Abstract
The anti-inflammatory, antioxidative, and antiarteriosclerosis activities of simvastatin along with its protective effects on the endothelium suggest that it may also have antiaging effects. The aim of this study was to investigate the antiaging effects of simvastatin as well as its effects on sirtuin 1 (SIRT1) expression in endothelial cells. Aged rats and human umbilical vein endothelial cells were treated with simvastatin in the presence and absence of oxidized low-density lipoprotein (OX-LDL). Aortic β-galactosidase staining was undertaken to determine senescence, and SIRT1 protein expression was evaluated using Western blot analysis. After simvastatin therapy, arterial endothelial cell aging was significantly reduced, and SIRT1 expression was significantly increased. The OX-LDL significantly accelerated the senescence of umbilical vein endothelial cells and decreased SIRT1 expression. The OX-LDL-induced downregulation of SIRT1 was blocked by simvastatin. Simvastatin treatment also reduced umbilical vein endothelial cell aging and increased SIRT1 expression.
Introduction
Our understanding of the mechanism of aging remains limited to some preliminary experiments and theories. For example, in yeast, the
The cholesterol-lowering agent, simvastatin, is widely used in clinical practice. It has anti-inflammatory, antiatherosclerotic, and antioxidant effects. 2 Because inflammation, atherosclerosis, and reactive oxygen species have all been reported to accelerate endothelial cell senescence, statins may have antiaging effects in endothelial cells.
The current study examined the effects of oral simvastatin on endothelial cell senescence in aged rats. In addition, the effects of simvastatin on SIRT1 expression and development of senescence in cultured human umbilical vein endothelial cells subjected to oxidized low-density lipoprotein (OX-LDL)-induced aging were also evaluated.
Materials and Methods
Animal Protocol
Male SD rats weighing 200 to 250 g were purchased from the Shanghai Experimental Animal Center of Chinese Academy of Sciences. Adult (control) rats were treated at 10 weeks of age. Aged rats were maintained in our laboratory until they were 60 weeks or older before treatment. Sixteen aged rats were randomly divided into 2 groups; 1 group received a normal diet and the other group was given oral simvastatin (5 mg/kg per d, Merck Co, Ltd, China) mixed as a powder in their food. Eight 10-week-old rats on a normal diet were included as a control adult group. The experimental (simvastatin or normal) diet was given for 16 weeks, 3 and the rats were then sacrificed after a 12-hour fast. Animals were anesthetized by intraperitoneal injection of ketamine hydrochloride (50 mg/kg) and diazepam (5 mg/kg). Five milliliters of arterial blood were collected, centrifuged, and serum aspirated and stored at −80°C until use. The abdominal aorta was obtained, washed 3 times with phosphate-buffered saline (PBS), and frozen at −80°C.
Measurement of Blood Lipids
Measurement of blood lipids was performed with a Hitachi 7600-110 automatic biochemical analyzer (Hitachi Co, Japan); total cholesterol and triglyceride (TG) assay kits (Hobsbawm Biological Engineering Co, Ltd, Australia) and high-density lipoprotein cholesterol and LDL cholesterol (LDL-C) assay kits (Weirikang Biotech Co, Ltd, China) were used. The OX-LDL kit was purchased from Shanghai Rongsheng Technology Co, Ltd, China.
β-Galactosidase Staining of Rat Arterial Endothelium
To measure senescence, β-galactosidase staining was undertaken as in previous studies
4
–6
using a kit purchased from Shanghai Biyuntian Biotechnology Co, Ltd, China. Cross-sectional slices of a 0.5-cm sample of the aorta were fixed with 3% formaldehyde +2% valeraldehyde at room temperature for 5 minutes and then rinsed with PBS 3 times. X-gal (5-bromo-4-chloro-3-indolyl-beta-
Oxidative Modification and Identification of LDL
To prepare OX-LDL, normal LDL was dialyzed in 0.01 mol/L PBS to remove the antioxidants added during the centrifugation. The PBS was then used to dilute the LDL to a concentration of 0.5 g/L. Freshly prepared PBS containing 10 μmol/L copper sulfate was added, and the sample was oxidized at 37°C for 12 hours. The samples were then dialyzed in PBS containing 100 mg/L EDTA at 4°C for 24 hours in order to terminate the oxidation reaction. The OX-LDL was sterilized by filtration using a 0.22-μm membrane after which it was stored at 4°C until later use. The degree of oxidative modification of LDL was determined by measuring the content of thiobarbituric acid reactive substances. This measurement showed that the malonaldehyde produced by OX-LDL was 4.1 times of that produced by LDL.
