Abstract
Obesity is associated with an increased risk of vascular thrombotic events. We sought to investigate how obesity and weight loss affect plasma fibrin clot properties. A total of 29 obese patients were studied before and after 3-month low-fat diet. Plasma fibrin clot parameters, including fibrin clot permeation coefficient (K s), the lag phase of the turbidity curve, clot lysis time (t 50%), maximum rate of increase in D-dimer levels, and maximum D-dimer concentrations, were determined. Low-fat diet resulted in the reduction of body weight (P < .0001), body mass index (P < .0001), fat mass (P < .0001), total cholesterol (P < .0001), low-density lipoprotein cholesterol (P = .0005), triglycerides (P = .008), and plasminogen activator inhibitor 1 (P = .02), but not in fibrinogen or C-reactive protein. The only change in fibrin clot variables was shorter t 50% (P = .02). Baseline t 50%, but not posttreatment, correlated with waist circumference (r = .44, p = .02). This study demonstrates that weight loss in obese people can increase the efficiency of fibrin clot lysis.
Introduction
Obesity increases the risk of cardiovascular disease (CVD) and venous thromboembolism (VTE) 1.5 to 2.5 times. 1,2 Compelling evidence indicates that obesity is associated with a prothrombotic state and a low-grade inflammatory state, characterized by enhanced production of several inflammatory cytokines, in particular interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α). 3,4
Adipose tissue is not an inert energy storage site but an active organ with endocrine and metabolic functions and thus could play a causal role in the prothrombotic state observed in obesity. Adipose tissue may contribute to altered hemostasis directly, by synthesis and secretion of tissue factor, plasminogen activator inhibitor 1 (PAI-1), and possibly thrombin-activatable fibrinolysis inhibitor (TAFI) and indirectly, by the effect of leptin and adiponectin on platelet activation.
5,6
Adipose tissue influences the liver function by the release of inflammatory cytokines such as TNF-
Impaired fibrinolysis is an abnormality often observed in obese patients. 8 –11 The PAI-1, a major physiological inhibitor of fibrinolysis, decreases fibrinolytic activity by acting as a rapid inhibitor of both tissue-type plasminogen activator (tPA) and urokinase-type plasminogen activator. 7 Excess of adipose tissue increases the production of PAI-1. 12,13 Plasma PAI-1 antigen and activity levels positively correlate with the body mass index (BMI) and the waist circumference. 14
Visceral fat produces more PAI-1 than subcutaneous or femoral fat. 15 Sam and coworkers showed that visceral adipose tissue is positively associated with PAI-1, even after adjustment of BMI. 16 Giltay et al shown that increased levels of PAI-1 are particularly evident in patients with abdominal obesity. 17 Treatment of obesity using diet, lifestyle intervention, or bariatric surgery leads to a decrease in PAI-1 levels. 18,19 McGill et al demonstrated that obese diabetic patients show 3-fold elevated concentrations of PAI-1 but no significant difference in plasma concentration of tPA compared to healthy lean individuals. 10
Fibrin formation is the final stage of blood coagulation. There is a correlation between fibrin structure and the efficiency of fibrinolysis. 20,21 Clot formation, morphology, function, and susceptibility to lysis are influenced by genetic and environmental factors affecting blood coagulation and fibrinolysis. 22,23 Clot lysis is affected by various factors including fibrin formation, characteristics of tPA/plasminogen, local concentrations of fibrinolysis inhibitors, and the structure of the clot. 22,24 Carter et al demonstrated that clot density and clot lysis time (CLT) increased progressively with increasing number of metabolic syndrome components. 25 However, the effect of weight loss on fibrin parameters has not been investigated in that study.
Different diets, including high or low protein, carbohydrates, and fat, show a similar final effect on weight. 26 Loss of weight depends on the diet type, time duration, and patients’ activity, but it is the change in nutritional habits that is of crucial importance. 26 –28 Standard diets have limited efficacy, because decrease by 5% to 10% of initial body weight beyond 6 months is the average long-term effect. 29 A large percentage decrease in weight is usually short lasting.
