Abstract
This is a first descriptive, retrospective, observational study aiming to evaluate the changes in bone turnover markers in pregnant women and to assess the effect of a long-term treatment with low-molecular-weight heparin (LMWH), specifically, enoxaparin. Study involved 50 pregnant Caucasian women with thrombophilia. The patients either received prophylactic enoxaparin once daily subcutaneously (N = 35) or were observed without treatment (N = 15). Concentrations of total serum alkaline phosphatase (total AP), bone alkaline phosphatase (bone AP), osteoprotegerin (OPG), and the receptor activator of nuclear factor κB ligand (RANKL) were measured at 15, 25, and 35 weeks of gestation. Total serum AP increased with gestational age. In the group treated with enoxaparin, the percentage of bone AP concentration was lower (P < .05) than in the control group. Serum OPG also increased with gestational age, but no significant difference was found between the groups with- and without treatment. Despite the OPG increased, RANKL did not change.
Introduction
The first report on osteoporosis in association with unfractionated heparin (UFH) treatment was published in 1965. 1 The treatment duration (8 month or longer) and dose (at least 15 000 IU per day) appeared to be important risk factors; the typical clinical manifestations of osteoporosis induced by heparin were vertebral and rib fractures.
In thromboprophylaxis during pregnancy, the UFH is now being replaced with low-molecular-weight heparin (LMWH), and a review of all published case and cohort studies indicate that safety is maintained. 2 Both UFH and LMWH can cause osteoporosis; however, most studies performed show that LMWHs have lower propensity for inducement of osteoporosis than UFH. 3,4 The adverse effect of both UFH and LMWH on bone metabolism has been demonstrated in several animal experiments 5,6 and in vitro studies on human osteoblasts. 6,7 However, only few clinical studies 4,8 have evaluated the risk of osteoporosis during pregnancy.
The effect of pregnancy on bone metabolism is complex. 9,10 Bone formation markers are direct or indirect products of highly active osteoblasts expressed during different phases of development. Alkaline phosphatase (AP), one of the bone formation markers, is an enzyme that plays a role in osteoid formation and calcification.
Osteoprotegerin (OPG) is a glycoprotein that has a heparin-binding site and is a decoy-receptor for receptor activator of nuclear factor κB ligand (RANKL), which is responsible for osteoclast development.
The RANKL/RANK/OPG system is one of the most important discoveries in bone biology in past decade. In addition to osteoblasts, OPG is also expressed in many other tissues. It is a member of the tumor necrosis factor superfamily, 11 which exerts its function as a soluble decoy receptor for RANKL. 12 Many reports support the assertion that the RANKL/OPG ratio is a major determinant of bone mass. 13 It was reported that binding of heparin to OPG inhibits OPG binding to RANK-RANKL complexes, and that OPG reduces the half-life of membrane-bound RANKL. 14 Recently, a significantly higher vascular mobilization of OPG was observed in UFH than in LMWH treatment, indicating that UFH has a higher affinity for OPG than LMWH. 15
Our study aimed to evaluate the changes in bone turnover markers in pregnant women who received thromboprophylaxis with LMWH, specifically, enoxaparin.
Patients, Materials, and Methods
A descriptive, retrospective, observational study involving 50 Caucasian pregnant women with thrombophilia (35 treated, 15 monitored) was performed at the General Teaching Hospital (2006-2008). All women were recruited consecutively from the outpatient clinic of the Thrombotic Center of the General Teaching Hospital in Prague. The study was approved by the Ethics Committee of the General Teaching Hospital. Pregnant patients at risk of thrombosis were treated as recommended by the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. 2 Informed consent was obtained from all trial participants.
