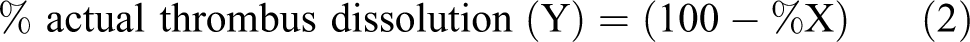Abstract
In the present study, in vivo thrombolysis efficiency of Brevithrombolase, a nontoxic fibrinolytic enzyme purified from Brevibacillus brevis strain FF02B, was affirmed by significant inhibition of thrombus formation in the k-carrageenan-induced rat tail, in a dose-dependent manner. Brevithrombolase at a dose of 600 µg/kg showed an efficacy that was comparable to streptokinase and plasmin, in dissolving in vivo thrombus of k-carrageenan-treated rats under identical conditions. The in vivo anticoagulant property of Brevithrombolase was demonstrated by its prolongation of activated partial thromboplastin time, prothrombin time, and thrombin time in Wistar rats. However, the Brevithrombolase-treated rats demonstrated an insignificant decrease in fibrinogen (Fg) level of plasma compared with Fg level of control group of rats corroborating in vivo as well as in vitro anticoagulant activity of Brevithrombolase is due to its hydrolytic action on thrombin. These findings unequivocally suggest that Brevithrombolase may serve a promising alternative to the commercial thrombolytic drugs.
Introduction
Intravascular thrombosis-associated arterial diseases, such as myocardial infarction and thromboembolic diseases, are leading causes of morbidity and mortality throughout the world. 1 The initial stage of arterial thrombus formation due to blood vessel injury, platelet adhesion, and aggregation is the root cause of these vascular disorders. 2 Usually, the unblocking of blood vessels can restore blood flow in occluded veins and arteries that may lead to the formation of deep vein thrombus. 3 For preventing and/or treating thrombosis-relevant disorders, approved thrombolytic drugs, such as streptokinase (SK), urokinase (UK), and tissue plasminogen activator (t-PA), have been used to effectively dissolve the thrombus; however, these drugs are associated with the risk of hemorrhage and other clinical complications. 1 In addition, these drugs are expensive, demonstrate low specificity toward fibrin, and have short half-lives. 1,4,5 To mitigate the thrombosis-associated disorders, cost-effective thrombolytic drugs of choice are urgently needed that do not show antigenicity, higher rates of occlusion, and intracranial or system bleeding; furthermore, the drugs must be devoid of procoagulant or other hypertensive effects. 5
To achieve the above-mentioned targets, the in vitro antithrombotic effect of several thrombolytic agents, including fibrinolytic serine proteases from Bacillus and other microorganism, has been evaluated. 6 Nevertheless, the in vivo toxicity, pharmacological properties (safety), and mechanism of anticoagulant action of most of these enzymes have not yet been studied in detail. Recently, we demonstrated that Brevithrombolase, a fibrinolytic serine protease purified from Brevibacillus brevis strain FF02B, is a noncytotoxic, nonhemolytic enzyme that does not exhibit lethality or adverse pharmacological effects on the Wistar strain of albino rats. 7 The combined biochemical and pharmacological properties indicate that Brevithrombolase could be developed as a superior thrombolytic drug to ameliorate and/or prevent intravascular coagulopathy. 7 Encouraged by earlier findings, in the present study, we assess the in vivo thrombolytic potency of Brevithrombolase with the k-carrageenan-induced rat tail thrombosis model.
A variety of physical and chemical methods have been applied to induce thrombus formation in experimental animals 3 ; however, because of the advantages of k-carrageenan (a mucopolysaccharide isolated from the cell walls of the red algae), it has been the chemical of choice. 3 The specific advantages include accurate continuous measurements of thrombus in a range, without killing the animals, and requirements for fewer animals, which supports their ethical use for in vivo experiments. 8 This method also avoids complicated surgery to expose blood vessels in the tail of animals. Moreover, k-carrageenan may influence the inactivation of Hageman factor, which is followed by endogenous coagulation. 3 The k-carrageenan-induced thrombus model has been found to be beneficial in assessing the efficiency of various clinically used thrombolytic agents like heparin, UK, SK, and aspirin. 3 Thus, in the present study, we chose to use the k-carrageenan-induced thrombus rat model for assessing the in vivo antithrombotic potential of Brevithrombolase.
