Abstract
We hypothesized that leukocytes have 2 opposing effects on patients with ischemic stroke treated with recombinant tissue plasminogen activator (rtPA). Patients with ischemic stroke treated with rtPA were divided into 2 groups using the peripheral leukocyte count: high leukocyte group (HLG) and low leukocyte group (LLG) and were evaluated with the National Institutes of Health stroke scale (NIHSS) during the first 24 hours. We defined significant improvement (SI) as NIHSS improving by more than 50% from the baseline, and deterioration following improvement (DFI) as the achievement of SI within 24 hours but its subsequent loss at 24 hours. Fifty-three patients were enrolled, and the rate of SI within 24 hours was higher in HLG than in LLG (85.2% vs 42.3%, P = .0011). However, the rate of DFI was significantly higher in HLG than in LLG (29.6% vs 7.7%, P = .0413). We found that leukocytes might have not only deleterious but also beneficial effects in intravenous rtPA treatment.
Introduction
Recombinant tissue plasminogen activator (rtPA) has been widely used in Japan for patients with ischemic stroke within 3 hours of onset. The efficacy and safety demonstrated by the Japan Post-Marketing Alteplase Registration Study 1 were compatible with those in the National Institute of Neurological Disorders and Stroke (NINDS) study. 2
Leukocytes play important roles in inflammation, which has deleterious effects on patients with acute coronary syndrome. 3,4 In terms of the relationship between leukocytes and stroke, it was also reported that higher leukocyte counts in peripheral blood were associated with the initial stroke severity and poor prognosis. 5,6 On the other hand, leukocytes have another role, namely, in thrombus resolution. Fibrinolytic enzymes such as neutrophil elastase, cathepsin G, and urokinase are produced by leukocytes and they act as key players. 7 Furthermore, leukocytes themselves in cooperation with singlet oxygen can find fibrin and destroy it. 8,9 Because there are a considerable number of leukocytes in human thrombus, 10 they can promote thrombolysis by plasmin. However, their contribution to thrombus resolution in patients with stroke is not fully understood. In addition, it is little known whether leukocytes can accelerate the thrombolytic effect of rtPA in ischemic stroke. We hypothesized in the present study that leukocytes in peripheral blood play 2 opposing roles in rtPA treatment in patients with ischemic stroke.
Materials and Methods
We registered consecutive patients with acute ischemic stroke of the anterior circulation who were treated with rtPA under the same protocol as recorded in the database of our hospital from January 2006 to December 2010, excluding the patients who had endovascular therapy just after rtPA infusion. Patients were treated with alteplase at 0.6 mg/kg using the inclusion and exclusion criteria of the NINDS. 2 Concomitant use of edaravone (a neuroprotective drug) 11 was permitted. We evaluated the patients using the National Institutes of Health stroke scale (NIHSS) just before rtPA and 33 times during the first 24 hours (15 minutes, 30 minutes, 45 minutes, 1 hour, then every half an hour until 7 hours, and hourly thereafter until 24 hours after rtPA). We defined significant improvement (SI) for rtPA therapy as NIHSS improving by more than 50% from the baseline at least once, and deterioration following improvement (DFI) as SI being achieved but then being lost at 24 hours. The data of leukocyte counts in peripheral blood on admission and at first blood test after rtPA infusion were collected. The association of NIHSS just before rtPA with leukocyte counts was analyzed and comparison of leukocyte counts between the SI group and the non-SI group was performed; sequential change in leukocyte counts in both groups was also evaluated. Then, patients were divided into 2 groups according to the median level of peripheral leukocyte counts on admission: high leukocyte group (HLG) and low leukocyte group (LLG). We compared the clinical backgrounds, concomitant therapies, and clinical outcomes of HLG versus LLG. In addition to NIHSS score, we adopted a modified Rankin scale (mRS) to assess clinical outcome at 14 days after rtPA, and defined mRS = 0 or 1 as good outcome. Hypertension, diabetes, and dyslipidemia were recorded if the patient had a past history or was under treatment. In addition, a patient was defined as diabetic with a glucose level of 200 mg/dL or more on admission. In order to clarify the stroke subtype, electrocardiogram, chest X-p, blood tests, head computed tomography (CT), head magnetic resonance imaging ([MRI] diffusion-weighted image [DWI], fluid-attenuated inversion recovery, T2*-weighted image, and MR angiography), and MR angiography of neck or ultrasonography of carotid artery were carried out for all patients on admission unless MRI was contraindicated. Holter electrocardiogram, transthoracic echocardiography, and transesophageal echocardiography were performed to determine the clinical category, when necessary. We used a modified version of the Alberta Stroke Programme Early CT Score (ASPECTS) system using the DWI-ASPECTS 12 to evaluate early ischemic brain damage before rtPA infusion. Patients were divided into 2 groups: cardioembolism group and noncardioembolism group using the trial of ORG 10172 in acute stroke treatment criteria. 13 Information about the use of any antiplatelet drug (aspirin, clopidogrel, or cilostazol) within 24 hours before onset and past history of stroke was collected.
