Abstract
The aim of the study was to analyze the relation between platelet reactivity and intramyocardial hemorrhage (IMH) in patients with ST-elevation myocardial infarction (STEMI) undergoing primary percutaneous coronary intervention. Platelet reactivity was measured in 49 patients with means of impedance aggregometry (Multiplate) before reperfusion and repeated in the subacute phase of STEMI. Cardiovascular magnetic resonance was used to detect IMH, which was found in 16 (33%) patients. There were no differences in platelet reactivity between patients with and without IMH before reperfusion. Reassessment in the subacute phase of STEMI demonstrated that patients with IMH had lower thrombin receptor activating peptide (TRAP)-induced platelet aggregation (P = .004) and trends toward lower values of ristocetin and collagen-induced platelet aggregation (P = .09 and P = .07). The TRAP-induced platelet aggregation and initial perfusion grade were the factors independently associated with IMH. Intramyocardial hemorrhage is related to more potent inhibition of platelet aggregation in the subacute phase of STEMI.
Keywords
Background
Benefits of the primary percutaneous coronary intervention (PCI) in ST-segment elevation myocardial infarction (STEMI) are limited by reperfusion injury. 1,2 It is hypothesized that the factors responsible for this phenomenon include microembolization with leukocyte–platelet aggregates or fragments of the atherosclerotic plaque, tissue edema, endothelial disruption, accelerated local inflammatory response, and metabolic changes. 1 These events manifest as microvascular obstruction (MVO) and/or intramyocardial hemorrhage (IMH) and can be observed with means of cardiovascular magnetic resonance (CMR) imaging. 3,4 The presence of MVO and/or IMH was shown to be an independent predictor of worse prognosis in STEMI. 5,6 However, the pathogenesis of IMH has not been determined.
Currently, most patients with STEMI undergoing primary PCI receive 3 antiplatelet drugs (aspirin, P2Y12 inhibitor, and glycoprotein [Gp] IIb/IIIa inhibitor). 7 One of the consequences of potent platelet inhibition with triple antiplatelet therapy is an increased frequency of clinically visible major bleeding events. 8,9 Nevertheless, until now there have been no studies on the association between platelet reactivity and IMH, which was a form of bleeding not detectable in vivo until the introduction of CMR.
Therefore, we have decided to analyze the relation between platelet reactivity and the occurrence of IMH in patients with STEMI undergoing primary PCI.
Methods and Results
Study Population
The study included a prospective group of 49 patients with first STEMI (age 60.5 ± 8.5 years, 73% male) treated with primary PCI. All patients received loading doses of 300 mg of aspirin and 600 mg of clopidogrel in the prehospital phase followed by intravenous administration of Gp IIb/IIIa inhibitor (abciximab 0.25 mg/kg bolus and 0.125 μg/kg per minute infusion for 12 hours) started in cath-lab. Maintenance doses of aspirin and clopidogrel were 75 mg daily. Platelet aggregation testing was performed in the cath-lab (after administration of all antiplatelet drugs but before PCI) and in the subacute phase of STEMI (days 3-5) along with CMR imaging.
The STEMI was defined as (1) presence of continuous chest pain for at least 30 minutes, (2) ST-segment elevation in 2 or more contiguous electrocardiogram leads (≥1 mm for the arm leads and ≥2 mm for precordial leads), (3) presence of coronary artery occlusion or flow-limiting stenosis on initial coronary angiogram. Each case of STEMI had to be eventually confirmed with the presence of elevated troponin I (TnI).
The study excluded patients with previous myocardial infarction, patients in critical clinical condition (ie, with balloon counterpulsation or mechanical ventilation), patients with acute or chronic renal failure defined as estimated glomerular filtration rate <50 mL/min (value suggested by the local ethics committee as a contraindication to gadolinium-based contrast agents administration in scientific studies), and other contraindications to CMR (such as temporary or permanent pacemakers, severe form of claustrophobia or metallic foreign objects in the body).
Informed consent was obtained from each participating patient. The study was approved by the local ethics committee.
