Abstract
Protein Z (PZ) deficiency may induce bleeding as well as thrombosis. The aim of our study was to estimate the concentration of PZ in patients with acute leukemia. Plasma levels of PZ were determined in 76 patients with newly diagnosed acute leukemia ([AML], n = 50; acute lymphoblastic leukemia [ALL], n = 26) and 62 healthy participants. In the patients, mean plasma concentrations of PZ were statistically lower than in healthy individuals: AML (1.24 ± 0.11 μg/mL vs 1.58 ± 0.05 μg/mL P = .01) and ALL (1.19 ± 0.16 μg/mL vs 1.58 ± 0.05 μg/mL P = .01). Levels of PZ below the fifth percentile (0.873 μg/mL) of normal value distribution in control participants were found in 30% of patients with AML and ALL and in 3% of controls (P < .0001). In this AML subgroup, we found statistically significant correlation between episodes of bleeding and PZ level (P = .01). There was no such correlation in ALL group. The results suggest that PZ can be a cofactor associated with an increased bleeding tendency in patients with AML.
Keywords
Introduction
Protein Z (PZ), a vitamin-K-dependent protein, was identified by Prowse and Esnouf in bovine plasma in 1977 1 and was isolated from human plasma by Broze in 1984. 2 The organization of the PZ gene, at chromosome 13q34, and molecule structure of PZ are homologous with other vitamin-K-dependent factors like VII, IX, X, protein C. 2–5 However, PZ differs from these other vitamin-K-dependent proteins, in that, there is lack of the His and Ser residues, which are necessary for serine protease activity. 3,5–7 Protein Z and protein Z-dependent protease inhibitor (ZPI) form a complex in plasma. In pooled normal plasma, which contains excess ZPI, all the PZs appear to be bound to ZPI. 8
Reported mean concentrations of PZ in adult plasma varied from 1.2 to 2.9 μg/mL, with an estimated plasma half-life of about 2.5 days. The concentration is reduced during oral anticoagulation with cumarin derivatives. 9
The potential role of alterations of PZ levels in the pathogenesis of disorders of coagulation has been investigated in several studies and has revealed conflicting results. Many of the clinical studies reported low PZ levels to be correlated with the occurrence and progression of several types of ischemic vascular diseases, whereas some studies did not observe that correlation or reported an association between low levels of PZ and bleeding tendency. 10 The occurrence of various coagulation abnormalities in acute leukemia is well established, 11,12 but there are no data assessing the role of PZ in hemostatic disorder in hematology cancers, including acute leukemia.
The aim of this study was to estimate the concentration of PZ in acute leukemia and to assess a correlation between PZ levels and the bleeding tendency. Additionally, we evaluated the influence of chemotherapy on PZ concentration.
Patients and Methods
Seventy-six patients (40 females and 36 males, aged 28-60 years) with acute leukemia: 50 with acute nonpromyelocytic myeloblastic leukemia (AML) and 26 with acute lymphoblastic leukemia (ALL) were studied. The diagnosis of acute leukemia was essentially based on World Health Organization (WHO) guidelines of conventional morphocytochemical criteria and immunophenototyping.
13
Patients were not under the oral anticoagulant treatment and had normal liver protein synthesis function. None of the patients had thromboembolic episode at the time of diagnosis nor clinical and laboratory evidence of disseminated intravascular coagulation. In patients with low hemoglobin concentration, amount of citrate was adjusted to hematocrit before blood sampling. Venous blood samples were drawn with butterfly cannula with a minimum stasis after at least 4 hours of fat fasting and before 10
Results
The significantly lower concentrations of PZ were observed in both groups of AML 1.24 ± 0.11 μg/mL and ALL 1.19 ± 0.16 μg/mL patients than in healthy individuals 1.58 ± 0.05 μg/mL, P = .01 and P = .01, respectively. There were no statistically significant differences in mean concentration of PZ between patients with AML and ALL (Figure 1).