Statistical Analysis
Values are presented as median and quartiles for continuous values. Spearman rank-order correlation was used for 2 variables. Repeated measurements were tested with Wilcoxon signed rank test, and comparison between 2 groups was carried out using Wilcoxon rank sum test for continuous values and chi-square test for comparison of proportions. The rate of cumulative SI during the observation period was calculated using the Kaplan-Meier method, and log-rank tests were used to compare 2 groups. Hazard ratio, adjusted for confounding factors, was calculated using Cox proportional hazard model. The data were analyzed with JMP 7.0.1 (SAS Institute Inc, Cary, North Carolina, USA), with P value <.05 considered significant.
Results
Fifty-three patients, excluding 1 patient with ischemic stroke of the posterior circulation and 2 patients who had endovascular therapy with Merci Retriever just after rtPA infusion, were enrolled between January 2006 and December 2010. The median peripheral leukocyte count on admission was 6940/mm3 (5355-8435). The first blood test after rtPA treatment was carried out at a median of the third day (4 patients on the day 2, 41 patients on the third day, 3 patients on the day 4, 1 patient on the day 5, 2 patients on the day 6, 1 patient on the day 7, and 1 patient on the day 11), and the median value was 8360/mm3 (7005-9645), which was significantly elevated from that on admission (P = .0003). The NIHSS scores just before rtPA were not correlated with leukocyte counts on admission (P = .7067), but were significantly correlated with those of the first blood test after rtPA treatment (P = .0009; Figure 1). Of all enrolled patients, 34 (64.2%) had SI within 24 hours. Figure 2 shows sequential change in leukocyte counts in the SI and non-SI groups. The SI group showed significant higher leukocyte counts on admission than the non-SI group (7635/mm3 [6205-8823] vs 6370/mm3 [4970-6880], P = .0072). In the non-SI group, leukocyte counts significantly increased from 6370/mm3 (4970-6880) on admission to 9000/mm3 (7330-11 150) on the day of the first blood test after rtPA (P < .0001). In contrast, leukocyte counts were hardly elevated in the SI group (from 7635/mm3 [6205-8823] to 8055/mm3 [6795-9003], P = .3080). Using the median leukocyte counts on admission, patients were divided into HLG and LLG groups. Comparisons of clinical background, concomitant therapies, and clinical outcomes between HLG and LLG are shown in Table 1. High leukocyte group had a significantly younger median age (72.0 [66.0-78.0] vs 78.0 [73.8-84.0], P = .0304) and a tendency for a lower rate of cardioembolism (37.0% vs 61.5%, P = .0745) than LLG. The rate of SI within 24 hours was higher in HGG than in LLG (85.2% vs 42.3%, P = .0011). However, the rate of neurological improvement at 24 hours was not significantly different between the 2 groups (55.6% vs 34.6%, P = .1258) because the rate of DFI was significantly higher in HLG than in LLG (29.6% vs 7.7%, P = .0413). There was no significant difference in the rate of good outcome at 14 days between HLG and LLG (37.0% vs 26.9%, P = .4304). Figure 3 showed the Kaplan-Meier curves of cumulative SI during 24 hours after rtPA, indicating significantly higher cumulative SI in HLG than in LLG using log-rank test (P = .0025). High leukocyte group (hazard ratio = 3.0531, P = .0036) was independently associated with SI during 24 hours, after adjustment for age and stroke subtype (cardioembolism or not; Table 2).
