Abstract
Patients with iliac deep vein thrombosis (DVT) have a poor prognosis and high incidence of postthrombotic syndrome (PTS). We evaluated the effect of low-molecular-weight heparin (LMWH; tinzaparin) versus usual care (tinzaparin plus warfarin for ≥12 weeks at home) in the development of PTS according to DVT location (iliac/noniliac) by retrospective analysis of the Home-LITE cohort (480 patients with proximal DVT). Patients with iliac DVT had an overall odds ratio of 0.53 (95% confidence interval [CI] 0.33, 0.83; P = .0079) for PTS (including ulcer data) in favor of tinzaparin. Patients with noniliac DVT had a similar odds ratio (0.79 [95% CI 0.67, 0.93], P = .0046) to that reported in the overall Home-LITE population (0.76 [95% CI 0.66, 0.89], P = .0004; including ulcer data), both in favor of tinzaparin. Long-term LMWH may be a suitable alternative for the prevention of PTS in patients with iliac DVT who are unlikely to undergo invasive thrombolysis.
Introduction
Deep vein thrombosis (DVT) of the lower extremities may occur in the proximal and/or distal veins of the leg. Apart from the risk of larger emboli, proximal venous obstruction is likely to have a more severe effect on patency and draining of the entire extremity and is therefore associated with a worse prognosis compared with distal thrombotic events. Prognosis is further exacerbated in patients with proximal DVT localized to the iliac veins, which is present in approximately 25% of the patients with proximal DVT 1 and is associated with a greater incidence of postthrombotic syndrome (PTS), venous ulcers, recurrent venous thromboembolism (VTE), and reduced quality of life. 1 –3 The PTS of the leg is a persistent condition that develops in 20% to 50% of the patients within 1 to 2 years after a symptomatic DVT event and is mainly characterized by limb swelling, pain, discomfort on walking, skin discoloration, and venous ulceration. 1,4,5
In patients with proximal DVT, the American College of Chest Physicians (ACCP) recommend the use of low-molecular-weight heparin (LMWH) and a vitamin K antagonist to prevent recurrent DVT and pulmonary embolism. 4 There are no published guidelines specifically for the management of patients with iliac DVT. To help prevent the onset of PTS, compression stockings or an intermittent compression device may be considered. Patients with iliac DVT have the greatest risk of developing PTS 1 and may benefit from a more aggressive treatment approach such as catheter-directed thrombolysis (CDT). 2,6 –8
The Home-LITE study reported a significantly lower rate of PTS signs/symptoms at 12 weeks in patients with proximal DVT treated with long-term tinzaparin compared with tinzaparin plus oral warfarin. 9 The aim of the subanalysis presented here was to assess whether the effect of long-term tinzaparin compared with tinzaparin plus oral warfarin (usual care) differed according to the anatomical localization of the thrombus in the Home-LITE cohort.
Methods
Home-LITE methods have been reported elsewhere. 9 Briefly, patients with acute proximal DVT (popliteal, femoral, or iliac vein thrombosis) were randomized to receive 175 IU/kg tinzaparin subcutaneously once daily for 12 weeks or “usual care,” defined as tinzaparin for ≥5 days plus oral warfarin, commenced on day 1, international normalized ratio adjusted, and continued for ≥12 weeks. Patients received the first injection in a clinic and subsequent treatment was administered at home. Patients requiring thrombolysis or thrombectomy were excluded from the study.
The primary end point of Home-LITE was the occurrence of recurrent VTE, and the primary safety outcome measure was the occurrence of bleeding during the treatment period. Secondary end points included mortality rate, patients’ self-reported treatment satisfaction, symptoms of PTS, and self-reported incidence of venous leg ulcers. The incidence of PTS at 12 weeks was measured using a questionnaire, in which patients replied “yes” or “no” with respect to 8 symptoms or signs (listed in Figure 1) whose presence in combination is commonly used to diagnose the presence and severity of PTS. 10 In an additional question, patients were also asked whether an ulcer was present in the skin above their ankle on the affected leg, and all cases of patient-reported venous ulcers were confirmed by a health care professional.
