Abstract
Objectives:
We investigated the relationship between coagulation assessed by thromboelastography (TEG) and myocardial damage in ST-segment elevation myocardial infarction (STEMI).
Methods:
We measured platelet activity with TEG-maximum amplitude (TEG-MA) in 233 patients undergoing urgent percutaneous coronary intervention (PCI). Infarct size and myocardial salvage index were evaluated using cardiac magnetic resonance, and the relation of these parameters to posttreatment coagulation was examined retrospectively. Adverse events were adjudicated and related to the coagulation status during the index event.
Results:
Hypercoagulation was found in 82 (35.2%) patients and was neither correlated to infarct size nor correlated to myocardial salvage index (P = .28 and .65, respectively) or clinical adverse events. Patients who experienced an adverse event during follow-up had a slightly higher TEG-MA value than patients with an event-free follow-up, but this was not statistically significant (68.1 vs 67.3, P = .44).
Conclusions:
The TEG-MA does not appear to be a sensitive predictor of reperfusion success and prognosis in urgent PCI for STEMI.
Keywords
Introduction
Acute reperfusion is known to reduce myocardial damage and thus improve outcome, which is why international guidelines recommend timely primary percutaneous coronary intervention (pPCI) in patients with ST-segment elevation myocardial infarction (STEMI). 1
In addition to revascularization, dual antiplatelet therapy with aspirin (ASA) and a P2Y12 ADP-receptor inhibitor such as clopidogrel is the gold standard in the medical management of patients undergoing PCI. 2,3 Interindividual variability in the platelet response to clopidogrel has been linked to an increased risk of early stent thrombosis. 4,5 This phenomenon has led to an increased focus on platelet hyperactivity using platelet function tests such as light transmittance aggregometry, vasodilator-stimulated phosphoprotein, multiplate whole-blood platelet aggregometry tests, and thromboelastography (TEG). 6 The TEG is a whole-blood viscoelastic assay that has been widely used in the assessment of coagulation and fibrinolysis. 7 –11 Gurbel evaluated a population of patients undergoing elective PCI with stent implantation and found that preprocedural TEG maximal clot strength was significantly associated with postprocedural ischemic events. 12
In patients with trauma and patients undergoing surgery, hemorrhage remains an important source of preventable deaths, and thus, early assessment of coagulopathy with point-of-care testing enables the prevention of these complications by guiding transfusion. 13,14 Reversely, TEG-hypercoagulable patients have a higher risk of thromboembolic complications and death after coronary artery bypass surgery. 15 A similar use of TEG to estimate the risk of thrombosis and the prognosis in patients with cardiovascular disease may prove to be useful. Ischemic heart disease has been linked to pronounced platelet activation and impaired hemostasis in which hypercoagulation compromises microcirculation. Indeed, patients with atherosclerosis exhibit increased platelet activity, a parameter that can be rapidly assessed by TEG. 16,17 Therefore, TEG may prove to be a useful tool to assess hemostasis and to predict the size of myocardial infarcts and to subsequently predict prognosis in patients with an acute myocardial infarction. 18
The degree of myocardial necrosis following a myocardial infarction can be measured as infarct size using cardiac magnetic resonance (CMR), 19 which also enables the evaluation of the degree of myocardial salvage, an indicator of successful reperfusion. 20
Distal embolization during PCI is linked to poor prognosis 21 and is believed to be related to a hypercoagulative state with the generation of new thrombi. 22 For this reason, it could be expected that patients in hypercoagulant state exhibit a poorer outcome after primary PCI expressed larger infarctions and a lower myocardial salvage index despite undergoing what is considered to be an optimal revascularization strategy with contemporary adjunctive medical therapy.
The aim of the present study was (1) to investigate the ability of TEG clot strength to estimate the overall coagulation status and (2) to establish a possible relationship between hypercoagulability as evaluated by TEG maximal clot strength 15 and prognosis as well as final infarct size and salvage index as assessed by CMR in patients with acute myocardial infarction.
Methods
Patients and Study Design
The following study is a substudy of a single-center randomized, double-blind, placebo-controlled trial performed from January to December 2009, involving 333 patients with STEMI referred for urgent reperfusion therapy. 23,24 Patients were enrolled in the main study after giving informed consent, and the study was performed according to the Helsinki Declaration of Good Clinical Practice.
