Abstract
Background: Endothelial dysfunction marker, von Willebrand factor (vWF), is physically connected with osteoprotegerin (OPG) in the Weibel-Palade bodies. We aimed to compare the effect of unfractionated (UFH) and low-molecular-weight (LMWH enoxaparin) heparin used as anticoagulants during hemodialysis (HD) on plasma levels and relationships of OPG, soluble receptor activator of nuclear factor κB Ligand (sRANKL), and vWF. Methods: Totally 21 clinically stable chronic HD patients were randomly assigned to either enoxaparin (n = 10) or UFH (n = 11) anticoagulation and followed prospectively for 12 weeks before crossing over to the alternate therapy for further 12 weeks. The OPG, RANKL, and vWF levels were measured at T0, T10, and T180 of HD session after each period of evaluation. Results: The baseline sRANKL level was higher under UFH treatment. Its over-HD level does not behave significantly different under enoxaparin and UFH treatment. Plasma OPG levels expressly changed during both enoxaparin (χ2 analysis of variance [ANOVA] = 31.13, P < .016) and UFH (χ2 ANOVA = 8.26, P = .016) anticoagulation, and its increment at T10 and T180 was significantly different between both the heparins. The main negative predictor of OPG concentration was the total cholesterol level (β = −.51, P = .025). von Willebrand factor concentration remained stable during UFH anticoagulation, whereas constant, no significant increments were noticed, under enoxaparin treatment. After 10 minutes of HD, especially under enoxaparin use, a positive correlation between OPG and vWF increase was noticed (P = .03, R = .45). Conclusions: Impact of heparin on endothelial cells and simultaneously on OPG/RANK/RANKL axis reinforces the presumption of the pathophysiological linkage between bone mineralization and endothelial dysfunction in end-stage renal disease.
Keywords
Introduction
Because of the coagulation system activation and risk for clot formation during extracorporeal circulation, hemodialysis (HD) procedure requires temporal anticoagulation. Introduction of unfractionated heparin (UFH) and later low-molecular-weight heparins (LMWHs) was the major breakthrough in HD evolution.
Unfractionated heparin is a series of mucopolysaccharide molecules composed of alternating
Patients with end-stage renal disease (ESRD) are exposed to UFH/LMWHs for many years. Question arises about the safety and side effects of this long-lasting treatment.
Recent years brought new data about osteoprotegerin/receptor activator of nuclear factor κB/receptor activator of nuclear factor κB ligand (OPG/RANK/RANKL) system. 2 –4 This axis is essential for bone remodeling and also involved in regulation of immune and vascular systems. 5 –8
The RANKL acts through its receptor RANK. After binding, RANKL induces intracellular cascade that regulates differentiation, function, and survival of many different cells—osteoclasts’ precursors, mature osteoclasts, dendritic cells, mammary glands, and some of the cancer cells. Osteoprotegerin expressed in different cells and many organs (osteoblasts, kidney, liver, spleen, bone marrow) acts as a decoy receptor that binds RANKL and therefore prevents its interaction with RANK. Therefore, OPG/RANKL ratio is an important determinant of bone mass and skeleton integrity. 9 In animals, OPG protects large vessels from medial calcification 5 and also limits calcification of atherosclerotic plaques. 10 Interestingly, human studies showed positive correlation between serum OPG levels and presence of cardiovascular disease (CVD). 11,12 The question arises whether OPG is just a marker of endothelial damage, it mediates progression of vascular disease, or it is a kind of protective mechanism aimed to limit vascular injury.
Interestingly, in Weibel-Palade bodies of endothelial cells, OPG is physically associated with von Willebrand factor (vWF)—well-known marker of endothelial dysfunction. 13 vWF is a weak predictor of CVD events in the general population, but it strongly predicts CVD mortality in high-risk patients. 14 It presents as a very good predictor of future cerebrovascular events in patients undergoing maintenance hemodialysis. 15 Patients with ESRD are at high risk of atherosclerosis and CVDs, which are the main cause of morbidity and mortality in that group.
During heparin treatment, after heparin injection, rise in OPG concentration is noted either in healthy volunteers 16 or in hemodialyzed patients, 17 and its amount depends on the heparin sort. 16 The OPG comes from both sources—endothelial cells and vascular smooth muscle cells (VSMCs). 18 If OPG and vWF form a complex, after heparin infusion we can expect the increase in blood concentration of both markers; however, the presence and significance of this process are currently unknown.
The aim of this study was to evaluate the influence of 2 different kinds of heparins (LMWH—enoxaparin and UFH) administered during HD procedure on OPG and RANKL plasma concentration and their relationship with vWF levels.
Methods
In all, 21 patients who were undergoing maintained hemodialysis for a median period of 41.5 months (range 4-111) were included. The causes of ESRD were glomerulonephritis (n = 6), interstitial nephritis (n = 5), hypertensive nephropathy (n = 1), diabetic nephropathy (n = 3), secondary amyloidosis (n = 1), APKD (n = 1), acute kidney injury (n = 1), and unknown (n = 3). None of these patients received immunosuppressive treatment (including steroids), oral anticoagulants, and contraceptive drugs. Infections of human immunodeficiency virus (HIV 1/2, hepatitis-B virus (HBV), and hepatitis-C virus (HCV) were excluded. None of these patients had an evidence of liver injury. Four patients had atrial fibrillation. All the patients received erythropoietin. There was no history of any neoplasmatic or pulmonary disease prior and during this study. Patients with acquired or hereditary deficiency of any coagulation factor, who experienced gastrointestinal bleeding during last 12 weeks and patients with deep venous thrombosis were excluded. Clinical and laboratory characteristics of these patients are presented in Table 1.
Clinical and Laboratory Characteristics of the Patients.