Culture of Human Umbilical Vein Endothelial Cells
Human umbilical vein endothelial cells were purchased from the Cell Institute, Chinese Academy of Sciences. The cells were cultured with Dulbecco modified eagle medium (DMEM) medium containing 15% fetal calf serum in an incubator at 37°C for 3 to 5 days. At confluence, the cells were digested with 0.05% trypsin and 0.02% EDTA. The cells were inoculated in a 24-well plate at a concentration of 3 × 108 cells/L and cultured. When the cells become confluent, the medium was replaced with serum-free and phenol red-free DMEM medium for subsequent analyses.
Endothelial Cell Treatment With OX-LDL and Simvastatin
Cells were divided into 2 groups: (1) an OX-LDL group that received 0, 25, 50, or 100 µg/mL of OX-LDL and (2) a simvastatin group that received 0, 1, 5, or 10 µmol/L of simvastatin. In the OX-LDL group, umbilical vein endothelial cells were cultured with different concentrations of OX-LDL for 24 hours, after which β-galactosidase staining and SIRT1 expression were undertaken. In the simvastatin group, the cells were pretreated with simvastatin for 1 hour prior to treatment with 100 mg/L OX-LDL for 23 hours. β-Galactose staining and SIRT1 expression were assessed.
Western Blot Analysis
Total protein was extracted from endothelial cells in each group, and protein concentrations were determined using the BCA method by following the manufacturer’s instructions (Tiangen Biochemical Technology Co, Ltd, China). A 30-μg sample of total protein was separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis, transferred onto nitrocellulose membrane, and stained with Ponceau S. The membrane was blocked with 20 g/L BSA and incubated with the primary anti-SIRT1 antibody (1:1000) followed by incubation of the secondary horseradish peroxidase–conjugated goat anti-mouse glyceraldehyde 3-phosphate dehydrogenase antibody. Luminescence analysis of the bands was carried out using Odyssey Two-Color Infrared Imaging System (LI-COR Biosciences, Lincoln, Nebraska).
Statistical Analysis
Normally distributed continuous variables were compared by 1-way analysis of variance. When between-group significance was found, multiple comparisons of means were performed using the Bonferroni procedure with type I error adjustment. Data were presented as mean ± standard deviation. All statistical assessments were 2-sided and evaluated at the .05 level of significance. Statistical analyses were performed using SPSS 15.0 statistics software (SPSS Inc, Chicago, Illinois).
Results
Reduced OX-LDL Levels in Aged Rats Treated With Simvastatin
Significant differences were observed in weight, cholesterol, TG, LDL, and OX-LDL between the control, untreated aged, and simvastatin-treated aged groups (
Comparison of Weight and Blood Lipids in the 3 Groups of Rats.a
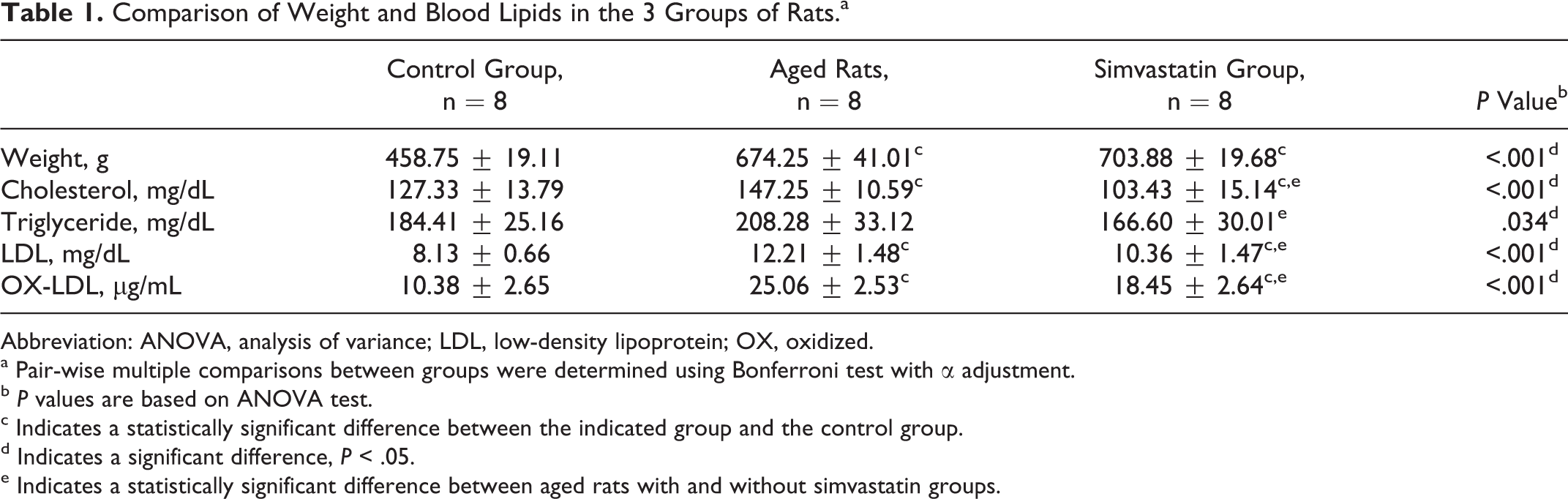
Abbreviation: ANOVA, analysis of variance; LDL, low-density lipoprotein; OX, oxidized.
