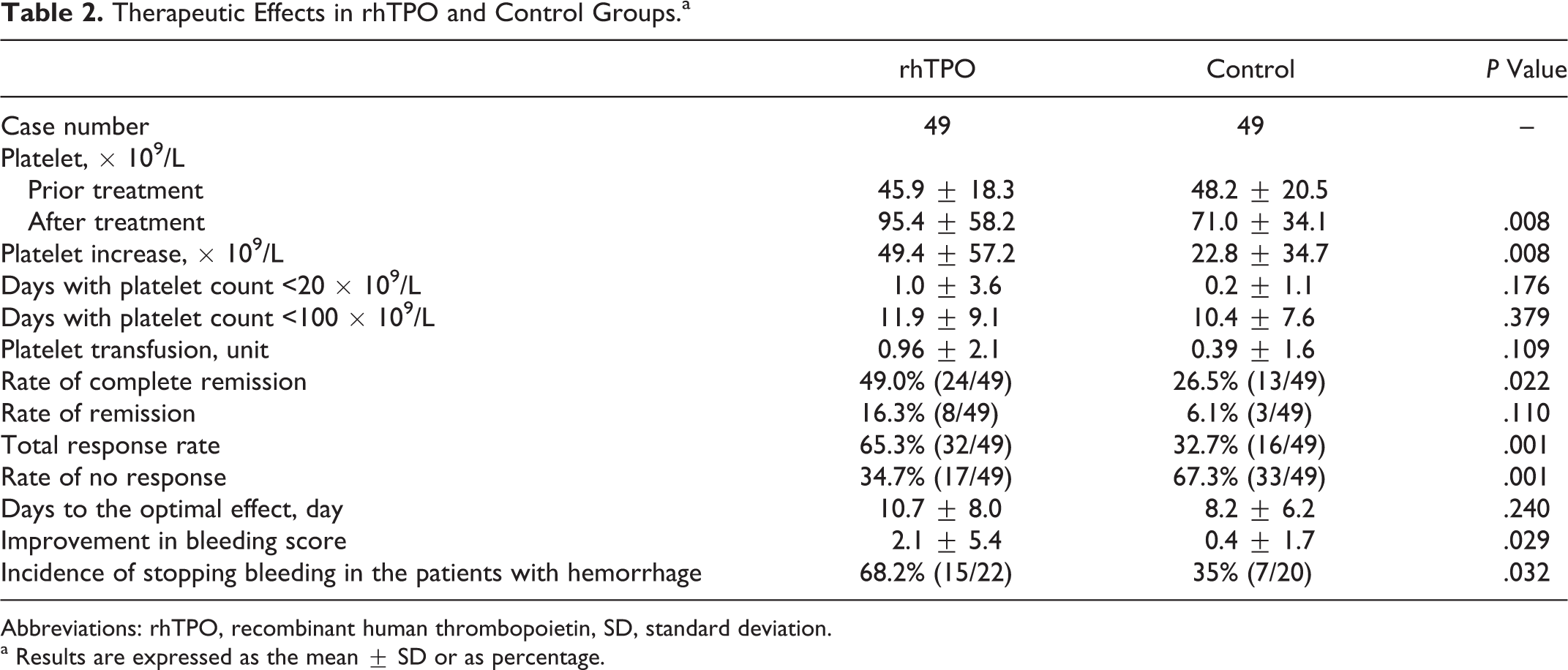
Abbreviations: rhTPO, recombinant human thrombopoietin, SD, standard deviation.
a Results are expressed as the mean ± SD or as percentage.
The 2 groups did not differ in the following measures: days with severe thrombocytopenia (platelet count < 20 × 109/L), days with platelet count <100 × 109/L, the amount of platelet transfusion, and days to achieve the optimal effect. Mortality rate is 28.6% (14 of 49) in the rhTPO group and 44.9% (22 of 49) in the control group. Although the mortality rate in the rhTPO group was lower than that in the control group, there was no statistically significant difference between the 2 groups (Pearson chi-square P = .094).