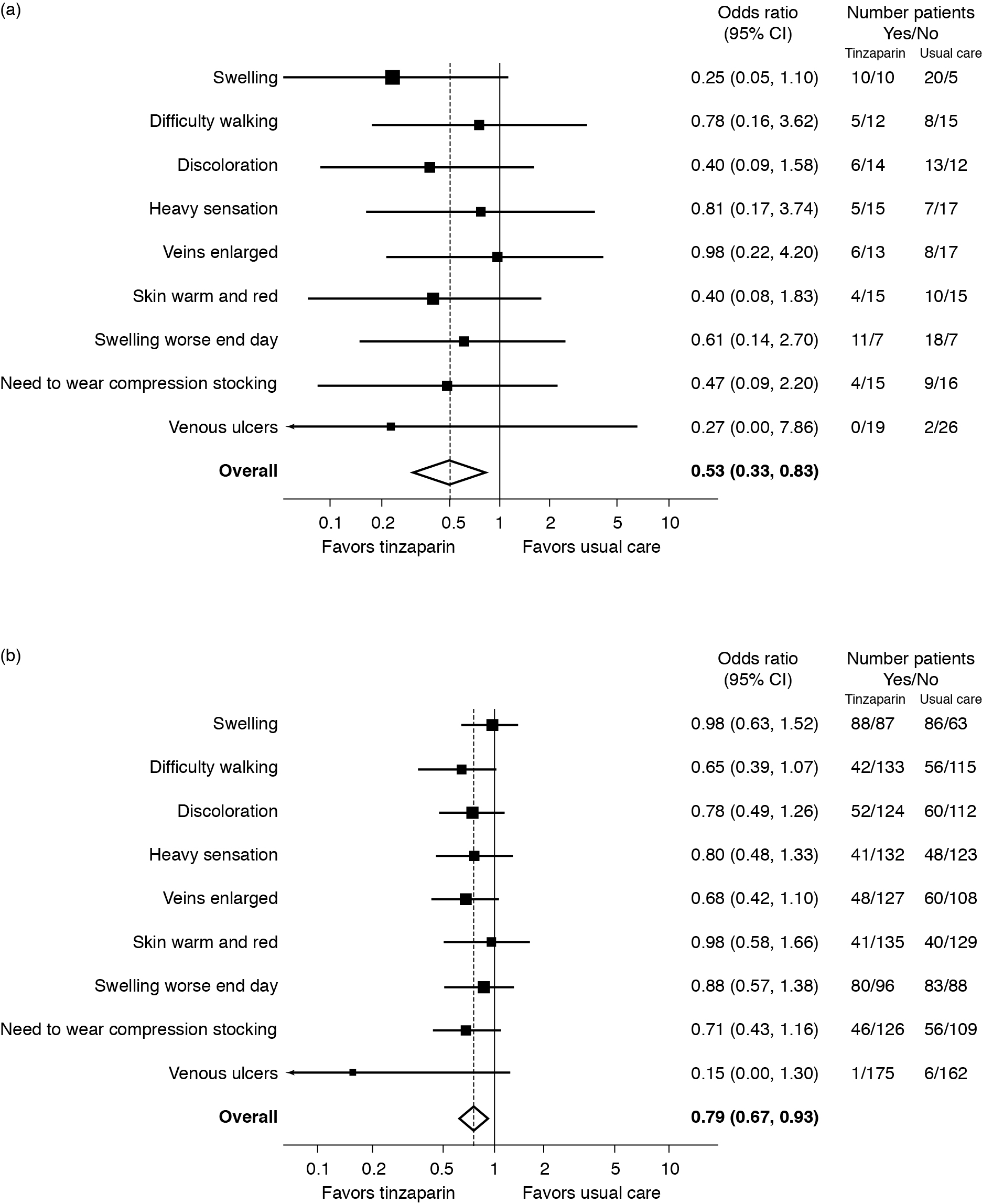
Symptoms/signs of the postthrombotic syndrome following 12 weeks of therapy. Patients replied “yes” or “no” concerning the presence of the symptoms/signs listed. Odds ratios <1 favor tinzaparin (ie, symptom/sign less likely to be present). (a) Patients with iliac involvement. Overall odds ratio = 0.53 (P = .0079); (b) patients without iliac involvement. Overall odds ratio = 0.79 (P = .0046).
