Abstract
Low-molecular-weight heparins (LMWHs) endure as important drugs for thromboprophylaxis. Although clinical use relies on the subcutaneous (SC) route, our previous studies show that single-dose orally administered LMWHs have antithrombotic activity. Since thromboprophylaxis requires long-term treatment, we examined antithrombotic effects of subacute oral LMWHs in a rat venous thrombosis model and compared results to SC or single-dose oral administration. We measured LMWH in endothelium and plasma, weight change and complete blood counts (CBC). Oral LMWH tinzaparin (3 × 0.1 mg/kg/12 or 24 hours) or reviparin (3 × 0.025 mg/kg/24 hours) significantly decreased thrombosis compared to saline. In the subacute study (60 × 0.1 mg/kg/12 hours), oral or SC tinzaparin significantly reduced thrombosis compared to saline but not to single or 3 × 0.1 mg/kg/12 hours oral tinzaparin. Antithrombotic effects were similar between oral and SC administration. LMWH was found on endothelium following oral but not SC administration. Endothelial concentrations were significantly correlated with incidence of stable thrombi (P = 0.021 and 0.04 for aortic and vena cava endothelium respectively, χ 2 test) and total thrombi (P = 0.003 for vena cava endothelium). Anti-Xa activity was significantly greater for oral or SC LMWH than saline and significantly greater for SC versus oral LMWH. Values for CBCs were within normal ranges (mean ± 2 SD). There was no evidence of bleeding. Weight gain was similar between groups. In conclusion, subacute oral and SC LMWH have similar antithrombotic effects. Antithrombotic activity with oral administration is correlated with endothelial LMWH concentrations but not with plasma anticoagulant activity.
Introduction
Despite the introduction of new oral anticoagulants, low-molecular-weight heparins (LMWHs) continue to be clinically important antithrombotic drugs. 1 Low-molecular-weight heparins are produced from unfractionated heparin by a variety of different methods; thus each LMWH is considered chemically unique, with a particular molecular weight profile. Minor differences are observed in biological activities most often measured by anticoagulant assays. 2 Uses of LMWH include the prevention of venous thromboembolism and the treatment of deep vein thrombosis and pulmonary embolism. 3 Low-molecular-weight heparins are considered the core therapy for thromboprophylaxis in patients with cancer and may improve survival in these patients by influencing the tumor biology. 4
Low-molecular-weight heparins are primarily administered by the subcutaneous (SC) route and are generally considered ineffective when given orally. 5 Our laboratory, however, has provided evidence that LMWHs are absorbed following oral administration. A dose-dependent decrease in thrombosis was observed with single doses of the LMWHs tinzaparin or reviparin in a rat jugular vein model when the vessel was examined 4 hours after thrombus initiation and drug administration by gastric tube. 6,7 We have also shown that thrombosis is prevented by oral tinzaparin in a rat carotid artery model 8 and in a rat venous stasis model. 9
Clinical thromboprophylaxis with LMWHs may require treatment over weeks or months. There is little data on the effectiveness or safety of LMWH when administered in repetitive oral doses. We have previously shown that repeated doses of orally administered unfractionated heparin and the LMWH tinzaparin prevented thrombosis in a rat carotid arterial model. Antithrombotic activity was similar for oral and SC administration. 10 In this study, we examine the antithrombotic effect of the LMWHs tinzaparin and reviparin, following repeated oral administration in a rat venous thrombosis model. We also determined whether an orally administered dose of LMWH repeated over 30 days had an antithrombotic effect similar to a single dose or to SC administration. In addition, the amount of LMWH found in endothelium, plasma, urine, feces, stomach, ileum, kidney, and lung was observed at 30 days following administration. We correlated the antithrombotic effect with that found in the endothelium. To identify any adverse effects of subacute doses of oral or SC tinzaparin, weight changes and complete blood counts (CBCs) were observed following 30 days of treatment.
Materials and Methods
Heparins
Tinzaparin sodium was obtained from Novo Nordisk (Denmark), with a peak maximum molecular mass of 5600 Da. Tinzaparin was dissolved in water at concentrations of 0.2 to 0.75 mg/mL. Reviparin sodium (average molecular weight of 4300 Da, anti-Xa activity 130 IU/mg; anti-IIa activity 29 IU/mg) was obtained from Knoll Pharmaceuticals (Ludwigshafen, Germany) and was dissolved in water at concentrations of 0.05 to 0.1 mg/mL.
Animals
A total of 262 male Wistar rats, average weight of 334.2 ± 5.2 g, were obtained from Charles River Canada (St Constant, Quebec, Canada) or from the Animal Resources Centre, University of Saskatchewan. All rats were handled and housed according to the Principles of Animal Care of the Canadian Federation of Biological Societies. Experimental procedures were approved by the Animal Care Committee of the University of Saskatchewan. Animals were anesthetized with barbital and methoxyflurane for experimental procedures.