Patients >18 years, presenting with chest pain and electrocardiogram (ECG) changes corresponding to STEMI, were referred to the catheterization laboratory for immediate coronary angiography, if they presented within 12 hours of onset of symptoms. Main reasons for exclusion were unconsciousness, cardiogenic shock, previous myocardial infarction or coronary artery bypass surgery, stent thrombosis, and presence of coronary lesions—other than the culprit lesion—with a stenosis >70% and thrombolysis in myocardial infarction flowgrade >1 on the angiography. Inclusion and exclusion criteria have been described in detail previously. 23
The TEG was performed in a subgroup of patients (n = 233) that constitutes the study population of the present substudy. Baseline characteristics of our study population did not differ from those of the main population (Table 1).
Baseline Characteristics.a
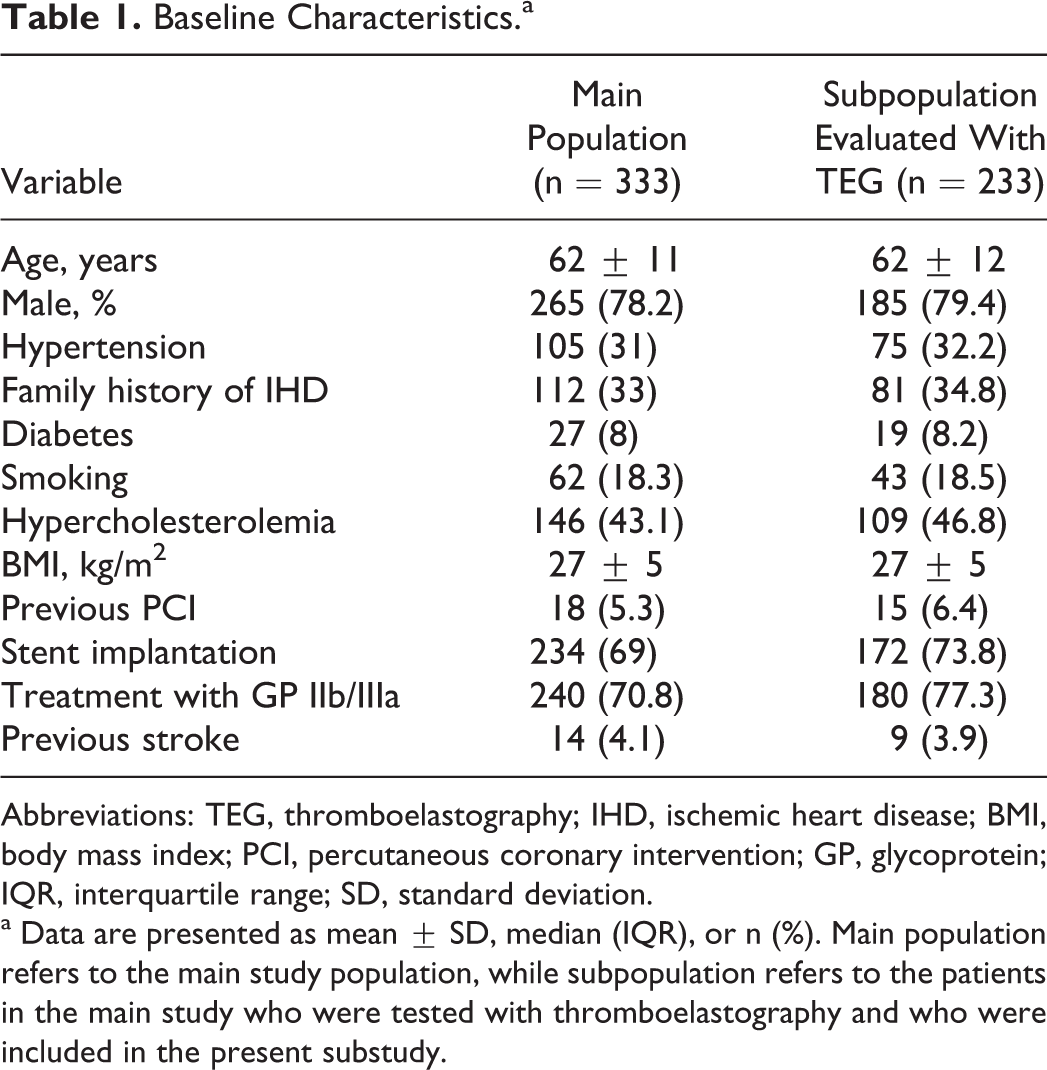
Abbreviations: TEG, thromboelastography; IHD, ischemic heart disease; BMI, body mass index; PCI, percutaneous coronary intervention; GP, glycoprotein; IQR, interquartile range; SD, standard deviation.
a Data are presented as mean ± SD, median (IQR), or n (%). Main population refers to the main study population, while subpopulation refers to the patients in the main study who were tested with thromboelastography and who were included in the present substudy.