Platelet Aggregation
Platelet reactivity testing was performed by means of a multiple electrode impedance aggregometry (Multiplate, Roche, Basel, Switzerland). 10 The instrument measured the change in impedance between 2 electrodes as platelets adhered and aggregated in response to specific agonist. Platelet aggregation was recorded continuously. The increase in impedance was transformed to arbitrary aggregation units (AUs) and plotted against time (AU × minute).
For the purpose of platelet reactivity testing, whole blood was withdrawn from antecubital venipuncture into a polyethylene tube containing a stabilized direct thrombin inhibitor. The following specific agonists and tests were used to analyze platelet reactivity: arachidonic acid (0.5 mmol/L)- ASPItest, adenosine diphosphate (ADP) plus prostaglandin E1 (6.4 μmol/L)- high-sensitivity ADPtest, thrombin receptor activating peptide 6 (32 μmol/L)- TRAPtest, collagen (3.2 μg/mL)- COLtest, ristocetin (0.2 mg/mL)- RISTOtest low.
The manufacturer reference values for Multiplate analysis using thrombin inhibitor blood were for ASPItest 745 to 1361 AU × minute, for high-sensitivity ADPtest 311 to 1071 AU × minute, for TRAPtest 941 to 1563 AU × minute, for COLtest 459 to 1166 AU × minute, for RISTOtest low 15 to 337 AU × minute.
Cardiac Magnetic Resonance
The CMR studies were performed with the use of 1.5-Tesla scanner (Avanto, Siemens, Erlangen, Germany). Scout images and electrocardiographic-gated breath-hold steady-state free precession (SSFP) cine images in 2- and 4-chamber views were registered to set up final short-axis imaging planes. Short-axis SSFP cine images were obtained from the mitral valve insertion point to the apex with 8-mm thick slices and 2-mm gap between subsequent slices to encompass the entire left ventricle (LV). The same short-axis and long-axis slice positions were used to obtain dark-blood T2-weighted short-tau inversion-recovery (STIR) fast-spin echo sequences. Finally, a 0.1 mmol/kg of body weight of gadolinium contrast (gadobutrol—Gadovist, Bayer Schering Pharma AG, Berlin, Germany) was administered via the antecubital vein and flushed with 30 mL of isotonic saline. Delayed enhancement (DE) images were obtained with a breath-hold segmented inversion recovery sequence performed 10 minutes after contrast injection and acquired in the same orientation as the cine images. The inversion time was adjusted to completely null normal myocardium.
Image Analysis
Cine images were analyzed with the use of a dedicated software (MASS 6.2.1, Medis, Leiden, the Netherlands). Initially, short-axis images were previewed from the base to the apex in a cinematic mode, then endocardial and epicardial contours for end-diastole and end-systole were manually traced. Delineated contours were used for the quantification of LV volumes normalized to body surface area (end-diastolic volume index [LVEDVI], end-systolic volume index [LVESVI]) and ejection fraction (LVEF). Left ventricular mass was obtained by multiplication of LV myocardial volume by 1.05 g/mL and normalized to body surface area (left ventricular mass index [LVMI]).