Materials and Methods
Materials
Brevithrombolase was purified by our previously described procedure. Male Wistar rats (200-250 g) were obtained from the Animal House, Defense Research Laboratory, Tezpur, Assam. The general conditions of captivity were maintained at 33°C to 37°C with relative humidity ≥75%. All experimental procedures were carried out in accordance with the National Institutes of Health (NIH) guide for the care and use of laboratory animals (NIH publication no 80-23 revised 1996), approved by the institutional animal ethical committee.
k-Carrageenan (sulfated plant polysaccharide extracted from algae), plasmin (catalog no: P1867-150 µg), and SK (catalog no: S3134-10 KU) were procured from Sigma Aldrich (USA). Bovine fibrinogen (Fg) and thrombin were purchased from Himedia, India. Liquiplastin kit (for prothrombin time [PT] determination) was purchased from Tulip diagnostics (P) Ltd, India. C.K. Prest (for activated partial thromboplastin time [APTT] determination), FIBRI-PREST (for quantitative estimation of Fg), and Thrombin kit (for determination of thrombin time [TT]) were procured from Stago Diagnestic, France. Animal foods were obtained from Pranav Agrotech, India. All other chemicals and commonly used reagents were analytical grade, obtained from Sigma-Aldrich.
Methods
Thrombus induction and thrombolysis activity
The in vivo thrombolytic activity of Brevithrombolase was assessed in the k-carrageenan-induced thrombosis rat model 9 with a slight modification. Before the thrombolytic assessment of Brevithrombolase, the optimal dose of k-carrageenan for rat tail thrombosis induction was determined. Briefly, different doses (0.2-1.1 mg/kg) of k-carrageenan were dissolved in 1× phosphate-buffered saline (PBS) and intravenously administrated in the tail vein of rats. The animals were observed after 24 hours for thrombus formation in the tail. The optimum concentration of k-carrageenan was determined on the basis of the saturation of a wine-color appearance in the tail, indicating thrombus formation.
To assess the in vivo thrombolytic activity, a total of 36 male rats were randomly subdivided into 6 groups, each group containing 6 rats. Group 1 rats were injected with 1× PBS (50 mmol/L sodium phosphate buffer, 100 mmol/L NaCl, pH 7.4), which served as the control (placebo). The animals in groups 2, 3, and 4 were treated with graded concentrations (200, 400, and 600 µg/kg in 1× PBS, respectively) of Brevithrombolase in a total volume of 500 µL. The rats in groups 5 and 6 were treated with a 600-µg/kg dose of SK (indirect thrombolytic agent that acts via plasminogen activation) and plasmin (direct-thrombolytic agent), respectively. After 30 minutes of intravenous doses of Brevithrombolase, the tails were ligated and 0.9-mg/kg body weight of k-carrageenan was induced through intravenous injection. Ligatures were removed after 15 minutes of injection. The length of the infarcted region was measured, and the appearance of the wine-colored thrombus formation in the tails was photographed after 24 hours of treatment by the above-mentioned thrombolytic agents. The percentage of in vivo thrombus dissolution after 24 hours of Brevithrombolase administration was calculated using the following formula
10
:
Substituting the %X value of Equation (1) to Equation (2):
Coagulation assay of Brevithrombolase
For the in vivo coagulation assay, the PT, APTT, TT, and Fg levels of plasma were determined using commercial diagnostic kits, following the instructions of the manufacturers. Briefly, after 2 hours of injection, blood samples were collected from the tail vein of Brevithrombolase-treated (200-600 µg/kg in 1× PBS) animals and control group (treated with 1× PBS) rats in microfuge tubes containing 3.8% sodium citrate solution (9:1, v/v). The platelet poor plasma (PPP) was obtained by centrifugation of the blood at 2000g for 10 minutes at 4°C. For each experiment, 100 µL of PPP was used, and the coagulation time (PT, APTT, TT, and Ca2+-clotting time) of the treated and the control PPP groups was recorded in a coagulometer (Sysmex CA-1500; Mittan Keynes, United Kingdom).
