Abstract
Background:
Hospitalized medically ill patients receiving antithrombotic medications experience increased risk of bleeding. We examined antithrombotic use, bleeding rates, and associated risk factors at 30 days post discharge.
Methods:
This retrospective database analysis included nonsurgical patients aged ≥40 years hospitalized for ≥2 days during 2005 to 2009. Previously cited, validated International Classification of Diseases, Ninth Revision, Clinical Modification codes for major bleeding were used to define clinically relevant bleeding.
Results:
Of the 327,578 patients, 9.1% received antithrombotic medications, of which 3.7% were anticoagulants. Rates of major and minor bleeding were 1.8% and 7.1%, respectively. Preindex gastroduodenal ulcer, thromboembolic stroke, blood dyscrasias, liver disease, and rehospitalization were the strongest predictors of major bleeding. Other risk factors included increasing age, male gender, and hospital stay of ≥3 days.
Conclusions:
Careful consideration of these demonstrated bleed-associated comorbidities before initiating anticoagulation or combining antithrombotic medications in medically ill patients may improve strategies for prevention of postdischarge thromboembolism.
Introduction
Venous thromboembolism (VTE) frequently complicates hospitalization for medical conditions and has historically been considered the most common cause of preventable hospital mortality. 1,2 Although pharmacologic prophylaxis effectively reduces the risk of VTE, only 13% to 34% of eligible medically ill patients receive guideline-recommended anticoagulation at the suggested dose and duration. 3,4 Key elements impacting effective use of thromboprophylaxis in the US health care settings include difficulty in recognizing risk factors for both VTE and bleeding, clinicians’ concerns about the risk of bleeding, and the absence of institutional protocols for VTE prophylaxis. 5 –7 Postdischarge bleeding may be related to the use of antithrombotic agents. Although several recent studies have investigated adverse bleeding events in hospitalized medically ill patients, 8 –13 few have examined risk factors for postdischarge bleeding in this unique patient population. 14 –16
The incidence of postdischarge major bleeding ranges from 0.2% 8 to 1.7% 9 in VTE prevention studies and from 2.8% to 4% in VTE treatment studies. 10,11 A number of factors increase the risk of a bleeding event after hospitalization. Advanced age, impaired renal function, concurrent use of antithrombotic medications, recent trauma or surgery, and comorbid conditions such as heart failure and active cancer have all been associated with an increased risk of inpatient and postdischarge bleeding in patients receiving anticoagulant agents for VTE prophylaxis or treatment. 12 –16 An increased bleeding risk of concurrent use of aspirin with warfarin has been convincingly demonstrated in several clinical trials, meta-analyses, and observational studies. 17 –21
In a bleeding risk assessment model (RAM) from a large registry in hospitalized medical patients, bleeding events were attributed to hypertension, gastroduodenal ulcer, bleeding 3 months prior to hospitalization, low platelet count, admission to intensive care or cardiac care unit, central venous catheterization, and rheumatic disease when these risk factors were present at hospital admission. 15 Although numerous risk factors have been reported, no independent predictors of major bleeding have been consistently identified across the studies. This may be attributed to the use of different data sources and inconsistent definitions of bleeding in this patient population.
Many RAMs have been used to quantify bleeding risk in patients with VTE. 14,16,22,23 Of those, only 1 model is fully applicable to the medically ill patient population, 15 but it is used only in the patient data evident at admission to the hospital. This study has been designed to address this evidence gap by examining the rates and identifying associated risk factors for clinically relevant bleeding at 14 and 30 days post discharge, along with the use of antithrombotic medications in hospitalized medical patients.
Methods
Data Source
This was a retrospective analysis of an administrative claims data set from the HealthCore Integrated Research Database (HIRD), consisting of eligibility, medical claims, and pharmacy claims of approximately 32 million patients from 14 geographically dispersed health plans in the Northeastern, Southern, Midwestern, and Western regions of the United States. The database contains claims information from the largest commercially insured population in the United States and includes health plan organizations such as health maintenance organizations, point of service plans, preferred provider organizations, consumer-directed health plans, and indemnity plans. Overall, the HIRD is representative of the US population in terms of age and gender, with the HIRD being slightly younger, consistent with the fact that the majority of patients are commercially insured (thus, the largest age and gender discrepancies are in the cohorts aged 65 years or older). Validity of the HIRD has been previously documented with refined claims algorithms for severe upper gastrointestinal bleeding, demonstrating up to an 87.8% positive predictive value in true outcomes. 24 Patient clinical settings included community hospitals (urban and rural), private hospitals, regional medical centers (academic and nonacademic), teaching hospitals, and other health care facilities. This analysis was conducted in compliance with state and federal laws, including the Health Insurance Portability and Accountability Act of 1996.
Patient Population
The study cohort included patients aged ≥40 years who had at least 1 inpatient nonsurgical hospitalization with a length of stay (LOS) of ≥2 days for an acute medical illness between January 1, 2005 and December 31, 2009. Medical conditions used to assess eligibility included heart failure, severe lung disease, ischemic stroke, active cancer, acute infection, rheumatologic inflammatory diseases, and endocrine disorders (diabetes mellitus, pancreatitis, and cholecystitis). Patients with these diagnoses were classified as acutely “medically ill patients,” while the other patients who met study eligibility criteria but were hospitalized for conditions different from those mentioned above were categorized as “other hospitalized patients.” Continuous health plan eligibility for the 12-month period prior to the index hospitalization date was also required. Exclusion criteria included a diagnosis of mental disorder, trauma, or VTE during the index hospitalization; hospitalization for obstetric conditions; a surgical procedure; diagnosis of atrial fibrillation during the 12-month preindex period; and use of warfarin (defined as a first prescription fill) 30 days or more post discharge.
