Abstract
Desirudin, a subcutaneously (SC) administered direct thrombin inhibitor, is indicated for prevention of venous thromboembolic events (VTEs) after total joint replacement surgery. DESIR-ABLE (multicenter trial of desirudin for the prophylaxis of thrombosis: an alternative to heparin-based anticoagulation) was a multicenter, open-label, single-arm study of hospitalized patients requiring VTE protection designed to extend the safety profile for desirudin to include a broad population of perioperative/critically ill patients. The primary end point was major bleeding. A total of 516 patients undergoing major surgery (378, 73%) or who were medically ill with prolonged immobility (138, 27%) were enrolled at 19 centers and received desirudin 15 mg Q12H. Many patients had high-risk features for bleeding and thrombosis such as thrombocytopenia (<100 × 109/mL, n = 50), severe obesity (body mass index >35, n = 145), and renal impairment (creatinine clearance <60 mL/min, n = 292). There were no major bleeds and no VTE-related deaths in this study. The DESIR-ABLE demonstrated the safety of desirudin in critically ill perioperative and medical patients. Trials in specific surgical or medically ill patients are needed to confirm these findings.
Introduction
Heparins are the most widely prescribed anticoagulant agents in the hospital and are used for various purposes including venous thromboembolic event (VTE) prophylaxis. 1 Therapy with heparins can be complicated by a variety of untoward events including thrombocytopenia (TCP), allergic reactions, and lack of efficacy. There are scant alternatives to heparin-based VTE prophylaxis for the hospitalized patient. The American College of Chest Physicians Guidelines for VTE prophylaxis recommends 5 different therapeutic modalities for VTE prophylaxis in various patient populations at risk of VTE: low-dose unfractionated heparin (LDUH), low-molecular-weight heparin (LMWH), fondaparinux, vitamin K antagonists, and intermittent pneumatic compression (IPC). 2 With the exception of warfarin that has limited utility in the hospitalized patient, all these recommended agents are natural heparins or synthetic heparin derivatives (eg, fondaparinux). All have similar limitations including an indirect mechanism of action, development of TCP with or without antibodies to platelet factor 4 (PF4), and lack of efficacy in higher-risk cohorts. In addition, the synthetic heparin derivative, fondaparinux, has a 17-hour half-life in patients with normal renal function which is further prolonged in patients with renal impairment, thus limiting its usefulness in perioperative and critically ill patients due to lack of a reversal agent. 2 Although widely used in hospitalized patients, IPC is known to be less effective than pharmacological therapies and provides inadequate protection for higher-risk patients. 2
Desirudin is a direct thrombin inhibitor administered by subcutaneous (SC) injection and approved by the European Medicines Agency in 1997 for the prevention of deep venous thrombosis (DVT) in patients undergoing total hip replacement (THR) and total knee replacement (TKR) surgery and by the US Food and Drug Administration in April 2003 for the prevention of DVT which may lead to pulmonary embolism (PE) in patients undergoing THR surgery. 3 Desirudin has been studied extensively for the prevention of VTE in high-risk surgical patients for the treatment of acute coronary syndromes (ACSs) and as a procedural anticoagulant in patients undergoing coronary angioplasty. 4 Desirudin, administered at a dose of 15 mg Q12H SC, was superior in preventing VTE in patients undergoing elective hip replacement surgery compared with either LDUH or LMWH with a similar bleeding profile. 5,6
The purpose of the current trial (DESIR-ABLE) was to investigate the safety of a fixed dose (15 mg Q12H, SC) of desirudin as an alternative to heparin-based anticoagulation in patients who required anticoagulation for the prophylaxis of thrombosis.
Methods
The DESIR-ABLE trial was a multicenter, open-label, single-arm study in patients requiring anticoagulation for the prophylaxis of thrombosis due to major surgery or prolonged immobility and who were expected to require VTE prophylaxis in the hospital for at least 3 days. Patients received desirudin 15 mg Q12H for as long as clinically required as determined by the enrolling investigator. Patients above 18 years of age were enrolled after obtaining informed consent. Patients were excluded if they were pregnant, had active bleeding, severe renal insufficiency (determined by either Cockcroft-Gault or Modification of Diet in Renal Disease equations), intracranial neoplasm or aneurysm or known allergy to desirudin, or a recombinant hirudin derivative. Participant safety was monitored by an independent Data Safety and Monitoring Committee. The protocol was approved by internal ethics boards as required by the Declaration of Helsinki.