a Pair-wise multiple comparisons between groups were determined using Bonferroni test with α adjustment.
b
c Indicates a statistically significant difference between the indicated group and the control group.
d Indicates a significant difference,
e Indicates a statistically significant difference between aged rats with and without simvastatin groups.
Reduced Arterial Endothelial Cell Senescence in Aged Rats Treated With Simvastatin
Figure 1 shows staining of the marker for endothelial cell senescence, β-galactosidase, in the arterial intima of each of the 3 groups. Significant differences were observed among the 3 groups in the percentage of cells staining positive for β-galactosidase (
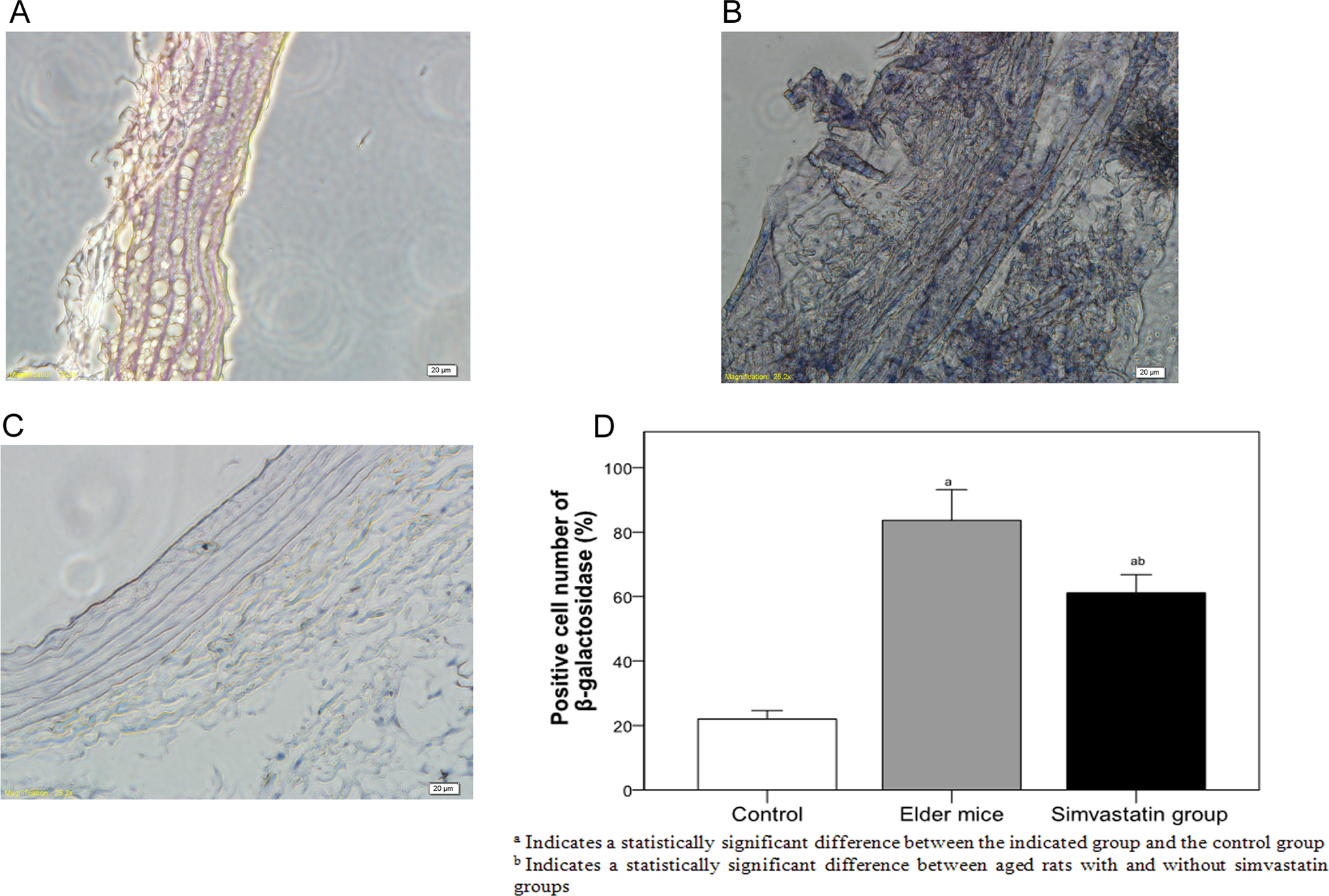
β-Galactosidase staining of the arterial intima of rats in different groups. (A) control, (B) aged rats, (C) simvastatin group, and (D) β-galactosidase-positive cells among the groups.