Study Analysis
The key outcomes of interest were major and minor bleeding observed at 14 and 30 days after discharge. The other outcomes including postdischarge VTE, rehospitalization (defined as the first postdischarge hospitalization with the same diagnosis as the index hospitalization), and all-cause mortality (captured through the Social Security Death Index database) were assessed at 90 days postdischarge. Because bleeding events were identified through the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes, the definitions of postdischarge major and minor bleeding (Appendix A) were based on previously published studies with minor modifications. 25 In our study, claims code diagnoses, including validated codes for bleeding derived from the literature 26 –29 were validated in a similar way and achieved similar results.
Patient preindex characteristics included demographics, risk factors for bleeding, comorbid conditions (acute infections; neoplasm; endocrine and metabolic disorders; diseases of the blood and blood-forming organs; immune, neurological, and musculoskeletal disorders; and digestive, genitourinary, circulatory, and respiratory system disorders), Deyo-Charlson Comorbidity Index (DCI) score, 30 and concomitant medication use. Evidence of antithrombotic drug use was determined by the presence of one or more pharmacy or medical claims for anticoagulant or antiplatelet agents in 14- and 30-day periods after hospital discharge.
Descriptive statistics were compiled for all the variables. Hazard ratios (HRs) for bleeding risk factors—including age, gender, major comorbidities, length of index hospitalization, postdischarge antithrombotic use, and rehospitalization—were estimated using multivariate survival analysis and Cox proportional hazards models. Statistical software SAS version 9.1 (SAS Institute, Cary, North Carolina,) was used to perform all analyses.
Results
A total of 24,361,751 health care claims records were screened for the study. Of these, 327,578 unique patients (141,628 medically ill patients and 185,950 other hospitalized patients) met the study entry criteria and were included in the analysis (Figure 1). Cohort median age was 68 years; 55.8% of the patients were female. The mean DCI score was 2.0 (standard deviation [SD] =2.2). A significant proportion of the patients had underlying endocrine, metabolic, or immune disorders (46%) and a history of blood dyscrasias (14%) during the preindex hospitalization period. The mean hospital LOS was 4.6 days (SD = 5.2 days; Table 1). Compared to patients who did not receive postdischarge antithrombotic medications at 30 days, those who did tended to be male, older with slightly higher DCI score, and had longer hospital stays.
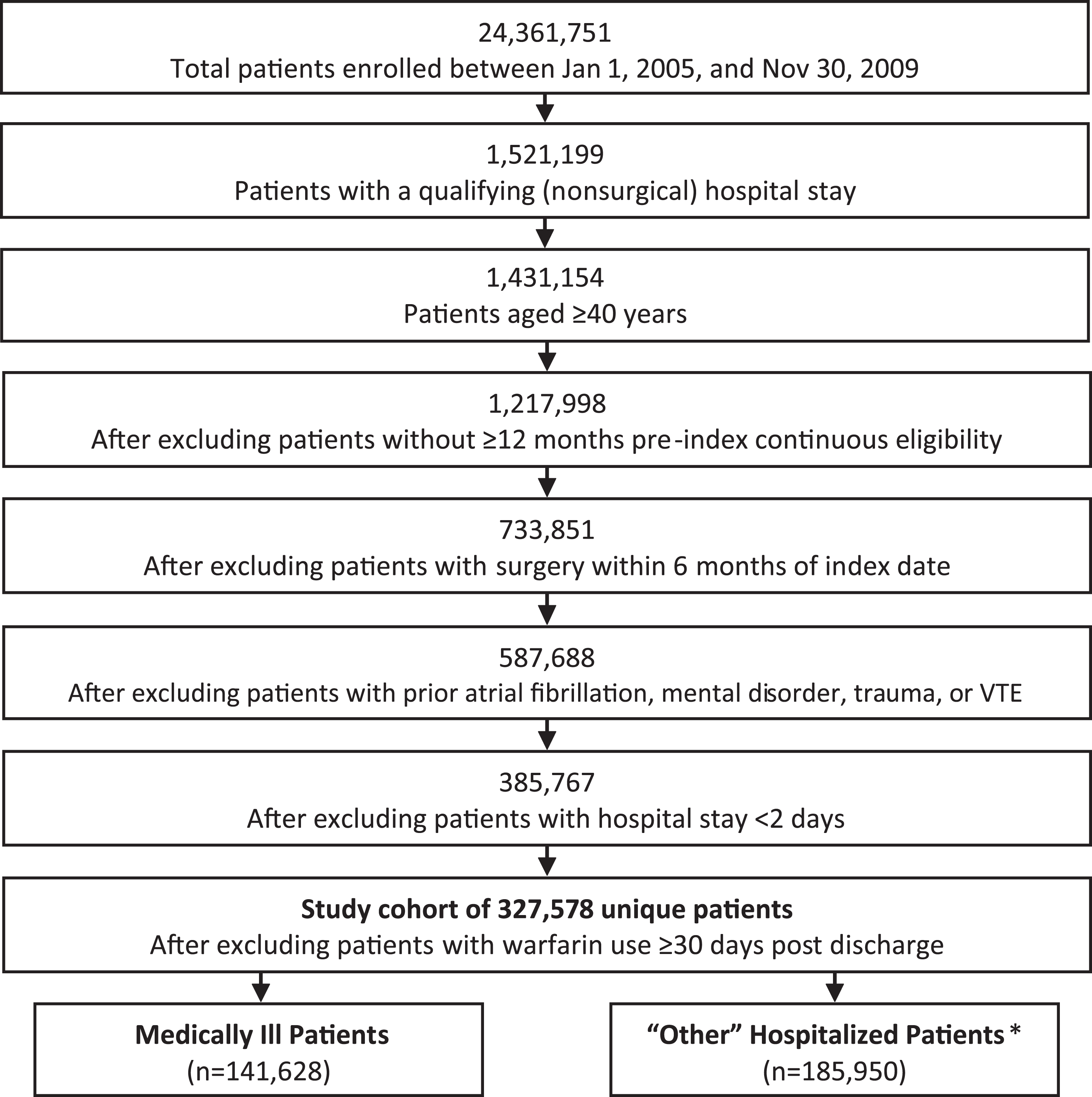
Patient selection flowchart. VTE indicates venous thromboembolism.*The most frequent diagnoses in the “other hospitalized patients” category were respiratory diseases, alteration of consciousness, hallucinations, syncope and collapse, and abdominal or pelvic pain or swelling.