Animal Model
Tinzaparin was administered as a single oral dose of 0.1 mg/kg to 20 rats; and in 3 repeated doses of 0.1 mg/kg each, 12 and 24 hours apart, with 20 and 27 rats per group, respectively. Reviparin was administered as a single dose of 0.025 mg/kg; and in 3 repeated doses of 0.025 mg/kg each, 12 and 24 hours apart, with 20 animals per group. In addition, tinzaparin was administered by the oral route or SC in 60 repeated doses of 0.1 mg/kg, each 12 hours apart, with 20 animals per group. Control animals were administered 0.4 mL oral saline in a single dose (51 rats) or in 60 doses over a 30-day period (43 rats). Oral administration was by stomach gavage using a steel needle 7.5 cm in length with a blunted end to which a 1-mL syringe was attached containing saline or the LMWH solution. Volumes administered consisted of 0.04 to 0.3 mL of the LMWH solution depending on rat weight, followed by 0.2 mL saline to ensure the complete drug dose was delivered. The SC injections, in volumes ranging from 0.11 to 0.19 mL depending on rat weight, were given into the nape of the neck using a 25-gauge needle on a 1-mL syringe. Saline or the last LMWH dose was given 4 hours prior to killing the animals. When repetitive doses were administered over 30 days, urine and feces samples were collected in metabolic cages for a 24-hour period, the day prior to, and on days 15 to 16 and 29 to 30 of the experiment. Animals were weighed prior to drug treatment and on days 15 and 30 in the 30-day experiments. Animals were examined for any signs of blood on the hair or paws. When animals were killed, the organs and the peritoneal and thoracic cavity were examined for any signs of bleeding.
Thrombosis Test
A thrombus was initiated in the rat jugular vein by a modified method given by Blake et al. 11 Several drops of 10% formalin in 65% methanol were applied to the exposed jugular vein and the incision closed. After 4 hours, the rat was anesthetized, the incision opened, and the exposed jugular vein inspected for a thrombus by pressing gently with a cotton pledget. It was scored positive (stable thrombus), if a thrombus was visible in the jugular vein and the blood flow was prevented. It was scored unstable, if the thrombus could be seen in the jugular vein, which moved on examination with the cotton pledget and the blood flow was maintained. It was scored negative, if a thrombus was not seen and the blood flow was not disrupted. The scoring was performed by the same observer who was blinded to the treatment. Percentage incidence of total thrombotic events and stable and unstable thrombi was calculated by dividing the number of thrombotic events by the total number of rats observed in the treatment.
Tissue Collection
Tissue samples were collected immediately after examination of the jugular vein for a thrombus. Blood samples were collected from the abdominal aorta in 3.8% sodium citrate (1 part sodium citrate and 9 parts blood). The thoracic aorta and inferior vena cava were removed and placed in saline to provide a source of endothelium. Immediately following blood collection, the activated partial thromboplastin time (APTT) was determined from plasma derived from blood samples obtained with a minimum of tissue trauma. Additional plasma aliquots were frozen for later determination of anti-factor Xa activity. In the 30-day studies, a whole blood sample was sent for CBCs to Prairie Diagnostic Services (Western College of Veterinary Medicine, University of Saskatchewan, Saskatoon, Canada). In the 30-day studies, the stomach, ileum, kidney, and lungs were collected. The lumen of the stomach and ileum were washed with distilled water. Tissues were then frozen for later extraction of LMWH.
Extraction of LMWH and Measurement From Endothelium and Tissues
Endothelium was obtained using a modified published procedure. 12 The collected aorta and vena cava were slit open, pinned to dental wax with the lumen side up, and rinsed in saline. Strips of cellulose acetate membrane (Schleicher & Schuell BioScience GmbH, Dassel, Germany) were applied to the luminal surface and when lifted, endothelium was removed. Mean areas for aortic and vena caval endothelium, measured from the monolayer on cellulose acetate paper, were 2.49 ± 0.03 cm2 and 0.45 ± 0.01 cm2 (standard error of the mean [SEM]), respectively. Cellulose acetate membrane was removed from the collected endothelium by 2 repeats of dissolution in cold acetone followed by centrifuging and discarding the supernatant. The precipitates were air-dried and then digested with 10 μL pronase (from Streptomyces griseus; Sigma-Aldrich, Canada; 40 mg/kg in 1 M Tris buffer) for 48 hours at 37°C. Digests were centrifuged at 10 000 rpm for 10 minutes, supernatant was collected and the precipitate washed twice with 100 μL of 26.8% NaCl with washes added to the supernatant. Glycosaminoglycans (GAGs) were precipitated from the supernatant, with 5 volumes of methanol, and the precipitate was dried.
Using modified methods previously described, LMWH was extracted from tissue and feces. 13 Minced tissues and feces were defatted with acetone and isopropanol/petroleum ether (1:1), and digested at 37°C by pronase, 40 mg/kg in 1 M Tris buffer in 0.1 M CaCl2 at pH 8. Digests were purified by precipitating with 1% NaCl in acetone and then methanol. Precipitates were dried, dissolved in water, and analyzed. Low-molecular-weight heparin was isolated from urine by exhaustive dialysis against water using 1000 molecular weight cutoff dialysis tubing (Spectrum Laboratories, Inc, RanchoDominguez, California).