Impact of Organ Failure
The infection was accompanied by one or more organ failure in 41 of 49 patients in the rhTPO group. In the rhTPO group, the baseline platelet count and the duration of rhTPO treatment did not differ between the 41 patients with organ failure and the remaining 8 patients without organ failure (Table 3). In patients with organ failure, the platelet count was 47.5 ± 18.6 × 109/L at the baseline, which increased to 88.2 ± 54.1 × 109/L after rhTPO treatment. The overall response rate in patients with organ failure was 58.5% (24 of 41), including the CR rate of 43.9% (18 of 41) and R rate of 14.6% (6 of 41). In the remaining 8 patients without organ failure, the platelet count was 37.9 ± 14.9 × 109/L at the baseline, which increased to 132.0 ± 68.3 × 109/L after rhTPO treatment. The overall response rate of rhTPO treatment was 100% (P < .05 vs. 58.5% in patients with organ failure) including the R rate of 25% (2of 8) and the CR rate of 75% (6 of 8). Both the absolute platelet count after the treatment and the platelet increase were significantly higher in patients without organ failure. The 2 subgroups did not differ in the days with platelet count at < 20 × 109/L, days with platelet count at < 100 × 109/L, platelet transfusion, improvement in bleeding score, and incidence of stopping bleeding in the patients with hemorrhage.
The Impact of Organ Failure on rhTPO Efficacy.a
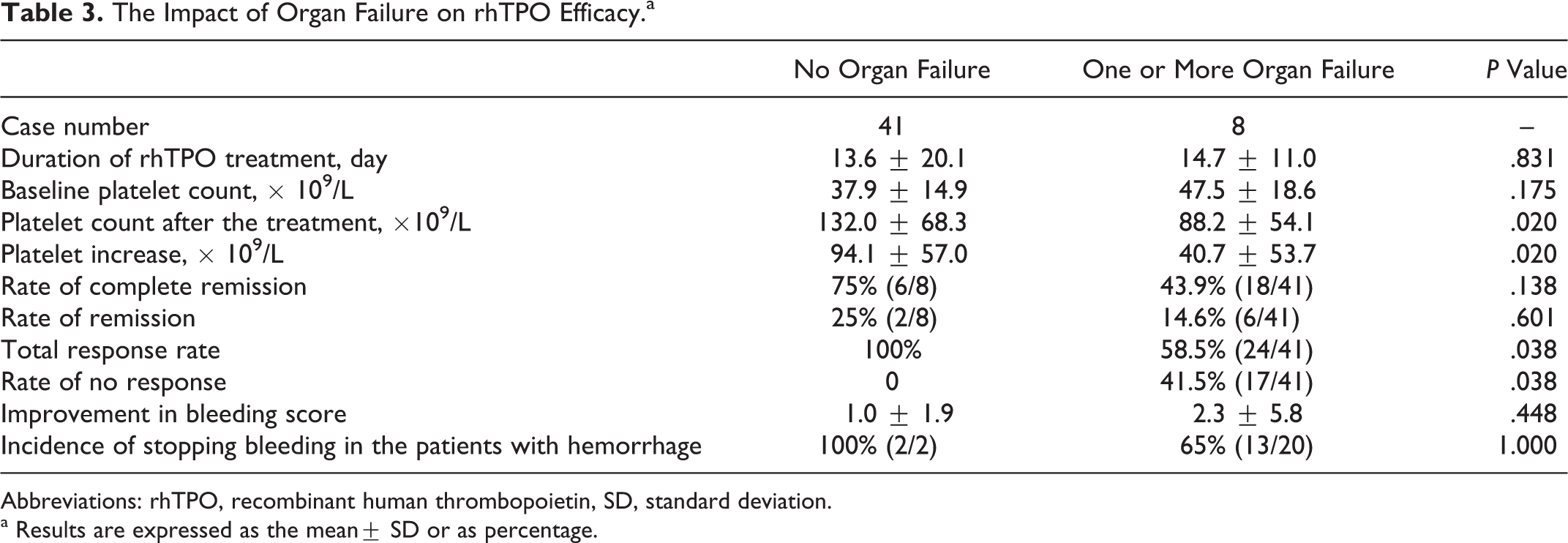
Abbreviations: rhTPO, recombinant human thrombopoietin, SD, standard deviation.
a Results are expressed as the mean± SD or as percentage.
Adverse Events
No dizziness, headache, high blood pressure, bone pain, and fever induced by rhTPO were recorded. There was no significant difference in white blood cell, hemoglobin, BUN, Cr, PT, APPT, Fbg, and ALT, TBIL, DBIL, and GGT prior to and after rhTPO treatment (Table 4, analysis of covariance using factors before treatment as covariance).