The IMH was defined as a hypointense area within the area at risk [AAR] defined as myocardial tissue with signal intensity of 2 standard deviations (SD) above mean signal obtained in the remote myocardium on STIR images 4 (Figure 1). Infarct size was defined as area above 50% of the maximal signal intensity within DE (full-width at half maximum) and expressed in grams (absolute infarct size) and as a percentage of LV mass (relative infarct size). 11 The MVO was defined as dark areas of absent contrast surrounded by DE. 3
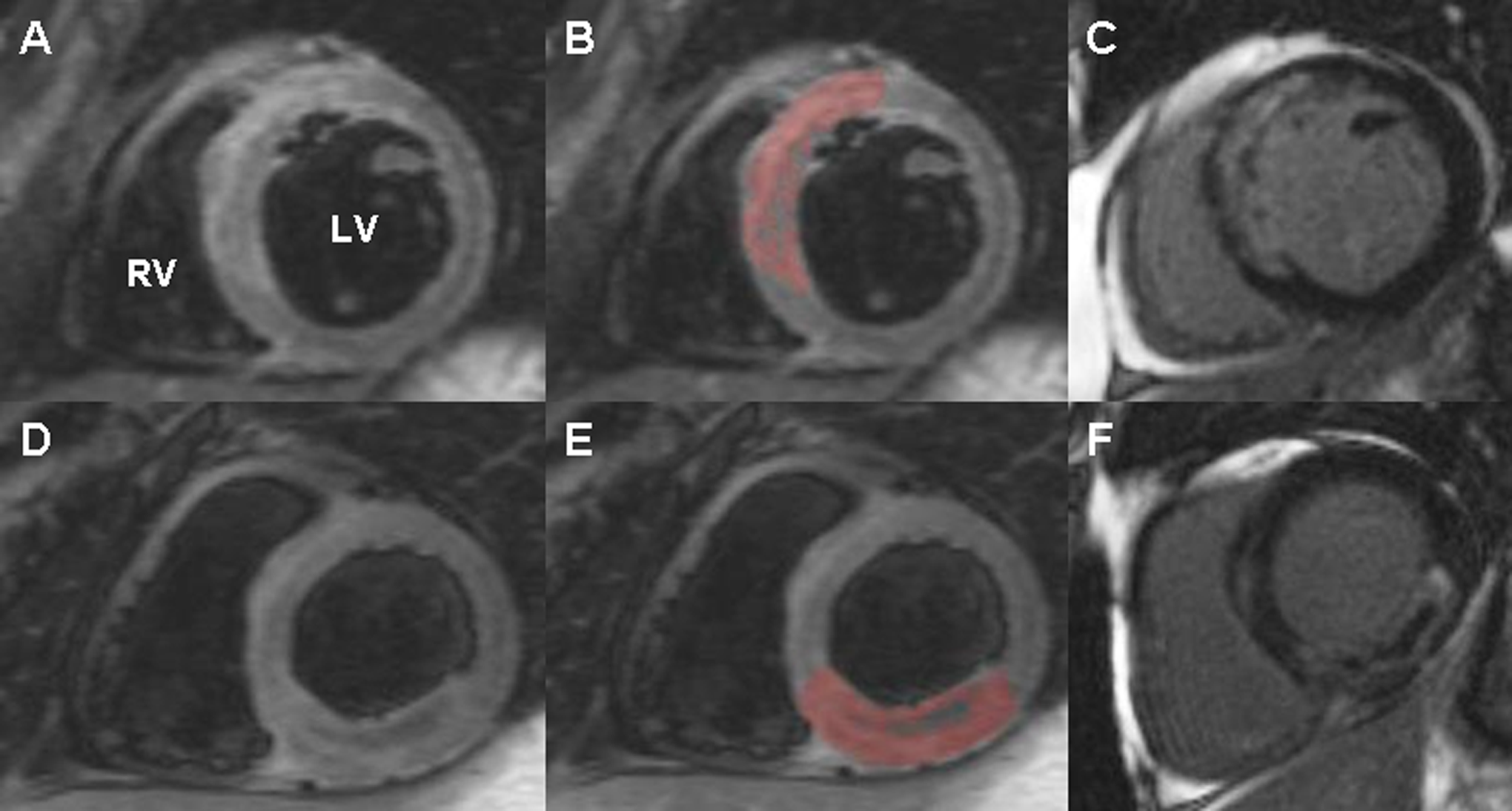
Short-axis cardiovascular magnetic resonance images demonstrating intramyocardial hemorrhage (IMH). A-C (ST-elevation myocardial infarction [STEMI]) of the anterior wall caused by proximal left anterior descending artery occlusion, (A) a dark-blood T2-weighted short-tau inversion-recovery fast-spin echo image showing IMH as a hypointense area surrounded by a hyperintensive area at risk, (B) same slice with painted area at risk (AAR), IMH is defined as the area within the area at risk that is not painted, (C) delayed enhancement image of the same slice showing diffuse infarct core with regions of microvascular obstruction(MVO)/IMH (dark areas). D-F, Same type of images in a patient with inferior STEMI caused by proximal right coronary artery occlusion. LV indicates left ventricle; RV, right ventricle.