Patient Characteristics and Antithrombotics Use After Hospitalization.
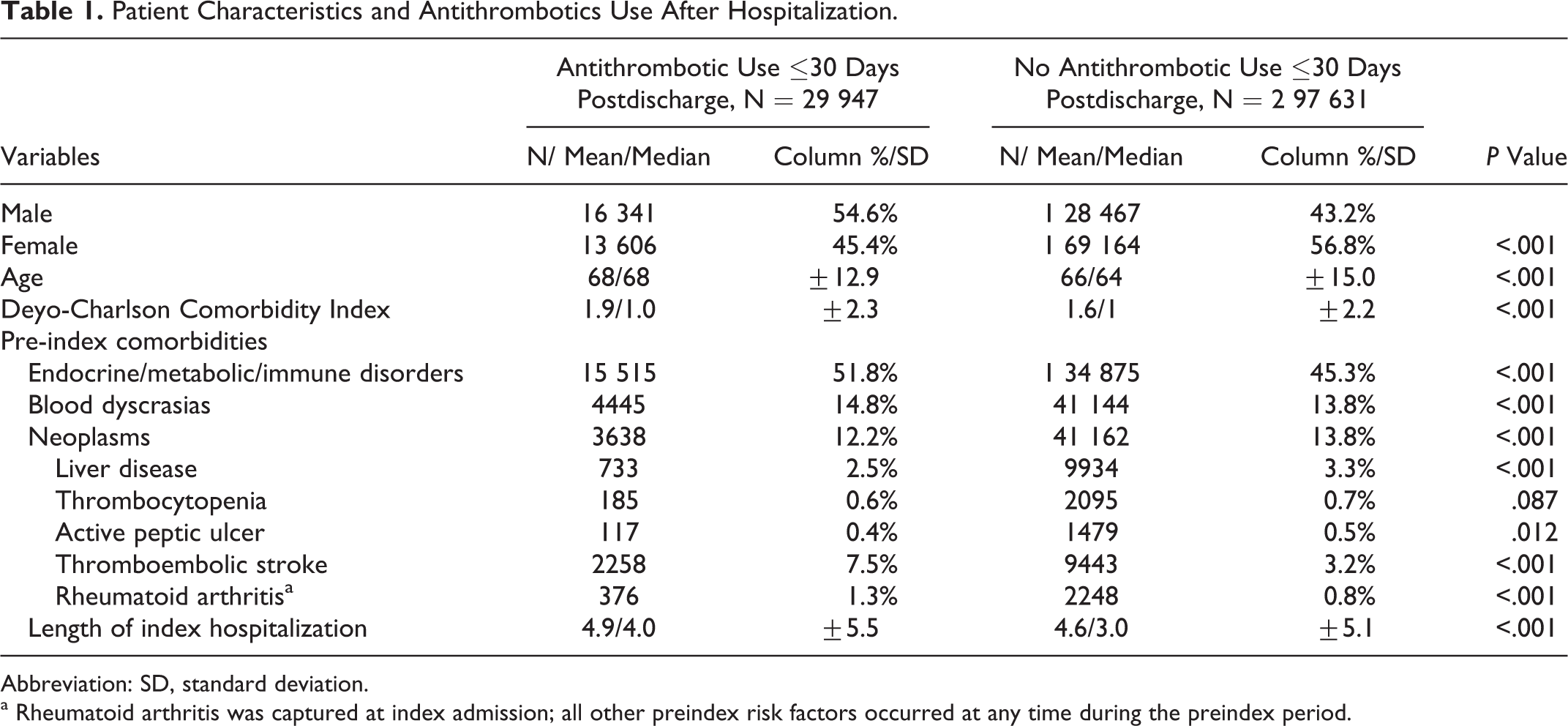
Abbreviation: SD, standard deviation.
a Rheumatoid arthritis was captured at index admission; all other preindex risk factors occurred at any time during the preindex period.