The occurrence and localization of VTE was assessed using vein compressibility and standard Doppler ultrasonography of the lower extremities, as described by Carpenter et al. 11 This technique has been extensively used in studies of venous thrombosis. 3,12 –15 The VTE localization data were acquired prospectively before treatment initiation and documented within the radiologists’ ultrasound imaging report. Localized iliac involvement was defined as any VTE that occurred in the iliac segments; any VTE occurring outside the iliac segments was defined as noniliac. For the purpose of this subanalysis, baseline and PTS data were analyzed according to the VTE localization, among the originally randomized patient treatment groups.
The statistical methods used for the Home-LITE study have been described previously. 9 For the present analysis, between-treatment statistical power could not be sought because this was a retrospective analysis and treatment groups were not originally stratified in terms of VTE localization.
Results
Study Populations
Clinical characteristics of the overall population and patients with iliac/noniliac VTE are shown in Table 1. Of the 240 patients who received tinzaparin during the Home-LITE study, 24 (10%) had iliac involvement and 211 (87.9%) had no iliac involvement. Similarly, of the 240 patients who received usual care, 28 (11.6%) had iliac involvement and 207 (86.2%) had no iliac involvement. The iliac and noniliac groups were broadly comparable at stratification, although the former included a greater number of older patients and had a higher incidence of cancer. In the iliac group, patients receiving usual care or tinzaparin also had broadly similar baseline characteristics.
Clinical Baseline Characteristics of Patients Treated With Long-Term Tinzaparin or Usual Care: Patients With/Without Iliac Involvement.
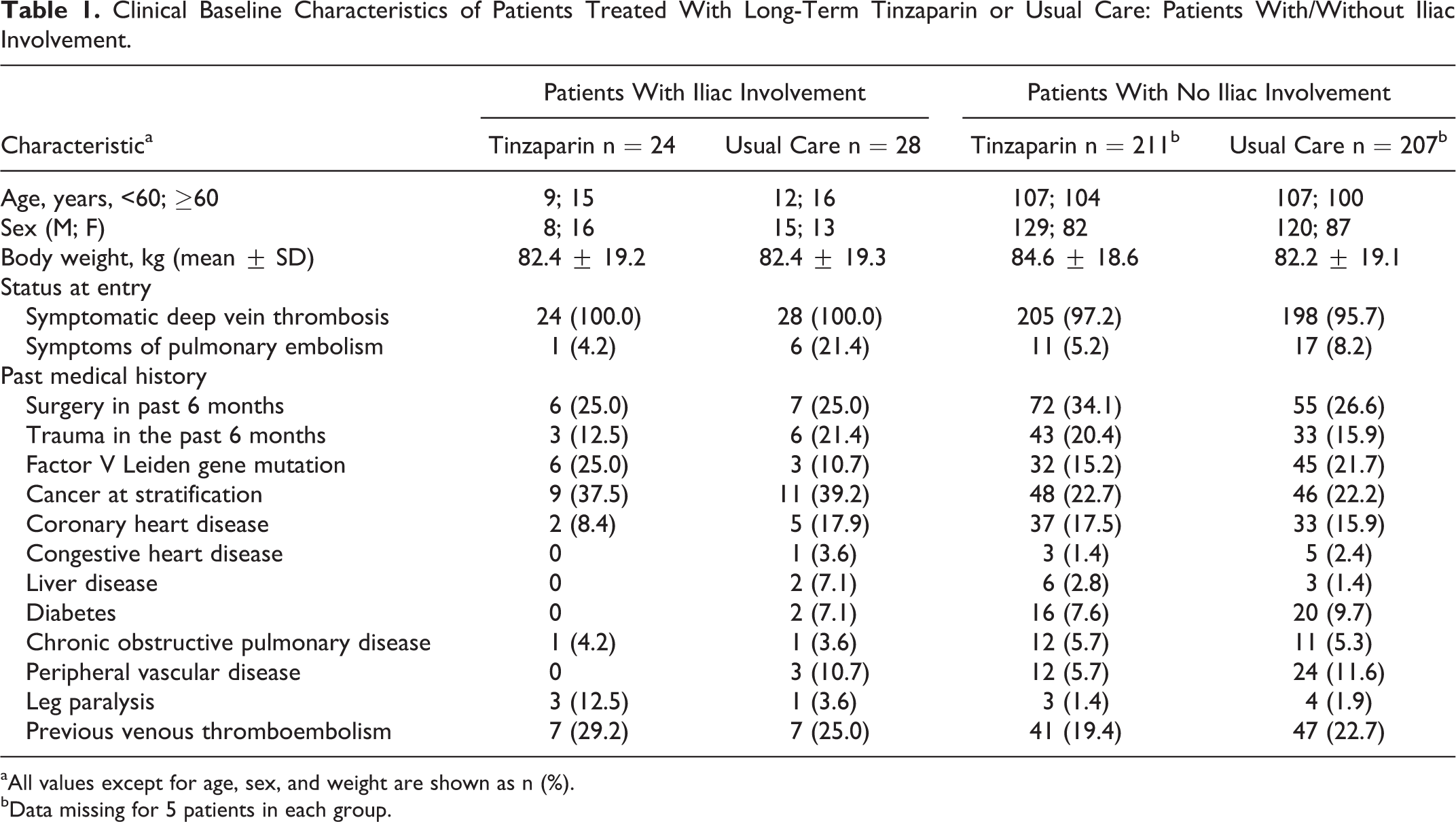
aAll values except for age, sex, and weight are shown as n (%).
bData missing for 5 patients in each group.
Recurrent VTE, Deaths, and Bleeding
The incidence of recurrent VTE, death, and bleeding after 12 weeks in the noniliac and iliac groups was broadly comparable with the overall Home-LITE population; however, 4 (7.7%) deaths occurred in the iliac group (n = 52) compared with 14 (3.3%) deaths in the noniliac group (n = 418). In patients with iliac involvement, the incidence of recurrent VTE and major bleeding was similar between treatment groups, although minor bleeding occurred in 4 (16.7%) patients in the tinzaparin group and in 2 (7.1%) patients in the usual care group (Table 2).
Outcomes of Patients Treated With Long-Term Tinzaparin or Usual Care: Patients With/Without Iliac Involvement.