Abbreviations: HD, hemodialysis; HDL, high-density lipoprotein; IU, international unit; LDL, low-density lipoprotein; U, unit.
All the patients were dialyzed for 4 to 5 hours, 3 times per week using the double-needle technique, native arteriovenous fistulas, standard bicarbonate buffer (bicarbonate and glucose in patients with diabetes
Prior to the study, all the patients had been anticoagulated with enoxaparin (Clexane, Bellon Rho^ne-Poulenc Rorer, Montrouge, France) during each HD session.
Additionally, fasting blood samples were collected from 16 healthy volunteers (mean age 55.8). The group consisted of 10 women (mean age 54.8) and 6 men (mean age 57.6)
Study Design
The study was performed in conformity with the Helsinki Declaration. Ethics committee approval was obtained and written informed consent was received from each patient. The study lasted for 6 months and was conducted as a prospective, open-label, crossover study. Totally, 21 patients were randomly assigned to UFH (n = 11) or LMWH (n = 10) anticoagulated HD for 3 months; then the patients were switched to another heparin for the next 3 months (Figure 1).
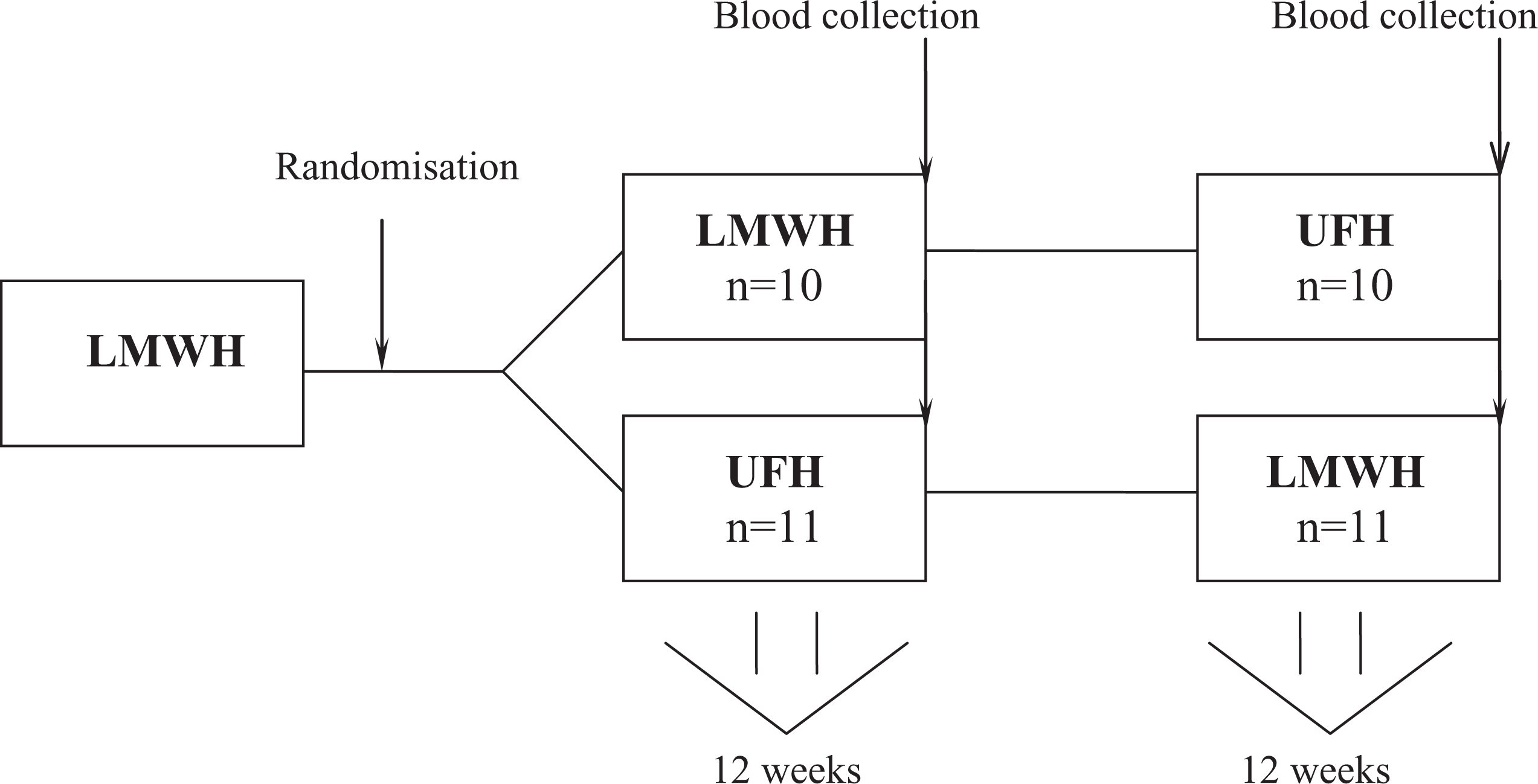
Study design. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin; n, number of patients.
The anticoagulation regiments were UFH—a median bolus of 1500 (range: 500-3500) international unit (IU) through the arterial line followed by a median infusion of 3000 (range: 1500-6500) IU via syringe pomp; and LMWH enoxaparin—a single dose of 50 (20-70) mg via the first-access needle at the onset of HD.
The total dose of UFH was 4881 ± 1916.3 IU (59.2 ± 19.5 IU/kg) per HD session. The infusion was started just after the bolus and stopped 1 hour before the end of HD. The UFH dosage had been individually titrated on the basis of whole-blood activated partial thromboplastin time (APTT) and established during the first 3 sessions after randomization. The goal was to obtain a 2-fold prolongation of APTT at both 30 and 120 minutes of HD compared with the baseline value.