Comparison of Clinical Backgrounds, Concomitant Therapies, and Clinical Outcomes Between High Leukocyte Group and Low Leukocyte Group.
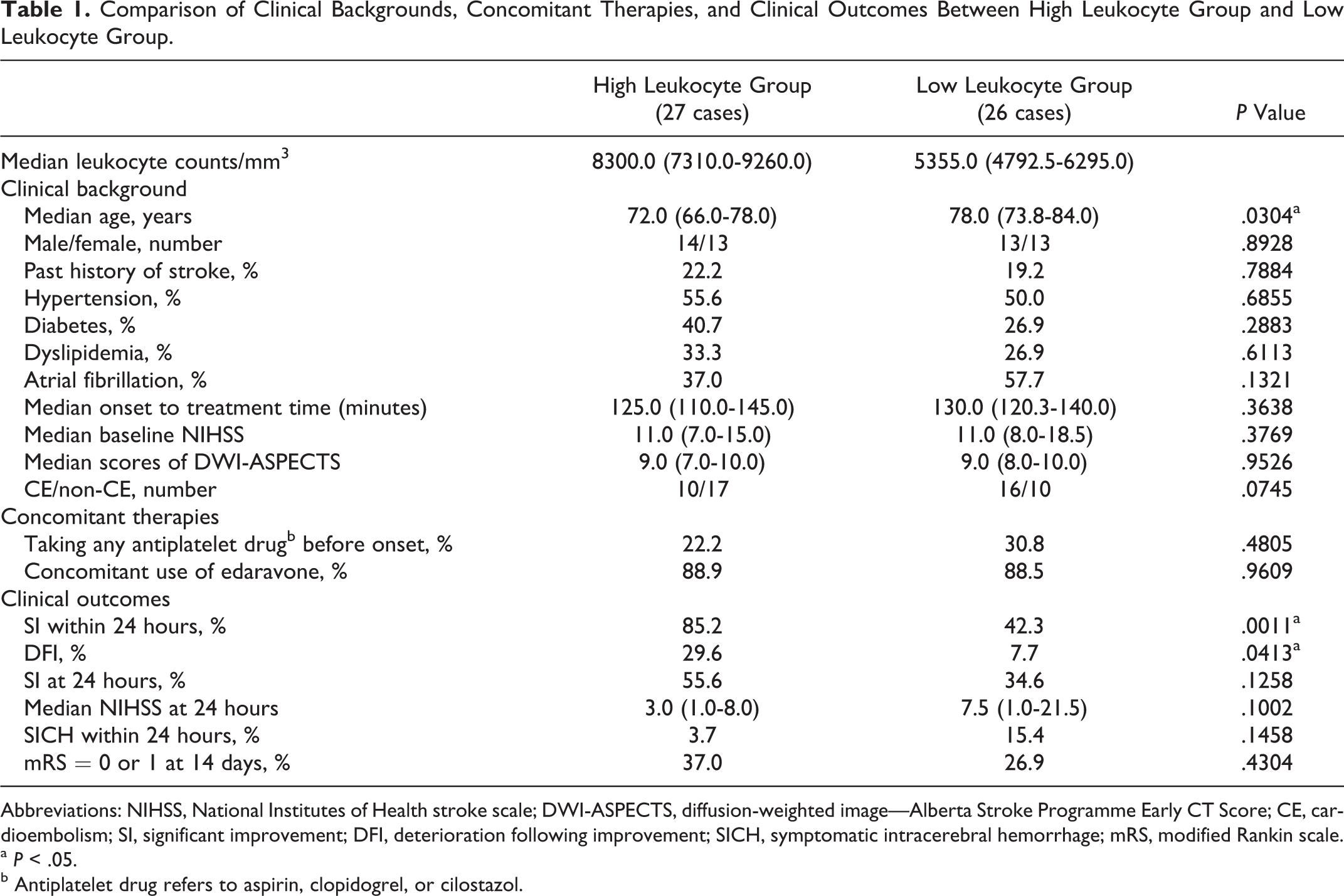
Abbreviations: NIHSS, National Institutes of Health stroke scale; DWI-ASPECTS, diffusion-weighted image—Alberta Stroke Programme Early CT Score; CE, cardioembolism; SI, significant improvement; DFI, deterioration following improvement; SICH, symptomatic intracerebral hemorrhage; mRS, modified Rankin scale.