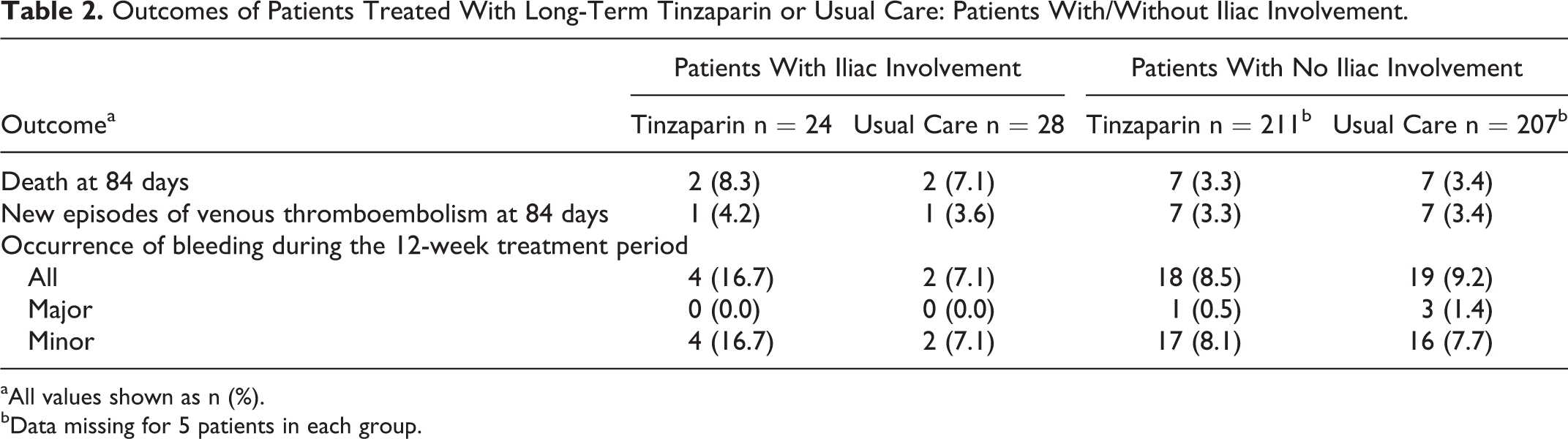
aAll values shown as n (%).
bData missing for 5 patients in each group.
Incidence of PTS and Venous Ulcers
Home treatment with tinzaparin or usual care in the overall Home-LITE population after 12 weeks resulted in a significantly lower incidence of PTS in the tinzaparin group (overall odds ratio of 0.76 [95% confidence interval [CI] 0.66, 0.89], P = .0004). For the purposes of this study, we included the incidence of leg ulcers in the comparison between tinzaparin and usual care.
In the current subanalysis, a similar odds ratio for the presence of PTS and venous leg ulcers was seen in patients without iliac involvement (overall odds ratio of 0.79 [95% CI 0.67, 0.93], P = .0046 in favor of tinzaparin). In patients with iliac involvement, the odds ratio for PTS and venous leg ulcers was significantly lower, 0.53 in favor of tinzaparin (95% CI 0.33, 0.83, P = .0079; Figure 1).
In the iliac group, 2 (7.7%) venous leg ulcers occurred in patients receiving usual care, and none occurred in patients receiving tinzaparin, while in the noniliac group, 6 (3.7%) venous leg ulcers occurred in patients receiving usual care and 1 (0.6%) occurred in patients receiving tinzaparin.
Discussion
In this subanalysis of the Home-LITE study, treatment with LMWH for ≥12 weeks rather than usual care in patients with iliac DVT reduced the overall risk of PTS even more than in patients with noniliac DVT.
Examination of specific subgroups of a study who may have different risks for an outcome, and may therefore benefit differently from an intervention, is considered a valid analytical approach by other authors. 16 Patients were grouped retrospectively according to the iliac involvement. Although VTE localization was not prespecified in the Home-LITE protocol and comparator groups were not stratified using VTE localization before randomization, it is important to note that efficacy data according to localization were collected prospectively (analysts were blinded to treatment efficacy data while determining iliac involvement). For the iliac group, patients treated with usual care presented a numerically higher incidence of comorbidities at baseline; however, with respect to both the treatments, the numbers in each comorbidity subgroup were very low (Table 1). We cannot rule out a confounding effect on the risk of developing PTS; nevertheless, these differences are unlikely to explain the significant reduction in risk observed in iliac patients.
Patients with iliac vein thrombosis are at greater risk of recurrent DVT and the development of PTS compared to patients with popliteal or distal vein thrombosis. 3,4 A possible approach for treating such patients is CDT, 6,17 –20 which is currently recommended by the ACCP and the Society of Interventional Radiology as an acceptable method for treating acute iliofemoral DVT in carefully selected patients. 17,18 The ongoing ATTRACT trial (NCT00790335) is comparing the effect of standard anticoagulation alone or standard anticoagulation plus CDT on the development of PTS in patients with DVT, stratified according to the iliofemoral or femoropopliteal location. 6
In the CLOT study of long-term use of LMWH to prevent recurrent VTE, the dose of dalteparin was reduced after 1 month. 21 It has been shown that full-dose tinzaparin can be safely used for long term, 22 and no dose reduction was applied in Home-LITE.