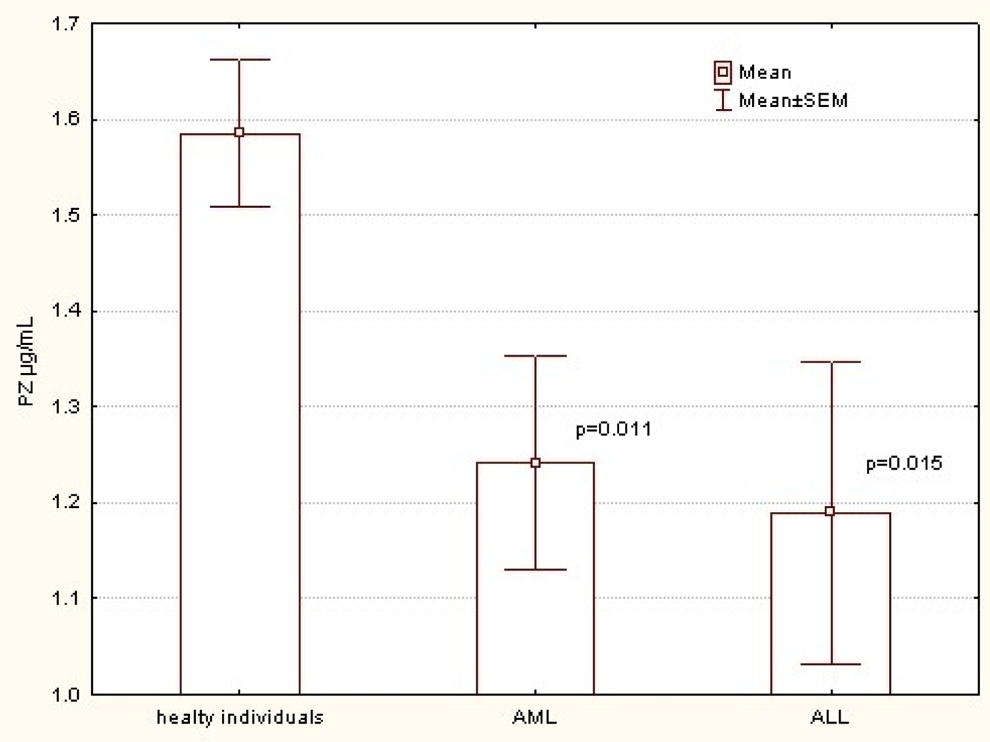
Mean concentration of PZ between controls and patients with AML and ALL. PZ indicates Protein Z; AML, acute myeloid leukemia; ALL, acute lymphoblastic leukemia.
Levels of PZ below the fifth percentile (0.873 μg/mL) of normal value distribution in control participants were found in 30% of patients with AML and ALL and in 3% of controls (P < .0001). The study revealed increased concentration of PZ during the treatment of patients with ALL, with statistically significant difference after treatment (P = .05). No differences in PZ concentrations during the treatment in patients with AML were observed (Table 1).
Mean concentration of PZ in acute leukemia patients before and after start of the treatment
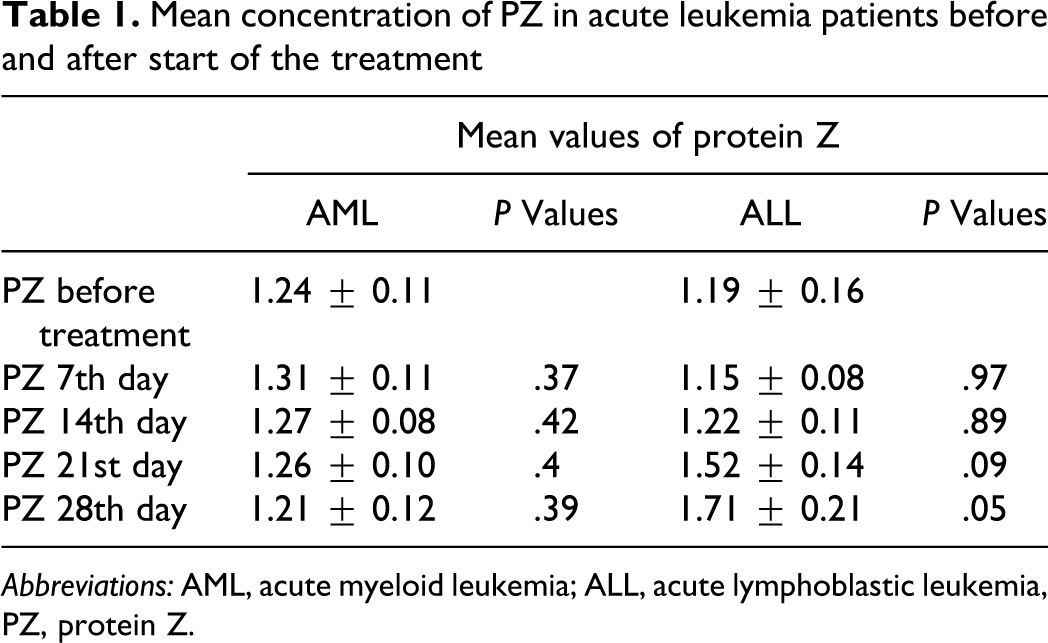
Abbreviations: AML, acute myeloid leukemia; ALL, acute lymphoblastic leukemia, PZ, protein Z.
All patients with acute leukemia, having a level of platelets under 10 × 10 3 /μL, received platelet concentrates, as a prophylaxis of the bleeding. Due to the stable concentration of PZ in patients with AML, we performed the correlation calculation using the mean concentration of PZ during the treatment and the episode of the bleeding or thrombotic episodes. We counted the bleeding when the number of platelets was above 30 × 103/μL, and the patient presented active bleeding (without the pinpoint hemorrhages on skin and mucous membranes). The APTT and PT were in normal range in all studied individuals at the time of PZ measurements. In the group of patients with AML with PZ concentration below fifth percentile of normal value distribution in control participants, we found statistically significant correlation between PZ level and episodes of bleeding (P = .01). There was no correlation between the concentration of PZ and the bleeding episodes in group of patients with ALL. One of the patients with AML and 2 patients with ALL had deep venous thrombosis (DVT) during the treatment. Concentrations of PZ in these individuals were not significantly different from the rest of the patients with leukemia. Patient with DVT had no evidence of pulmonary embolism (PE).