The effective dose of enoxaparin was 0.66 ± 0.14 mg/kg (1 mg = 100 IU of antifactor Xa). It was established by visual inspection of the air trap and the dialyzer on the basis of the clinical guidelines. 19 Clinical clotting was evaluated by nursing stuff by visual inspection after blood draining of the air trap every 60 minutes (1 = no clotting in the trap, 2 = fibrin ring, 3 = clot formation, and 4 = coagulated system) and by visual inspection of the dialyzer at the middle and at the end of each session (1 = clean filter, 2 = a few blood stripes affecting less than 5% of the fibers seen at the surface of the dialyzer, 3 = many blood stripes affecting more than 5% of the fibers, and 4 = coagulated filter).
After 3 months since the randomization procedure, plasma OPG, sRANKL (total), and vWF levels were measured predialysis (T0), at 10 minutes (T10) and 180 minutes (T180) after beginning of the HD session in patients anticoagulated with either UFH or LMWH. Then heparins were switched and the measurements were next repeated after the further 3 months. Each patient was tested against himself or herself.
The study was designed to keep the heparin doses, other HD prescriptions, and pharmacologic treatment stable during the follow-up. All the patients uneventfully completed the study.
Laboratory Procedures
Fasting blood was drawn without the use of a tourniquet through a wide-gauge butterfly needle into monovettes containing a 1.2 mL EDTA. In each case, blood was obtained during a mid-week morning HD session: at T0 from the arterial needle (before heparinization) and at T10 and T180 from the pre-dialyzer port after slowing the blood flow to 100 mL/min for 1 minute. The first 1 mL of the blood was discarded; then 4.5 mL of blood was collected. Tubes were centrifuged 15 minutes later at 2000g for 30 minutes. One-third of the plasma was collected from middle region of the supernatant, aliquoted, and immediately frozen at −80°C.
Plasma OPG and sRANKL levels were determined using enzyme-linked immunosorbent assay (ELISA) kits purchased from BioVendor (Modrice, Czech Republic; Human Osteoprotegerin ELISA and Human sRANKL (Total) ELISA, respectively). Plasma vWF levels were measured using ELISA kit purchased from American Diagnostica Inc (Stamford, CT-USA; Imubind vWF Elisa). Activated partial thromboplastin time was measured using an automated coagulation system and reagents from BioMeárieux (Marcy-I’Etoile, France).
For calculation of the results, a computer and a curve-fitting program were used. The within-and between-assay coefficients of variations were <10%. The T180 values were corrected for hemoconcentration according to serum albumin levels.
Statistical Analysis
The statistical analysis was performed using Statistica software (version 6.0 PL, StatSoft, Tulsa, Oklahoma). Shapiro-Wilk W test of normality was used for data distribution analysis. The normally distributed data were presented as means ± standard deviation (SD), and the skewed data as median (full range).
Normally distributed data were tested using Student t test for dependent and independent data. Skewed data were tested using Mann-Witney and Wilcoxon tests. Analysis of variance (ANOVA) was used to check intragroup comparisons between variables. Bivariate correlations were assessed using Pearson or nonparametric Spearman regression analysis. Stepwise multiple linear regression analysis with both forward selection and backward elimination was employed to evaluate any associations between OPG and multiple independent variables. P values <.05 were considered statistically significant.
Results
Comparision of Study Group and Healthy Controls
Comparison with the control group revealed higher T0 OPG and vWF concentrations in HD patients, regardless of the anticoagulation regimen (P < .10−5 and P = .006, respectively) Concentrations of sRANKL were comparable in HD and controls (P > .05; Table 2).
Comparison of Control Group With Hemodialyzed Patients.
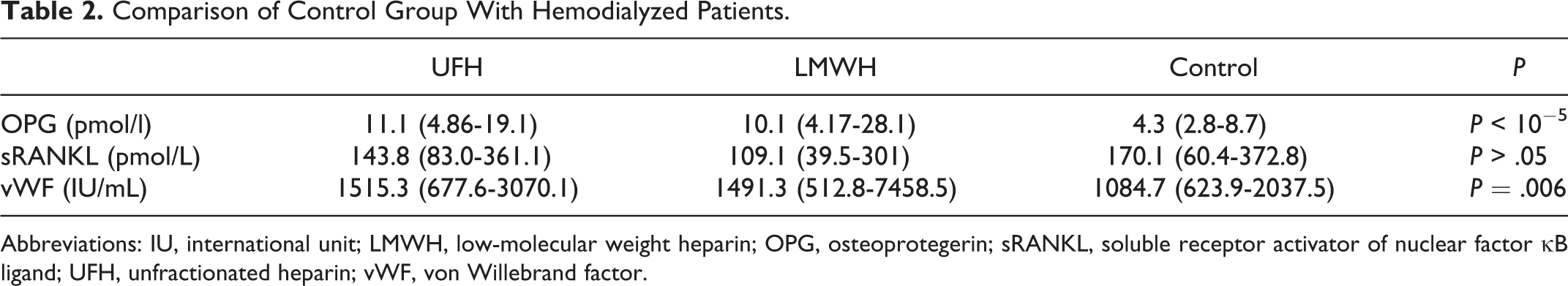
Abbreviations: IU, international unit; LMWH, low-molecular weight heparin; OPG, osteoprotegerin; sRANKL, soluble receptor activator of nuclear factor κB ligand; UFH, unfractionated heparin; vWF, von Willebrand factor.
T0 Plasma OPG, sRANKL, and vWF Levels in Hemodialysed Patients
T0 plasma OPG and vWF levels were similar in both UFH and LMWH. T0 sRANKL was significantly higher during UFH treatment (P = .006; Table 3, Figure 2).