The primary end point of major bleeding was defined as clinically evident hemorrhage associated with a hemoglobin decrease ≥2 g/dL that led to a transfusion of ≥2 units of whole blood or packed red cells outside of the perioperative period (time from the start of the surgery or procedure and up to 12 hours after), or hemorrhage that is intracranial, retroperitoneal, or into a prosthetic joint. Secondary end points included new onset symptomatic thrombosis requiring medical or surgical intervention or death due to thrombosis defined as fatal PE, ischemic stroke, mesenteric thrombosis, or myocardial infarction. Participants were assessed for adverse events (AEs) during treatment and for 30 days following the last dose of study drug. Bleeding-related AEs were determined by querying the AE database for bleeding or anemia-related terms (Table 1).
Bleeding-Related Adverse Event Terms.

Abbreviations: IV, intravenous; GI, gastrointestinal; SM, small; AMT, amount; ABD, abdomen; RT, right.
Statistical Analysis
End points were assessed from initiation of study drug up until 24 hours after study drug completion. All statistical tests used a significance level of P = .05. Two-tailed tests were performed for all analyses that used statistical testing. Summary statistics included the number and percentage of responses in each category for discrete variables, and the mean, median, standard deviation, minimum, maximum, 25th percentile, and 75th percentile for continuous variables. Version 9.1 of SAS (SAS Institute, Inc., Cary, NC) or higher was the statistical software package used to produce all summaries, listings, statistical analyses, and graphs. No formal sample size calculations were conducted given the descriptive nature of the study design.
Results
A total of 516 patients were enrolled at 19 centers with the following VTE prophylaxis indications: major surgery (378, 73%) or; medically ill with prolonged immobility (138, 27%). Comorbidities were frequent and included diabetes mellitus (27.9%), hypertension (67.2%), coronary artery disease (24.6%), and malignancy (23.4%). Many patients had high-risk features for bleeding and thrombosis such as TCP (<100 × 109/mL, n = 50), severe obesity (body mass index [BMI] >35, n = 145), moderate renal impairment (creatinine clearance [CLcr] = 30-59 mL/min, n = 269), and severe renal impairment (CLcr <30 mL/min, n = 23; Table 2). Within the major surgery cohort, 93 patients underwent orthopedic surgery including 65 patients with elective TKR, 76 patients underwent cardiothoracic, or other major vascular surgery, and the remaining 209 patients underwent mostly abdominal surgical procedures with 77 of these involving the removal of various types of cancers (ovarian, cervical, colon, and lung). Of the 285 patients who underwent nonorthopedic surgery, 10.5% (30) were enrolled from intensive care units. Medically ill patients were more likely than surgical patients to have diabetes (36% vs 25%, P < .05) or congestive heart failure (15% vs 7.1%, P < .05) and were more than twice as likely to be enrolled from an intensive care unit (16.7% vs 8.2%, P < .05).
Demographics.