All images were analyzed by a single cardiologist with 4 years of training in CMR (Ł.A.M.) and supervised by a senior expert with 8 years of experience in CMR (J.M.).
Statistical Analysis
All results for categorical variables were expressed as number and percentage and for continuous variables as mean and SD or median and interquartile range (IQR), depending on the normality of distribution assessed with the use of a Kolmogorov-Smirnoff test. Platelet aggregation assessed with multiple electrode impedance aggregometry was not normally distributed and therefore was presented as median and IQR. Chi-square test or Fisher exact test was used for comparison of categorical variables when appropriate. Student t test or Mann-Whitney test was applied to compare continuous variables depending on the normality of distribution. Correlation was assessed with the Spearman test. Multivariable logistic regression model was used to test for an independent association of thrombin receptor activating peptide (TRAP)-induced platelet aggregation and IMH. Besides TRAP-, collagen-, and ristocetin-induced platelet aggregation measures, the model included all the significant variables from the univariate analysis (lesion location in infarct-related artery [IRA], initial Thrombolysis in Myocardial Infarction [TIMI] flow, peak TnI, LVESVI, and LVEF) with an exception of inclusion of only 1 infarct size measure—peak TnI. Also, because of collinearity between MVO and IMH, we decided to drop MVO from the multivariable analysis. All tests were 2-sided with a significance level of P < .05. Statistical analyses were performed with MedCalc statistical software 10.0.2.0 (MedCalc, Mariakerke, Belgium).