To identify and measure quantities of LMWH in tissue, urine, feces, and endothelial samples, extracts were applied to agarose gel electrophoresis. Slides were stained with 0.04% toluidine blue in 80% acetone and the background color was removed with 1% acetic acid. 14 Low-molecular-weight heparin was identified by electrophoretic migration as compared to reference material and amounts determined by densitometry. To obtain endothelial samples, 5 μL of distilled water was added to each endothelial extract and 2 μL of the dissolved extract was added to 2 lanes on the agarose gel electrophoresis slide. After determining the density of the band and comparing to the reference LMWH, the total amount of LMWH in each endothelial sample was calculated and expressed in microgram per square centimeter (μg/cm2) of endothelial surface area. The percentage of LMWH-positive endothelial samples was calculated by dividing the number of samples which had LMWH by the total number of samples examined in each experimental group multiplied by 100.
Analyses
All results are expressed as mean ± SEM. Thrombosis data are expressed as a percentage with 95% confidence intervals. The χ2 test for difference between proportions was used to compare thrombotic incidences and the percentage of LMWH-positive endothelial samples. Differences in LMWH concentrations in the endothelium and in anticoagulant activity between groups were determined by the Kruskal-Wallis nonparametric test followed by Dunn’s multiple comparison test. A Pearson’s correlation was used to correlate mean endothelial concentrations with the total and stable thrombotic incidence. Differences in hematological values between groups were analyzed using a 1-way analysis of variance (ANOVA) followed by Tukey multiple comparison test. Single-dose and 30-day controls were combined when the endothelial and plasma LMWH concentrations were considered. Differences in tissue heparin concentrations were determined using a paired t test and a 2-tailed t test; P < .05 was considered significant.
Results
Antithrombotic Activity
To determine suitable time intervals for repetitive dosing with LMWHs to achieve an antithrombotic effect, the incidence of thrombosis was determined after 3 repetitive oral doses of the LMWHs tinzaparin or reviparin at 12- or 24-hour intervals. The doses chosen were those that reduced the incidence of thrombosis by at least 50%, compared to rats given saline, based on previous studies wherein the vessels were examined 4 hours after a single oral dose. 6,7 Compared to saline administration, the incidence of both stable and total thrombi decreased significantly with single-dose administration of tinzaparin (0.1 mg/kg) and with 3 doses of tinzaparin of 0.1 mg/kg each (3 × 0.1 mg/kg), at 12-hour or 24-hour intervals (Figure 1). There was no significant difference in thrombotic events between single or repeated doses of oral tinzaparin. Compared to control, both stable and total thrombi decreased significantly when reviparin was administered orally as a single dose (0.025 mg/kg) or as 3 oral doses (3 × 0.025 mg/kg) at 24-hour intervals. When reviparin was administered (3 × 0.025 mg/kg) at 12-hour intervals, the incidence of stable thrombi, but not total thrombi, was significantly less than control. When compared to single dose reviparin administration (0.025 mg/kg), the incidences of stable and unstable thrombi were significantly greater than for 3 doses of reviparin (3 × 0.025 mg/kg) at 12-hour, but not 24-hour intervals.

Antithrombotic activity of the LMWHs tinzaparin and reviparin administered orally in single or 3 repeated doses. Tinzaparin and reviparin at 0.1 and 0.025 mg/kg, respectively, were administered by stomach gavage as a single dose or in 3 repeated doses 12 or 24 hours apart. Control rats were given saline. The final dose was given immediately following thrombus initiation. Vessels were examined for a thrombus 4 hours later. Upward deflecting bars show 95% confidence intervals for total thrombotic events; downward deflecting bars show 95% confidence intervals for the incidence of hard clots. Significantly different than (a) control and (b) single-dose reviparin (χ2 test for differences between proportions). LMWHs indicate low-molecular-weight heparins.
A subacute 30-day study was then done comparing the antithrombotic effects of oral versus SC administration of tinzaparin. Based on the observation with 3 repetitive oral doses (Figure 1), tinzaparin was administered orally and SC at a dose of 0.1 mg/kg for a 30-day period at 12-hour intervals (60 × 0.1 mg/kg/12 hours). As shown in Figure 2, thrombotic incidence, both stable and total, for repetitive oral or SC tinzaparin treatment was significantly less than the single-dose saline or 30-day repetitive dose oral saline (0.4 mL/12 hours). There was no difference in thrombotic incidence when tinzaparin was administered as a single oral dose; or in 3 or 60 oral, or 60 SC doses (all at 0.1 mg/kg) at 12-hour intervals.