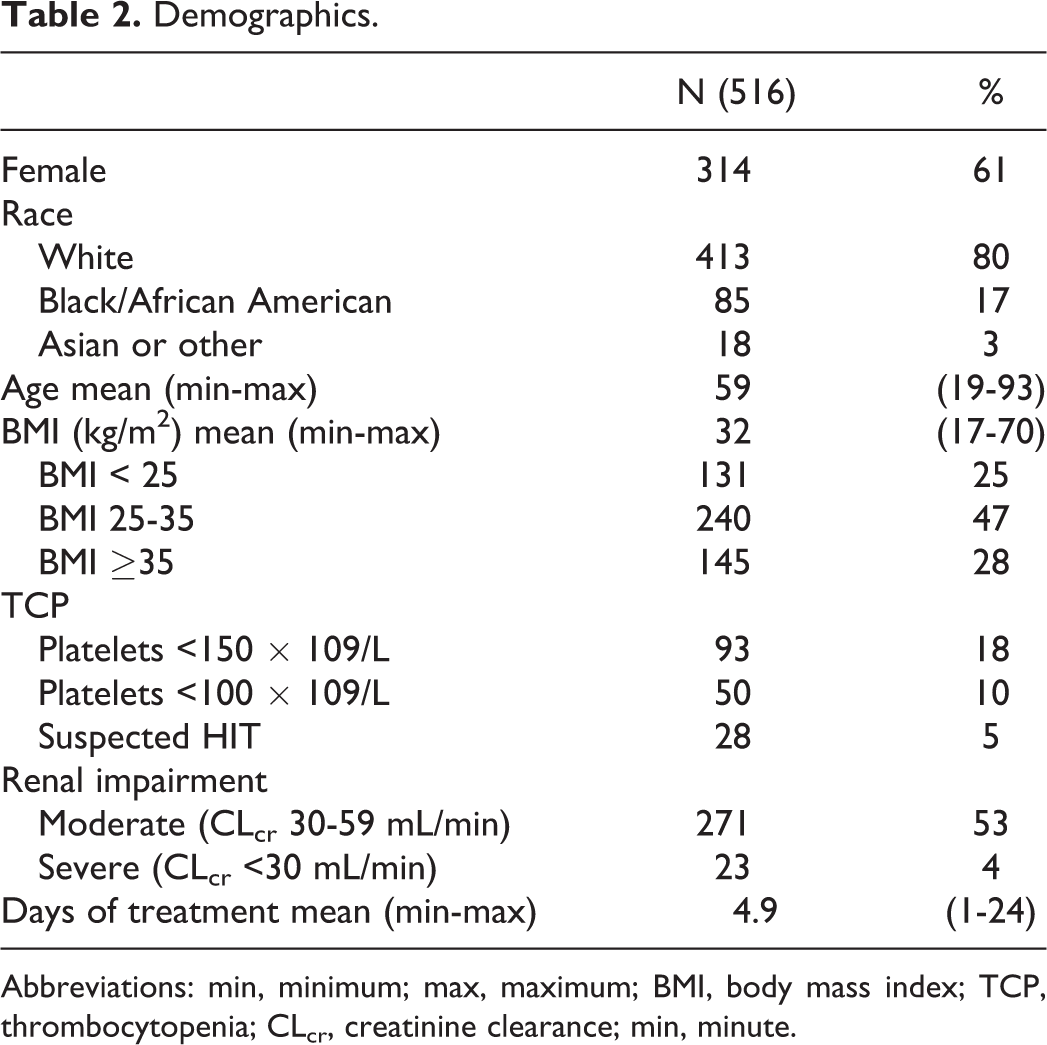
Abbreviations: min, minimum; max, maximum; BMI, body mass index; TCP, thrombocytopenia; CLcr, creatinine clearance; min, minute.
No patient experienced the primary end point of major bleeding (Table 3). There were 58 drug-related bleeding AEs occurring in 10% of patients; 6 events (1.2%) were considered “definitely” or “probably-related” to study drug. None of these patients required study drug discontinuation. Eleven (2.1%) patients discontinued study drug secondary to a bleeding AE considered possibly drug-related, all of which were of mild or moderate severity. There were 2 fatal bleeding events, 1 of which was classified as possibly related to study drug. This patient was a 93-year-old woman admitted for heart failure exacerbation. Relevant concomitant medications included clopidogrel 75 mg once daily. She was on study drug for 5 days until hospital discharge. Two weeks after discontinuation of study drug, she was readmitted to hospital with a 1 to 2 week history of mental status changes. A computed tomography (CT) scan and subsequent neurosurgery consult diagnosed bilateral subacute subdural hygromas, but a magnetic resonance imaging (MRI) scan performed on the next day revealed bilateral subacute subdural hematomas. The investigator determined that the MRI findings were possibly related to study drug administered during the previous hospitalization. The patient was discharged to hospice and expired approximately 24 days after cessation of study drug. The other fatal bleeding event was a massive gastrointestinal hemorrhage occurring 48 hours after cessation of study drug. The patient underwent surgical repair of a 4-mL perforation in the first portion of the duodenum and received 3 doses of study drug beginning 5 days postoperatively. Autopsy revealed a large peptic ulcer in the first portion of the duodenum which had eroded into a 0.3-cm artery causing massive gastrointestinal hemorrhage. There were 9 symptomatic VTEs requiring intervention in 8 patients (1.6%; Table 4). Serious AEs occurred in 80 (15.5%) of participants with 8 events leading to study drug discontinuation. There were 10 (1.9%) deaths during the study, none of which were thrombosis related.