Changes in Plasma Osteoprotegerin, Soluble Receptor Activator of Nuclear Factor κB Ligand, and von Willebrand Factor Levels During Hemodialysis Procedure.
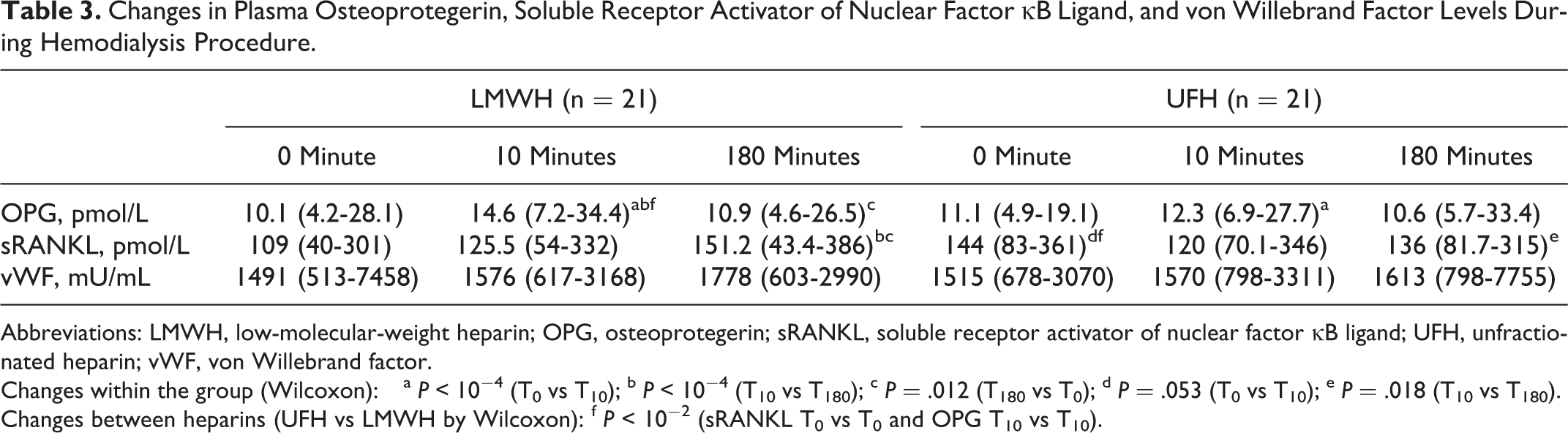
Abbreviations: LMWH, low-molecular-weight heparin; OPG, osteoprotegerin; sRANKL, soluble receptor activator of nuclear factor κB ligand; UFH, unfractionated heparin; vWF, von Willebrand factor.
Changes within the group (Wilcoxon): a P < 10−4 (T0 vs T10); b P < 10−4 (T10 vs T180); c P = .012 (T180 vs T0); d P = .053 (T0 vs T10); e P = .018 (T10 vs T180).
Changes between heparins (UFH vs LMWH by Wilcoxon): f P < 10−2 (sRANKL T0 vs T0 and OPG T10 vs T10).
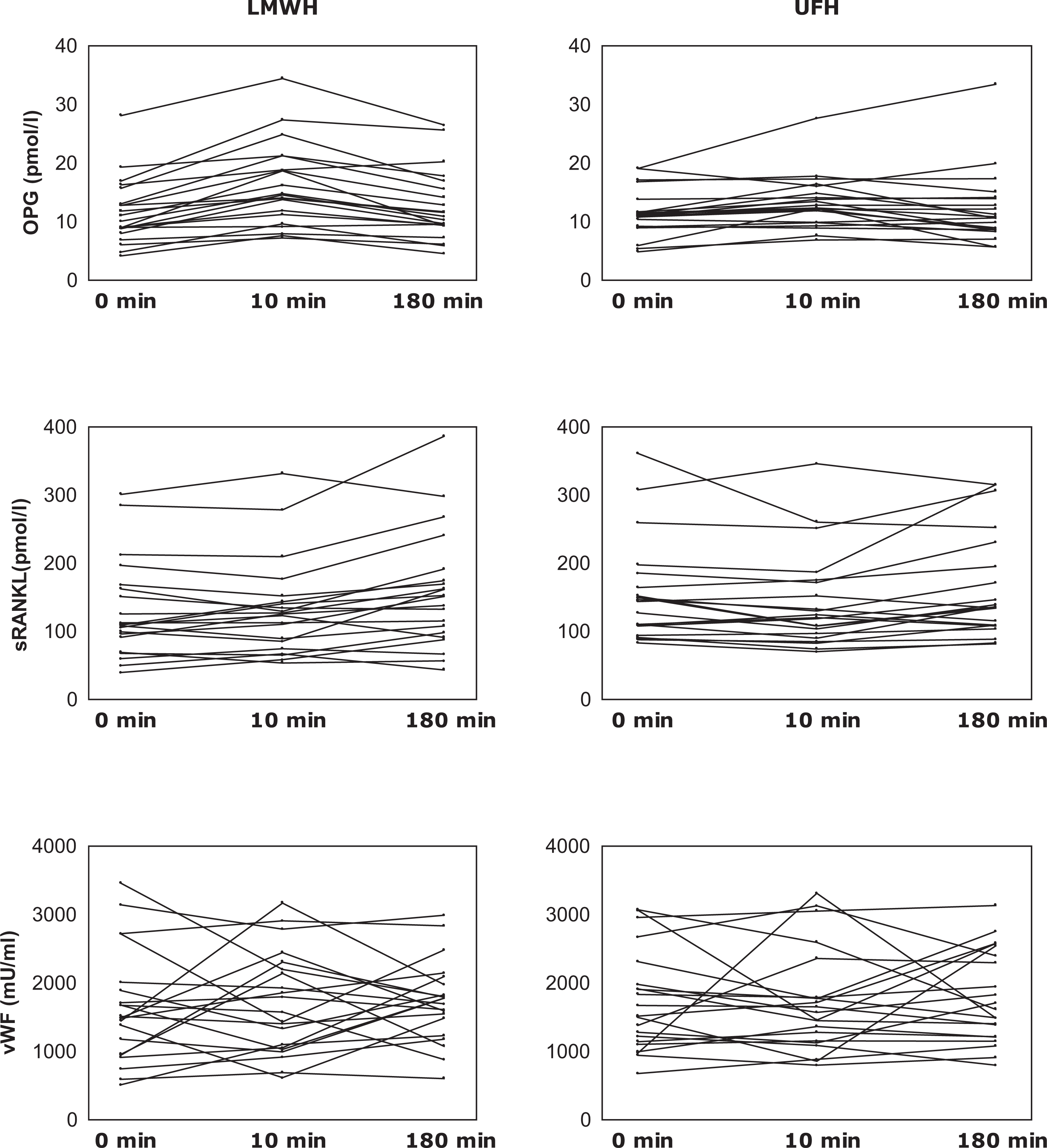
Changes in plasma osteoprotegerin, soluble receptor activator of nuclear factor κB ligand, and von Willebrand factor concentration during hemodialysis. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin; min, minutes.