Patients
A total of 50 pregnant patients with thrombophilia were divided in 2 groups according to their risk of thromboembolic disease and pregnancy complications. In a control group (N = 15), observation without pharmacological thromboprophylaxis was indicated. In the intervention group (N = 35), pharmacological thromboprophylaxis was administered as recommended by the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. 2 Exclusion criteria were: preexisting known bone disease (osteoporosis, osteoarthritis, osteogenesis imperfecta, Paget’s disease, multiple myeloma), metabolic disease (hyperthyroidism, hyperparathyroidism, hypercortisolism, diabetes mellitus), or use of drugs interfering with bone metabolism (corticoids, antiepileptic drugs, agonist or antagonist of luteinizing hormone) or smoking. The baseline characteristics of pregnant women with and without LMWH prophylaxis are shown in Table 1 . These women did not take a calcium/vitamin D supplement. There were no multiple pregnancies. No women in control group received ever an unfractionated heparin or Coumadin. In the intervention group had 13 women previous Coumadin therapy (with mean lasting 6 month) . None of them has anticoagulation therapy at the beginning of pregnancy.
Baseline Characteristic of Study Participants Comparing Women Who Received Antepartum Enoxaparin (Intervention) With Women Who Did Not Receive Antepartum Enoxaparin Prophylaxis (Control); (Mean, SD)
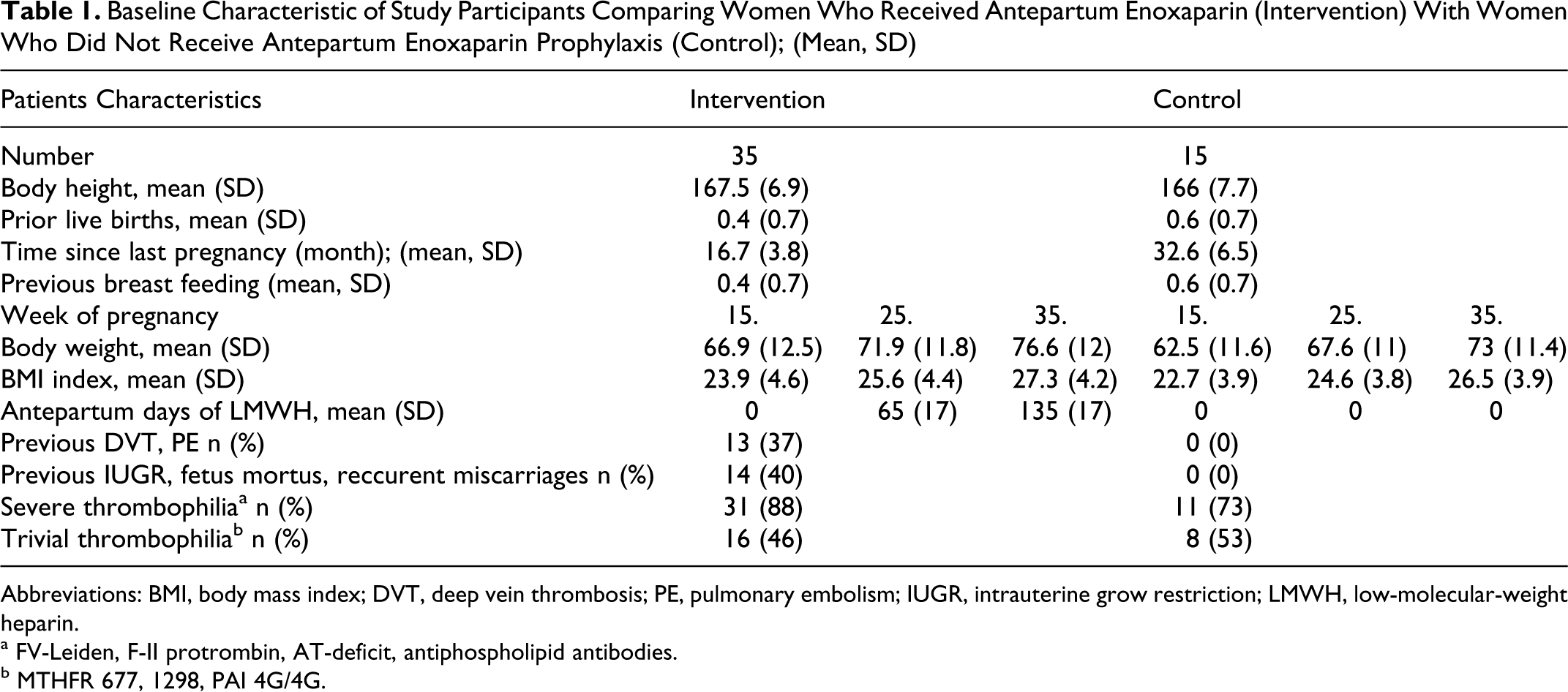
Abbreviations: BMI, body mass index; DVT, deep vein thrombosis; PE, pulmonary embolism; IUGR, intrauterine grow restriction; LMWH, low-molecular-weight heparin.