Results.
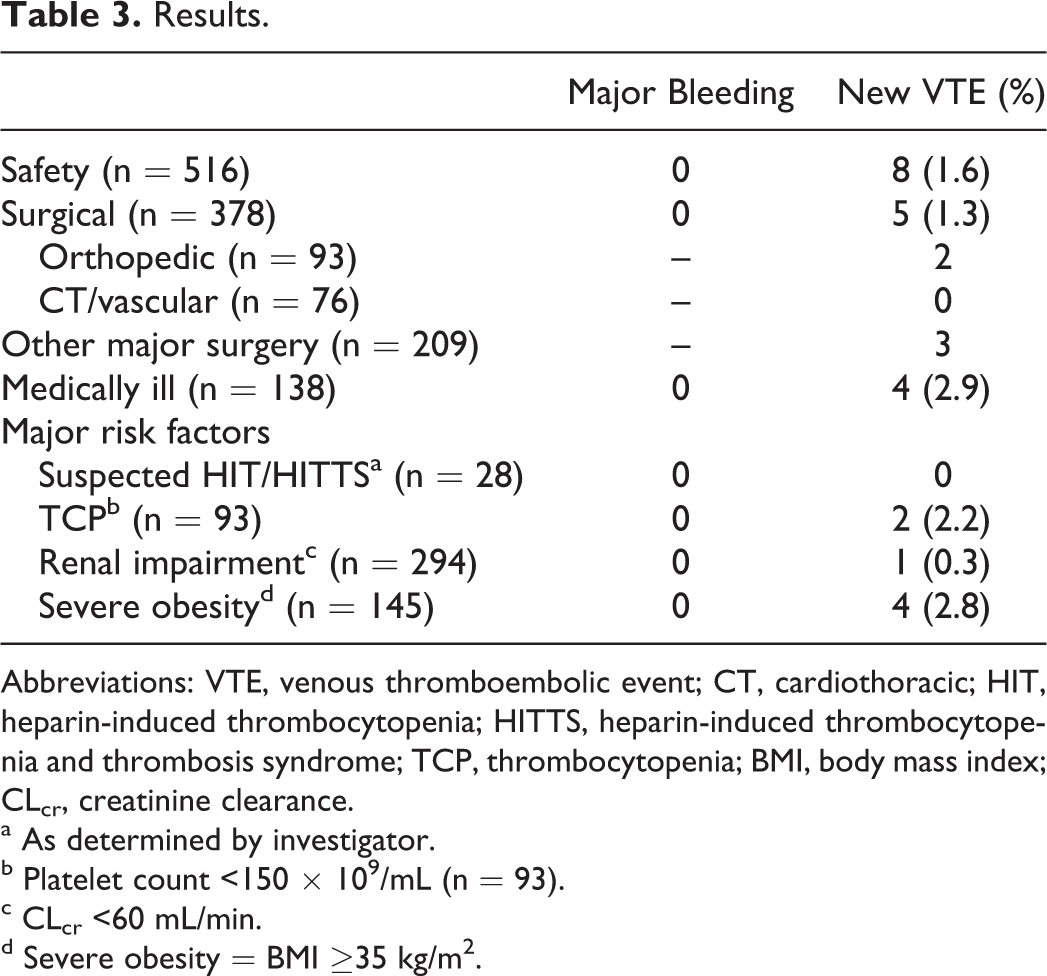
Abbreviations: VTE, venous thromboembolic event; CT, cardiothoracic; HIT, heparin-induced thrombocytopenia; HITTS, heparin-induced thrombocytopenia and thrombosis syndrome; TCP, thrombocytopenia; BMI, body mass index; CLcr, creatinine clearance.
a As determined by investigator.
b Platelet count <150 × 109/mL (n = 93).
c CLcr <60 mL/min.
d Severe obesity = BMI ≥35 kg/m2.
Demographics of Patients With a Thrombosis End Point.