Oxidized LDL Increases Senescence and Decreases SIRT1 Protein Expression in Human Umbilical Venous Endothelial Cells
To determine the degree of cell senescence induced by OX-LDL, β-galactosidase staining was examined in human umbilical venous endothelial cells incubated with different concentrations of OX-LDL. As the concentrations of OX-LDL increased, the number of β-galactosidase-positive cells increased significantly (Figure 2), reaching a maximal value at 100 µg/mL OX-LDL (Figure 2E). At this concentration of OX-LDL, the SIRT1 protein expression was significantly decreased as compared to control cells (
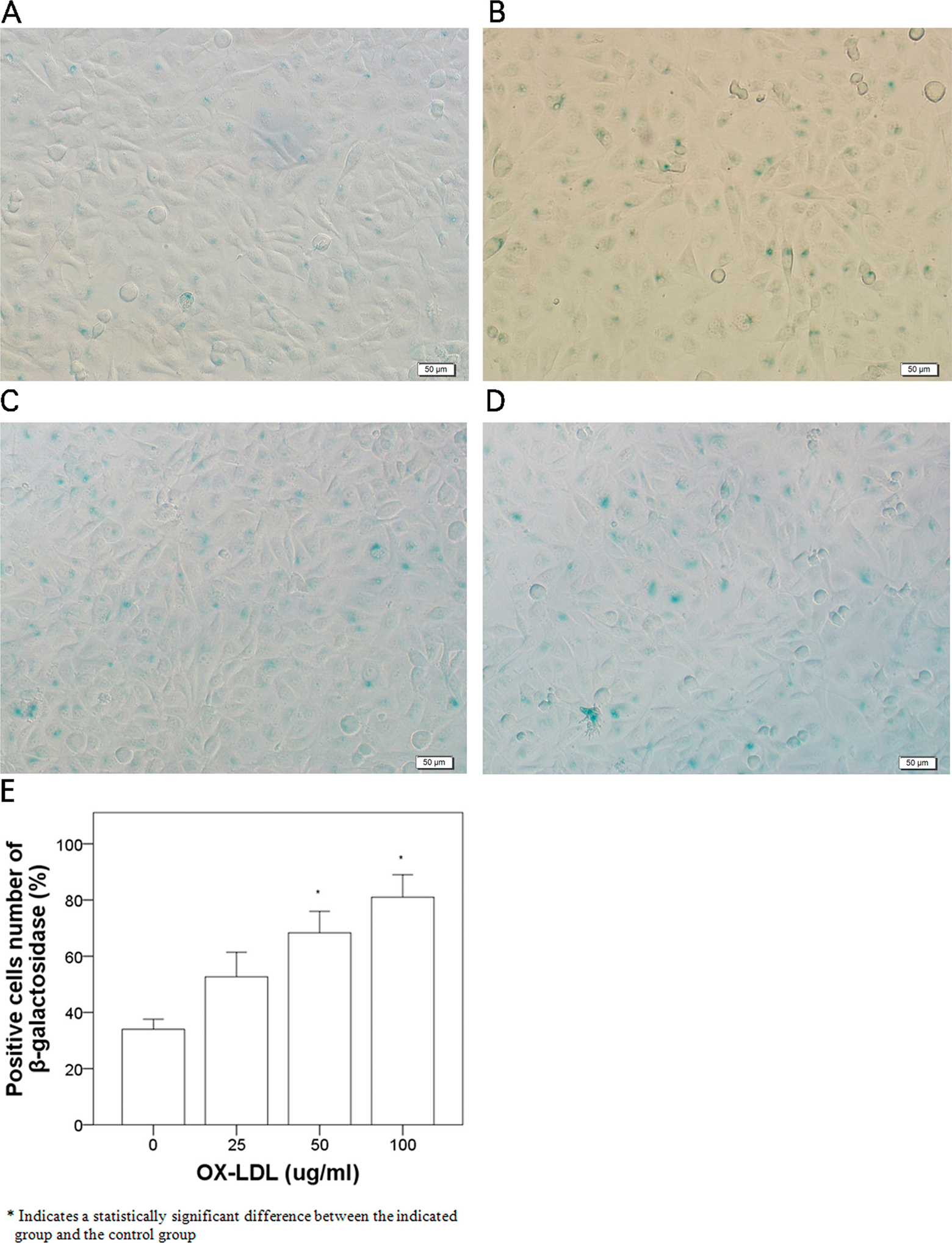
β-Galactosidase staining in human umbilical vein endothelial cells incubated with 0, 25, 50, and 100 µg/mL OX-LDL for 24 hours. OX-LDL indicates oxidized low-density lipoprotein.
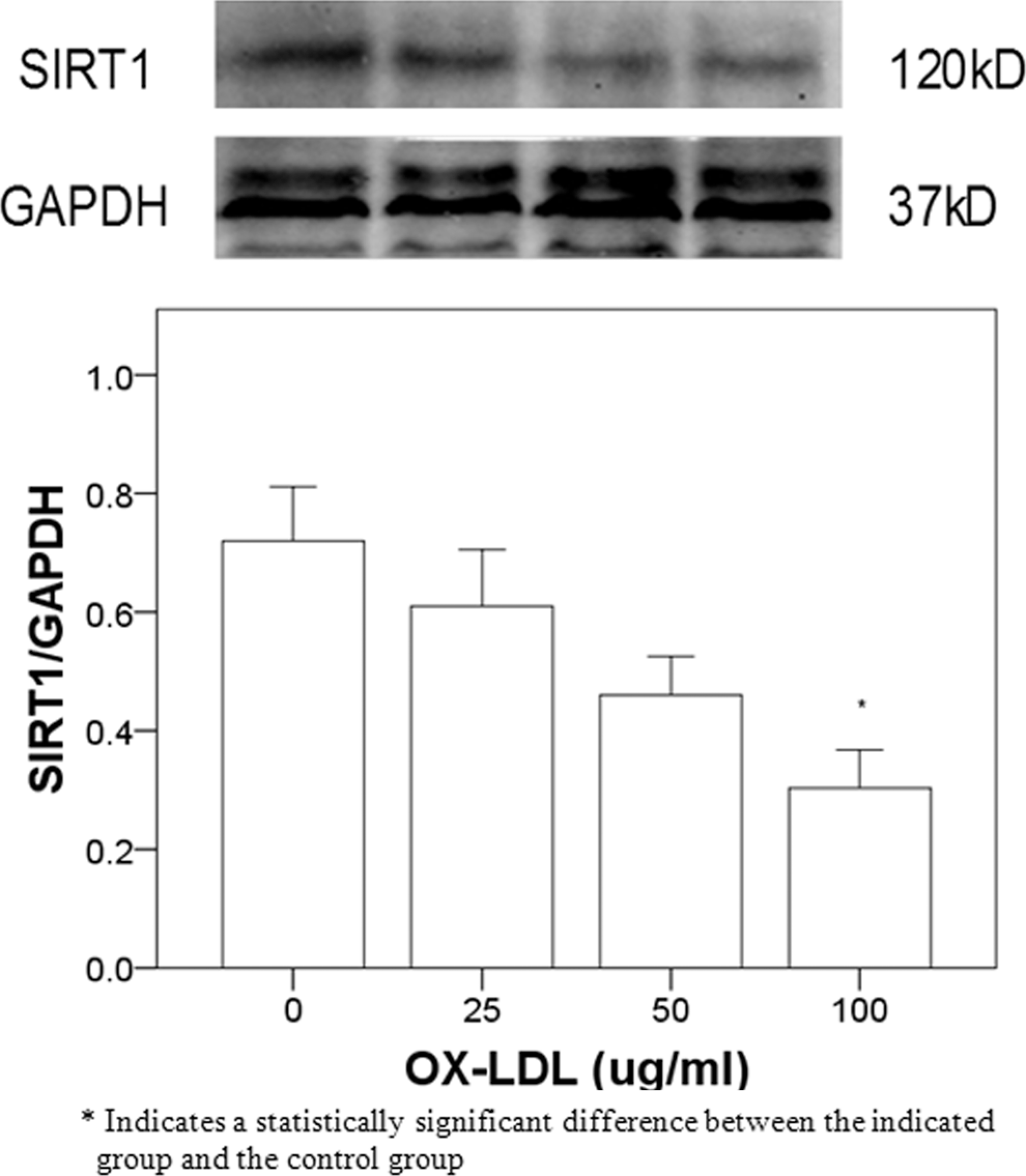
The SIRT1 protein expression in human umbilical vein endothelial cells incubated with 0, 25, 50, and 100 µg/mL OX-LDL for 24 hours. OX-LDL indicates oxidized low-density lipoprotein; SIRT1, sirtuin 1.