a FV-Leiden, F-II protrombin, AT-deficit, antiphospholipid antibodies.
b MTHFR 677, 1298, PAI 4G/4G.
Intervention
The treatment started at a concluded 15th week of gestation, as confirmed by date of last menstruation, and continued for 6 weeks after delivery. The patients received enoxaparin (N = 35) once daily subcutaneously. The control group (N = 15) was not treated. Enoxaparin (Clexane, Sanofi-Aventis, Paris, France) was administered at a starting dose of 4000 IU, and the dose was later adjusted based on plasma anti-Xa activity measurements. If the anti-Xa was less than 0.2 IU/mL 4 hours after injection, the enoxaparin dose was increased to 6000 IU. Increasing of doses was necessary in 8 patients with enoxaparin in 35th week of gestation.
Plasma levels of total AP, bone AP, OPG, and RANKL were measured at the 15th, 25th, and 35th week of gestation. In the treated group, blood samples were taken 4 hours after the enoxaparin injection.
Methods
Total alkaline phosphatase was measured by the optimized IFCC method (Bio-la-test®, Pliva-Lachema Diagnostika s.r.o., Prague, Czech Republic). Wheat-germ lecithin activity test−ecithin affinity separation of AP isoforms on an agarose gel−was performed using a commercially available electrophoresis system (Hydragel, ISO-PAL, Sebia, Issy-les-Moulineaux, France); bone specific AP-Elpho was calculated as a fraction of the skeletal isoform multiplied by total AP level obtained from the same sample.
OPG was measured with the Luminex100 analyzer using Human Bone Panel Milliplex Assay Kit (Millipore, St. Charles, MO); the detection limit was 1.42 pg/mL; intra-assay and interassay coefficients of variation (CV) were 6.5% and 6.3%, respectively. RANKL was measured with the Luminex100 analyzer using Human Bone Panel Milliplex Assay Kit (Millipore, St. Charles, Missouri); the detection limit was 4.8 pg/mL; intra-assay and interassay coefficients of variation (CV) were <5% and <6%, respectively
Anti-Xa measurements were performed using the BCS analyzer (Dade Behring, Wien, Austria), using chromogenic factor Xa inhibition assay Coamatic Heparin (Chromogenix Instrumentation Laboratory S.p. A., Milano, Italy).
Statistical Analysis
The statistical program SigmaStat for Windows (Aspire Software International, Ashburn, Virginia) version 3.5 was used for statistical analysis. Descriptive statistics data were determined, and changes of parameters in time were analyzed by 2-way repeated measures analysis of variance using the Holm-Sidak method for data with normal distribution. Friedman repeated measures analysis of variance (ANOVA) on ranks with Tukey test was used for data without normal distribution. Delta scores (postvalues and prevalues) were calculated on selected variables and analyzed by ANOVA for repeated measures. Results were considered significant if the probability of type I error was .05 or less. The correlation coefficient was calculated using the multiple regression test to examine the relationships between the variables (total AP, bone AP, OPG, RANKL) and the anti-FXa activity of enoxaparin. The level of statistical significance was determined to be P ≤ .05 for each test.
Results
The baseline characteristics of pregnant women with and without LMWH treatment are shown in Table 1. No adverse events (eg, heparin-induced thrombocytopenia or allergic reaction) or bleeding complications were observed. No osteoporotic fracture was found during the study. Findings from descriptive analysis of the variables of bone markers at the 15th, 25th, and 35th week of gestation are shown in Table 2 .