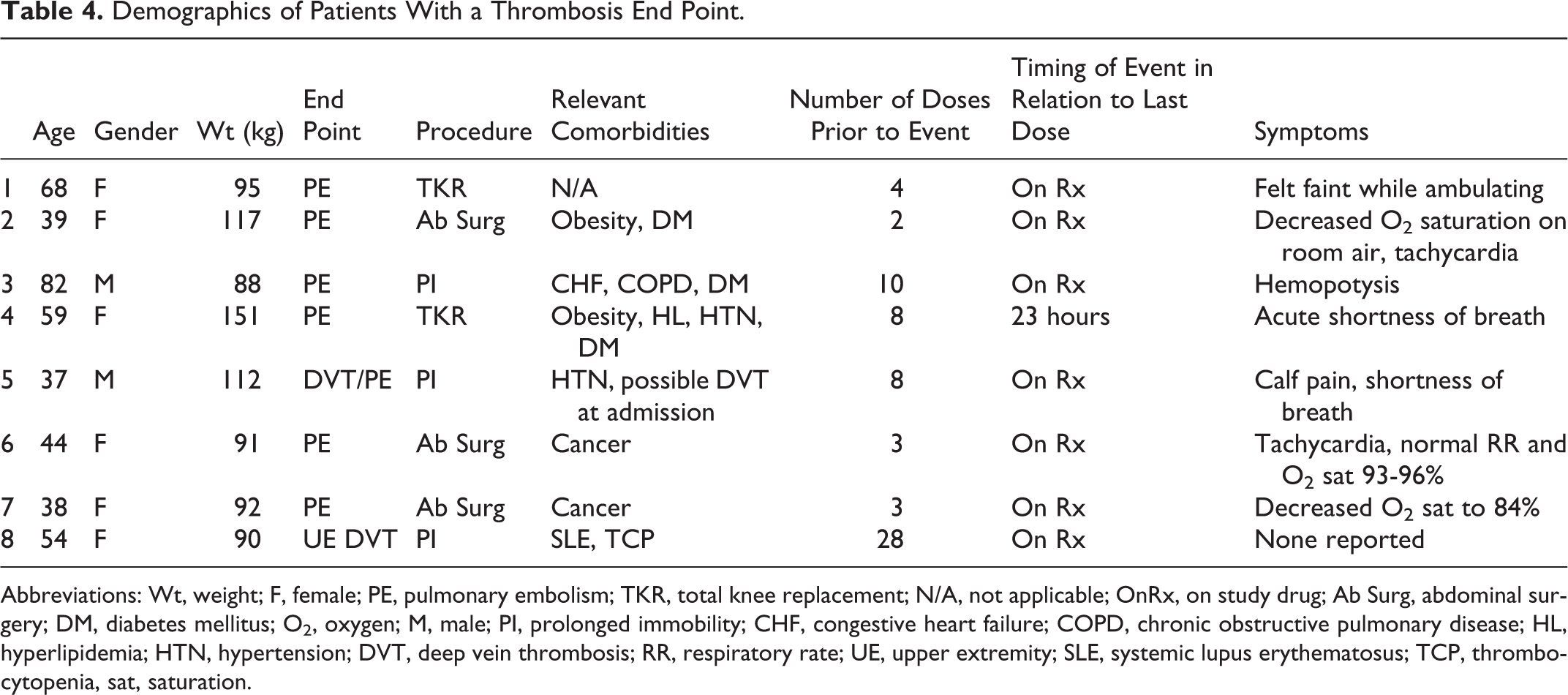
Abbreviations: Wt, weight; F, female; PE, pulmonary embolism; TKR, total knee replacement; N/A, not applicable; OnRx, on study drug; Ab Surg, abdominal surgery; DM, diabetes mellitus; O2, oxygen; M, male; PI, prolonged immobility; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; HL, hyperlipidemia; HTN, hypertension; DVT, deep vein thrombosis; RR, respiratory rate; UE, upper extremity; SLE, systemic lupus erythematosus; TCP, thrombocytopenia, sat, saturation.