Simvastatin Pretreatment Inhibits the OX-LDL-Induced Senescence and SIRT1 Downregulation in Human Umbilical Vein Endothelial Cells
Pretreatment of simvastatin with 10 µmol/L significantly decreased OX-LDL-induced aging in cells treated with 100 µg/mL OX-LDL (Figure 4). Pretreatment with simvastatin also significantly inhibited OX-LDL-induced SIRT1 protein downregulation (Figure 5).
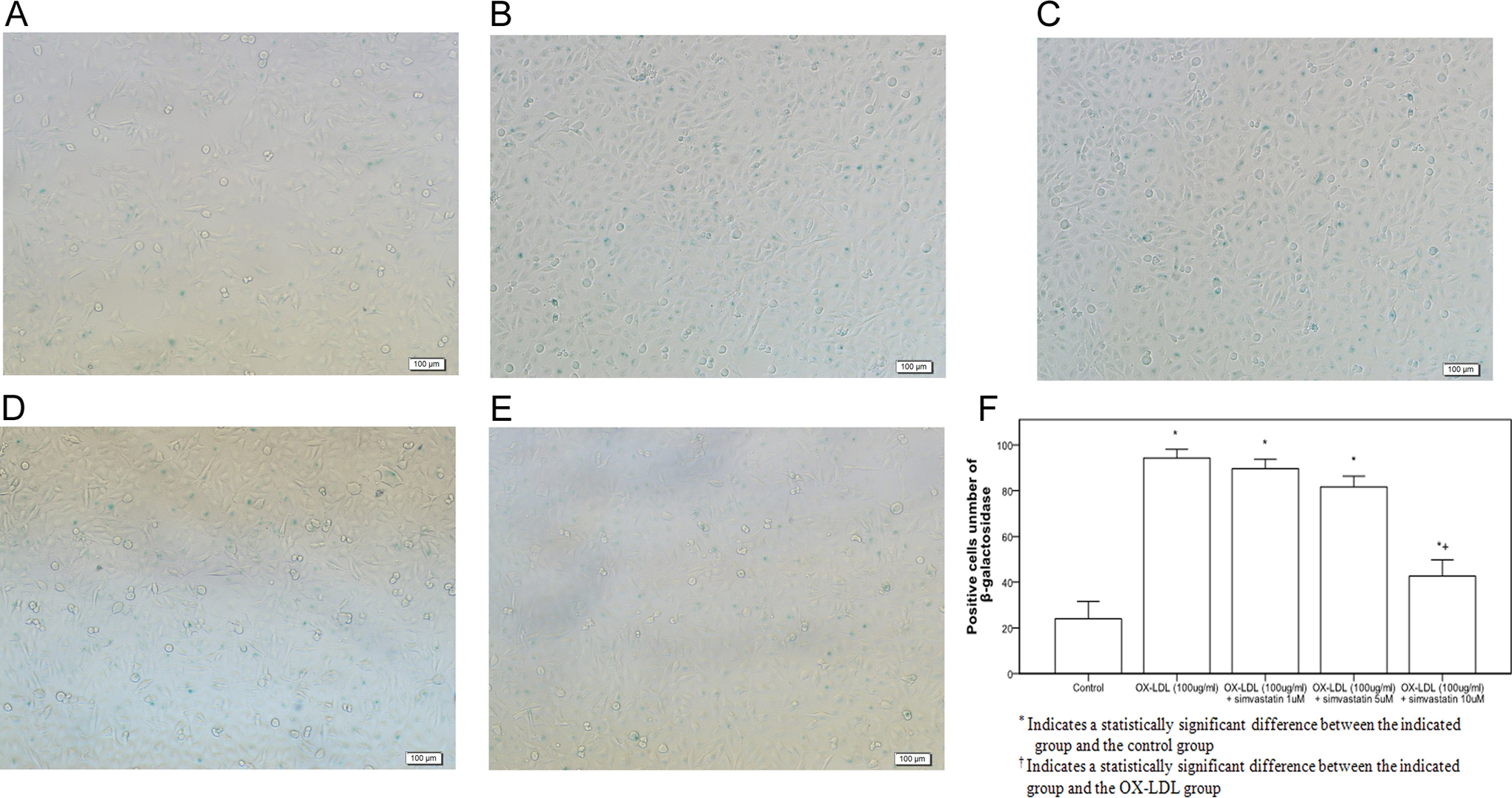
Simvastatin inhibited OX-LDL-induced endothelial cell aging. Cell senescence was determined by β-galactosidase staining. A,: control group; B, OX-LDL (100 µg/mL) group, C, simvastatin (1 µmol/L) group, D, simvastatin (5 µmol/L) group, E, simvastatin (10 µmol/L) group, and F: β-galactosidase-positive cells among the groups. OX-LDL indicates oxidized low-density lipoprotein.
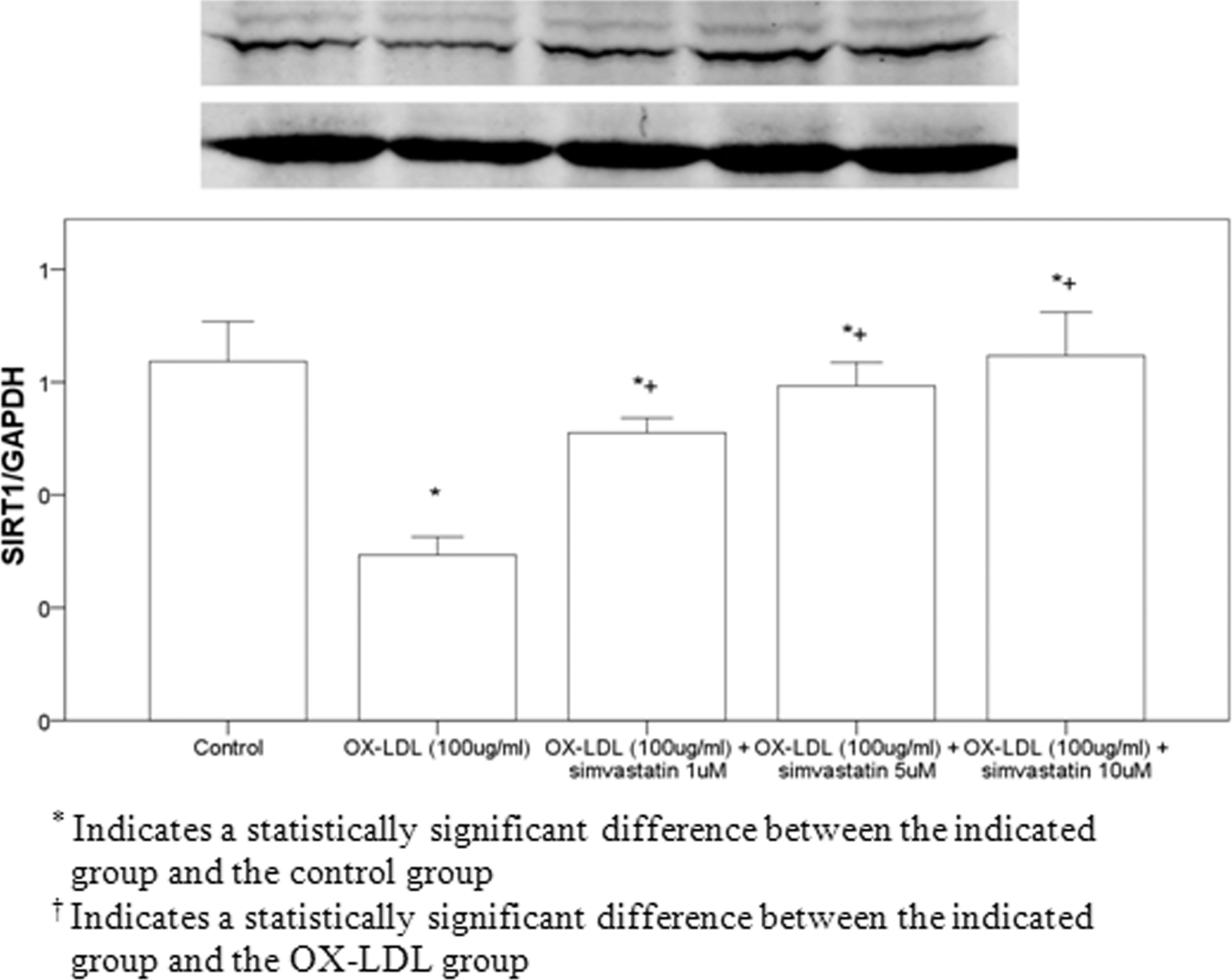