To our knowledge, there have been no studies regarding the effects of a large weight loss on plasma fibrin clot formation and degradation. We sought to test the hypothesis that weight loss results in improved fibrin clot properties assessed ex vivo in plasma.
Materials and Methods
We enrolled 29 apparently healthy, obese patients (17 female and 12 male, age range 20-72 years) with no medication treatment in outpatient obesity clinic in Krakow, Poland. The inclusion criterion was obesity defined as BMI ≥ 30 kg/m2. The exclusion criteria were as follows: previous history of coronary artery disease, cancer, diabetes, hyper- or hypothyroidism, chronic kidney disease with glomerular filtration rate < 60 mL/min, increased asparagine aminotransferase (AST) or alanine aminotransferase (ALT) level (≥10% above the upper limit of reference values), heart failure, arterial hypertension requiring pharmacological therapy, acute infection, autoimmune disorders, alcohol and drug abuse, and pregnancy. Patients were advised against the use of nonsteroidal anti-inflammatory drugs. Four visits were scheduled during the study. During all visits, each patient completed questionnaires regarding eating and living habits and questionnaires regarding his/her mood. A comprehensive medical history was collected, and the following examinations were carried out: medical examination and determination of the body mass, weight, body fat mass (FM), nonfat tissue mass, and basal metabolic rate (BMR) using bioelectrical impedance analysis technology (Tanita, Tokyo, Japan) and the anthropometric measures—waist and hip circumference and BMI ratios. The study was approved by regional medical board ethics committee. All patients signed informed consent to participate in the study.
Patients followed a 3-month low-fat diet, containing about 18% of fat. The diet and reduction in calories were individually established based on the BMR, sex, patient’s physical condition, and activity. Nutrient goals for the patients were low fat, average protein diet (18% fat, 15% protein, and 65% carbohydrate). The goal for dietary fiber was 20 g/d minimum and for dietary cholesterol was 150 mg/1000 kcal. Carbohydrate-rich foods were used having a lower glycemic index. Estimated energy needs were calculated from BMR and physical activity. Each patient’s diet prescription represented a 750 to 1000 kcal/d deficit depending on the initial body mass and physical activity. The individual recommendations were 1200 to 2000 kcal/d. Patients had precisely described menu and were asked to weigh the foodstuffs. Patients had moderate physical activity, that is, 60-minute march twice a week.
We have arbitrarily chosen weight reduction in 2.5% of the initial value as the cutoff for inclusion to the final analysis assuming that low-fat diet if followed should provide a certain weight loss.
Laboratory Investigations
Fasting blood samples were drawn between 08.00 and 10.00
The PAI-1 antigen and tPA antigen were determined in plasma with an enzyme-linked immunosorbent assay (Diagnostica Stago, Asnieres, France).
Plasma fibrin clot variables were determined in duplicate by technicians blinded to the origin of samples (intra-assay and interassay coefficients of variation, <8%), as described.
30
Fibrin clot permeation using a pressure-driven system as described previously,
31
with the calculation of a permeation coefficient (K
s), which indicates the pore size. Lower K
s values indicate reduced permeability. Plasma samples were diluted 1:1 with 0.05 mol/L Tris-HCl, 0.15 mol/L NaCl, pH 7.4, and addition of 1 U/mL human thrombin (Sigma) and 15 mmol/L calcium chloride to plasma-initiated polymerization. Absorbance was read at 405 nm for 15 minutes with a Perkin-Elmer Lambda 4B spectrophotometer (Molecular Devices Corp, Menlo Park, California). The lag phase of the turbidity curve, which reflects the time required for initial protofibril formation, and maximum absorbance at plateau reached by all individuals (ΔAbs), which reflects the number of protofibrils per fiber, were recorded. Each sample was analyzed twice. The interassay and intraassay coefficients of variation were 5.8% to 7.1%, respectively.