Results
Intramyocardial hemorrhage was observed in 16 (33%) patients as demonstrated in Table 1. Patients with IMH were more likely to have total occlusion of the IRA on the initial angiogram (P = .006) and a higher frequency of proximal lesion location (P = .02) in comparison to patients without IMH. The IMH was also related to larger infarct size as demonstrated with higher peak TnI concentration (P = .0001) and higher absolute or relative infarct size measured with CMR (P = .002 and P = .0001, respectively). As a consequence of a larger infarct size patients with IMH had lower LVEF (P = .001) and higher LVESVI (P = .006). The MVO closely accompanied IMH. It was present in all patients with IMH and in 52% of the patients without IMH (P = .0002).
Characteristics of Patients With and Without Intramyocardial Hemorrhage (IMH).
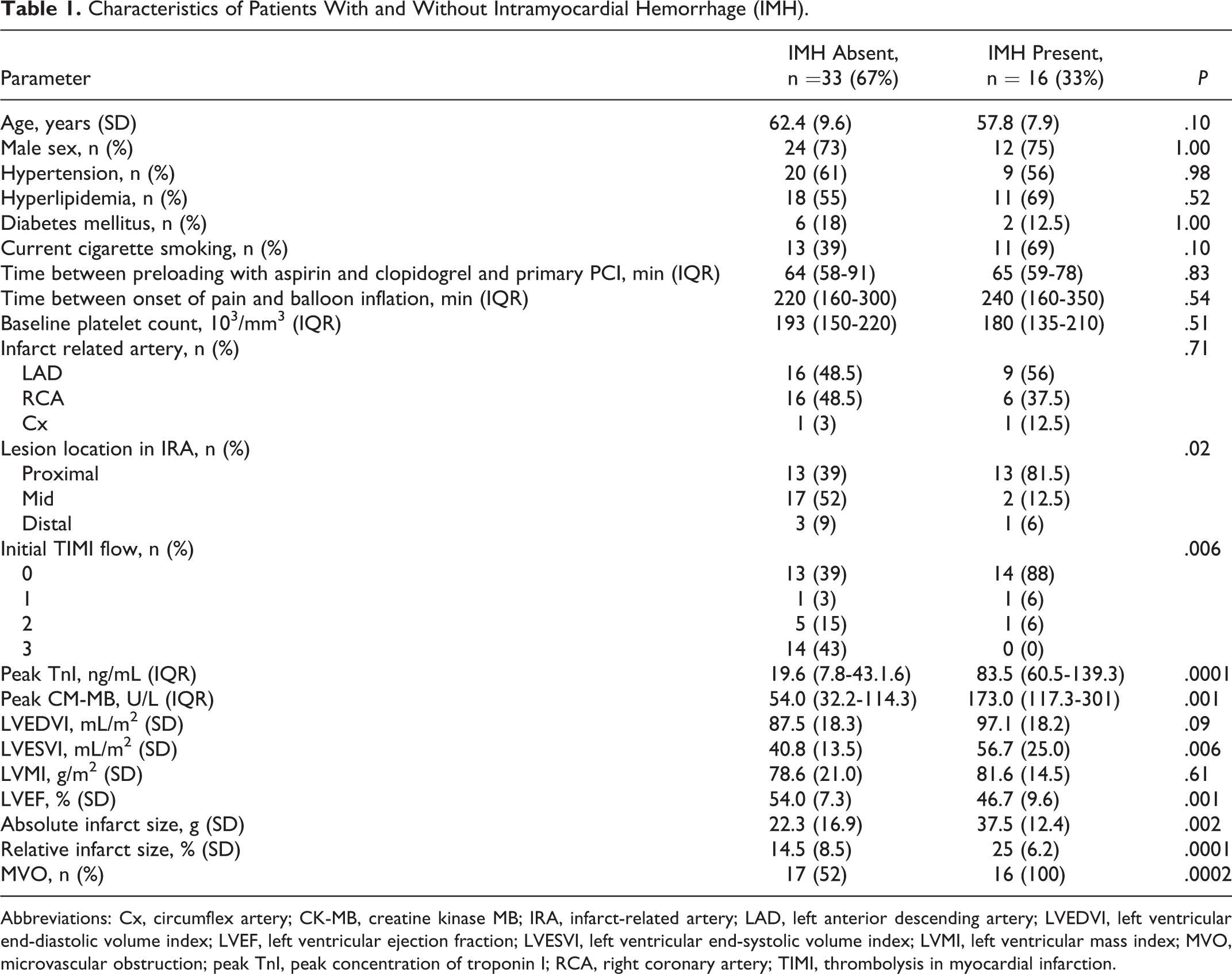
Abbreviations: Cx, circumflex artery; CK-MB, creatine kinase MB; IRA, infarct-related artery; LAD, left anterior descending artery; LVEDVI, left ventricular end-diastolic volume index; LVEF, left ventricular ejection fraction; LVESVI, left ventricular end-systolic volume index; LVMI, left ventricular mass index; MVO, microvascular obstruction; peak TnI, peak concentration of troponin I; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction.
Before reperfusion there were no differences in platelet aggregation between patients with and without IMH as demonstrated in Table 2.
Platelet Aggregation Stimulated With Various Agonists in the Cath-lab After Administration of All Antiplatelet Drugs but Before Reperfusion and in the Subacute Phase of ST-elevation myocardial infarction (STEMI).