Study Protocol
Patients eligible for urgent PCI were pretreated with ASA (300 mg orally or 500 mg intravenously), heparin 10 000 U intravenously, and clopidogrel 600 mg bolus orally as soon as the diagnosis of possible STEMI had been established.
Following identification of the culprit lesion by angiography, the patients were randomized to placebo or adjuvant exenatide—a glucagon-like peptide 1 analogue—prior to intervention and according to the purpose of the main study. The choice of stent and the administration of glycoprotein IIb/IIIa inhibitors (GPIs) were left to the discretion of the operator. However, at the time of the study, abciximab and eptifibatide were routinely used at our center unless contraindicated. After PCI, all patients received a daily maintenance dose of 75 mg of clopidogrel for 12 months and lifelong 75 mg ASA daily.
Thromboelastography
Standard kaolin-activated TEG was used to assess maximal clot strength and detect hypercoagulation, using 3.2% citrated whole blood in a TEG 5000 Hemostasis Analyzer System (Haemonetics Corp, Massachusetts). The samples were analyzed within 2 hours from collecting the blood, as recommended by the manufacturer.
Blood was drawn from the study patients during initial hospitalization within the first 3 days after pPCI for STEMI and at least 12 hours after the procedure. Four values can be determined by standard kaolin-activated TEG: (1) the R value (3-8 minutes), also called reaction time, indicates the time until the initial thrombin generation; (2) the K value (1-3 minutes) represents the speed of clot formation, while (3) the angle (55°-78°) is the tangent of the curve as K is reached. The parameter (4) maximum amplitude (MA [51-69 mm]) is often used to assess hemostasis and bleeding risk, and we mainly focused on this parameter as it reflects the maximum clot strength and hence the degree of platelet activity. 25,26
Hypercoagulation was defined as TEG-MA values above 69 mm, a cutoff used in previous cardiac and noncardiac studies. 27 Measuring hypercoagulation on anticoagulant-naïve patients was not possible, since patients had already been given antithrombotic treatment with ASA, heparin, and clopidogrel prior to admission and also received GPI during the index procedure and before the study sampling.
Infarct Size and Salvage Index by CMR
In the absence of contraindications, a CMR scan was performed at a median (interquartile range) of 2 (1-3) days after the STEMI, and CMR was repeated after a median of 89 (80-93) days as described previously. 24 Patients who did not undergo the initial scan were still considered for the second scan. The myocardial area at risk was assessed on the first CMR scan as edema, using a T2-weighted short tau inversion recovery sequence. 20,28,29 Multiple slices in the short-axis image plan were acquired to cover the entire left ventricle. The endocardial and epicardial borders were manually traced in each short-axis image, and the left ventricle mass was calculated without incorporation of the papillary muscles and slow flowing blood in the trabeculae. The area at risk was defined as the hyperintense area on T2-weighted images. A myocardial area was regarded as hyperintense when the signal intensity was higher than 2 standard deviations of the signal intensity in the normal myocardium. Hypointense areas within the area at risk were considered a part of the area at risk. The salvage index was calculated as follows: (area at risk [g] − infarct size [g])/area at risk (g). On the second CMR examination, delayed enhancement images were obtained to determine the final infarct size using a standard ECG-triggered inversion recovery sequence. 30 Images were obtained approximately 10 minutes after the intravenous injection of 0.1 mmol/kg body weight gadolinium-diethylenetriamine pentaacetic acid (Gadovist; Bayer Schering, Berlin, Germany). In a single slice, the inversion time was adjusted to null the signal from the normal myocardium. Multiple slices in the short-axis image plan were acquired to cover the entire left ventricle. The final infarct size was assessed using the freely available software Segment v1.8. 31
Myocardial salvage index is considered as an indication of successful reperfusion representing jeopardized myocardium “saved” by revascularization. 32 The calculation of the salvage index requires 2 CMR scans, which is why the number (n) of patients where this value is given in Table 2 is lower than the number (n) of patients where the final infarct size was calculated.