In patients with AML, 29 (58%) achieved the complete remission, 12 (24%) partial remission, 5 (10%) no remission, and 4 (8%) patients died during the treatment from infectious complication. There was no significant correlation between the PZ concentration at day 28 and the stage of the diseases. In patients with ALL, 24 (92.4%) achieved the complete remission, 1 (3.8%) partial, and 1 (3.8%) no remission. There was no significant correlation between PZ concentration at day 28 and the stage of diseases after the treatment, as well.
Discussion
Patients with cancer are at high risk of both thrombosis and hemorrhage. 16 Venous and arterial thromboembolism (VTE and ATE) are more frequently observed in patients with solid tumors, whereas hemorrhage, usually attributed to thrombocytopenia and uncompensated disseminated intravascular coagulation (DIC), occurs more often in patients with hematologic malignancies, particularly acute leukemia. 17 Chemotherapy, as well as common risk factors for thromboembolism, further complicated the natural history of patients with cancer, including acute leukemia. 18 The molecular pathogenesis of thrombosis and DIC has been studied in several models of human malignancy. 19,20 Major determinants of the pathogenesis of clotting activation in hematologic malignancies include tumor cell-derived procoagulant, fibrinolytic and proteolytic factors, and inflammatory cytokines, 16 cytotoxic therapies, 21 and infectious complications. 22
In patients with thrombosis, the deficiency of antithrombotic factors was investigated in several studies. Tsumita et al reported a case of AML with multiple thrombophlebitis due to hereditary protein C deficiency. 23 Another study concerning AML with DVT as a result of antithrombin (AT) deficiency has also been reported. 24 Significantly lower protein C antigen and activity has been reported by Troy et al in active patients with AML in comparison to patients in remission or in normal controls. In contrast, patients with ALL, both with active disease and in remission had normal concentration and activity of protein C. 25 Levels of total protein S were normal in all patients with leukemia, but free protein S was significantly lower in patients with active AML than in patients in remission or controls. Levels of free protein S were normal in patients with ALL. The evidence which connects PZ to blood coagulation is its structural similarity to 6 other vitamin-K-dependent plasma proteins and the unique sensitivity to chronic cumarin therapy. 9
The role of PZ in the pathogenesis of hemostatic disorders in humans remains to be established. From investigations performed so far, no clear link was evidenced between PZ concentrations and bleeding or thrombosis tendency. However, a significant association was found between low PZ levels and arterial vascular diseases, pregnancy complications, and venous thromboembolic diseases in the last published meta-analysis by Sofi et al. 26 On the other hand, it has been suggested that PZ deficiency is associated with hemorrhagic disorders, perhaps related to capillary fragility. 27 Kemkes-Matthes et al studied 36 individuals with the bleeding disorders of unknown etiology and correlated the bleeding with a decreased level of PZ. In additional studies, Greten et al used prothrombin complex concentrate, containing PZ, to prevent perioperative hemorrhage in patients whose level of PZ was under 600 µg/L and who had an undiagnosed history of bleeding. 28 It should be noted that 10% of healthy individuals, blood donors, have PZ levels of less than 50%, with no trace of bleeding tendency. 29 Furthermore, PZ-null mice have normal bleeding times and do not have a hemorrhagic phenotype. 30 In the study of Ravi et al, it was suggested that PZ can be a weak cofactor associated with an increased bleeding tendency. 31 On the other hand, a relationship between venous thrombosis and PZ plasma levels has been excluded and revealed the opposite results. The studies indicated that only very low levels of PZ, arbitrarily considered as lower than 5.0 percentile of controls, have been associated with a moderate thrombotic risk. 32 These results suggest that low PZ levels may be a mild prothrombotic risk factor, but its procoagulant consequences might increase when combined with additional risk factors.
There is only one study in which the concentration of PZ in acute leukemia was assessed. Undar et al studied only 16 patients (11 with AML and 5 with ALL) and found no differences between patients and healthy individuals. 33 In our analysis, we found statistically significant lower concentration of PZ in groups of patients with ALL and AML in comparison to healthy individuals. The study revealed an increase in concentration of PZ during the treatment of patients with ALL to the concentration observed in healthy individuals. No differences in PZ concentrations during the treatment in patients with AML were observed.
In patients with AML, the correlation calculation was done based on the mean concentration of PZ during the treatment and the number of bleeding and the thrombotic episodes. The study revealed in the group of AML patients with PZ concentration below fifth percentile of normal value distribution in control participants, statistically significant correlation between PZ level and the number of bleeding episodes. There was no correlation between the concentration of PZ and the bleeding episodes in the group of patients with ALL. We did not find any differences in PZ concentration in AML group of patients with thrombosis.
In the ALL group, 2 patients had venous thrombosis at the time of treatment. The influence of
In conclusion, our results suggest that PZ deficiency can be a cofactor associated with an increased bleeding tendency in patients with acute myeloid leukemia. Further studies are required to determine the hemostatic role of the ZP/ZPI complex and the possible hemostatic dysfunction consequences of its abnormalities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