The Impact of rhTPO on Hepatic, Renal, and Coagulation Functions.a
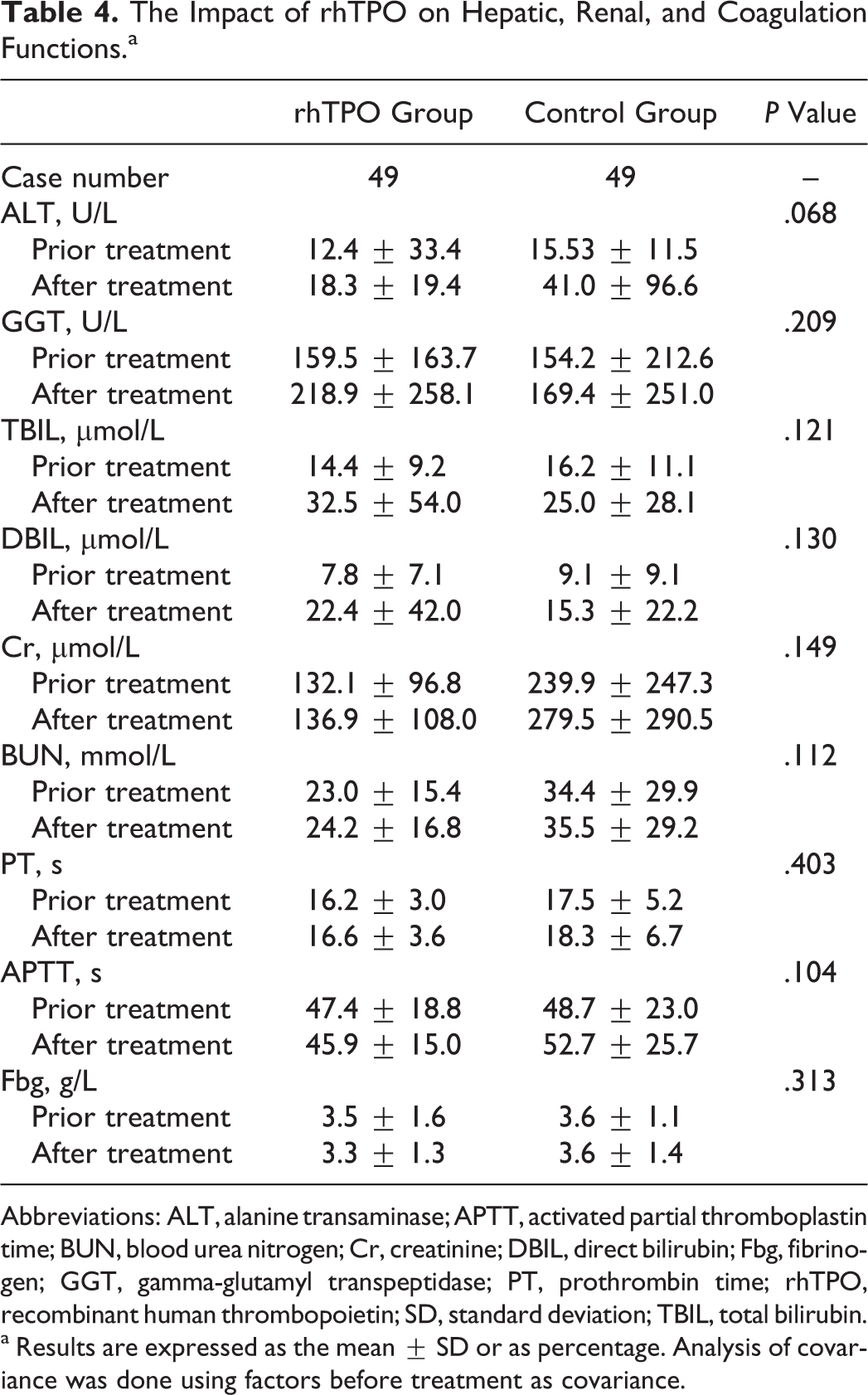
Abbreviations: ALT, alanine transaminase; APTT, activated partial thromboplastin time; BUN, blood urea nitrogen; Cr, creatinine; DBIL, direct bilirubin; Fbg, fibrinogen; GGT, gamma-glutamyl transpeptidase; PT, prothrombin time; rhTPO, recombinant human thrombopoietin; SD, standard deviation; TBIL, total bilirubin.
a Results are expressed as the mean ± SD or as percentage. Analysis of covariance was done using factors before treatment as covariance.