Correlation Between the Degree of Coagulation Assessed by TEG and Myocardial Damage Assessed by CMR.a

Abbreviations: CMR, cardiac magnetic resonance; LV, left ventricle; MA maximum amplitude; TEG, thromboelastography.
aFinal infarct size: in % of the left ventricular mass in g. Hypercoagulants have a TEG-MA value > 69 U.
Clinical Study End points
Clinical outcome was assessed as all-cause and cardiovascular mortality, reinfarction, stroke, and stent thrombosis during the entire follow-up period. Major adverse cardiac events (MACE) were also evaluated within the first year of follow-up. Mortality events, stent thrombosis, and reinfarction were registered according to the previous definitions. 33 Clinical events were adjudicated by an independent committee unaware of the treatment assignment.
Statistics
Statistical analysis was performed using the SPSS software version 18 (SPSS Inc, Chicago, Illinois). Two-sided P values <.05 were considered statistically significant. Continuous data were evaluated for normal distribution and analyzed using the T test or Mann-Whitney, as appropriate. Categorical variables were compared using the chi-square and Fischer tests. The distribution of clinical events is illustrated by the Kaplan-Meier method and was compared by the log-rank test for univariate analysis.
Results
Baseline Characteristics
A total of 233 patients were evaluated with TEG during the inhospital period. The TEG-MA values ranged from 50 to 85 mm. According to the defined cutoff value, 82 (35.2%) patients were hypercoagulant. Of these, 80.5% received IIb/IIIa inhibitors prior to intervention, compared to 76% of the normocoagulants. The baseline characteristics comparing the 2 coagulation groups are described in Table 3. It has been shown that platelet aggregation determined by platelet function tests is influenced by platelet count. 34 Only 4 (1.9%) patients exhibited thrombocytosis (2 in each coagulation group) while 1 (0.5%) exhibited thrombocytopenia. However, we did find a significantly lower mean platelet count of 233 × 109/L (±50) in the normocoagulant group compared to a mean platelet count of 272 × 109/L (±69) in the hypercoagulant group (P < .0001). Thus, although thrombocytosis and thrombocytopenia were not associated with the overall coagulation status assessed with TEG, hypercoagulants exhibited a higher numerical platelet count (Table 3).