32
CLT is defined as the time required for a 50% decrease in clot turbidity at 405 nm (t
50
%), induced by 1 μg/mL recombinant tPA (Boerhinger Ingelheim, Germany). Maximum rate of increase in D-dimer levels (American Diagnostica, Stamford) and maximum D-dimer concentrations measured in a buffer containing 0.2 μmol/L recombinant tPA (Boerhinger Ingelheim) percolating through fibrin clots.
Statistical Analysis
Normality of distribution was tested with the Shapiro-Wilk test. Data were shown as mean ± standard deviation or median and interquartile range as appropriate. Comparisons between the groups were carried out by Student t test for paired samples for parametric variables or Wilcoxon-matched pairs test otherwise. Independence of categorical variables was verified by the chi-square test. The correlation analysis was performed with Pearson test for normally distributed data or Spearman test otherwise. The univariate and multivariate linear regression analysis with forced entry of PAI-1 as the standardizing variable was used to eliminate the impact of this potential confounder on the correlation between t 50% and waist circumference. A P value < .05 was considered statistically significant.
Results
We studied 29 apparently healthy obese patients, mean age 38.9 (range: 20-72 years), 17 women and 12 men, with BMI ranging from 30 to 48.1 kg/m2. Baseline characteristics of the study participants are presented in Table 1.
Comparison of the Study Participants Before and After a 3-Month Low-Fat Diet.
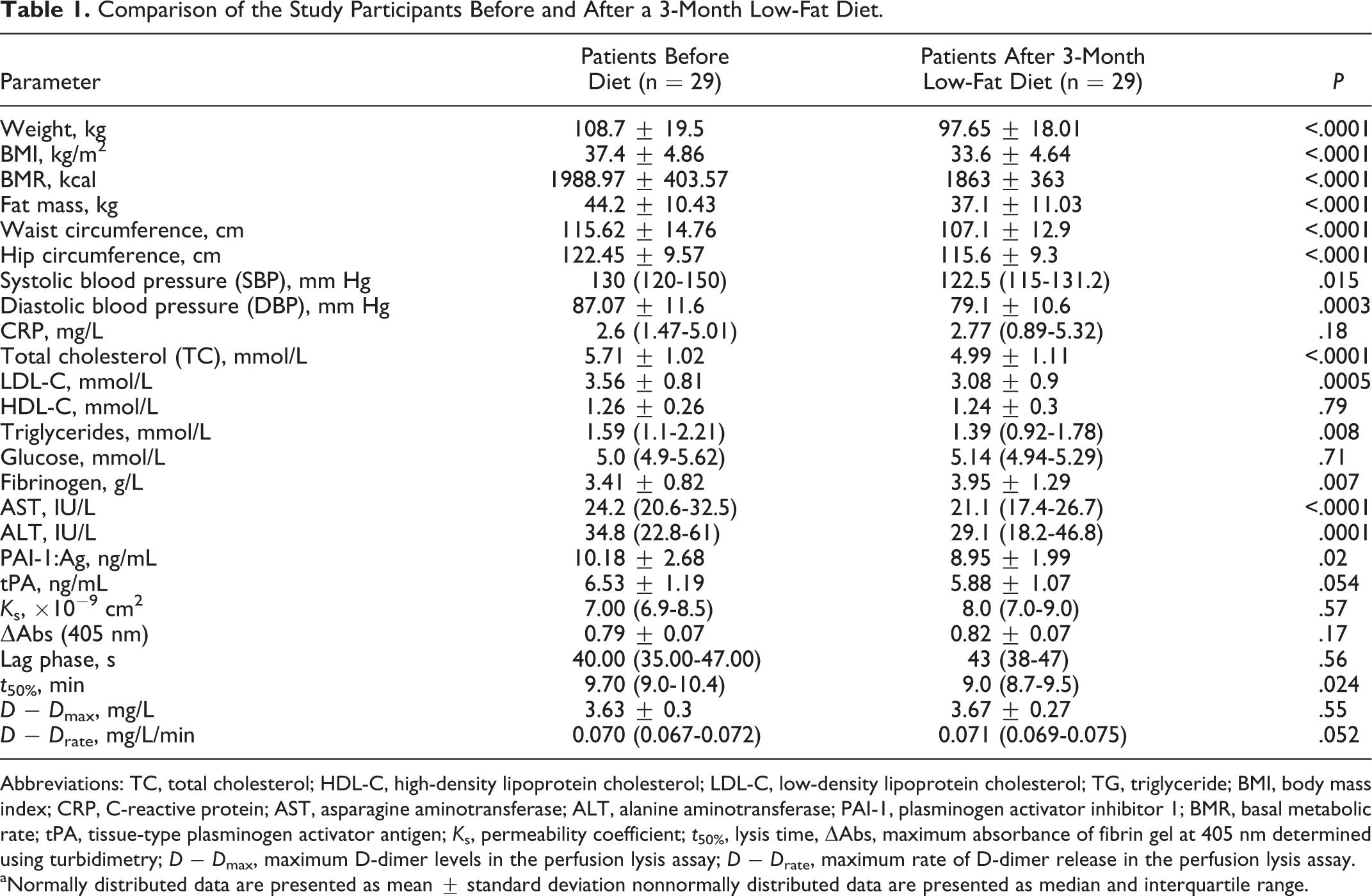
Abbreviations: TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglyceride; BMI, body mass index; CRP, C-reactive protein; AST, asparagine aminotransferase; ALT, alanine aminotransferase; PAI-1, plasminogen activator inhibitor 1; BMR, basal metabolic rate; tPA, tissue-type plasminogen activator antigen; K s, permeability coefficient; t 50%, lysis time, ΔAbs, maximum absorbance of fibrin gel at 405 nm determined using turbidimetry; D − D max, maximum D-dimer levels in the perfusion lysis assay; D − D rate, maximum rate of D-dimer release in the perfusion lysis assay.