a P < .05.
b Antiplatelet drug refers to aspirin, clopidogrel, or cilostazol.
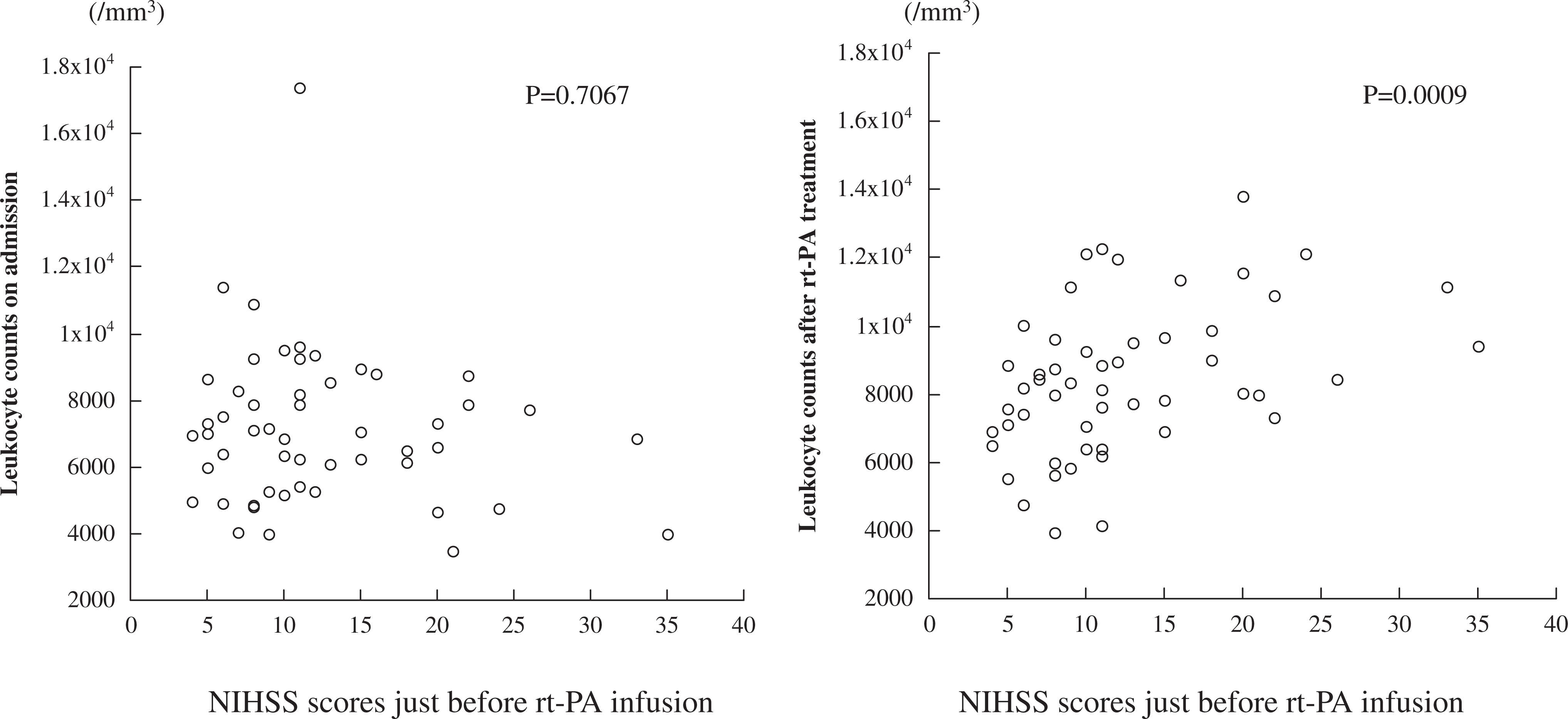
Correlation between leukocyte counts on admission (left) or at first blood test after rtPA treatment (right) and NIHSS scores just before rtPA treatment. Spearman rank-order correlation was used to test the 2 variables. NIHSS indicates National Institutes of Health stroke scale; rtPA, recombinant tissue plasminogen activator.