Comparison of Baseline and Procedure-Related Variables Between Categories of Coagulation (N = 233).a
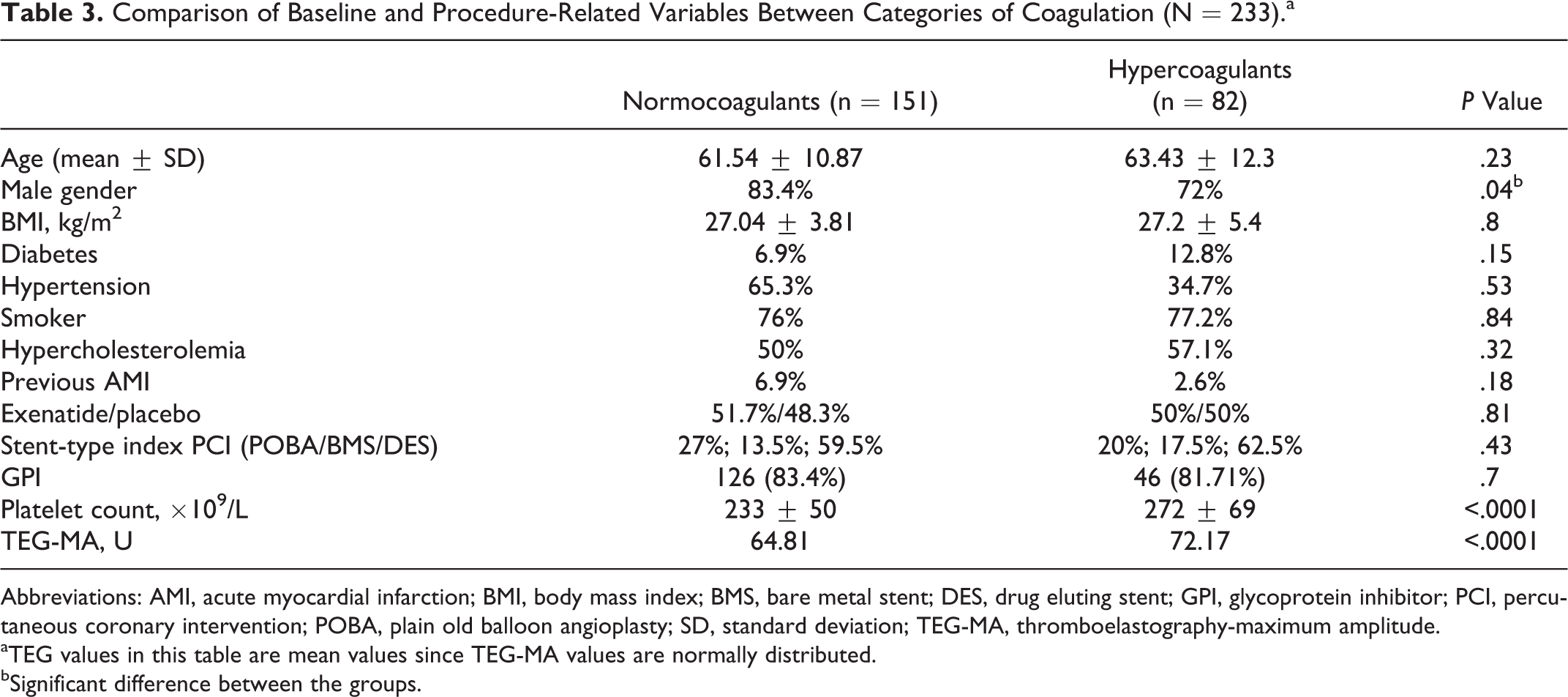
Abbreviations: AMI, acute myocardial infarction; BMI, body mass index; BMS, bare metal stent; DES, drug eluting stent; GPI, glycoprotein inhibitor; PCI, percutaneous coronary intervention; POBA, plain old balloon angioplasty; SD, standard deviation; TEG-MA, thromboelastography-maximum amplitude.
aTEG values in this table are mean values since TEG-MA values are normally distributed.
bSignificant difference between the groups.
Cardiac Magnetic Resonance Evaluated Infarct Size and Salvage Index
Myocardial salvage index and final infarct size were found to be of equal magnitude among both the subgroups (Table 2). Neither a linear relationship between TEG-MA and final infarct size nor an inverted linear relation with the salvage index was found.
Clinical End points
Patients were followed for a median of 831 days, and the primary end point was reached in a total of 14 (6%) patients. We found a larger proportion of all-cause deaths and patients with stroke in the hypercoagulant group compared to the normocoagulant group (7.3% vs 4% and 2.5% vs 1.3%, respectively), but the difference was not statistically significant (Table 4).
Rates of Clinical Events According to Patients’ Coagulation Status (During Follow-Up).
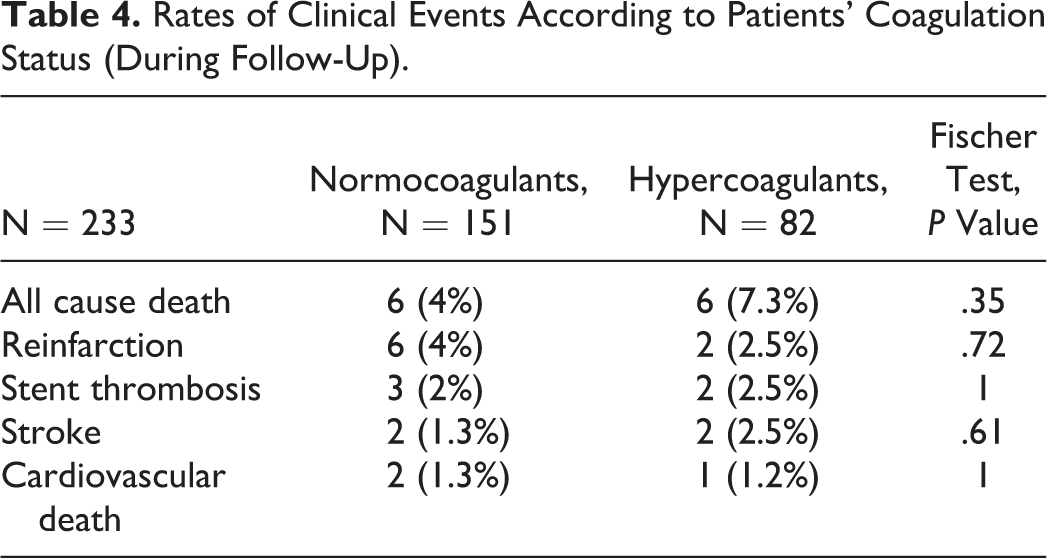
Although the incidence of MACE was numerically higher in the hyper- versus normocoagulant group (7.4% vs 5.3%, respectively), there was no statistically significant difference between the groups (P = .568). Throughout the entire follow-up period, stent thrombosis occurred in 2.5% of the hypercoagulants versus 2% of the normocoagulants, but the difference was not significant (P = 1.0). The median TEG-MA value in the group of patients experiencing an adverse event was numerically higher than that found in the event-free patients (68.1 vs 67.3 mm, P = .44), but no significant difference was detected. Furthermore, events occurred at similar time points in the 2groups (P = .63). Figure 1 shows the Kaplan-Meier curve of clinical events during the entire follow-up period.