aNormally distributed data are presented as mean ± standard deviation nonnormally distributed data are presented as median and interquartile range.
After 3 months of low-fat diet, the median reduction in weight was 9.1 kg (range 2-35.7 kg). We recorded 10% reduction in weight, 10% in BMI, 16% in FM, 7.4% in waist circumference, and 5.6% in hip circumference. Moreover, there were 5.8% decreases in systolic and 9% in diastolic blood pressure, 12.6% in total cholesterol, 13.4% in low-density lipoprotein cholesterol, 15.3% in asparagine, and 16.3% in ALT levels. The level of fibrinogen increased after the follow-up by 15.8%.
After low-fat diet, PAI-1 level was reduced by 12% when compared to the baseline, and a tendency to lower tPA (mean reduction by 7.5%) was also observed.
The only plasma fibrin clot parameter that was altered following a 3-month low-fat diet was t 50%. There was a small, but significant, reduction by 7.2%. There were no significant posttreatment changes in other fibrin clot parameters.
The complete comparison of all analyzed parameters before and after 3 months of low-fat diet is presented in Table 1.
The initial but not posttreatment t 50% correlated with waist circumference even when adjusted for PAI-1 level as a potential confounder (r2 = .19, P = .01 and r2 = .183, P = .008, respectively). Other significant correlations between the parameters of plasma fibrin clot and anthropometric and laboratory results in the baseline and after 3 months of dietary intervention are presented in Table 2.
Correlations Between Clinical Parameters, Laboratory Results, and Fibrin Clot Properties (n = 29).