Of the study cohort of 327,578 patients, 9.1% received postdischarge antithrombotic medications, of which 3.7% were anticoagulants. Among the 29,947 patients who used antithrombotic medications within 30 days postdischarge (Table 2), 59.1% used at least 1 antiplatelet agent and 39.1% used at least 1 anticoagulant agent. A small proportion (1.8%) of patients used both anticoagulants and antiplatelets. Clopidogrel and aspirin-dipyridamole were the most frequently prescribed antiplatelets (15,745 and 1863 patients, respectively) while warfarin and the low-molecular-weight heparin (LMWH) enoxaparin were the most frequently prescribed anticoagulants (11,330 and 1332 patients, respectively). The highest utilization of warfarin was observed among patients with underlying heart failure (6.3%), stroke (5.4%), and rheumatic inflammatory disease (4.8%), whereas patients with ischemic stroke had the highest rate of clopidogrel use (18.1%; data not shown). The overall rates of antithrombotic use between the disease categories of “acutely ill medical patients” and “other hospitalized patients” were similar (9.4% and 9.0%, respectively).
Utilization of Antithrombotic Medications Within 30 Days After Hospitalization.a
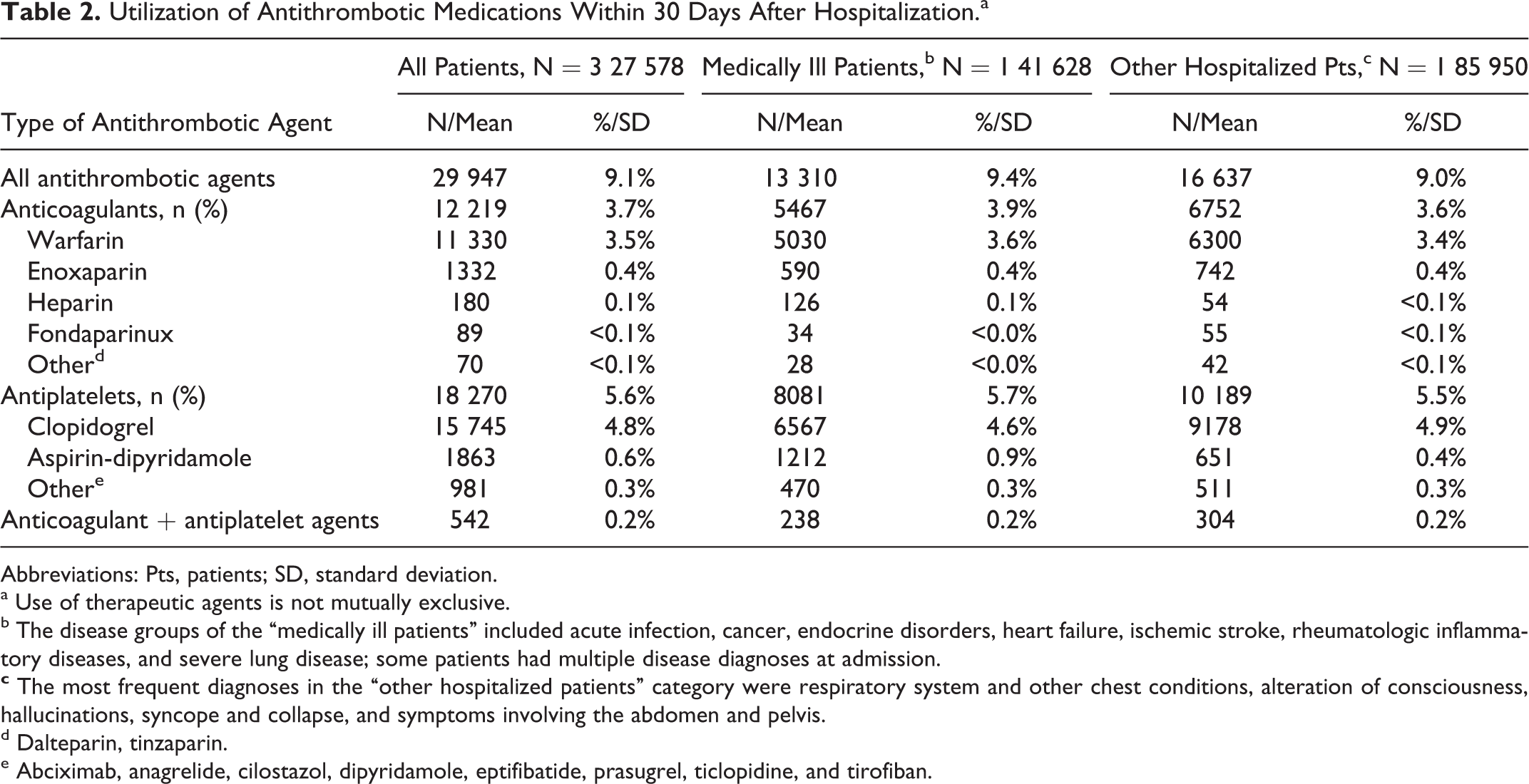
Abbreviations: Pts, patients; SD, standard deviation.
a Use of therapeutic agents is not mutually exclusive.
b
d Dalteparin, tinzaparin.
e Abciximab, anagrelide, cilostazol, dipyridamole, eptifibatide, prasugrel, ticlopidine, and tirofiban.
At 14 days of discharge 3815 (1.2%) patients had a major bleeding event; this number increased to 5951 (1.8%) patients at 30 days. Of the 29,947 patients who received antithrombotic medications postdischarge, 404 (1.4%) and 658 (2.2%) of the patients experienced major bleeding at 14 and 30 days, respectively. Among the patients who did not use antithrombotics, the rates of major bleeding at 14 and 30 days were 1.2% and 1.8%, respectively. The mean times to occurrence of a major bleeding event at 14 and 30 days were 6 and 10 days, respectively (Table 3). Of those patients who received antithrombotic agents and experienced major bleeding at 30 days postdischarge, two-thirds received antiplatelets, nearly one-third received anticoagulants, and a few patients received both anticoagulants and antiplatelets (data not shown).