Statistical Analysis
The data were statistically analyzed by Student t test, using Sigma Plot 11.0 software for Windows (version 7.0). A P value of <.05 was considered as the significant difference between 2 values.
Results and Discussion
In this study, an optimum dose of k-carrageenan for thrombus formation in the rat tail vein was initially determined using different doses of k-carrageenan (from 0.2 to 1.1 mg/kg). With increasing doses of k-carrageenan, the length of thrombus in the rat tail vein also increased, up to 0.9 mg/kg (Figure 1). With further increases in the dose of k-carrageenan; however, the length of the thrombus did not increase (Figure 1). The optimum dose of k-carrageenan for thrombus formation in the rat tail vein was found to be 0.9 mg/kg.
Determination of thrombus formation in rat tail at various concentrations (0.2-1.1 mg/kg) of k-carrageenan. Data represent mean ± standard deviation (SD) of triplicate experiments (n = 3). Significance of difference with respect to control group of rats, *P < .001, and **P < .05.
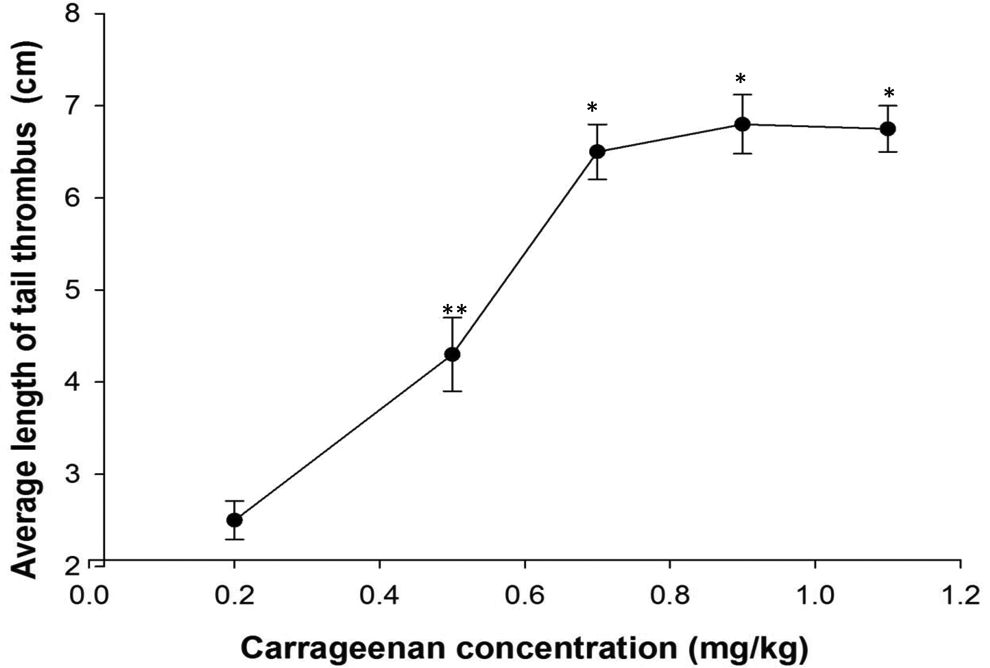
In our previous study, the biochemical and pharmacological properties of a fibrinolytic serine protease (Brevithrombolase) were explored and its in vitro thrombolytic activity was found to be superior to that of plasmin or SK. 7 Our study was extended to demonstrate that Brevithrombolase also exhibited a potent anticoagulant effect in a k-carrageenan-induced rat tail thrombus model. Moreover, the in vivo thrombolytic effect of Brevithrombolase was comparable to that of SK and plasmin (Table 1).
A Comparison of In Vivo Thrombolytic Activity of Brevithrombolase, Plasmin, and Streptokinase After 24 Hours of Treatment on Wistar Strain Rat.a
Abbreviations: PBS, phosphate-buffered saline; SD, standard deviation; -do-, Brevithrombolase.
aThrombus was induced in rat tail by injection of 0.9 mg/kg dose of k-carrageenan. Values are mean ± SD of triplicate determinations. Significance of difference with respect to blood clot lysis activity of Brevithrombolase at a dose of 600 µg/kg.
bP < .05.
cP < .001.
cdenotes significance of difference p < 0.001 value.