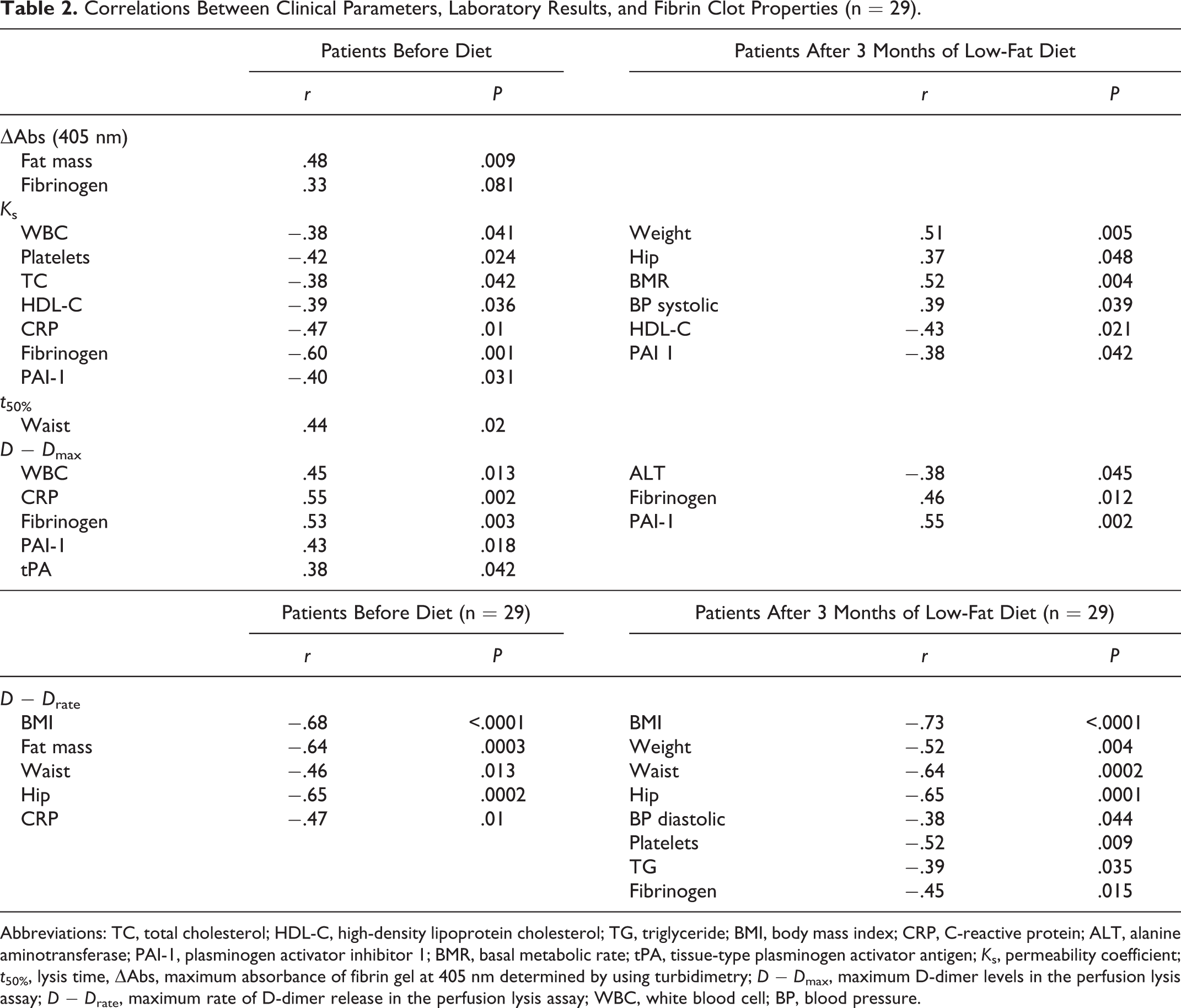

Abbreviations: TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglyceride; BMI, body mass index; CRP, C-reactive protein; ALT, alanine aminotransferase; PAI-1, plasminogen activator inhibitor 1; BMR, basal metabolic rate; tPA, tissue-type plasminogen activator antigen; K s, permeability coefficient; t 50%, lysis time, ΔAbs, maximum absorbance of fibrin gel at 405 nm determined by using turbidimetry; D − D max, maximum D-dimer levels in the perfusion lysis assay; D − D rate, maximum rate of D-dimer release in the perfusion lysis assay; WBC, white blood cell; BP, blood pressure.
We did not find any correlations between the degree of improvement of CLT and the change in PAI-1 levels or waist circumference before and after the treatment.