Discussion
Infection could decrease the platelet count via the following mechanisms: (1) increased antiplatelet antibodies 19 ; (2) increased platelet clearance due to increased macrophage colony-stimulating factor and consequently hemophagocytosis 20 ; (3) inhibition of megakaryocyte formation; and (4) increased adhesion of platelet in microcirculation due to the action of endotoxin. 21 –23
The estimated incidence of thrombocytopenia in patients with community-acquired pneumonia is 25%. 2 Sharma et al showed that the incidence of thrombocytopenia is as high as 55% in patients with septic shock. 3 Due to declined organ reserve, elderly patients tend to develop organ failure upon infection. Our previous study in 836 elderly patients with pneumonia demonstrated that thrombocytopenia occurs in a significant proportion of the patients (Jie Lin, unpublished observations, 2012). Specifically, the platelet count was <100 × 109/L in 24.5% of the patients and <50 × 109/L in 10.2% of the patients. The rate of thrombocytopenia in patients with severe pneumonia was 68.7%. Previous study also showed that thrombocytopenia was an independent prognostic factor in patients with infection. Brogly et al observed a significant increase in mortality rates due to severe community-acquired pneumonia, which were 30.8% in patients with a platelet count ≥150 × 109/L, 44.1% in patients with a platelet count of 51 to 149 × 109/L, and 70.7% in patients with a platelet count ≤50 × 109/L.2 More importantly, correction of thrombocytopenia was an independent factor associated with a reduced risk of death.4
Currently, TPO is primarily used to manage chemotherapy-induced thrombocytopenia and ITP. Marketed formulations include pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF), 24 –27 full-length rhTPO, 13,14,28,29 and TPO receptor agonists (Romiplostim, Eltrombopag). 30 –32 The PEG-rHuMGDF was a first-generation TPO molecule. The PEG-rHuMGDF could reduce the extent of thrombocytopenia in nonmyeloablative chemotherapy regimens and improve the platelet counts in patients with ITP or myelodysplastic syndrome. 24 –27 However, subsequent larger safety studies with PEG-rHuMGDF in healthy volunteers demonstrated the appearance of a neutralizing antibody to TPO. So the development of PEG-rHuMGDF was stopped. 33,34 The results of the clinical trials with the first-generation TPO molecules were sufficiently encouraging to prompt a search for nonimmunogenic TPO molecules. The second-generation TPO molecules were developed including romiplostim and eltrombopag that have now been approved by the US Food and Drug Administration for the treatment of ITP. Recombinant human TPO could also induce the production of antibodies. However, the antibodies are temporary and not neutralizing in nature. 16 Since the approval in 2006 in China, the therapeutic effects of rhTPO have been established for chemotherapy-induced thrombocytopenia and ITP. 13,14,16
The results of this retrospective analysis indicated that rhTPO is effective in alleviating infection-associated thrombocytopenia and hemorrhage. The overall response rate was 65.3%. In addition, we considered that rhTPO could improve the quality of life by improvement of hemorrhage, though it did not improve mortality significantly. Since platelet transfusions maybe a confounding variable for the efficacy of rhTPO, analysis of covariance was also done using platelet transfusion as the covariance. But results showed that units of platelet transfusion did not affect platelet count after treatment (P = .38). Further analysis indicated that rhTPO is particularly effective in patients without organ failure. Such findings encourage the early use of rhTPO, preferably prior to the emergence of organ failure. The current study failed to demonstrate an efficacy of rhTPO in terms of thrombocytopenia duration and the amount of platelet transfusion. Furthermore, our study showed rhTPO did not affect hepatic function, renal function, and coagulation. Based on these findings, we recommend using rhTPO inpatients with infection when the platelet count was <50 × 109/L until complete platelet recovery or 14 days if NR is observed.
Circulating TPO level typically decreases with increasing turnover of platelets (eg, ITP) and increases with decreasing platelet production (eg, chemotherapy-induced thrombocytopenia and aplastic anemia). 35,36 A previous study by Wolber et al documented increased level of messenger RNA in the liver upon acute infection. 37 In patients with infection, the 2 opposing conditions (increasing platelet turnover and decreasing platelet production) may coexist, with varying levels and differential causes of thrombocytopenia. Previous studies in neonates suggested that the level of TPO is positively correlated with the severity of the infection. 5,38 We are currently planning a study to examine the level of TPO and its association with thrombocytopenia in senile patients with infection and to explore the mechanisms by which rhTPO alleviate infection-associated thrombocytopenia.
In summary, the current study indicated that rhTPO could alleviate infection-associated thrombocytopenia and hemorrhage in senile population. The therapeutic effects are particularly prominent if rhTPO is used prior to the development of organ failure. Prospective studies are needed to confirm these findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Special Funds for Central Health Projects (No. B2009B115); Innovation Funds in PLA general hospital (No. 12KMM34); and Clinical Research Supportive Funds in PLA general hospital (No. 2012FC-TSYS-1020).