Changes in Plasma OPG, sRANKL, and vWF Concentrations During Enoxaparin and UFH Anticoagulated HD
During HD with enoxaparin anticoagulant, OPG concentration changed (ANOVA χ2 = 31.13; P < 10−5). It rapidly increased after 10 minutes (P < 10−3) then declined after 180 minutes (P < 10−4) but reminded still higher than in T0 (P = .012). Similarly, in UFH anticoagulated HD plasma OPG concentration significantly increased in the first 10 minutes and then declined (ANOVA χ2 = 8.27, P = .012). The OPG increment during the first 10 minutes of HD was much more prominent after enoxaparin infusion (ΔT10 − T0 for UFH = 1.07 pmol/L; ΔT10 − T0 for LMWH = 4.71 pmol/L; P = .001; Table 3, Figure 2).
Progressive increase in sRANKL level during enoxaparin anticoagulated HD (ANOVA χ2=7.24, P = .026) was noticed. During UFH anicoagulated HD, sRANKL concentration fell in the first 10 minutes after UFH infusion (T0 vs T10, P = .053), then rose until T180 (T10 vs T180, P = .018) to similar concentration like at the beginning of the HD (ANOVA χ2 = 5.56, P = .062). Significant change was observed in the T0 sRANKL levels after switching from enoxaparin to UFH treatment (P = .005). During UFH anticoagulation, vWF concentration remained stable during the whole HD procedure (ANOVA χ2 = 0.08, P = .953). Intradialytic rise in vWF concentration was noticed during enoxaparin anticoagulation but did not