Outcomes, Time to Event, and Antithrombotic Use After Hospitalization.
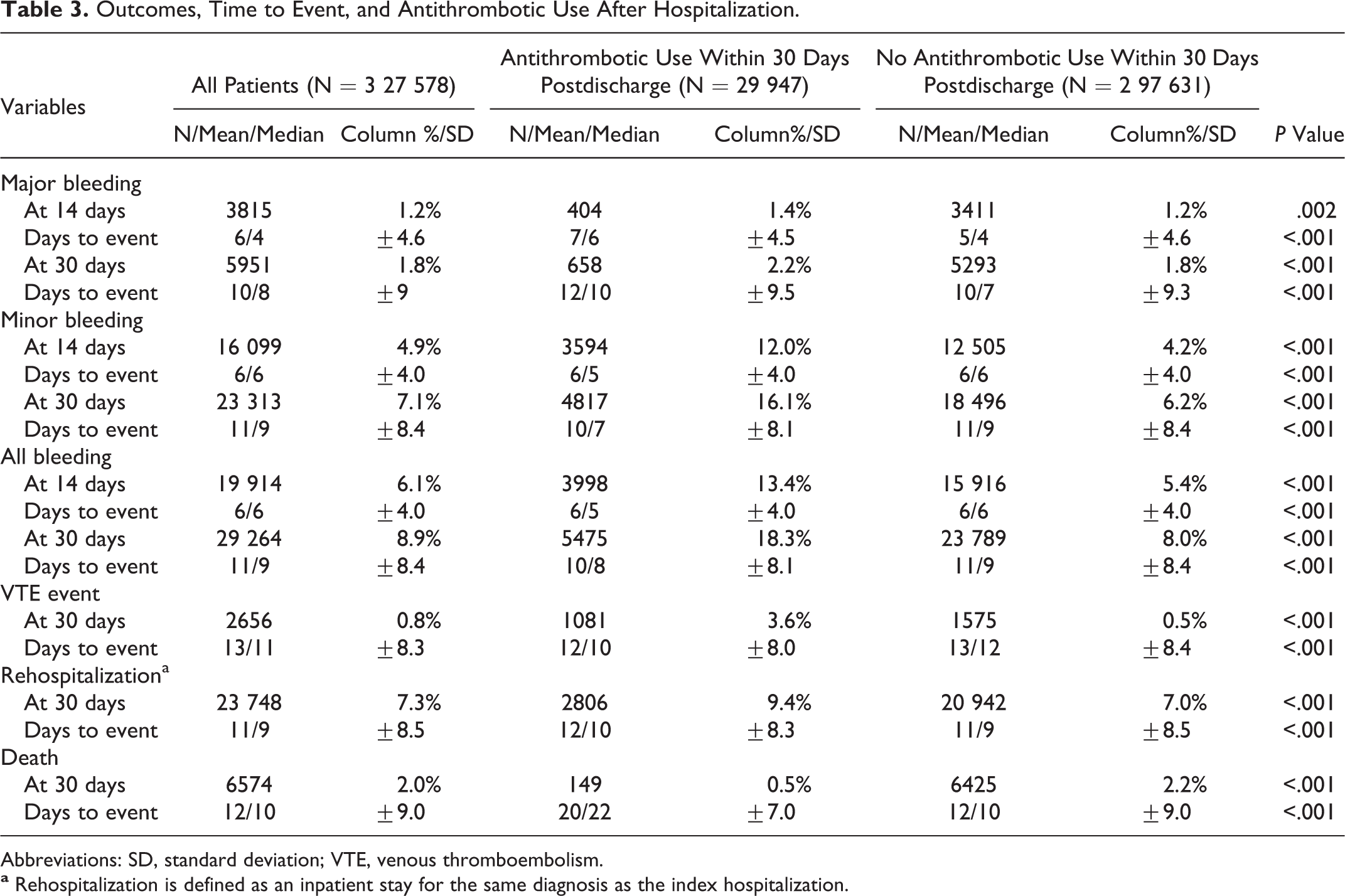
Abbreviations: SD, standard deviation; VTE, venous thromboembolism.
Patients exposed to antithrombotics had a slightly longer mean time to occurrence of a major bleeding event within 30 days postdischarge, compared to those who were not exposed to such therapeutics (12 vs 10 days; Table 3). Overall, at 30 days, the mean time to a postdischarge major bleeding event was 10 days (SD = 9.4 days). Figure 2 shows Kaplan-Meier curves for cumulative survival without major bleeding up to 30 days, stratified by the use of antithrombotic medications.