In Vivo Thrombolytic Potency of Brevithrombolase
To determine whether or not Brevithrombolase exhibits thrombolytic effect in vivo, different concentrations of the enzyme were intravenously injected in the k-carrageenan-induced rat tail thrombus model. The mean length of the infarcted region of the tail significantly decreased with time and nearly disappeared after 24 hours of Brevithrombolase infusion, in a dose-dependent manner (Figure 2). Conversely, the thrombus that formed in the tail vein of the control group of rats did not decrease with time (Figure 2). The in vivo thrombolytic potencies of Brevithrombolase, SK, and plasmin are shown in Table 1. At a dose of 600 µg/kg, the thrombus dissolution properties of Brevithrombolase, SK, and plasmin were found to be identical (Table 1). Therefore, we assume that, like the currently available thrombolytic drugs (ie, plasmin and SK), Brevithrombolase also has in vivo clot-dissolving properties, making it suitable for use as a thrombolytic drug. Notably, at 600 µg/kg dose, the in vivo thrombus dissolving efficacy of Brevithrombolase surpassed the same activity displayed by nattokinase, 11 and fibrinogenase 12 suggests superiority of the former thrombolytic enzyme.
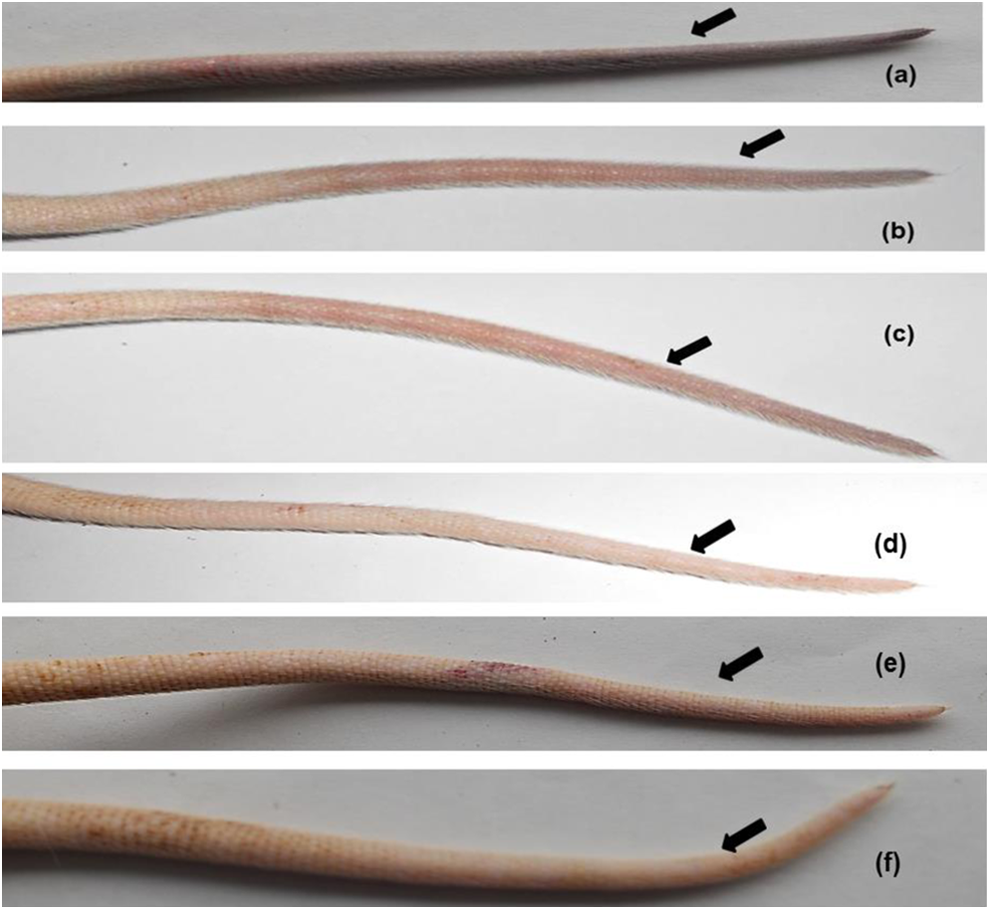
The dose-dependent thrombolytic effect of Brevithrombolase and comparison of its thrombolytic potency with streptokinase and plasmin after 24 hours of intravenous administration in Wister strain rat (n = 6). The arrows indicate thrombus formation area (wine color) in the tail of rats; (a) group 1: k-carrageenan (0.9 mg/kg) dissolved in physiological saline; (b) group 2: k-carrageenan (0.9 mg/kg) and 