Markers of Bone Metabolism at 15th, 25th, and 35th Week of Gestation in Women Who Received Antepartum Enoxaparin Prophylaxis (Intervention) and Women Who Did Not Receive Antepartum Enoxaparin Prophylaxis (control)
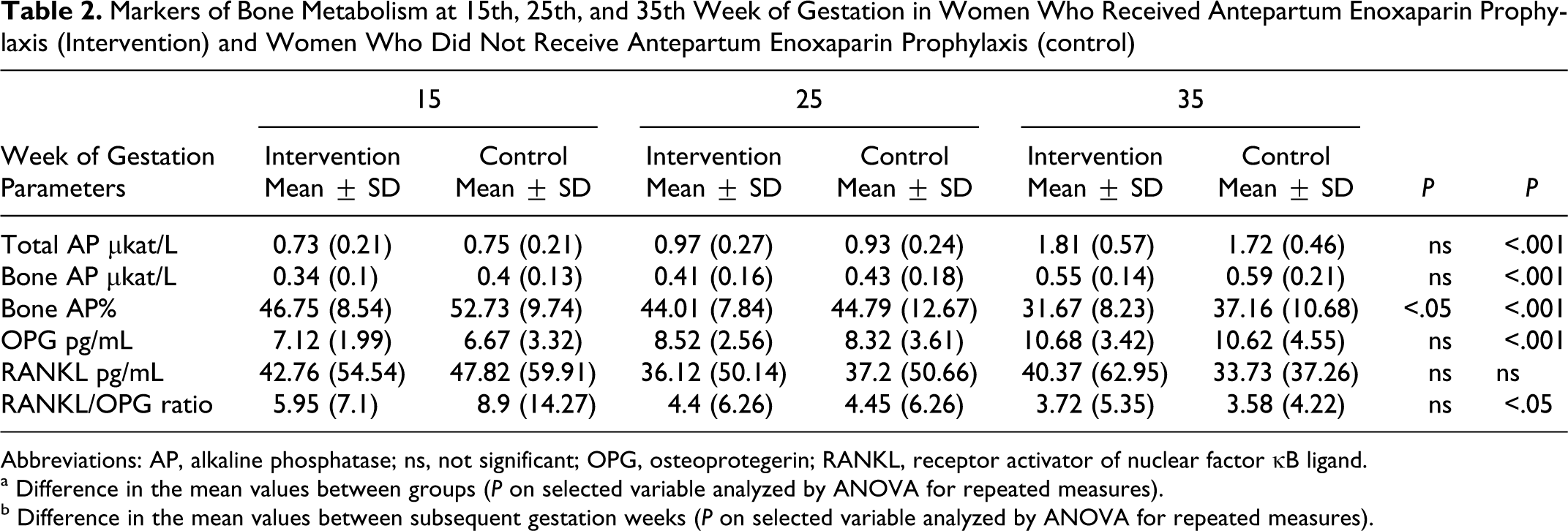
Abbreviations: AP, alkaline phosphatase; ns, not significant; OPG, osteoprotegerin; RANKL, receptor activator of nuclear factor κB ligand.
a Difference in the mean values between groups (P on selected variable analyzed by ANOVA for repeated measures).
b Difference in the mean values between subsequent gestation weeks (P on selected variable analyzed by ANOVA for repeated measures).
There were no significant differences between the 2 groups in age and body height. Women in intervention group had significantly higher body weight (P < 0.05). This concern the weight at 15th week of gestation, the weight evolution along pregnancy was analogous.
Bone Formation
Changes in the levels of bone formation markers are shown in Table 2. Total serum AP increased with gestational age. A significant increase in each assessed gestation week was detected in the following bone formation markers: total AP (ukat/L) and bone AP (ukat/L; P < .001, Table 2). The concentration percentage of bone AP (bone AP% = bone AP [ukat/L]/ total AP [ukat/L]) was significantly lower in the group with enoxaparin treatment (P < .05) than in the control group (Table 2).