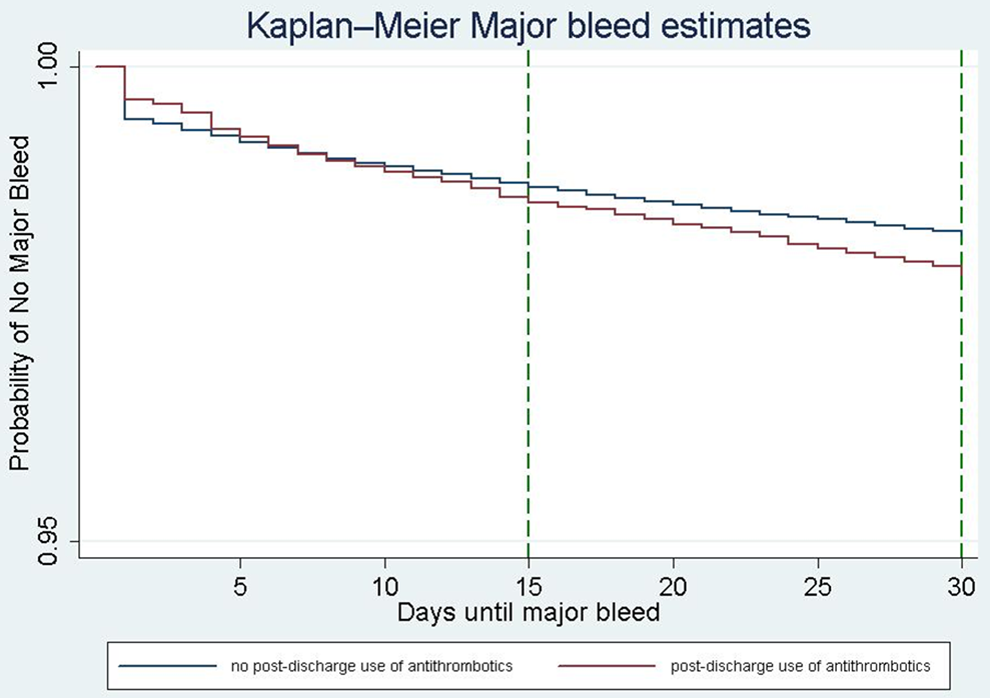
Kaplan-Meier estimation of cumulative survival without major bleeding up to 30 days following discharge.
The rates of minor bleeding at 14 and 30 days were 12% and 16% among patients who used antithrombotics and 4.2% and 6.2% among those who did not use these therapeutics postdischarge, respectively. The time to occurrence of minor bleeding events at 14 and 30 days was similar for both the patient groups, 4 and 8 days, respectively.
Approximately 1 of 14 (7.3%) of patients was readmitted to the hospital with the same diagnosis as the index hospitalization (ie, other than bleeding) during the 30 days postdischarge. A higher proportion of the patients who received antithrombotics was rehospitalized than those who did not receive such therapeutics (9.4% vs 7%; P<.001). The opposite pattern was observed with respect to postdischarge short-term (at 30 days) all-cause mortality; a higher death rate occurred in those without post-discharge antithrombotic drug exposure than in those who were exposed to it (2.2% vs 0.5%; P <.001; Table 3). Among patients who received antithrombotic medications compared to those who did not, the VTE rates at 30 days were 3.6% vs 0.5%, a difference that was statistically significant (P < .001).
After adjusting for differences in patient characteristics via multivariate analyses, several independent predictors of major bleeding at 30 days postdischarge were identified (Table 4). At 30 days, the strongest predictor of major bleeding was rehospitalization (with the same diagnosis as the index hospitalization; HR = 2.37, 95% confidence interval [CI] 2.23-2.56), followed by preindex gastroduodenal ulcer (HR = 2.08, 95% CI 1.67–2.59), thromboembolic stroke (HR = 1.7, 95% CI 1.55-1.88), blood dyscrasias (HR = 1.69, 95% CI 1.59-1.8), and liver disease (HR = 1.4, 95% CI 1.24-1.58). Other risk factors included increasing age, male gender, and an index hospital stay of ≥3 days. Interestingly, a preindex cancer diagnosis appeared to have a protective effect (HR = 0.68, 95% CI 0.61-0.75), while postdischarge use of antithrombotic medications did not reach statistical significance as a predictor of major bleeding at 30 days (Table 4).
Multivariate Survival Analysis: Predictors of Major Bleeding Within 30 Days After Hospitalization.
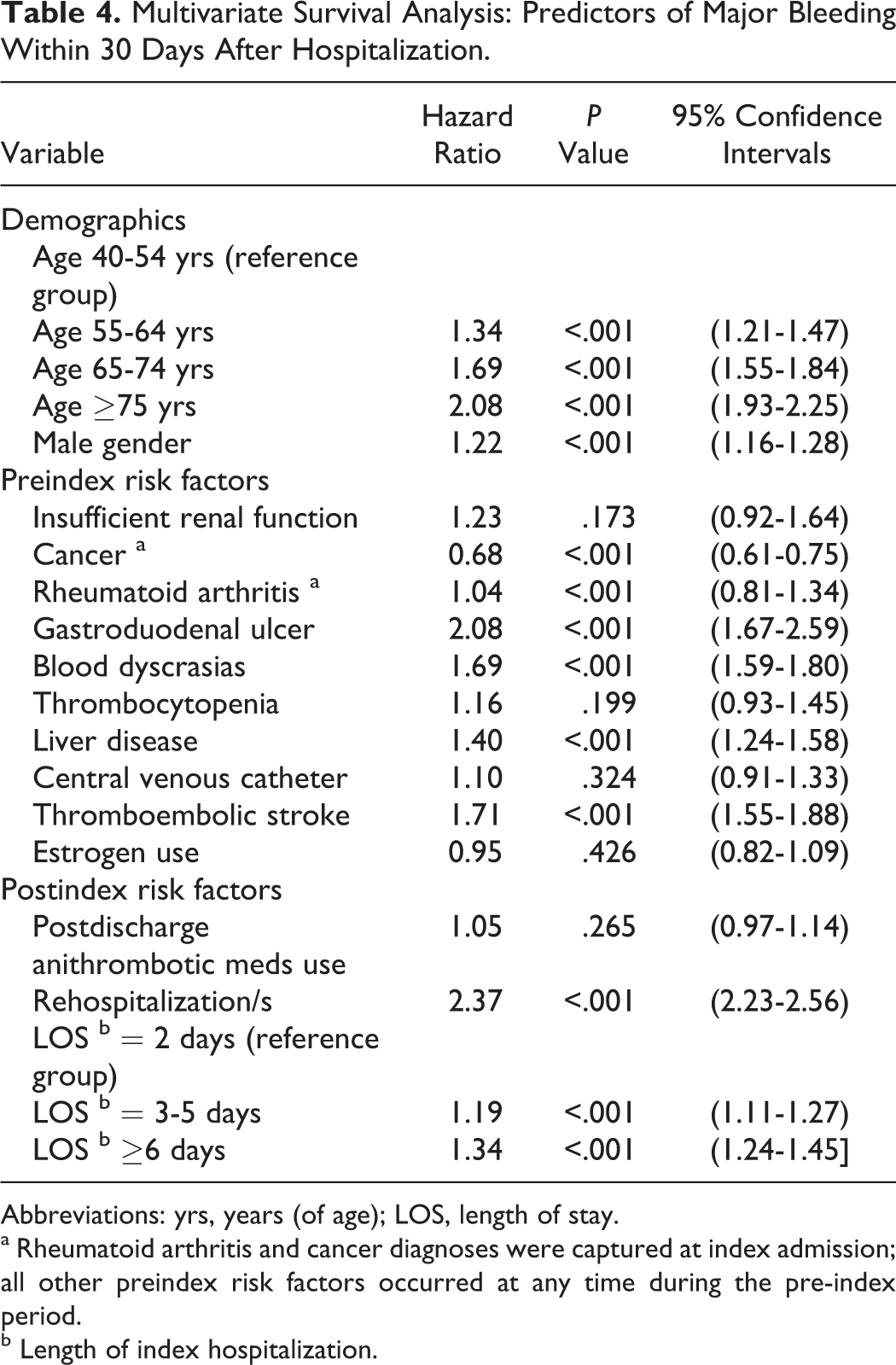
Abbreviations: yrs, years (of age); LOS, length of stay.
a Rheumatoid arthritis and cancer diagnoses were captured at index admission; all other preindex risk factors occurred at any time during the pre-index period.
b Length of index hospitalization.
Discussion
The current study extends our understanding of the risk factors for major bleeding and the therapeutic decisions for prevention of postdischarge thromboembolism following hospital discharge in medically ill patients. At 30 days, all and major bleeding rates of 8.9% and 1.8% were observed, with 9.1% of the patients receiving postdischarge antithrombotics in the study cohort. The primary predictors of postdischarge major bleeding identified in this study were rehospitalization for reasons other than bleeding, preindex gastroduodenal ulcer, thromboembolic stroke, blood dyscrasias, and liver disease along with several other risk factors such as increasing age, male gender, and an index hospital stay of ≥3 days. These results add to the findings of previous work in this field. 