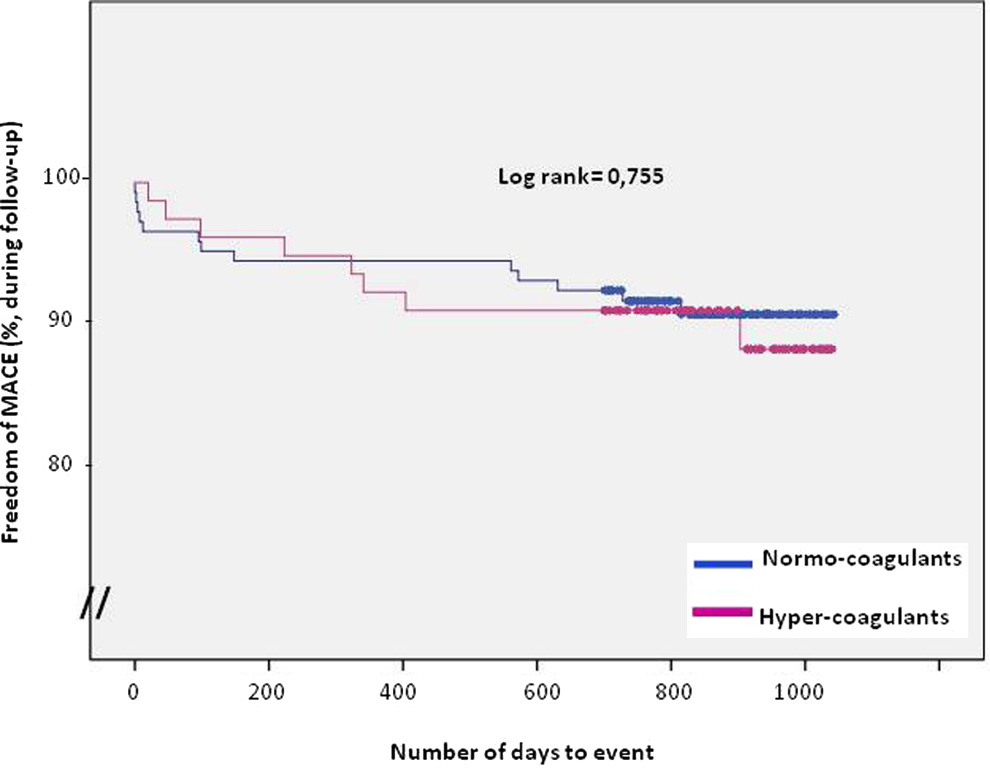
Kaplan-Meier estimates of survival and freedom of MACE in patients with ST-segment elevation myocardial infarction (STEMI) according to their coagulation status assessed by thromboelastography. Hypercoagulants are defined by thromboelastography-maximum Ampl-itude (TEG-MA) values >69 mm. MACE indicates major adverse cardiac event (reinfarction, stroke, stent thrombosis, and death).