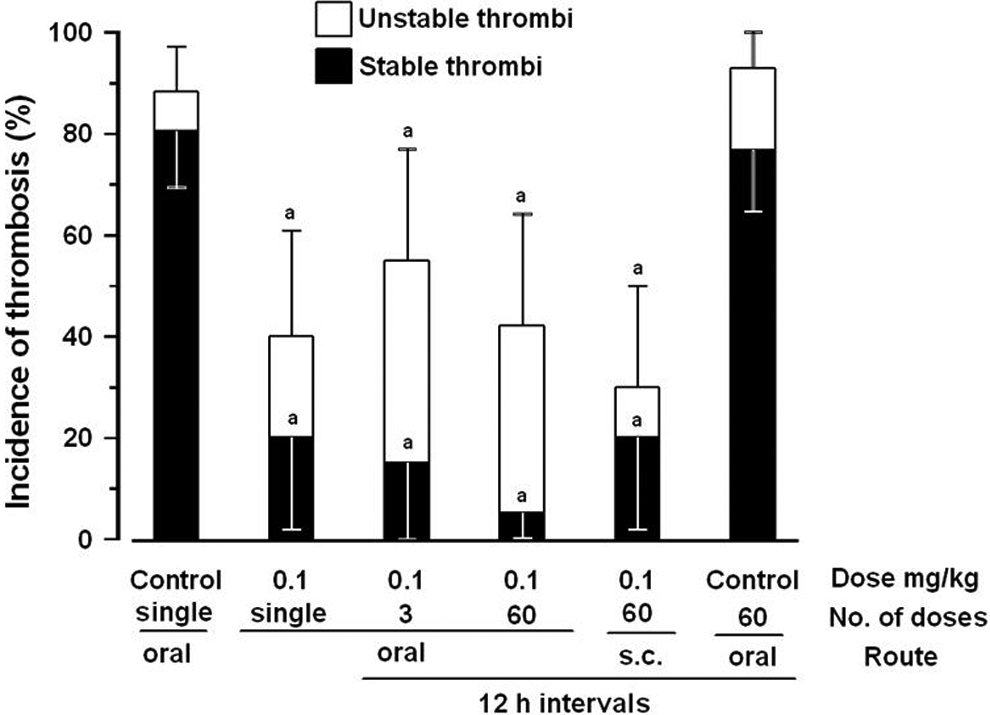
Antithrombotic activity of the LMWH tinzaparin following subacute oral or subcutaneous administration. Tinzaparin was given in repeated doses of 0.1 mg/kg by stomach gavage or subcutaneously 12 hours apart for a 30-day period (60 × 0.1 mg/kg/12 hours). Incidence of thrombosis was compared to that in control rats given oral saline as a single dose or oral saline every 12 hours over a 30-day period and to single-dose tinzaparin (1 × 0.1 mg/kg) or 3 doses of tinzaparin (3 × 0.1 mg/kg/12 hours). The final LMWH or saline dose was given immediately following thrombus initiation. Vessels were examined 4 hours later. Upward deflecting bars show 95% confidence intervals for total thrombotic effects; downward deflecting bars show 95% confidence intervals for the incidence of hard clots. Significantly different than (a) single or 30-day controls (χ2 test for differences between proportions). LMWH indicates low-molecular-weight heparin.
Low-Molecular-Weight Heparin on Endothelium
In all groups, LMWH was found on aortic and vena caval endothelium following oral administration of tinzaparin or reviparin (Table 1). After repeated SC administration of tinzaparin (60 × 0.1 mg/kg/12 hours), LMWH was not found on endothelium. The GAGs, with migration and staining characteristics similar to LMWH, were found in 1 of 44 control aortic and vena caval endothelial samples. The percentage of endothelial samples positive for LMWH was significantly greater in the oral tinzaparin subacute group (60 × 0.1 mg/kg/12 hours) than all other groups. For aortic endothelium, the percentage of endothelial samples positive for LMWH was significantly greater in all oral tinzaparin or reviparin repeated dose groups and the reviparin single-dose group compared to saline administration. The percentage of LMWH-positive samples in the oral tinzaparin 12-hour repeated dose group (3 × 0.1 mg/kg/12 hours) and the reviparin single and 12 hours repeated dose group (3 × 0.025 mg/kg/12 hours) was significantly greater than the SC tinzaparin group. When vena caval endothelium was considered, the percentage of LMWH-positive samples in the 12-hour interval tinzaparin group (3 × 0.1 mg/kg/12 hours) and the single and 24-hour interval reviparin group (3 × 0.025 mg/kg/24 hours) was significantly greater than control and subacute SC administration.
Heparin With Endothelium Following Administration of LMWHs in Repetitive Doses.a

Abbreviations: LMWHs, low-molecular-weight heparins; SC, subcutaneous.
aSignificantly different than bsaline, csingle, d12 hours, e24-hour intervals, fSC tinzaparin 60 doses. χ2 for differences between proportions for percentage positive; Kruskal-Wallis nonparametric test for differences in concentrations (μg/cm2).
Amounts of tinzaparin found on both aortic and vena caval endothelium following 60 oral tinzaparin doses (60 × 0.1 mg/kg/12 hours) were significantly greater than that in control, single oral, and repeated SC tinzaparin groups. Amounts found on vena caval endothelium following 60 oral tinzaparin doses were also significantly greater than the 24-hour interval oral tinzaparin group.