14 –16 The postdischarge major bleeding risk factors from this analysis show a great degree of overlap on 5 variables at 30 days with those included in the inhospital bleeding RAM derived from the International Medical Prevention Registry On Venous Thromboembolism (IMPROVE), 5 and they also coincide with some of the risk factors contained in the IMPACT-ILL 5 and Padua Prediction Score VTE RAMs. 31,32 Additional risk factors listed in the IMPROVE bleeding RAM include a prior history of bleeding, renal failure rheumatic disease, a stay in the intensive care unit, central venous catheter placement, and cancer. 5 This study also found that an index hospitalization of ≥3 days, rehospitalization for reasons other than bleeding, blood dyscrasias, and thromboembolic stroke are independent predictors of postdischarge bleeding. Interestingly, postdischarge antithrombotic use was not a predictor of major bleeding at 30 days. This may be explained by the fact that usually the postdischarge VTE prophylaxis with anticoagulants in the US patients diagnosed with an acute medical illness is suboptimal and provided for a rather short duration. 33 In addition, only 0.2% of the study patients used a combined anticoagulant–antiplatelet therapy after hospitalization. It was somewhat surprising to find out that the diagnosis of cancer during the preadmission period was a protective factor against major bleeding at 30 days. This finding is, however, consistent with previous reports, which showed that the use of VTE prophylaxis in patients with cancer is complicated by the fact that although they are at an increased risk of VTE, they are also at an increased risk of bleeding. 34,35 Therefore, patients with cancer are significantly less likely than noncancer patients to receive thromboprophylaxis prior to VTE occurrence. 36 In summary, our findings about the risk factors for major bleeding have important implications for existing bleeding RAMs for medically ill patients. After validation by other studies, they can be used to refine the existing RAMs for use in other clinical settings. 15,32
The rates of postdischarge major bleeding reported here are consistent with previously noted clinical studies. 8,9,15 However, overall bleeding rates appear somewhat higher than those described in the literature. This can be explained by the type of therapeutic exposure that, in addition to anticoagulants, included frequent use of antiplatelet agents, known risk factors for gastrointestinal bleeding, especially when used concomitantly with steroids and nonsteroidal anti-inflammatory drugs. 37,38 Additionally, this may be due to the difference in “real-world” rates of bleeding compared to the bleeding events reported by carefully controlled clinical studies.
In this study, many patients who were at risk of bleeding were prescribed antithrombotic medications after hospital discharge. This finding highlights the need for improved bleeding risk assessment approaches and further research to maximize patient care and safety. Use of antithrombotic agents in this patient population, and likely in other patient groups, should be guided by risk stratification for both VTE and bleeding in order to identify the best therapeutic approach to patient care.