Detection of SIRT1 protein expression in cells using Western blot analysis. SIRT1 indicates sirtuin 1.
Discussion
In the current study, aged rats treated with oral simvastatin had reduced serum OX-LDL concentrations and signs of senescence in arterial endothelial cells. Simvastatin also inhibited OX-LDL-induced senescence in a human umbilical vein endothelial cell culture through increasing SIRT1 expression.
Radiation, 7 hypoxia, 8 peroxidation, 9 hyperglycemia, 10 and high blood lipid concentrations 11 have been shown to cause aging. Oxidized LDL is also an important contributor to the aging process; it increases with the degree of aging in endothelial progenitor cells. 12 In humans, OX-LDL levels gradually increase with age, 13 and inhibition of OX-LDL reduces the degree of cellular aging. 14 Therefore, reductions in OX-LDL concentrations and OX-LDL antagonists have antiaging effects. Tavridou et al 15 showed that treatment with 40 mg/d simvastatin for 3 months significantly decreased the OX-LDL concentrations in patients with dyslipidemia. In addition, oral simvastatin treatment (1 mg/kg per d) in adult and aged rats for 12 weeks significantly decreased OX-LDL levels. 16 Simvastatin significantly lessened the age-related decrease in endothelial function, 16 suggesting that simvastatin has antiaging effects. In the current study, oral simvastatin for 16 weeks significantly slowed the arterial endothelial cell aging. Simvastatin also decreased OX-LDL-induced aging in umbilical vein endothelial cells. However, the effects of simvastatin on endothelial cell function were not determined in the present study and will therefore be the subject of future analyses.
Aging is not determined by a single gene. A number of markers of aging have been reported, including telomerase, β-galactosidase, and
Endothelial cell senescence plays an important role in the pathophysiology of atherosclerosis. Fenton et al 19 showed that atherosclerosis was closely related to aging. In their model of atherosclerosis, the injured intima showed significant aging as determined by β-galactosidase staining as compared to the control group, indicating that accelerated aging occurs simultaneously with atherosclerosis. 19 Stein et al 20 found that SIRT1 can prevent high-fat-induced atherosclerosis in animals. The current study found that simvastatin has antiaging effects; it can also prevent oxidative stress-induced decrease in SIRT1 expression. Further studies will determine whether the mechanism through which simvastatin decreases the formation of atherosclerosis is through increasing SIRT1 expression.
Conclusions
In summary, simvastatin shows antiaging effects, which may be medicated through restoration of the decreased SIRT1 expression induced by OX-LDL in endothelial cells.
Footnotes
Authors’ Note
The authors Junping Lei and Xiaosong Gu contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