Correlation Between Heparin on Endothelium and Thrombotic Incidence
When tinzaparin and reviparin were given in repeated oral doses, mean amounts of LMWH found on endothelium were plotted against percentage of stable thrombotic incidence and total thrombotic incidence for each group (Figure 3). Control groups consisted of single oral dose administration of saline (51 rats) and repeated dose administration where saline was given every 48 hours over 30 days (43 rats). There was a significant negative correlation between the mean amount of LMWH found on vena caval endothelium and the percentage of total thrombotic incidence (P = .003, r 2 = 0.851) and percentage of stable thrombi (P = .021, r 2 = 0.689) and between the mean amount of LMWH found on aortic endothelium and percentage of stable thrombi (P = .040, r 2 = 0.603). The correlation between mean amount of LMWH on aortic endothelium and percentage of total thrombi (P = .119, r 2 = 0.419) did not reach significance although a trend was evident.
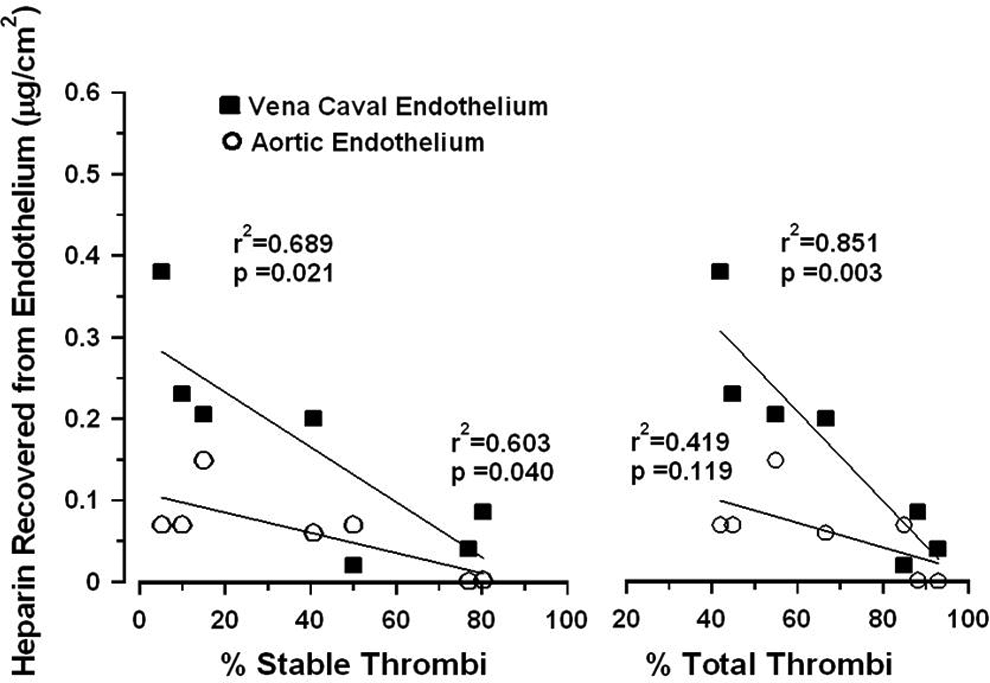
Correlation between antithrombotic activity and mean concentrations of endothelial heparin after repeated oral administration of the LMWHs tinzaparin and reviparin. Tinzaparin (0.1 mg/kg) or reviparin (0.025 mg/kg) was given in 3 repeated doses at 12- or 24-hour intervals immediately following thrombus initiation. Tinzaparin was also given in 60 doses (0.1 mg/kg) 12 hours apart. Control rats were given saline for 30 days. A thrombus was initiated by application of 10% formalin in 65% methanol to the exposed jugular vein. The thrombus was scored as stable or unstable if the blood flow resumed on examination. Aorta and superior vena cava were harvested 4 hours after thrombus initiation. Mean values are shown for concentrations on aortic and vena caval endothelium obtained from at least 19 rats per group. Pearson correlation was used for the calculation of r 2 and P values.
Low-Molecular-Weight Heparin in Plasma
To determine whether LMWH could be found in blood, APTT and anti-Xa activity were measured in plasma samples. Mean values of APTT for groups ranged between 15.6 and 27.3 seconds (Table 2). There was no significant difference in mean APTT between rats treated with a single dose or multiple doses (0.1 mg/kg) of tinzaparin or single dose of reviparin (0.025 mg/kg) compared to control. Rats treated with 3 doses of reviparin at 12- or 24-hour intervals had a significantly lower APTT compared to the oral saline group. Mean APTT was significantly less in groups receiving 3 repeated doses of tinzaparin or reviparin at 12- or 24-hour intervals compared to single doses of the same drug. Groups administered 60 SC or oral doses of tinzaparin (60 × 0.1 mg/kg/12 hours) and 3 doses of oral tinzaparin at 12-hour intervals (3 × 0.1 mg/kg/12 hours) showed a significantly greater mean anti-Xa activity compared to oral saline and single dose tinzaparin groups. As well, anti-Xa activity was significantly greater in the group receiving SC tinzaparin than the group receiving oral tinzaparin at the same dose and time interval (60 × 0.1 mg/kg/12 hours). With reviparin treatment, there was no difference in anti-Xa activity between single or 3 repetitive doses, compared to controls.