Osteoprotegerin/RANKL system
During pregnancy, OPG levels increased markedly (P < .001), but there was no significant difference between groups with or without treatment (Table 2). The plasma OPG level in treated women at 15th week of gestation was 7.12 ± 1.99 pg/mL, which was not significantly higher than that in the control participants (6.67 ± 3.32 pg/mL, P > .05).
Despite the OPG increase, RANKL did not change (P > .05; Table 2).
Anti-Xa activity (IU/mL) was measured 4 hours after the 40-mg enoxaparin administration. There was no significant correlation between anti-Xa inhibition and concentration of the bone turnover markers (P > .05).
Discussion
We evaluated bone turnover marker levels in pregnant women with or without LMWH prophylaxis.
Pregnancy is one of the few situations in modern therapeutic where LMWH is recommended for prolonged use. Antithrombotic therapy is frequently prescribed in pregnancy, and LMWHs, despite their higher cost, are becoming more popular due to their convenient use with prefilled syringes, more predictable dose response, greater bioavailability, and a possibly lower risk of complications. Bleeding complications and heparin-induced thrombocytopenia seem to occur less frequently with the LMWH use than with the UFH 2
Hence, pregnancy is the only situation in which LMWH-induced osteoporosis has been evaluated.
Pregnancy is a particular time of risk of fracture because of the growing fetus deriving all its calcium from maternal sources. 16 Although many clinical factors (eg, age, family history, body mass index, use of steroids, or previous fractures) contribute to a patient’s susceptibility to fracture, the level of bone resorption markers has proved to be an important predictor of a fragility fracture risk. 17 In contrast to bone resorption, the markers of new bone formation showed both increase and a decrease during pregnancy, which is consistent with a state of high bone turnover in pregnancy. 16
In the current study, total AP rapidly increased with each week of pregnancy. The main changes of total AP were seen between the 25th and 35th week of gestation. Triple increase in total AP in pregnancy was due to the increase in placental and bone-specific AP. In reports by others, bone AP was found to be low in the first trimester and either remained low or rise to normal or above normal level in the last trimester. 18 We observed a gradual increase in bone AP (ukat/L) until the 25th gestational week in both groups, but without a significant difference between groups with or without enoxaparin treatment. However, in the concentration percentage of bone AP (bone AP % = bone AP [ukat/L]/ total AP [ukat/L]), there was a significant difference between groups with and without treatment. In the enoxaparin group, the percentage concentration was lower (P < .05) than in the control group.
In a study performed on human osteoblasts, 7 a decline in proliferation and protein synthesis was observed after incubation with dalteparin. The study analyzed osteoblast-specific quantitative gene expression using the semiquantitative Real Time Polymerase Chain Reaction (RT-PCR). There was impaired bone alkaline phosphatase expression due to the dalteparin treatment, whereas fondaparinux showed no such effect. 7 Another study showed that mitochondrial activity and protein synthesis in osteoblasts treated with enoxaparin, dalteparin, or UFH were significantly lower than in those treated with fondaparinux. Similar effects were demonstrated for the matrix collagen type II content and calcification: enoxaparin, dalteparin, and UFH treatment led to a significant decrease of matrix collagen type II content and calcification in concentrations equal or higher than the therapeutic one. 19 In a study of bone nodule formation in a mouse osteoblast model, enoxaparin was found to be less active than standard heparin 20 ; in this study, the molecular size was a major determinant of heparin’s ability to inhibit the bone nodule formation.
It is also possible that pregnant women treated with LMWH have better placental perfusion and higher concentration of placental AP than women without this treatment.
