Abstract
Increased levels of factor VIII (FVIII) are a prevalent and independent risk factor for deep venous thrombosis (DVT). After a median of 10 years of the first DVT, we evaluated FVIII coagulation levels in 55 patients with DVT of the lower limbs and previous high levels of FVIII and in 74 controls. Subsequently, we analyzed the presence of post-thrombotic syndrome (PTS) in patients and its relationship with FVIII levels. After a median of 10 years of the first DVT, the FVIII levels were still significantly higher in patients when compared to controls (P < .001). Patients with severe PTS showed increased levels of FVIII when compared to patients with moderate or absent PTS (P < .001). We demonstrated a persistent increase in FVIII levels in a subset of patients with DVT, but in a lower magnitude after 10 years of the first DVT episode. Moreover, we observed a significant association between increased FVIII levels and severe PTS.
Introduction
Deep venous thrombosis (DVT) is a multifactorial disease, and increased levels of coagulation factor VIII (FVIII) have been established as a risk factor for DVT. 1 –4 The main determinants of FVIII in plasma are the von Willebrand factor (VWF) and the ABO blood group. 5,6 We recently demonstrated that increased levels of FVIII is a risk factor for DVT in Brazilian patients (Odds ratio [OR] = 3.0; 95% confidence interval [CI 95%] 1.6-5.7; P < .001). 7 Although FVIII is an acute phase protein, results from previous studies suggested that increased FVIII levels in patients with DVT are independent of an acute reaction. 8,9
The nature of the relationship between inflammation and DVT is not yet established. C-reactive protein (CRP) does not appear to be useful in predicting future or diagnosis of acute DVT. 10,11 Several markers of inflammation are elevated in acute DVT, and this altered inflammatory profile is more likely to be a result rather than a cause of DVT. 12,13
The median follow-up of previous studies that analyzed persistent increase in FVIII in patients with DVT was about 1 to 5 years, 9,14,15 and in this study we evaluated a subset of patients with high levels of FVIII after a median of 10 years of the first thrombotic episode.
Post-thrombotic syndrome (PTS) is a long-term complication of DVT of the lower limbs, which is present in 20% to 50% of symptomatic patients. 16,17 The many factors that contribute to the incidence and severity of PTS, such as age, gender, obesity, residual DVT, location, and extent of DVT, are controversial. 18 The PTS can be a confounding effect in studies evaluating the association of DVT and inflammatory biomarkers because it is associated with a chronic inflammatory process and despite the increase of FVIII levels are not associated with acute reactions, may be the stimulus for this alteration may contribute to this increase. The relationship between PTS and inflammatory markers is still controversial. 19,20
In the present study, we evaluated FVIII levels in a subset of patients with DVT, after a median of 10 years of the first thrombotic episode, and previous high levels of this coagulation factor. Moreover, since chronic inflammation of affected member can be present, and constitute a stimulus to increase FVIII levels, we also analyzed the association between clinical PTS and FVIII levels.
Design and Methods
Patients and Controls
Our initial cohort consisted of 230 adult patients with a first episode of acute DVT of lower limbs, between January 1990 and September 2004, followed up at the outpatient clinic of University of Campinas. Patients with cancer, liver, renal, or systemic inflammatory diseases were excluded. Deep venous thrombosis was confirmed by imaging tests. In 2004, in a first assessment, FVIII levels were evaluated in all these patients and in 230 matched controls. In 2011, all patients from this initial cohort who originally presented with FVIII levels above the 90th percentile (n = 81) were recruited for a second assessment of FVIII activity. In total, 55 patients (68%) agreed to participate and provided informed consent. Totally, 74 healthy controls were again selected according to age, gender, ABO blood group, and the same exclusion criteria were used for the study entry.
Laboratory Methods
Factor VIII levels were measured by a 1-stage clotting assay with FVIII-deficient plasma (Siemens, Marburg, Germany). The FVIII tests were performed in duplicate on an automated coagulation analyzer (BCS XP, Siemens). C-reactive protein levels were determined in the same samples by a nephelometric method (Siemens). Factor VIII levels were considered only in the presence of normal CRP levels. ABO blood group was determined by agglutination and adsorption-elution test.
Determination of PTS
The severity of PTS was evaluated by the clinical score (range 0-6) of the revised Clinical, Etiologic, Anatomic and Pathophysiologic (CEAP) classification. 21 In the CEAP classification, patients with class 0 represent no visible or palpable signs of venous disease symptoms of PTS; class 1 telangiectasias, reticular veins, or malleolar flare; class 2 varicose disease; class 3 edema without skin changes; class 4 skin changes ascribed to venous disease (pigmentation, lipodermatosclerosis); class 5 skin changes with healed ulcer; and class 6 skin changes with active ulceration. Post-thrombotic syndrome was defined as absent for classes 0 to 2, moderate for classes 3and 4, and severe for classes 5 and 6.