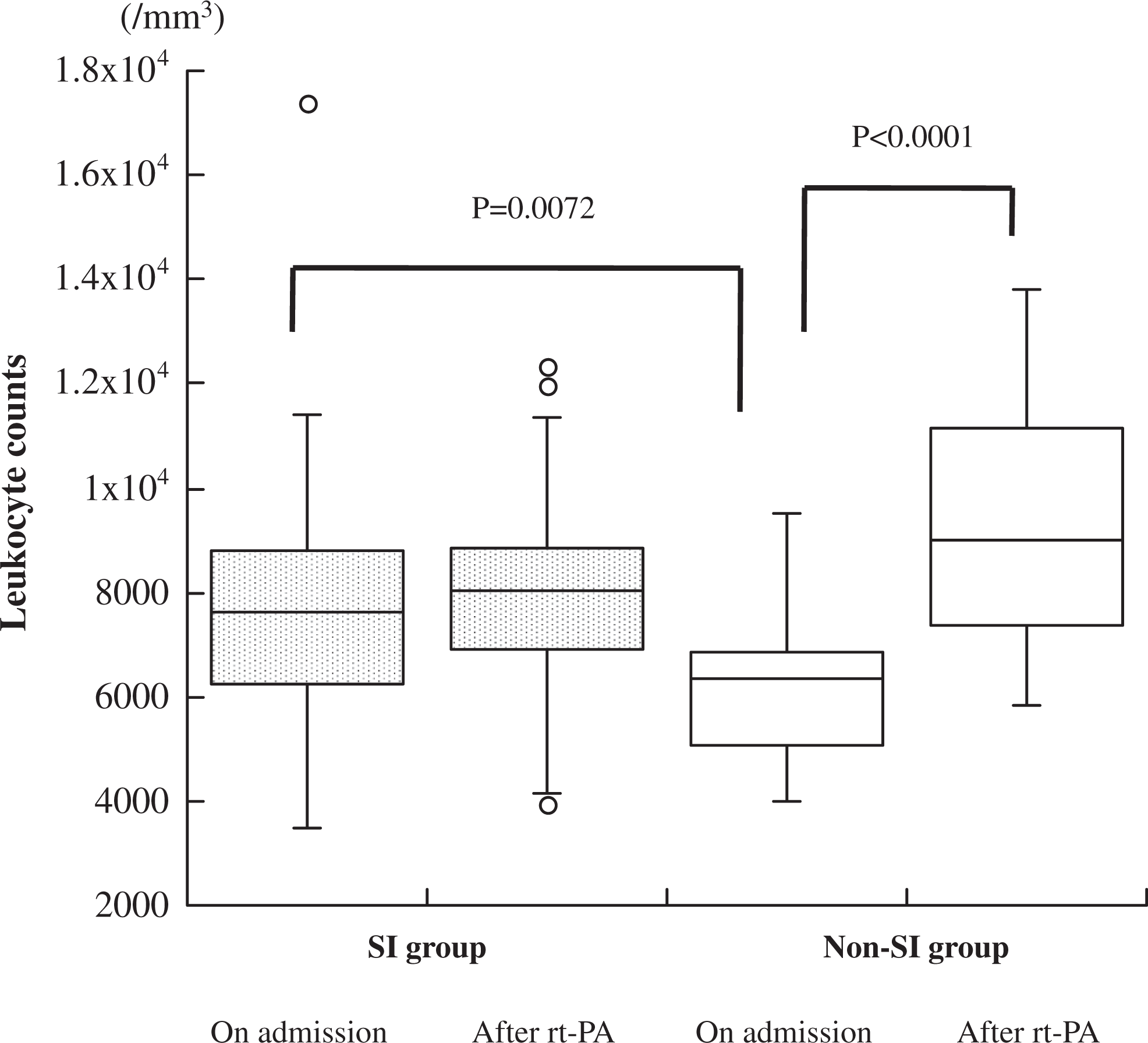
Change in leukocyte counts before and after rtPA treatment in SI and non-SI groups. Comparison between 2 groups was carried out using Wilcoxon rank sum test, and sequential changes were tested with Wilcoxon signed rank test. rtPA indicates recombinant tissue plasminogen activator; SI, significant improvement.