Nearly 20% (93 of 516) of patients had TCP defined as a platelet count of less than 150 000/mm3. Of these, 50 (9.7%) had a platelet count of less than 100 000/mm3, a level that necessitates cessation of heparin-based anticoagulation according to prescribing information for heparins and fondaparinux. Compared to participants with normal platelet counts, patients with TCP were older (median age 66 vs 59, P < .05), more likely to be male (65% vs 34%, P < .06) and underweight (BMI <25: 37% vs 23%, P < .05), and less likely to be morbidly obese (BMI >35, 14% vs 31%, P < .05). Patients with TCP were also more likely to have a recent (<3 months or concurrent) history of cardiac surgery (coronary artery bypass graft surgery or valve replacement surgery, 24% vs 3%, P < .05), coronary artery disease (47% vs 20%, P < .05), congestive heart failure (18% vs 7%, P < .05), and/or atrial fibrillation (16% vs 4%, P < .05). Heparin-induced TCP (HIT) was suspected in 27 (29%) patients with TCP, although the presence of heparin–PF4 antibodies was not confirmed by serological testing and no participants had evidence of thrombosis at enrollment. Treatment duration was similar among TCP patients and the overall cohort (5.1 ± 3.3 days vs 4.9 ± 3.2 days, P = NS). The incidence of new VTE was similar among patients with TCP and non-TCP: 2 (2.2%) patients with TCP developed a VTE compared with 6 in 423 patients with non-TCP (1.4%, P = NS). There were no bleeding events meeting criteria for the primary end point in either group. Serious AEs occurred in 16 patients with TCP, all considered unrelated to study drug.
A majority of participants had renal insufficiency defined as an CLcr of less than 60 mL/min (n = 302, 59% of the entire cohort). Mean CLcr for patients with renal insufficiency and those with normal renal function (CLcr ≥ 60 mL/min) were 46 ± 10 and 78 ± 15 mL/min, respectively. Compared to patients with normal renal function, those with renal insufficiency were older (mean age 65 vs 51 years, P < .05), more likely to be female (81% vs 33%, P < 0.05), and to have hypertension (74% vs 58%, P < .05) and coronary artery disease (28% vs 20%, P < .05). Treatment duration was similar in both the groups. Bleeding-related AEs considered possibly related to study drug occurred in 1 patient with normal renal function and 2 patients with renal insufficiency (P = NS). The incidence of VTE was similar among patients with normal renal function and those with renal impairment: 4 (1.9%) versus 5 (1.7%, P = NS).
Discussion
This is the first report on the use of desirudin in a large cohort of patients requiring VTE prophylaxis outside of elective orthopedic surgery. The overall rate of major bleeding and bleeding AEs was low in this trial, similar to previous reports in patients undergoing elective THR surgery. These data suggest that desirudin has an acceptable safety profile in a broad population of surgical and medically ill patients requiring VTE prophylaxis, including important subgroups such as patients with TCP, suspected HIT, and patients with moderate renal impairment (CLcr 30-59 mL/min).
Thrombocytopenia is a known side effect of heparin treatment and is listed as a warning in the approved prescribing information of all heparin-based anticoagulants (UFH, LMWH, and fondaparinux), indicating that platelet counts should be monitored closely and anticoagulation discontinued if TCP with a platelet count of less than 100 000/mm3 develops. 1,7,8 In this cohort of high-risk, hospitalized patients requiring VTE prophylaxis, nearly 1 in 5 patients had a platelet count of less than 150 000/mm3 and 10% had a platelet count of less than 100 000/mm3. Previously thought to be benign, a recent report published by University of North Carolina and Duke University found that TCP in patients receiving UFH or LMWH for VTE prophylaxis was frequent and associated with increased mortality. 9 In the Complications After TCP Caused by Heparin registry, 1017 patients received heparin or LMWH for VTE prophylaxis and 190 (19%) developed TCP. Acquired TCP was the strongest predictor of in-hospital mortality (3.7% vs 0.7%, P = .004) and predicted both thrombotic and bleeding complications (11.1% vs 7.1%, P = .07 and 16.5% vs 4.9%, P < .0001). 10 Time from initial heparin exposure to diagnosis of TCP was 55 hours, showing that TCP occurs early after heparin exposure. 9