A systematic review concluded that long-term treatment of DVT with LMWH is more beneficial than usual care in preventing the development of PTS-related end points (symptoms, ulcers, degree of recanalization, and degree of reflux). 23 Our findings show that the risk of developing symptoms of PTS was reduced in patients receiving long-term LMWH regardless of thrombus location. In patients with iliac involvement, the risk was approximately halved with long-term tinzaparin (odds ratio = 0.53 vs usual care, P = .0079), and despite a small study population (only 24 and 28 patients in the tinzaparin and usual care groups had iliac involvement, respectively), this effect reached statistical significance. Another study has shown that natural thrombolysis may occur earlier and more extensively in response to tinzaparin compared with usual care, although this effect was nonsignificant in patients with iliac thrombosis at the time points studied (1, 3, 6, and 12 months) but significant in patients with superficial femoral thrombosis from the third month of treatment onward. 24 Pharmacological studies have shown that LMWH can exhibit thrombolytic activity 25,26 ; for example, an in vivo experiment using a rabbit jugular vein clot lysis model demonstrated that LMWH had a similar dose-dependent effect to urokinase on thrombolysis. 26 Further studies will be helpful.
A limitation of our study is that the diagnosis of PTS was not confirmed by a health care professional but was assessed in terms of odds ratios based on symptoms reported by patients in response to a questionnaire, the “Patient-Tracked Symptoms Questionnaire.” 9,10 The questionnaire, developed during the 1990s when the Home-LITE study was planned, is based on 8 symptoms or signs whose presence in combination is commonly used in various scoring systems such as the Villalta scale 27 to diagnose the presence and severity of PTS, 10 and the questionnaire has clear face validity. Furthermore, all cases of patient-reported venous ulcers were confirmed by a health care professional. The second limitation is the short observation period; if thrombus regression was slower in the patients with iliac than with noniliac DVT, then longer follow-up may have shown even greater benefit. With respect to compression stockings, while these are often prescribed for at least 3 months in patients with iliac DVT, in Home-LITE patients were allowed to stop using them if free of PTS symptoms at 3 months.
In patients with iliofemoral DVT or other severe conditions related to DVT (phlegmasia), prognosis in terms of the PTS is worse than in patients with noniliac DVT. Our findings suggest that LMWH treatment could be an alternative to these patients. The LMWH should be considered as the treatment of choice in patients who have experienced a DVT with iliac involvement who are unlikely to undergo CDT due to clinical, financial, or other factors.
Footnotes
Acknowledgments
The authors would like to thank Steven Barberini and Gabrielle Parker of Watermeadow Medical, Witney, UK, for their writing and editorial assistance, which was supported by LEO Pharma.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Russell Hull has received grants/research support from Bayer Pharmaceuticals Corp., LEO Pharma Inc., and Sanofi-Aventis; been a consultant for Bayer Pharmaceuticals Corp., LEO Pharma, Inc., Pfizer Inc., GlaxoSmithKline, Wyeth Pharmaceuticals and Portola Pharmaceuticals; and sat on advisory boards for Bayer Pharmaceuticals Corp., Pfizer Inc., and Sanofi-Aventis. Jane Liang has no conflicts of interest to declare. Tazmin Merali has received research support from LEO Pharma Inc, Sanofi-Aventis, Pfizer, Boehringer-Ingelheim, and Bristol-Myers Squibb.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Home LITE was supported by grants from the Medical Research Council (now Canadian Institutes for Health Research) and industry (LEO Pharma A/S). Additional funding was provided by Pharmion and Dupont Pharmaceuticals. LEO Pharma provided the study drug and drug safety monitoring. The industry sponsors did not have any influence on the design or analysis of the study.