reach statistical significance (ANOVA χ2 = 1.52, P = .466). Regardless of no significant vWF changes during enoxaparin anticoagulated HD, we observed its positive correlation with OPG increment during the first 10 minutes (R = .44, P = .038; Figure 3). However, we did not observe such associations under UFH treatment.
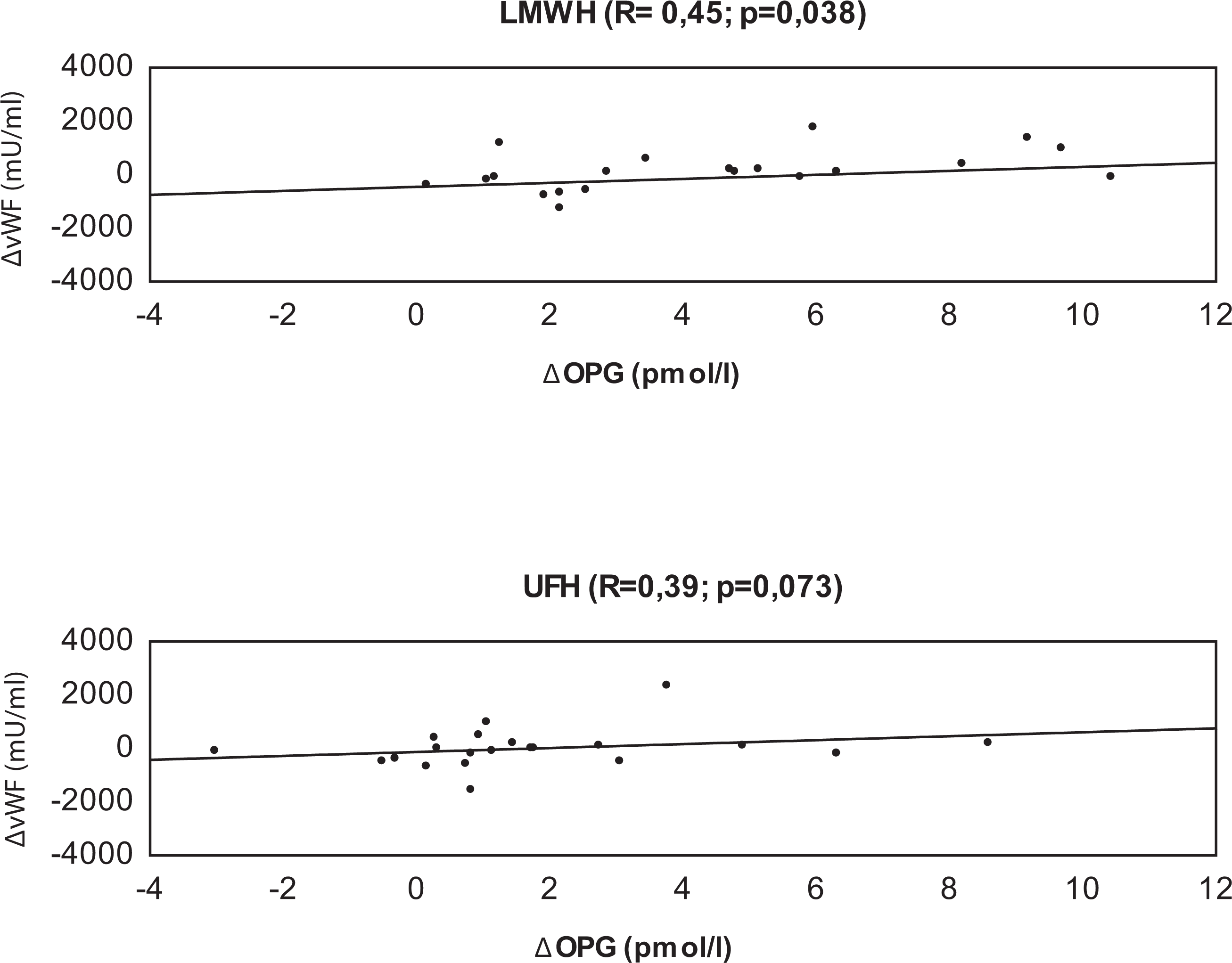
Association between von Willebrand factor and osteoprotegerin increment during the first 10 minutes of hemodialysis procedure. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin; Δ, plasma von Willebrand factor; osteoprotegerin changes during the first 10 minutes of HD compared with baseline (T10 − T0).
The Other Plasma OPG, sRANKL, and vWF Associations
The OPG/sRANKL ratio correlated negatively with cholesterol (Figure 4) and triglyceride (Figure 5) levels. Significant negative correlation, independent of heparin type and period of evaluation, also existed between OPG and cholesterol levels (data not shown). Stepwise multiple linear regression analysis confirmed cholesterol as a main predictor of OPG increment mainly after the first 10 minutes of HD (β = −.51; P = .025).
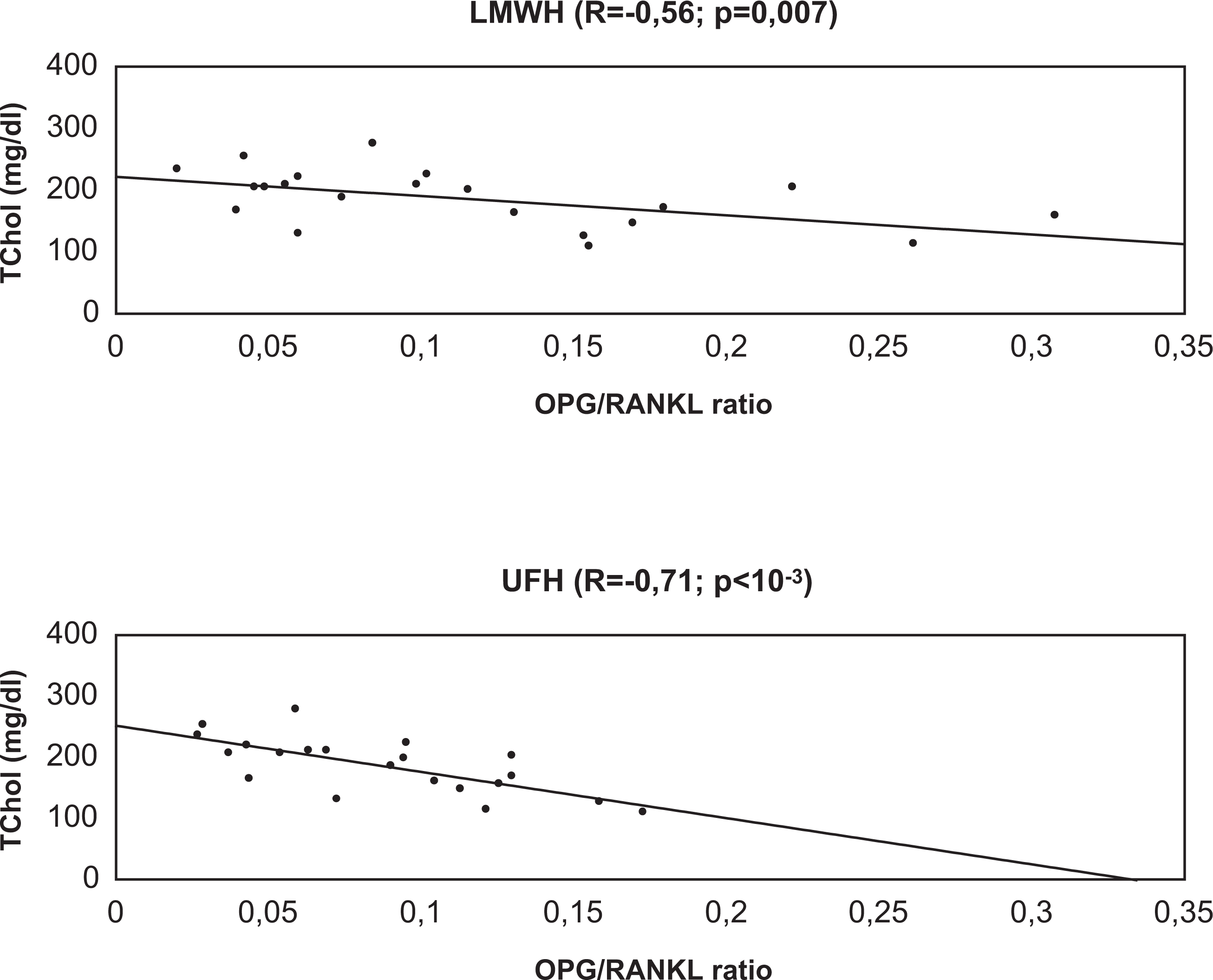
Association between total cholesterol level and osteoprotegerin/receptor activator of nuclear factor κB ligand ratio. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin; TChol, total cholesterol.