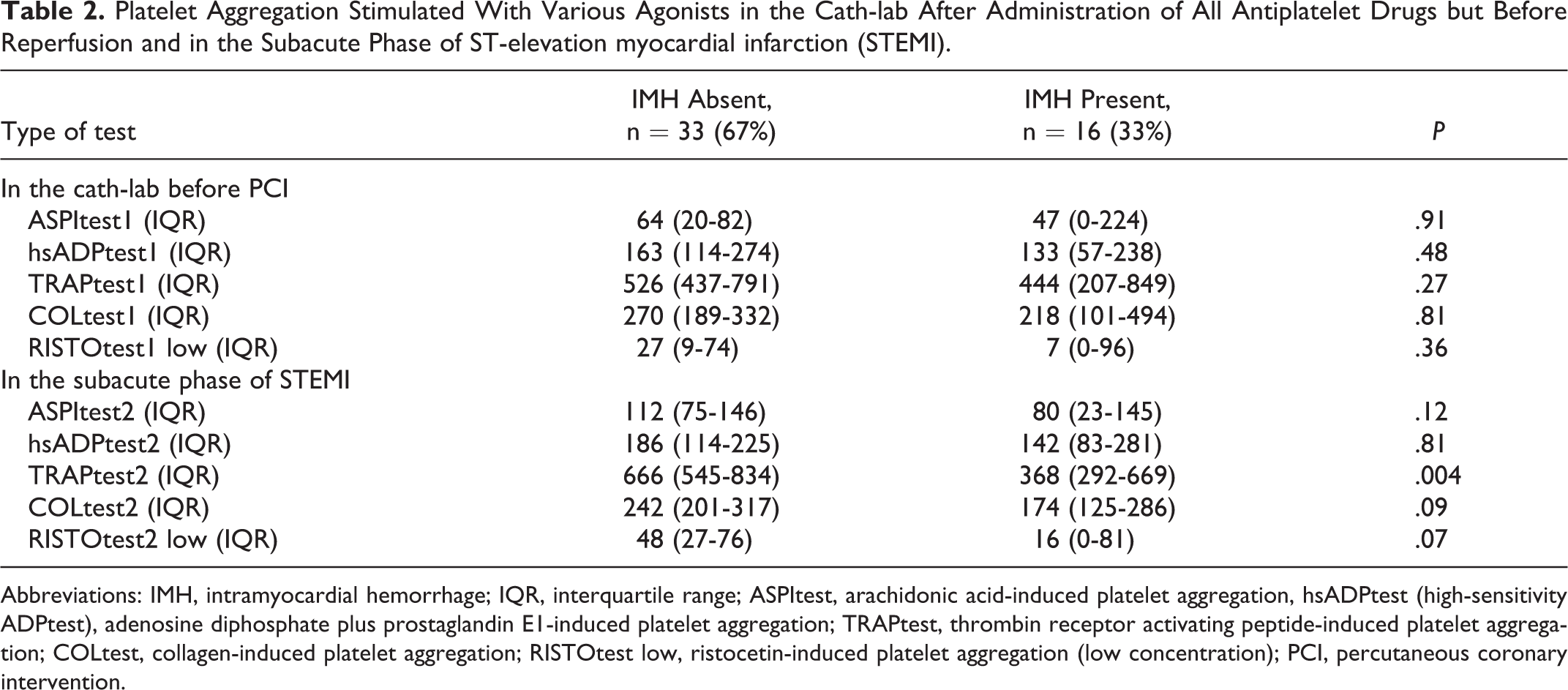
Abbreviations: IMH, intramyocardial hemorrhage; IQR, interquartile range; ASPItest, arachidonic acid-induced platelet aggregation, hsADPtest (high-sensitivity ADPtest), adenosine diphosphate plus prostaglandin E1-induced platelet aggregation; TRAPtest, thrombin receptor activating peptide-induced platelet aggregation; COLtest, collagen-induced platelet aggregation; RISTOtest low, ristocetin-induced platelet aggregation (low concentration); PCI, percutaneous coronary intervention.
Platelet reactivity testing performed in the subacute phase showed that all patients had ASPItest values below 5th percentile of the manufacturer reference values and only 2 (4%) patients had ADPtest values above 5th percentile of the reference values confirming good response to antiplatelet drugs in majority of the patients. Blood sampling at this time point revealed that patients with IMH have a lower platelet aggregation after stimulation with TRAP in comparison to patients without IMH and a trend toward lower platelet aggregation after stimulation with ristocetin and collagen (Table 2).
There was also a weak negative correlation between TRAPtest performed in the subacute phase of STEMI (but not for TRAPtest performed in the cath-lab) and an absolute and relative infarct size (rho = −0.315, P = .03 and rho = −0.351, P = .01, respectively).