Anticoagulant Activity Following Administration of LMWHs in Repetitive Doses.a

Abbreviations: APTT, activated partial thromboplastin time; LMWHs, low-molecular-weight heparins; SC, subcutaneous.
aSignificantly different than bsingle-dose same drug, csaline, d60 × 0.1 mg/kg/12 hours oral, Kruskal-Wallis nonparametric test.
Recovery From Tissue
Amounts of LMWH in urine and feces, and in stomach, ileum, kidney and lung were determined after 60 repetitive doses of tinzaparin (60 × 0.1 mg/kg/12 hours) given SC or orally. Urine and feces were collected 24 hours immediately prior to treatment (day 0) and beginning on days 15 and 29 of the experiments. When SC tinzaparin was administered, LMWH was found in 24-hour urine samples collected on days 29 to 30 and in feces samples collected on days 15 to 16 and 29 to 30 (Figure 4). When oral tinzaparin was administered, LMWH was found in urine and only in trace amounts in feces samples collected on days 29 to 30. In urine, LMWH was significantly greater in samples collected on days 29 to 30 compared to day 0 samples (P < .0001 and <.0289, 2-tailed paired t test) for SC and oral administration, respectively. Low-molecular-weight-heparin in urine samples, collected on day 29 to 30 following SC administration, was significantly greater than in those following oral administration and day 29 to 30 controls (P < .0001 and <.0009, respectively, 2-tailed t test), but amounts following oral administration was not significantly different than days 29 to 30 controls although a trend was evident (P < .059, 2-tailed t test). With SC administration, LMWH found in feces was significantly greater than that found in samples collected on day 0 (P < .0001 and .0392 for days 15 to 16 and 29 to 30, respectively, paired t test). Amount of LMWH found in feces on days 15 to 16 was significantly greater than the same day control (P < .0001), same day oral administration (P = .0001), and days 29 to 30 feces samples (P = .0004; 2-tailed t test). Amount of LMWH found in feces on days 29 to 30 was significantly greater than oral administration (P = .0216) but not the same day control although a trend was evident (P = .09, 2-tailed t tests).
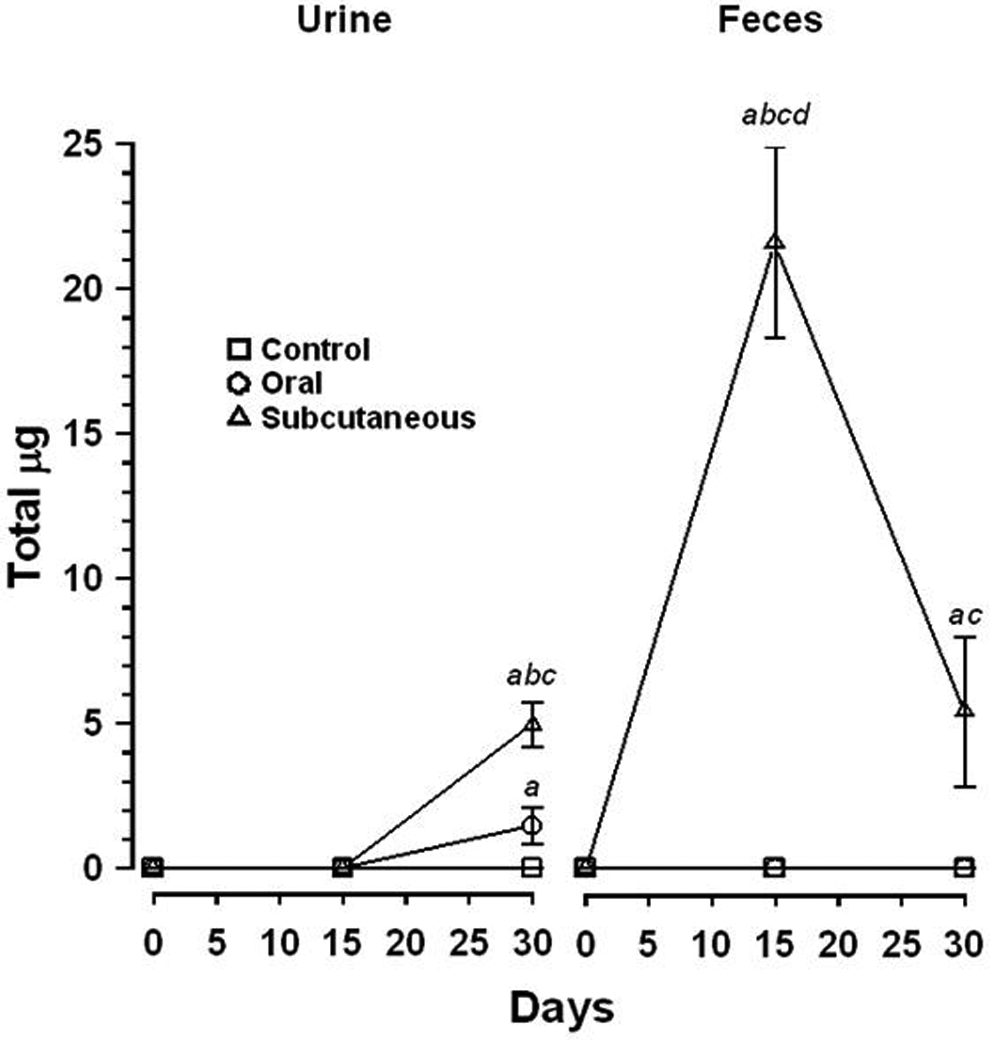
Heparin recovered from urine and feces after repeated oral or subcutaneous administration of the LMWH tinzaparin. Tinzaparin was given by oral or subcutaneous administration in 60 doses 12 hours apart. Feces and urine were collected in metabolic cages for a 24-hour period beginning prior to days 0, and days 15, and 29. Mean values and SEM are shown. Results include 14 to 20 rats per group for urine and feces. Significantly greater than (a) prior to day 0, paired t test, (b) same day control, (c) same day oral administration, (d) day 29 to 30 feces, 2-tailed t test. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
Trace amounts of LMWH were recovered from stomach and lung following oral administration (0.77 ± 0.49 and 16.13 ± 16.13 μg/g wet weight, mean ± SEM) and from lung following SC administration (30.64 ± 28.31 μg/g wet weight), although the qantity was not significantly different compared to saline-treated rats (P = .089, .300, and .264, respectively, 2-tailed t tests, 6-8 tissues/group). Low-molecular-weight heparin was not found in ileum or kidney after oral or SC administration and in stomach after SC administration.