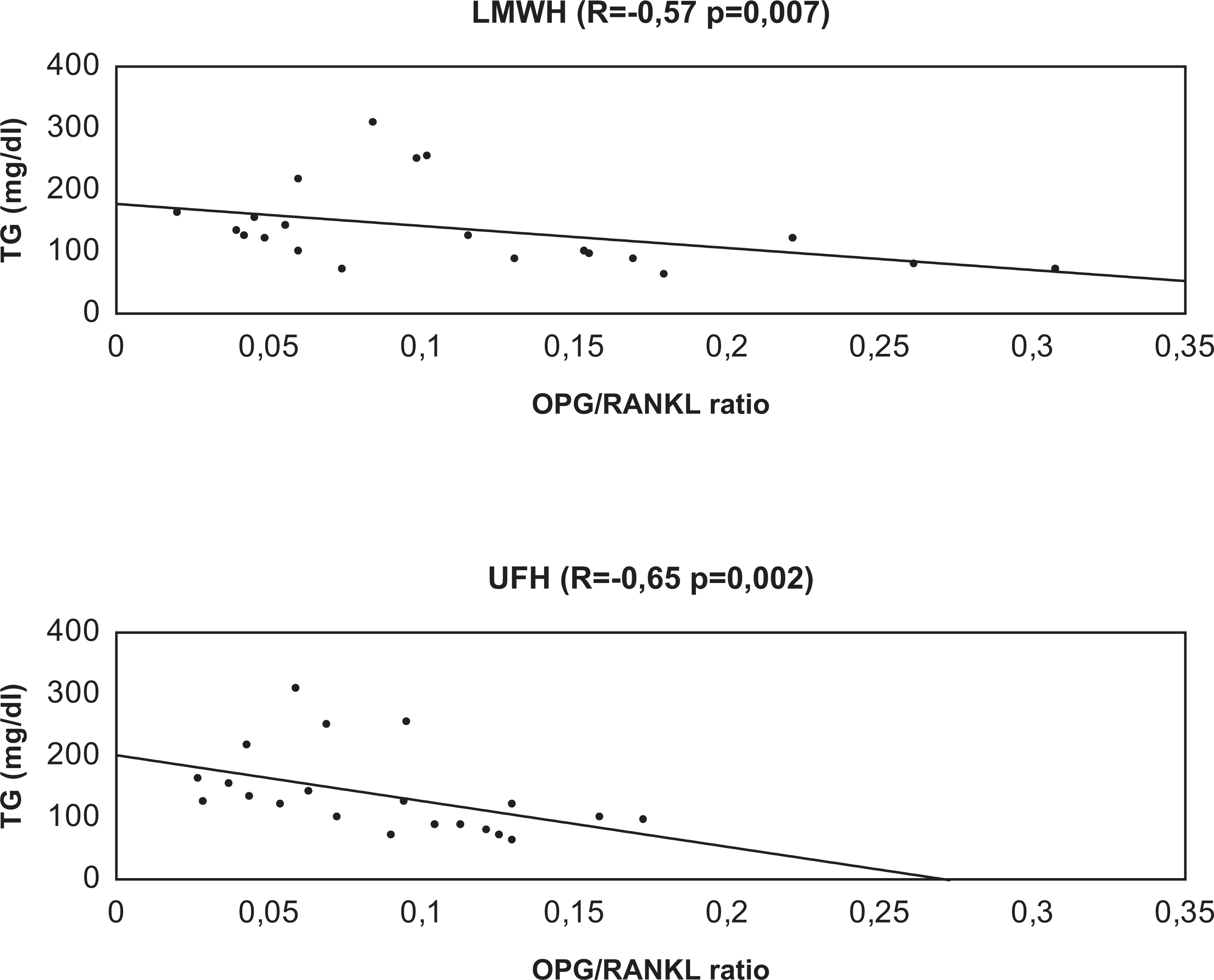
Association between triglyceride level and osteoprotegerin/receptor activator of nuclear factor κB ligand ratio. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin; TG, triglyceride.
Baseline (T0) sRANKL levels correlated positively with triglycerides under both heparin anticoagulation (LMWH: R = .54, P = .012; UFH: R = .47, P = .029).
Interestingly, in patients with atrial fibrillation (n = 4), independent of the type of coagulation, in each period of evaluation we have found significantly lower levels of sRANKL (for LMWH with/without AF: T0 = 64/113 pmol/L, T10 = 71/127 pmol/L, and T180 = 82/153 pmol/L; for UFH with/without AF: T0 = 92/149 pmol/L, T10 = 86/130 pmol/L, and T180 = 106/139 pmol/L).
Discussion
Our work is consistent with many previous studies reporting higher levels of OPG 20 –24 and vWF 25 –27 in patients with chronic kidney disease. As vWF is an endothelial dysfunction marker and OPG is considered to be one, increased levels of these glycoproteins identify population with high cardiovascular risk.