Statistical analysis
Factor VIII levels were described as median (minimum-maximum). Medians between patients and controls were compared by the Mann-Whitney test. Medians of FVIII between the first and second analyses in both groups were compared by the analysis of variance test for repeated measures. Clinical characteristics (age, gender, ethnicity, ABO blood group) between patients and controls were compared by the Fisher exact and Mann-Whitney tests. A P value <.05 was considered statistically significant. All analysis was performed using the R Foundation for Statistical Computing, version 2.13.2.
Results
Median age of the 55 patients (16 male and 39 female) was 47 years (range 20-70), and median of the first DVT episode was 10 years (range 8-15). In the first measure of FVIII, in our initial cohort, the median of the first DVT episode was 3 years (range 1-8). The control group consisted of 74 participants (28 male and 46 female) with a median age of 45 years (range 21-70). There was no significant difference between patients and controls related to gender, age, ethnicity, and ABO blood group (see Table 1).
Clinical Characteristics of Patients With Deep Venous Thrombosis and the Control Group.

Abbreviation: DVT, deep venous thrombosis.
Deep venous thrombosis was spontaneous in 17 (31%) patients. In the remaining 38 (69%) patients, associated risk factors were surgery (8.3%), immobilization (20.5%), oral contraceptive use (22.5%), pregnancy and puerperium (13.5%), among other less prevalent causes (4.2%).
Factor VIII levels were significantly higher in patients when compared to controls (158.0 IU/dL, 82.0-216.0 vs 126.1 IU/dL, 83.4-187.1; P < .001). Those patients also showed higher plasma FVIII when compared to controls (235.8 IU/dL, 200.3-510.0 vs 127.2 IU/dL, 80.1-211.0; P < .001) when previously analyzed with a median of 3 years after venous thrombosis. Factor VIII levels were significantly lower at the moment when compared to the initial assessment (P < .001), with a mean reduction of 33%.
Importantly, only 17 (30.9%) of 55 patients included in this study still presented increased FVIII levels (above P90). C-reactive protein levels in patients with DVT were between normal values (0.13 mg/dL) but significantly lower when compared to the first measure with 3-year median after venous thrombosis (0.22 mg/dL; P = .003).
Unfortunately, only 68% of the patients analyzed in the first assessment and with FVIII levels above 90th percentile were included in this study. So, we also compared the FVIII levels between this group and those who did not respond to our invite, and there was no significant difference (235.8 IU/dL vs 256.1 IU/dL; P = .26).
egarding PTS, a total of 31 (56.3%) of 55 patients presented PTS and 8 (14.5%) were classified as severe PTS. Table 2 shows the median of FVIII and CRP levels according to the PTS classification. Classes 5 and 6, representing severe PTS, were associated with significantly increased levels of FVIII (182.0 IU/dL, range 171-216), when compared to patients with moderate PTS (155.5 IU/dL, 82-184; P = .002) or no PTS (154.0 IU/dL, 112-189; P < .001). There was no statistical correlation or difference between CRP levels and PTS classification.
Factor VIII and C-Reactive Protein Levels According to the Clinical, Etiologic, Anatomic and Pathophysiologic Classification.a
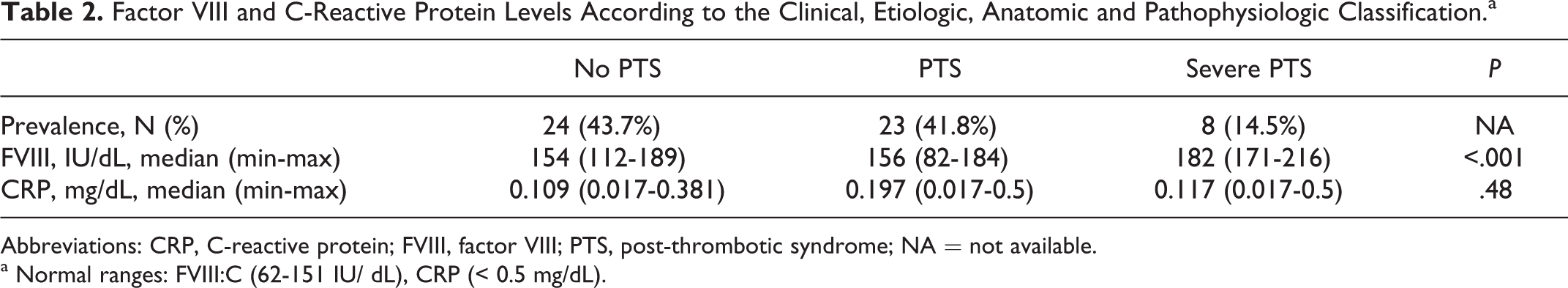
Abbreviations: CRP, C-reactive protein; FVIII, factor VIII; PTS, post-thrombotic syndrome; NA = not available.
a Normal ranges: FVIII:C (62-151 IU/ dL), CRP (< 0.5 mg/dL).