Results of a multivariable linear regression model (Table 3) showed that TRAP-induced platelet aggregation and initial TIMI flow were the only factors independently associated with IMH.
Results of a Multivariable Linear Regression Model.a
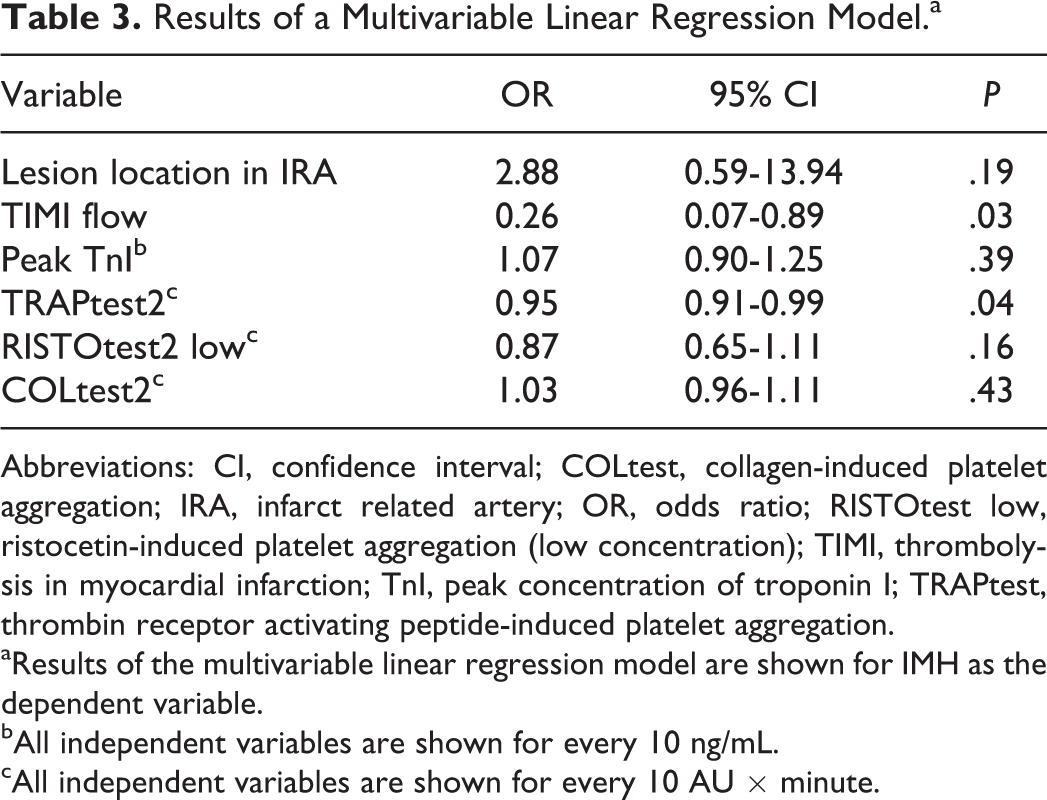
Abbreviations: CI, confidence interval; COLtest, collagen-induced platelet aggregation; IRA, infarct related artery; OR, odds ratio; RISTOtest low, ristocetin-induced platelet aggregation (low concentration); TIMI, thrombolysis in myocardial infarction; TnI, peak concentration of troponin I; TRAPtest, thrombin receptor activating peptide-induced platelet aggregation.
aResults of the multivariable linear regression model are shown for IMH as the dependent variable.
bAll independent variables are shown for every 10 ng/mL.
cAll independent variables are shown for every 10 AU × minute.
Discussion
To our knowledge this is the first study demonstrating that lower platelet reactivity in the subacute phase of STEMI is independently associated with IMH.