200 µg/kg of Brevithrombolase; (c) group 3: k-carrageenan (0.9 mg/kg) and 400 µg/kg of Brevithrombolase; (d) group 4: k-carrageenan and 600 µg/kg of Brevithrombolase; (e) group 5: k-carrageenan (0.9 mg/kg) and 600 µg/kg of streptokinase; and (f) group 6: carrageenan (0.9 mg/kg) and 600 µg/kg of human plasmin.
Brevithrombolase Displays In Vivo Anticoagulant Activity
To evaluate the in vivo therapeutic potential of Brevithrombolase in managing thrombotic diseases, we examined its anticoagulant activity in the k-carrageenan-induced rat thrombus model. When k-carrageenan was injected alone, swelling and reddening of the tail were seen first, which was followed by the tail becoming auburn colored. Brevithrombolase inhibited the k-carrageenan-induced thrombus formation over 15 minutes of being intravenously infused (in a rapid dose-dependent manner). The dose-dependent treatment of Brevithrombolase showed a prolongation of the APTT and PT in ex vivo (Figure 3A), compared to the placebo-control group of rats. These results suggest that the inhibition of intrinsic pathways is due to a reduction in coagulation factors, such as VIII, IX, XI, and von Willebrand factor, 13 whereas the delayed PT suggests obstructed extrinsic pathways possibly due to the depletion of coagulation factors such as V, VII, and X. 13 Brevithrombolase may block the activation of the coagulation cascade and the synergistic process of thrombus formation by inhibiting extrinsic as well as intrinsic pathways.
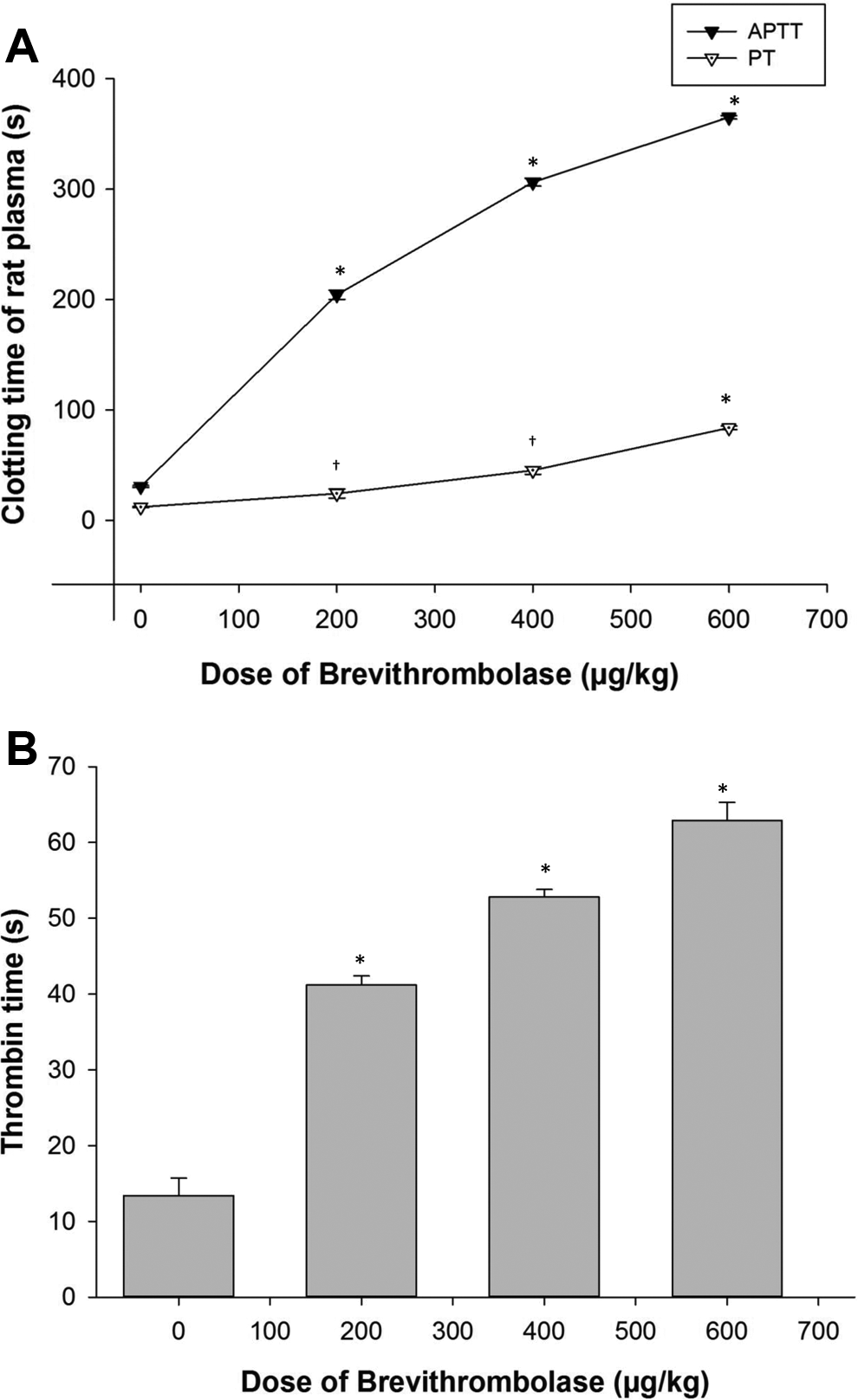