Deep venous thrombosis recurrence was observed in 12 (21.8%) of 55 patients, but there was no difference in FVIII levels between patients with and without recurrence (160.0 IU/dL vs 158.0 IU/dL; P = .14). The majority of the patients with DVT recurrence presented PTS (83%), and this prevalence was significantly different from the patients without recurrence (49%), (P = .049).
Discussion
Previous studies demonstrated increased FVIII levels in patients with DVT after a median of 5 years of follow-up. 9,14,15 In this study, we showed that patients with a median of 10 years after the first episode of DVT presented higher FVIII levels when compared to healthy controls, but the magnitude of this difference was significantly lower than that observed at earlier time points, and only a minority of patients still presented FVIII levels above the 90th percentile. It is important to point out that in our study we established an appropriate reference range of high FVIII levels, using a percentile value. These findings suggest the presence of stimulus to increase FVIII levels that could be stronger during the initial years of the DVT episode. On the other hand, we could not rule out the possibility that those patients present an increased basal FVIII level that could contribute as a causal factor for the first episode of DVT.
There is general consensus agreement that increased FVIII is a risk factor for the first DVT episode and that in several cases, these increased FVIII levels represent a genetic characteristic of the patient, as opposed to a mere inflammatory response. 22 However, whether higher FVIII levels increase the risk of recurrent DVT is still controversial. 14,23 A possible explanation for these apparent discrepant results is the fact that in a proportion of patients followed in these studies, increased FVIII resulted from acquired causes such as PTS or other more subtle proinflammatory state, not revealed by CRP levels.
In a recent prospective study, Tichelaar et al demonstrated that following acute venous thrombosis, although FVIII:C levels remained persistently elevated in 72% of participants for at least 6 months, the CRP levels had corrected to within the normal range by 3 months in the majority of patients. These findings clearly suggest that in patients with elevated plasma FVIII levels, the post-thrombotic acute phase response is of minor importance, and other more important unidentified factors are responsible for maintaining high FVIII levels in the longer term. 24
So, we investigated the possibility that PTS, a common complication of DVT characterized by a chronic inflammation of the affected member, such as edema, increased temperature, and skin lesions, could be related to the FVIII levels. We used CEAP classification, which is a sensitive and specific method to define PTS. According to this method, we demonstrated that 56.3% of the patients presented PTS, with 14.5% classified as severe. The most interesting finding was the significant association between increased FVIII levels and severe PTS. Post-thrombotic syndrome was not associated with CRP levels, in accordance with a previous study. 19 The VWF plays a critical role in regulating plasma FVIII levels. A possibility is that in severe PTS, the vascular injury with increased endothelial cell secretion of VWF could contribute to the increased plasma FVIII levels found in those patients. 25
In this subset of patients the recurrence was 21.8% in a median of 10 years, which is similar to that described in other populations. 26 However, the number of patients in our study is much smaller than previous studies. Although there was no difference in FVIII levels between those with and without recurrence, PTS was more common in the first one. One explanation for this association might be the fact that PTS and recurrent DVT share similar risk factors. We could also speculate that recurrence could contribute to the PTS development. Most of the patients (10 of 12) with recurrent DVT presented with mild PTS, which is not clinically associated to an important local limb inflammation, and may be this could explain why they did not show higher FVIII in comparison to those without recurrence. The number of patients of this study is small and studies with larger numbers of patients with DVT are necessary to confirm and expand these data and results.
Conclusions
In conclusion, we demonstrated a persistent increase in FVIII levels in a subset of patients after 10 years of the first DVT, but in a much lower magnitude than that observed in the first 3 years of the disease. In addition, according to our predefined criteria, only one-third of patients with initially increased FVIII levels still presented this alteration. Finally, we observed a significant association between increased FVIII levels and severe PTS. These data highlight the importance that PTS is to be regarded as a potential confounding effect in studies that evaluate the association of DVT with FVIII.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP) and Instituto Nacional de Ciências e Tecnologia do Sangue (INCTS) for the financial support.