Complete Blood Count and Weight Gain
To help determine the possible side effects of subacute LMWH administration, hematological parameters were compared between control rats and those given 60 repetitive doses of tinzaparin (60 × 0.1 mg/kg/12 hours) by oral or SC routes (Table 3). All hematological values were within normal range defined as mean ± 2 SD. 15 Nevertheless, the hemoglobin (Hgb) concentration and packed cell volume were significantly lower in rats treated with LMWH by the SC or oral route versus controls. The Hgb concentration was significantly less in rats receiving LMWH by the SC route compared to the oral route. The red blood cell count in rats administered SC LMWH was significantly less than that in control rats but not in rats administered oral LMWH. Mean corpuscular hemoglobin concentration (MCHC) was significantly less following SC compared to oral LMWH administration but not compared to controls. There was no difference in the mean corpuscular volume, mean corpuscular hemoglobin, percentage of reticulocytes, white blood cell or platelet counts between groups. There were no signs of external or internal bleeding.
Hematological Values in Rats Administered 60 Doses of Saline or 0.1 mg/kg Tinzaparin Every 12 Hours by Oral or Subcutaneous Routes.a

Abbreviations: Hgb, hemoglobin concentration; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PCV, packed cell volume; Platelets, platelet count; RBC, red cell count; Retics, percentage of reticulocytes; WBC, white cell count.
aSignificantly different than bcontrol, coral administration. One-way ANOVA. Saline-treated rats were administered 0.4 mL saline by gastric tube.
There was no difference in the 30-day weight gain between rats given oral or SC tinzaparin or saline (190.1 ± 3.8 g, 180.0 ± 6.9 g, and 172.5 ± 10.6 g [mean ± SEM], respectively, P = .3126, 1-way ANOVA).
Discussion
Low-molecular-weight heparins, important for thromboprophylaxis, are primarily administered SC. It is assumed that LMWHs are not effective orally due to their high charge and molecular weight. Nonetheless, an oral LMWH would prevent the bruising and discomfort associated with SC injection and could decrease health-care costs. Recently oral antithrombotic agents, specific for anti-Xa and antithrombin activity, have been introduced for clinical use. Although these new drugs have the potential to reduce the need for oral heparin, heparins provide a combination of mechanisms that contribute to antithrombotic activity and are unlikely to be replaced by these new anticoagulant drugs. 16 As well, heparins continue to be investigated for use in thrombotic conditions such as large artery occlusive disease 17 or as an adjunct to cancer therapy 4 and for the treatment of other chronic conditions such as interstitial cystitis, 18 asthma, 19 inflammatory bowel disease, 20 and control of angiogenesis, 21 that have little to do with specific anti-Xa or antithrombin activity. Heparins have shown efficacy in many additional conditions including allergic rhinitis, 22 arthritis, 23 transplant rejection, 24 and hypertension. 25,26,27,28 Therefore, oral administration of LMWHs for long-term use remains an important consideration.
Our laboratory has provided evidence that orally administered unfractionated heparins and LMWHs are absorbed and are effective antithrombotic agents in animal models. Single-dose unfractionated heparins and the LMWHs reviparin and tinzaparin prevented thrombosis in a rat jugular vein thrombosis model. 6,7,29 Repeated doses of orally administered unfractionated heparins and the LMWH tinzaparin prevented thrombosis in a rat carotid arterial model where the effects were equal or similar to SC administration. 10 The results presented here, using a rat jugular vein thrombosis model, support this observation and indicate that the LMWH tinzaparin, administered by gastric tube at 0.1 mg/kg/12 hours over a 30-day period, prevents thrombosis, and the antithrombotic effect is equal to that following SC administration. This reveals that repetitive orally administered LMWH has antithrombotic effects in both arterial and venous thrombosis models. It also implies that oral tinzaparin is absorbed as effectively as SC tinzaparin.