A, Activated partial thromboplastin time (APTT) and prothrombin time (PT) of Brevithrombolase (200-600 µg/kg, 2 hours after intravenous [iv] injection) was measured against citrated platelet poor rat plasma in a coagulometer at 37°C. Data represent mean ± SD of triplicate experiments. Significance of difference with respect to control group of rats, *P < .001; † P < .05. B, Dose-dependent thrombin time (TT) of Brevithrombolase. Blood was collected from tail vein of Brevithrombolase (200-600 µg/kg)-treated rats, and thrombin (200 µL of 1.5 National Institutes of Health [NIH] U/mL) was added to 300 μL PPP to determine TT using STA-THROMBIN kit reagents. Data represent mean ± SD of triplicate experiments. Significance of difference with respect to control group of rats, *P < .001. SD indicates standard deviation.
Like many other fibrinolytic enzymes, 1,14 Brevithrombolase also showed a significant increase in the TT with increasing concentrations in treated rats, in comparison to control rats (Figure 3B). Moreover, the insignificant reduction in Fg level in rat plasma suggests that Brevithrombolase poorly degrades Fg (data not shown) that has been described previously. 7 This suggests that the hydrolysis of thrombin and/or other coagulation factors by Brevithrombolase may impair fibrin formation in the coagulation cascade rather than depletion of the Fg level in Brevithrombolase-treated rat plasma. 7 In addition, the defect in fibrin polymerization was not only reflected in the prolonged thrombin and PTs but also implicated in the prolonged recalcification time by the progressively increased Ca2+ clotting of PPP by Brevithrombolase in a concentration-dependent manner (Figure 4). These findings, taken together, further advocate the in vivo anticoagulant potential of Brevithrombolase.