Discussion
This study is the first to show that in apparently healthy obese patients, weight reduction following a 3-month low-fat diet leads to improved fibrin clot lysis and does not alter clot permeability or other variables describing clot structure and function. It appears that clot lysis can be favorably altered after as few as 3-months’ low-fat diet, while other parameters of fibrin clots are relatively “resistant” to such a short dietary intervention. Given the fact that prolonged CLT has been observed in patients with deep vein thrombosis and premature CVDs, 33,34 it might be hypothesized that weight reduction may decrease thromboembolic risk in the arterial and venous circulation in part through improved velocity of fibrin clot degradation.
Our study provides the evidence that the reduction in adipose tissue, most likely in relation to lower PAI-1 levels, accelerates clot lysis. In the current study on obese patients, loss of weight following low-fat diet resulted in significant reductions in t 50%, PAI-1, and tPA. Decrease in PAI-1 level, which was associated with FM loss, and tPA—the marker of endothelial injury, can also modulate fibrin clot properties. 21 –23 In our study, the initial CLT correlated with waist but not with weight or BMI. This may suggest the association with abdominal-type obesity. Using other experimental approaches to determine CLT, PAI-1 has remained the strongest determinant of CLT and explained the majority of the variance in CLT, followed by TAFI levels, 35 and PAI-1 accounted for up to 13% of variance in turbimetric lysis variables. 25
Majority of the previous studies on the fibrin clot properties concerned the population with comorbidities related to obesity, for example, diabetes or CVDs. In our study, we demonstrate that in obese individuals without these comorbidities, reduction in weight leads to changes in CLT what augments the importance of weight reduction in primary cardiovascular prevention.
It is unclear whether other types of diet (high protein, low carbohydrate, and high in fat or vegetarian) influence clot properties to a similar extent. It might be speculated that the final loss of weight and change in patients’ nutritional habits may contribute to the alterations in fibrin properties.
Various diets, calories restrictions, and time of observation result in different rates of the weight loss. 26,36 –38 Most of the weight loss occurs in the rapid phase within the first 6 months of diet, and after 12 months usually patients slowly regain body weight. Positive alterations in fibrinolysis are likely to disappear with the subsequent weight gain. In our study, the majority of patients after 3 months still had BMI more than 30 kg/m2 and high FM quantity despite weight loss. It is probable that larger weight reduction will result in more pronounced changes in plasma fibrin clot characteristics.
Elevated serum levels of fibrinogen are usually observed in obese patients. Fibrinogen is an acute phase reactant, and its elevated levels may, at least in part, reflect a proinflammatory state closely associated with insulin resistance and obesity. 39 Studies on changes in fibrinogen levels during weight loss have brought conflicting results. Pardina et al reported a significant decrease in fibrinogen level in severely obese patients after bariatric surgery. 19 Garanty-Bogacka et al have demonstrated that the successful 6-month lifestyle intervention leads to the reduction in fibrinogen levels in obese children. 40 Other studies on nondiabetic obese individuals demonstrate that moderate weight loss does not alter fibrinogen levels. 41 –43 An increase in fibrinogen levels observed in our study may be, at last in part, associated with a small number of participants and relatively short observation.
Our study has several limitations. The number of patients is relatively small, and analysis of correlations must be interpreted with caution. The observation period was only 3 months, leading to a rapid weight loss in a large proportion of the patients studied. Long-term effects of low-fat diet were not evaluated in the present study. We did not measure other variables that might affect fibrin clot properties, for example, TAFI or plasminogen. Microscopic images of plasma clots were not analyzed. However, given no difference in fibrinogen and permeability, significant posttreatment changes in fiber thickness or architecture are unlikely.
In conclusion, in addition to benefits resulting from lipid profile changes, blood pressure reduction, physical fitness, and improved quality of life, weight loss in obese people can increase the efficiency of fibrin clot lysis. Our findings suggest a novel mechanism underlying beneficial effects of weight reduction in thrombotic risk. It might be speculated that faster clot lysis associated with weight loss may decrease VTE and cardiovascular risk in obese individuals.
Footnotes
Acknowledgments
We thank Anna Walach and Jerzy Walach, Renata Kostrz, Małgorzata Morawska and Joanna Tkaczewska from Obesity Treatment Center in Krakow for their help during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by a grant of the Jagiellonian University.