Although 9% of the study population received antithrombotic agents, only 3.7% of the patients received an anticoagulant agent alone (predominantly warfarin or enoxaparin). These findings are consistent with previous reports of low rates of anticoagulant use for postdischarge prevention of thromboembolism in this patient setting. 3 –5,39,40 The American College of Chest Physicians (ACCP) recommends LMWH, unfractionated heparin, or fondaparinux as VTE prophylaxis for hospitalized medically ill patients, with mechanical thromboprophylaxis recommended only for patients in whom anticoagulants are contraindicated. Furthermore, the ACCP does not recommend extending VTE prophylaxis after discharge. 41 In our study, despite the strict exclusion of patients already receiving chronic warfarin therapy, warfarin was still the primary antithrombotic agent used postdischarge.
A key finding was that higher rates of postdischarge adverse outcomes, including bleeding, VTE, and rehospitalization (for reasons other than bleeding), were observed in patients who received antithrombotic agents compared to those who did not. This appears to suggest that clinicians tend to prescribe postdischarge antithrombotics for patients they consider to be at higher risk of postdischarge morbidity. In support of this interpretation, the preindex DCI score clearly showed statistically significant higher comorbidity scores, older age, and longer hospital stay in patients with postdischarge antithrombotic use than in those without such use. Hence, we hypothesized that practitioners prescribe antithrombotic agents, including anticoagulants for VTE prophylaxis, more frequently for patients with more severe medical conditions than for those with less severe medical conditions. On the other hand, the fact that the all-cause mortality was significantly lower among patients with the use of postdischarge antithrombotic agents than among those without such usage may in part reflect reluctance among physicians to administer antithrombotic medications to patients with terminal illnesses.
The strength of this study was the use of an integrated health care claims database that allowed access to a large, diverse patient population. This enabled robust multivariate analyses over multiple disease categories in hospitalized medical patients. Study limitations included the retrospective design, which may have allowed for classification bias. For example, the use of aspirin is likely underrepresented in this study due to the fact that it is an over-the-counter drug in the United States, and therefore is not well captured by pharmacy claims data. Certain comorbid conditions may have not been captured or may have been underreported because health care claims may contain data gaps and coding errors. Reliance on ICD-9-CM codes to identify bleeding events and use grouping antithrombotic drugs all together made the study more inclusive, but it also may have affected the specificity of the findings. Finally, although we adjusted for preindex characteristics in the multivariate analysis, unmeasured confounders, such as the use of over-the-counter medications like aspirin, may still limit statistical inferences from the data. Despite these limitations, we believe the results provide important new evidence on postdischarge utilization of antithrombotics and risk factors for bleeding following hospitalization of medically ill patients which can guide future studies.
Conclusion
To our knowledge, this is the first study to specifically examine postdischarge bleeding rates and clinically relevant bleeding risk in hospitalized medically ill patients. Several identified predictors of postdischarge bleeding overlap with those reported for postdischarge VTE. Use of a risk stratifying approach for both bleeding and VTE would help clinicians guide therapeutic decisions for prevention of postdischarge thromboembolism. It must be recognized, however, that bleeding is multifactorial, and careful consideration of associated comorbidities and concomitant therapies needs to be taken into account prior to either initiating anticoagulation or combining potent antithrombotic drugs in medically ill patients. Physician concerns about increased bleeding risk with the use of antithrombotic medications are not unfounded, as any amount of clinically relevant bleeding is known to prognosticate significant increases in the risk of postdischarge complications, as well as death. Further research aimed to evaluate the benefit–risk ratio of antithrombotics use is warranted.
Footnotes
Appendix A
Acknowledgments
The authors would like to acknowledge Ruth Sussman, PhD, for providing editorial support with funding from Janssen Scientific Affairs, LLC.
Authors’ Note
Dr Mahan contributed to the study design, interpretation of the data and statistical results, and preparation of the manuscript. He will serve as the guarantor of the article, taking responsibility for the integrity of the work as a whole. Dr Spyropoulos contributed to the study design, interpretation of the data and statistical results, and critical review of the manuscript. Dr Fisher contributed to the study design, interpretation of the data collection and processing, and interpretation of the statistical analysis. Ms Stephenson contributed to the study design, protocol development, and interpretation of the data and statistical results. An-Chen Fu performed the data collection, processing, and statistical analyses. Dr Fields contributed to the interpretation of the data and statistical results and critical review of the manuscript. Dr Mills contributed to the study conception, interpretation of the data and statistical results, and critical review of the manuscript. Dr Klaskala contributed to the study conception and design, protocol development, interpretation of the data and statistical results, preparation and critical review of the manuscript, obtaining funding, and supervising the project, before his untimely death in December 2012. Data analyses were performed at HealthCore, Inc. All authors had full unrestricted access to the data. All authors participated in drafting and revising the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Maxine Fisher, An-Chen Fu, and Judith Stephenson have no conflicts of interest to declare. Alex Spyropoulos has served as a consultant for Boehringer Ingelheim, Johnson & Johnson (J&J), Eisai, Bayer, and Astellas, and has served in advisory committees for Bristol-Myers Squibb, Boehringer Ingelheim, and J&J. Charles Mahan has received investigator-initiated grants from Sanofi-Aventis and funding from the North American Thrombosis Forum Traveling Fellowship Award, served as a consultant for Sanofi-Aventis, Boehringer Ingelheim, J&J, Polymedix Inc., Leo, and Eisai, and served as a speaker for Sanofi-Aventis, Boehringer Ingelheim, and Eisai.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Janssen Scientific Affairs, LLC.