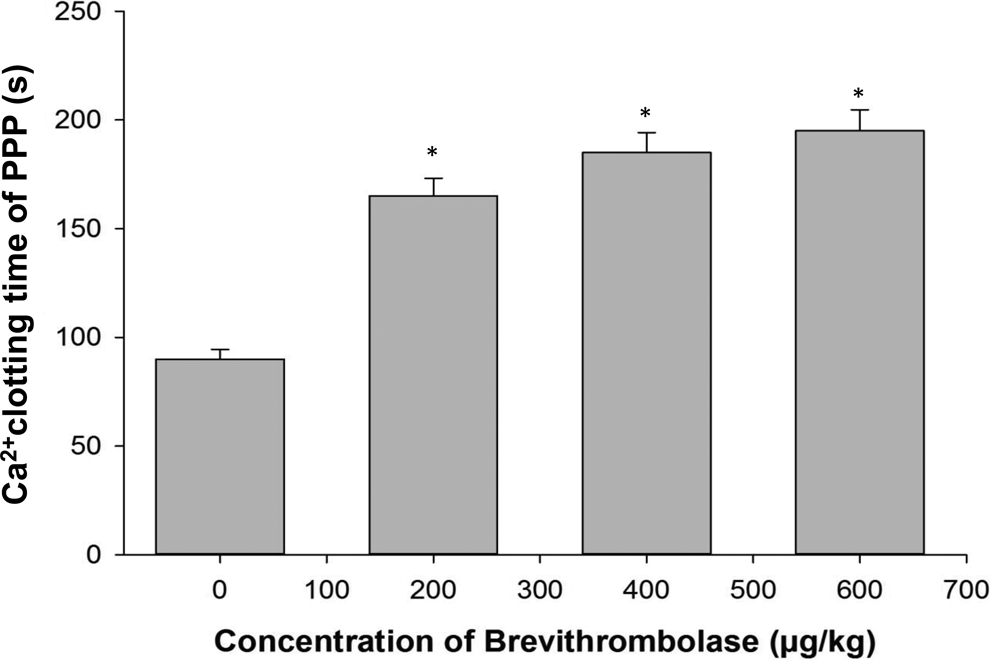
Determination of recalcification time of PPP from control and Brevithrombolase-treated groups of rats. The experiment was done as described in the text. Data represent mean ± standard deviation (SD) of triplicate determinations. Significance of difference with respect to control group of rats, *P < .001.
The application of anticoagulant drugs in treating thromboembolic disease is conceptually attractive and their pharmacological importance has been evaluated based on their mechanisms for preventing thrombus formation. For example, the limited ability of heparin to inhibit thrombin on the thrombus surface and in the surrounding plasma could explain its failure to inhibit the accretion of new fibrin on the clot. 15 Moreover, the addition of intravenous heparin to t-PA was reported to have an improved potency rate when compared with aspirin. 15 Although the feasibility of all anticoagulant drugs has been demonstrated, with regard to frequent complications like antigenicity, toxicity, and defective coagulation, Brevithrombolase overcomes these drawbacks, in terms of in vivo toxicity and lack of bleeding complications, as described earlier. 7 Overall, these results suggest that Brevithrombolase may have a dual role in antithrombotic and thrombolytic activities. 1,16 The results of the present investigation therefore suggest that Brevithrombolase has a thrombolytic function in vivo that is as effective as plasmin or SK, and due to lack of toxicity it may be a promising alternative to the commercial thrombolytic drugs.
Conclusion
The remarkable anticoagulant and thrombolytic activities of Brevithrombolase were compared to the activities of the commercially available drugs, and the enzyme was evaluated for its possible use as a thrombolytic agent. The results of the present study warrant more detailed pharmacokinetics and pharmacodynamics studies before the phase II clinical trial of Brevithrombolase. We also recommend additional bioavailability studies using drug delivery systems that target thrombus for developing more effective cardiovascular drugs.
Footnotes
Acknowledgments
Authors sincerely thank Dr Glen D. Wheeler, University of Alberta, for carefully editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SM was the recipient of SRF in DBT-twining project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by financial assistance to AKM under the DBT-twinning project (BT/38/NE/TBP/2010) from the Department of Biotechnology, Ministry of Science and Technology, Govt. of India.