Repeated oral dosing with the LMWH tinzaparin at 0.1 mg/kg at 12-hour intervals for 30 days (60 doses) had the same antithrombotic effect as 3 repeated doses at 12-hour intervals and single-dose administration. This observation suggests that there is no accumulation of antithrombotic activity following repeated doses at 12-hour intervals of tinzaparin. It also suggests that the LMWH tinzaparin is rapidly absorbed and reaches the peak pharmacodynamic activity within a 12-hour period. Results suggest that 3 doses of tinzaparin at 0.1 mg/kg administered at 24-hour intervals are not as effective as that at 12-hour intervals (Figure 1), supporting the idea of rapid absorption and distribution of tinzaparin. Reviparin was administered at 0.025 mg/kg, a dose that reduced thrombosis by approximately 50% compared to single-dose saline administration. 30 In contrast to tinzaparin, the antithrombotic effect at 24-hour intervals was superior to that at 12-hour intervals. This difference may be due to the reduced dose of reviparin versus tinzaparin (0.025 mg/kg vs 0.1 mg/kg, respectively), and difference in molecular weight between the drugs which along with other factors may contribute to different pharmacokinetic properties of these 2 LMWHs.
We have used anticoagulant activity to estimate the amount of LMWH found in plasma. There was little or no increase in APTT for all treatment groups including those that received 60 repeated doses. This agrees with our earlier findings following oral administration of heparins. 6,7,30 Changes in anti-Xa activity were seen between groups. The highest value was observed following SC administration for 30 days. Anti-Xa activity increased significantly following oral administration; however, the increase in anti-Xa activity was the same whether it was given in 3 or 60 doses at 12-hour intervals, indicating no accumulation of anti-Xa activity over time when administered at 12-hour intervals.
There is a correlation between the amount of LMWH found on endothelium and the antithrombotic effect following oral administration. This correlation is significant for percentage of stable thrombi for both vena caval and aortic endothelium and percentage of total thrombi for vena cava endothelium while the correlation approaches significance for percentage of total thrombi and aortic endothelium (Figure 3). This agrees with our previous observations regarding single-dose unfractionated heparin, where a correlation existed between heparin recovered from aortic and vena caval endothelium and the percentage of total thrombi. 30 This suggests that distribution of LMWH on endothelium may be a better predictor of antithrombotic activity than LMWH in plasma following oral administration.
After repetitive doses, there was a small but significant decrease in the Hgb concentration and hematocrit in rats treated with SC and oral LMWH, but all values were within the normal limits. The changes observed with SC LMWH were more pronounced than those with oral LMWH, where both the Hgb and MCHC were significantly lower than controls receiving SC but not oral LMWH. There was no evidence of internal or external bleeding by any route. This indicates that orally administered LMWH is as safe as that administered SC.
There were some differences observed between the 2 routes of administration. In contrast to oral administration, LMWH was not found on endothelium following SC administration following 30-day administration. This is similar to our previous observations following SC administration of the LMWH tinzaparin in a rat carotid arterial model where 30-day administration (60 × 0.1 mg/kg/12 hours) resulted in less endothelial recovery from aortic endothelium compared to oral administration. 30 As well, anti-Xa activity found in plasma was significantly greater following SC administration versus oral administration. Greater amounts were found in urine at 30 days following SC versus oral administration and LMWH could be found in feces at 15 and 30 days after SC administration. The presence of LMWH in urine and feces following administration supports previous observations. 31,32 The increased anti-Xa activity and LMWH found in urine and feces following SC versus oral administration suggest a minor increase in circulating LMWH with the SC route compared to the oral route. However, this difference does not translate into an increase in antithrombotic activity which may be offset by the increased concentrations in endothelial LMWH for oral versus SC administration. It is still not known why there is more LMWH in plasma following SC administration and more LMWH in endothelium following oral administration. Perhaps LMWH exposed to the environment of the stomach and intestine is altered so that it binds better to the endothelial surface or LMWH exposed to the SC environment may not be able to bind as readily to endothelium. Alterations could include direct changes to LMWH or more likely to different counterions on the LMWH molecule gathered from the gut and SC environment.
Conclusion
The observation that repeated doses of LMWH have antithrombotic effects and can be found in endothelium and urine after oral administration, despite low amounts found in plasma, agrees with our previous observations and suggests that heparins, including LMWHs, are absorbed. This is further supported by the observation that SC heparin given at the same dose and time interval had similar antithrombotic effects. Antithrombotic activity for repeated oral administration correlated with amounts found on endothelium. Repeated doses of oral LMWH were as safe as SC when safety was assessed by CBCs and weight gain in this rat model. Thus, the use of oral LMWHs for thromboprophylaxis requires further study.
Footnotes
Acknowledgments
The author gratefully acknowledges the contributions of Sandra Wice and Tilly Ping for the collection of data.
Author Contributions
L.M. Hiebert contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author received financial support from the Heart and Stroke Foundation of Saskatchewan for this work.