Rarely is TCP in patients receiving heparin-based anticoagulation caused by an immune-mediated reaction known as HIT. Heparin-induced TCP is a devastating adverse drug reaction, often accompanied by thrombosis, characterized by a decreased platelet count and development of heparin antibodies to PF4. Morbidity and mortality rates are reported to be 30% to 50% and standard of care dictates immediate cessation of heparin therapy and treatment with an alternative anticoagulant. 11 Heparin-induced TCP is a clinical syndrome and a definitive diagnosis is often difficult, frequently requiring days of data collection and specialized laboratory testing, all while patients are exposed to potent anticoagulation that increases the risk of bleeding. 12 In our cohort, 5% of patients had suspected HIT, none of which were confirmed with serological testing. Since desirudin is a direct-thrombin inhibitor, it neither causes the formation of nor interacts with heparin antibodies to PF4. Previous reports indicate that it may be effective in preventing thrombosis in patients with isolated HIT (eg, TCP and PF4 antibodies without thrombosis) with a better safety profile than argatroban. 13
In the absence of TCP, heparin-based anticoagulation can lead to the development of heparin/PF4 antibodies. Notably, although it has been shown that fondaparinux rarely interacts with heparin-PF4 antibodies to precipitate the clinical syndrome of HIT, it is equally immunogenic to LMWHs. 14 Although previously thought to be “asymptomatic,” a mounting body of evidence indicates that heparin-PF4 antibodies are associated with poor long-term outcomes in a variety of patient populations including those treated with heparins for ACSs 15,16 and those undergoing cardiothoracic surgery. 17 Additional heparin therapy in patients immunized with preexisting heparin-PF4 antibodies can lead to the clinical syndrome of HIT. 18 Patients undergoing cardiac and vascular surgery are known to have a high rate of PF4 seroconversion in the postoperative period. Avoidance of additional heparin exposure in these patients through the use of an alternative anticoagulant for VTE prophylaxis may decrease the rate of development of HIT in these highest risk patients.
Patients with renal impairment represent another important subpopulation not well served by current therapies. Historically, focus has been on safety in patients with severe renal impairment (CLcr
The occurrence of 7 PEs in this study with only 1 documented concomitant DVT is unexpected. We are aware of previous reports of isolated PE without precedent DVT in patients with trauma, 21 but the finding of a majority of PE among these VTEs appears unusual. Data do indicate a high number of comorbidities in this patient population suggesting an especially high risk of VTE. Indeed 2 of the 7 PEs occurred in patients undergoing cancer surgery, and the 1 case of upper extremity DVT occurred in a patient with systemic lupus erythematosus—all conditions known to increase the risk of thrombosis. All the PEs were documented by CT scan as is standard of care, but it is possible that the threshold for obtaining a CT scan may have been lower than is typical due to the open-label nature of the study. Indeed, in 3 of the 7 patients with documented PE, the preceding symptomatology was atypical, with 1 patient “feeling faint” as a sole reported symptom, 1 with isolated hemoptysis without shortness of breath or oxygen desaturation, and 1 with acute onset shortness of breath possibly attributed to a history of congestive heart failure. Finally, in the 1 patient with DVT preceding a documented PE, it was noted that right calf pain was present on admission, so it is possible this patient had a baseline DVT which not unexpectedly, progressed while the patient was treated with a prophylactic dose of desirudin. Randomized controlled clinical trials are necessary to determine whether the relative efficacy of desirudin in these subgroups is similar to that observed in orthopedic surgery.
These data are limited by the lack of a comparator group and the open-label nature of the study. The primary end point of major bleeding is based on objective laboratory evidence unlikely to be biased by the open-label design, but reporting of adverse bleeding events not meeting the criteria for major bleeding could be biased by knowledge of drug therapy. As mentioned previously, the open-label design may have biased enrolling physicians to obtain CT scans more readily than they would otherwise in normal clinical practice.
In summary, this study evaluated the safety profile of desirudin in a broad population of hospitalized patients requiring VTE prophylaxis due to major surgery or prolonged immobility. No patient experienced the primary end point of major bleeding and the overall safety profile appears to be similar to that noted in randomized controlled trials conducted in patients undergoing elective hip replacement surgery. Additional randomized, controlled clinical trials are necessary to confirm these findings and to determine the efficacy of desirudin in preventing DVT compared with standard therapy in these populations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