All biochemical bone metabolism markers increased during pregnancy as it is naturally a state of high bone turnover. Elevated bone turnover may explain trabecular bone loss during pregnancy. 21 In mouse bone marrow cell culture (lasting 9 days) standard heparin at 0.1 IU/mL intensified osteoclast formation, whereas at higher concentration (1 and 10 IU/mL), it suppressed osteoclast formation. Low-molecular-weight heparins (nadroparin, enoxaparin, parnaparin, dalteparin) suppressed the formation of osteoclasts at all used concentrations (1-100 anti-Xa IU/mL). 22 In a rat bone marrow cell cultures, however, both standard heparin and LMWH increased osteoclast formation. 22
In our study, OPG levels increased markedly (P < .001) during pregnancy, but there was no significant difference between groups with or without treatment. Irie at al. 5 have shown that heparin inhibits OPG activity by binding to this cytokine, resulting in stimulation of osteoclastic bone resorption. Although it is not known whether OPG is responsible for heparin-induced osteoporosis in vivo, these results suggested that OPG could play a pivotal role in this condition. Osteoprotegerin is expressed in many tissues apart from osteoblasts. It is a member of the tumor necrosis factor superfamily, which exerts its function as a soluble decoy receptor for RANKL. 11 RANKL is necessary for development, maturation, and activation of osteoclasts. 23 Many reports support the assertion that the RANKL/OPG ratio is a major determinant of bone mass. 24 Osteoprotegerin contains a highly basic heparin-binding domain, suggesting that affinity interactions could be used to control the rate of its ion exchange-driven release from drug-delivery devices. 14 Some reports demonstrate that glycosaminoglycans strongly interfere with OPG binding to RANKL and to the reformed complex RANK–RANKL. Moreover, as OPG reduces the half-life of membrane RANKL, glycosaminoglycans inhibit this mechanism and thus maintain a high level of RANKL at the cell membrane and modulate the bioavailability of this factor involved in bone remodeling. 14,25 Recently, a significantly higher vascular mobilization of OPG was found in UFH than in LMWH, indicating that UFH has a higher affinity for OPG than LMWH. And the higher release of OPG, then in normal pregnancy, was measured when higher prophylactic dosage was used. 15 Clinical studies have shown that serum OPG levels in humans are about 0.1 ng/mL, 26 while 0.2 to 0.4 U/mL of heparin in serum is considered safe and effective for antithrombotic therapy. 2 Because it was shown that 0.1 to 1 U/mL of heparin effectively inhibits 0.1 ng/mL of OPG in vitro, 5 OPG activity in vivo could be inhibited by heparin treatment. Furthermore, OPG is important for maintaining serum levels of RANKL by binding to it. Therefore, heparin could elevate the serum concentration of free RANKL by inhibiting the RANKL/OPG complex formation and thereby induce osteoporosis.
A decrease in bone mineral density (BMD) has been reported to occur in patients treated with LMWH. In some studies, decrease in BMD was observed after long-term LMWH use. 4,27,28 Long-term treatment and prophylaxis of venous thromboembolism caused a modest but progressive decrease in BMD, more evident in patients using LMWH than acenocoumarol. 27 There was no difference in mean BMD between a dalteparin intervention group (1.11 g cm [−2]) and control groups (1.14 g cm [−2]). These results suggest that a long-term use of prophylactic dalteparin in pregnancy is not associated with significant decrease in BMD. 4 In another study, the incidence of clinically significant bone loss (> or = 10%) in the femur in women who received thromboprophylaxis in pregnancy was approximately 2% to 2.5%, and it was similar in patients receiving either LMWH or UFH therapy. 28 The effects on bone demineralization require further investigation.
The limitations of our study were largely due to a special state of its participants; the decrease in bone mass could not be evaluated densitometrically because, naturally, the pregnant women could not be exposed to radiation. Additionally, as deviations of bone marker levels occur physiologically during pregnancy, the values had to be compared individually against each previous measurement. Results of systematic review are ambiguous. 29
Conclusion
In our study, OPG levels increased markedly (P < .001) during pregnancy, but there was no significant difference between groups with or without treatment and study showed that enoxaparin prophylaxis decreases the concentration percentage of bone specific AP, suggesting a possible etiology of the heparin-induced osteoporosis. Further clinical and experimental studies are warranted to investigate this pathophysiological mechanism as well as the impact of LMWH on bone metabolism.
Footnotes
Acknowledgments
Authors thank all laboratory staff of the Biochemical and Central Hematological Laboratories for their assistance with the study.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Research Project No.MZ0VFN2005 under the patronage of the Ministry of Health of the Czech Republic.