We showed that heparin infusion, independent of its sort, promotes rapid release of OPG into the circulation. Then, in spite of constant exposure to heparin, OPG falls constantly during HD session. Interestingly, during enoxaparin anticoagulation, changes in OPG concentrations were much more prominent in each point of the study. These findings are partly consistent with the previous one provided by Vik et al relating to healthy volunteers. 16 They found that intravenous (IV) UFH bolus as well as subcutaneous (SC) administration of LMWH (dalteparin) causes a prompt increase in OPG. They observed superiority of IV UFH over SC LMWH administration in the mobilization of OPG into the circulation, despite the fact that LMWH injection revealed 3-fold higher anti-Xa activity. 16 In our study, rise in OPG levels was more prominent in LMWH group that probably could be caused by IV administration of enoxaparin.
The sRANKL concentrations under UFH treatment significantly fell in the first 10 minutes and then raised slightly. Conversely, in LMWH group we noticed constant rise in sRANKL levels during HD session. The RANKL stimulates osteogenic differentiation and calcification of VSMCs. 11 Endothelial cells and VSMCs physiologically produce OPG, whereas RANKL is not detected in normal vasculature. 28 Some amounts of RANKL can be detected in the initial stages of atherosclerosis, whereas in calcified vessels and valves expression of RANKL increases. 29 Kiechl et al showed that higher levels of RANKL predicted CVD risk. 30 Taken together, our data of sRANKL increase after heparin infusion may be epidemiologically important. It is likely that OPG rise could be a compensatory response to RANKL increment. Finding of lower sRANKL levels in patients with atrial fibrillation was unexpected, especially in the light of study of Kiechl et al. 30 Because of the small quantity of our group (only 4 patients with atrial fibrillation), these findings need to be confirmed.
Similar design study was conducted by Cianciolo et al relating to hemodialyzed patients anticoagulated with UFH or nadroparin. 10 They found identical increase in OPG concentration just after heparin infusion, regardless of the heparin type, whereas sRANKL did not show significant changes with both heparins used. After heparin administration, OPG returned to basal values between 4 and 8 hours later and the only predictive factor of OPG increase was anti-Xa activity. Interestingly, in our study, multiple linear regression analysis revealed total cholesterol level as the main predictor of OPG rise in the first 10 minutes of HD session (heparin dose was totally eliminated by this statistical model).
Endothelial dysfunction together with dyslipidemia, among others, is strongly related to atherosclerosis development. The association between cholesterol level and mortality is supposed to be reversed in ESRD when compare to general population. 31 The higher the level of cholesterol, the better the prognosis of patient with ESRD. Epidemiological studies demonstrated that higher OPG concentration was strictly associated with higher CVD risk. 11,32 In this study, we observed the rule—the higher the cholesterol was, the lower the levels of OPG. This may probably depict 2 situations—endothelium is less damaged (if we consider OPG as a dysfunction marker) or needs less protection (when we think of OPG as a protective factor to prevent vascular damage). Similarly, OPG/sRANKL ratio correlated negatively with cholesterol and triglyceride levels. If this ratio is the determinant of bone stability; similar dependence could also exist in vasculature.
We did not find a direct association between heparin usage and overdialysis vWF concentration. However, prominent increase of OPG and slight changes in vWF concentration during the first 10 minutes after heparin injection were positively correlated. This effect was especially legible under enoxaparin treatment. In endothelial cells, OPG is physically associated with vWF and in response to for example inflammatory stimuli, this complex is probably rapidly secreted. 13 Moreover, release of OPG into the circulation from Weibel-Palade bodies in response to stimuli seems to be dependent on its association with vWF through OPG’s A1 domain. 33 Although as shown in the in vitro study, 18 most of the circulating OPG are derived from VSMCs, simultaneous changes in OPG and vWF concentrations that we observed in vivo might partially confirm endothelial origin of OPG. Probably, this is the first portion of OPG which leads to the peak concentration derived from endothelial cells, whereas a higher constant level is kept by VSMCs. 33 Physiological relevance of this assumption is not well understood, but it is plausible that vWF may be carrier protein for OPG just like for factor VIII, and in this way OPG may be engaged in vascular injury, inflammation, and also hemostasis.
Concluding, emptying of Weibel-Palade bodies is a response to heparin treatment (especially LMWHs) similar to inflammatory stimuli. It is a logical consequence of its action as the modulator of endothelial function. Heparin empties Weibel-Palade bodies and binds to OPG which inhibits the interaction between OPG and RANKL. The basal OPG level in HD patients is higher than in healthy controls 22,23,34,35 because of impaired endothelial function in uremia and can further increase after UFH or LMWHs infusion.
One of the adverse effects of heparin usage, especially UFH, is osteoporosis. 36 Its pathogenesis is related to OPG/RANK/RANKL axis. OPG-deficient mice develop osteoporosis because of unopposed actions of RANKL to stimulate osteoclastic cell formation, activity, and survival. 4 The RANKL/OPG ratio is an important determinant of bone mass and skeleton integrity. 9 Heparin binds to C-terminal domain of OPG molecule and inhibits the interaction between OPG and RANKL, which results in enhanced osteoclastic bone resorption. If heparin can influence OPG causing osteoporosis, similar interaction could take effect on cardiovascular system as well, for example exacerbating calcification of the blood vessels.
Rise in OPG concentration after heparin infusion may be a protective mechanism. Heparin binds some amount of OPG, so VSMCs and ECs are stimulated to enhance production of protective OPG to restore balance between OPG and RANKL. Similar to bone metabolism, where OPG/RANKL ratio presents as a determinant of bone mass, the same relationship may exist in vasculature.
Conclusion
The role of OPG/RANK/RANKL system in the development of ESRD complications may be crucial. Heparin usage, regardless of its sort (UFH or LMWH) and impact on OPG/sRANKL system, should be considered as a part of pathophysiological process that leads to mineral and vascular disorders, especially in maintenance hemodialysed patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant No 3-54708 (2010) from the Medical University, Bialystok, Poland.