Discussion
The present study reports biochemical, functional, and clinical data in patients undergoing pPCI with the main findings: (1) following grouping of patients based on coagulability, we found a similar myocardial salvage index and final infarct size among patients in hyper- and normocoagulant states; (2) the combined primary clinical end point was reached in a total of 14 (6%) patients, without differences between groups, although there was a larger proportion of all-cause deaths and stroke in the hypercoagulant group. Throughout the entire follow-up period, stent thrombosis occurred with equally low rates in both the groups. Thus, the expected relationship between clot strength as assessed by TEG and myocardial damage could not be verified in this population of patients undergoing pPCI for STEMI. Previously, Gurbel and colleagues investigated 84 patients undergoing PCI with stenting and found that preprocedural TEG clot strength was significantly associated with postprocedural ischemic events. 35 Patients in the highest quartile of clot strength more often exhibited an ischemic event than patients in the low MA quartile (48% vs 13%, P = .02). This is in alignment with a recent study by Rafiq and colleagues who investigated 200 patients undergoing coronary artery bypass grafting and found that a presurgical clot strength above 69 mm was associated with a significantly increased incidence of the combined MI, stroke, and all-cause mortality (17.2% vs 6.6%, P = .019). The association of increased incidence of thromboembolic events in patients with a TEG-MA above the normal reference of 69 mm has also been reported in major noncardiac surgery 27 and trauma, 36 and consequently, the findings presented in our study are somewhat surprising. A potential explanation may be that the blood was sampled after the administration of the GPI, since this treatment greatly inhibits the ability of platelets to form clots and may cause thrombocyte dysfunction. Consequently, the patients’ native platelet reactivity, and hence clot strength, could not be established. This is further illustrated by the newly developed TEG assay functional fibrinogen, where the GPI Abciximab is added to prevent the platelets from participating in clot formation, thus enabling evaluation of the sole fibrin contribution to the clot formation. 37 An in vitro study performed by Bailey et al in 2005 also showed that platelet function could not be adequately interpreted by TEG on a population sample treated with IIb/IIIa inhibitors. 38
Although not statistically significant, in hypercoagulants the percentage of deaths for all causes and strokes is almost double than
that of normocoagulants. There is a possibility that larger studies can lead to different results. Contrary to platelet function tests such as Multiplate and Verify-Now, TEG seems to have a greater prognostic value in a population of antiplatelet-naïve patients than in a population pretreated with antiplatelet drugs such as GPI and also seems to translate hemostasis more than a potential risk of thrombosis. In order to evaluate the degree of platelet activation and estimate the true platelet function, TEG platelet mapping is preferable to TEG. The TEG-platelet mapping, a newer TEG assay, is designed to evaluate the platelet inhibition following administration of antiplatelet therapy and could have been beneficial in this setting, but this assay was not available in our institution at the time of study initiation.
Study Limitations
The sample size was small, and the rate of clinical events was very low, thus the study did not carry statistical power to enable reliable conclusions with regard to clinical end points such as cardiovascular death, stroke, and stent thrombosis. We found a greater proportion of all-cause deaths and stroke in the hypercoagulant group, and larger studies may be able to show a significant correlation between coagulation status and clinical outcome. Blood samples were drawn and analyzed during day time only, implying a certain variability related to the time of the index procedure and the administration of antithrombotic medication. A more rigid sample schedule could have possibly altered the results. Larger studies are therefore needed in order to further investigate the possible relationship between coagulation as assessed by TEG and clinical outcome as well as infarct parameters in patients with STEMI.
In the current local guidelines, it is imperative to administer antithrombotic drugs to patients with STEMI as soon as the diagnosis is suspected; therefore, these drugs are often administered either on the field by the paramedics or at the referring hospital prior to transferring the patient to our institution for PCI. This prevented the evaluation of platelet function prior to inhibition of platelet aggregation by antithrombotic drugs.
The coadministration of several very potent antithrombotic drugs prior to TEG testing may have blurred the results of platelet function, since the sensitivity of TEG is not sufficient to discriminate among the different platelet-inhibiting drugs.
Conclusions
On-treatment hypercoagulation determined by the TEG parameter MA was neither related to final infarct size nor to myocardial salvage or clinical end points in a population of patients with STEMI undergoing urgent PCI, although this study was not powered to determine a potential relationship to clinical end points.
Larger observational studies are needed to establish a true possible relationship between hypercoagulation assessed by TEG and clinical end points. Furthermore, more sensitive assays may be required in order to (1) discriminate between several antithrombotic drugs and (2) obtain a reliable evaluation of the degree of platelet hyperactivity in a population of acute myocardial infarction.
Footnotes
Acknowledgments
The authors thank the staff at the Section of Transfusion Medicine at Rigshospitalet for skillful assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Jaascha Foundation, the Arvid Nilsson Foundation and the Danish Heart Association for financial support.