At this point we are unable to clearly determine which of the antiplatelet drugs might have been responsible for this phenomenon. Platelet reactivity testing in the cath-lab was performed before the full onset of action of at least some of the administered antiplatelet drugs (around 30 minutes for aspirin, 2-6 hours for clopidogrel and several minutes for Gp IIb/IIIa inhibitors). This time of sampling however reflected the status of platelet inhibition during mechanical reperfusion of the infarct zone. Second time of sampling was set to occur at the time of CMR detection of IMH (3-5 days). At this point, patients exhibited full effect of aspirin and clopidogrel, but the action of Gp IIb/IIIa inhibitors should have been largely diminished. Nevertheless, the difference in platelet reactivity between patients with and without IMH was observed for TRAPtest, which is defined as the test reflecting platelet inhibition with Gp IIb/IIIa inhibitors. Therefore, the most probable explanation of our findings is a stronger and/or prolonged antiplatelet effect of Gp IIb/IIIa inhibitors in patients with IMH. We cannot fully exclude the potential role of clopidogrel and/or aspirin in this process as the degree of platelet activation through ADP or arachdonic acid pathways may influence the final stage of platelet aggregation measured by means of TRAPtest.
The rationale for the use of Gp IIb/IIIa inhibitors in STEMI was to improve coronary perfusion after reopening of the occluded IRA. Indeed initial studies performed before the era of prehospital loading with clopidogrel demonstrated the reduction of short- and long-term mortality with Gp IIb/IIIa inhibitors. 12,13 At that time, addition of Gp IIb/IIIa inhibition before or during primary PCI was filling the time gap between the initiation of therapy with clopidogrel and acquisition of adequate platelet inhibition. Recent data question the value of Gp IIb/IIIa inhibitors in elective and primary PCI in the era of prehospital treatment with high-loading doses of clopidogrel. 14 According to BRAVE 3 a randomized clinical trial, in patients with STEMI treated with primary PCI who received pretreatment with high-loading dose of clopidogrel addition of abciximab did not reduce the infarct size and tended to increase minor bleeding events. 15 We have previously demonstrated that propensity-adjusted analysis did not show an additive effect on 1-year survival for combination therapy with Gp IIb/IIIa inhibitors and clopidogrel in comparison to treatment with clopidogrel alone or GP IIb/IIIa inhibitors alone on top of aspirin. 16 A recent meta-analysis after inclusion of results of new studies demonstrated that routine use of Gp IIb/IIIa inhibitors does not reduce mortality but is related to higher frequency of major bleeding complications. 17 In our opinion, the above findings may indirectly support our hypothesis on the role of Gp IIb/IIIa inhibitors in the pathogenesis of IMH.
An issue of reliability of noninvasive assessment of IMH should be discussed. The CMR is a well-validated, noninvasive method of infarct zone characteristics including detection of myocardial edema (area at risk), IMH, MVO, and infarct zone. 18,19 Studies on animals showed that hypointensive areas within myocardial edema observed on T2-weighted images in acute myocardial infarction depict zones of IMH as confirmed with histopathological studies. 20 It has been demonstrated in humans that the presence of IMH in the subacute phase of STEMI is an independent marker of left ventricular remodeling and worse clinical outcome. 3 –6 This is in line with our findings demonstrating that the presence of IMH is related to larger myocardial infarction and therefore lowers left ventricular systolic performance.
Our study has some limitations. It was performed on a relatively small group of patients; however, most of the studies analyzing patients with STEMI who underwent CMR included a similar number of patients. 18,19 The differences in TRAP-induced platelet aggregation observed in the subacute phase of STEMI are highly significant which lowers the possibility of random finding. For logistic reasons, we assessed platelet reactivity only twice. Additional samples would better depict the platelet reactivity curve on different antiplatelet drugs and could have provided valuable data. Finally, IMH may be potentially related to the overdose of antithrombotic drugs. However, we did not assess the activated clotting time or activated partial thromboplastin time that prevented this kind of analysis.
In conclusion, IMH is related to lower platelet reactivity in the subacute phase of STEMI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the scientific grant of the Institute of Cardiology, Warsaw, Poland no. 2.17/III/10 and financially supported by the Foundation for Polish Science (FNP).