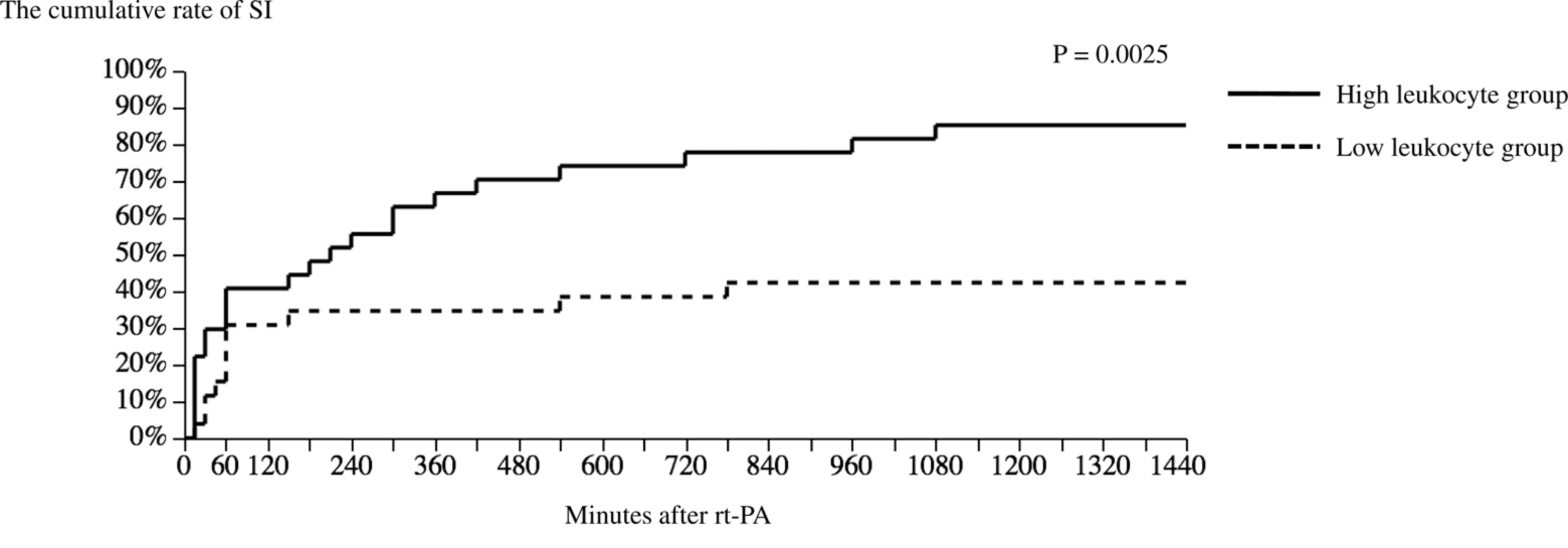
Comparison of cumulative significant improvement during 24 hours after rtPA between high leukocyte group and low leukocyte group. Kaplan-Meier curves and log-rank test were used for analysis. rtPA indicates recombinant tissue plasminogen activator; SI, significant improvement.
Hazard Ratio for Significant Improvement Using Cox Proportional Hazard Model.
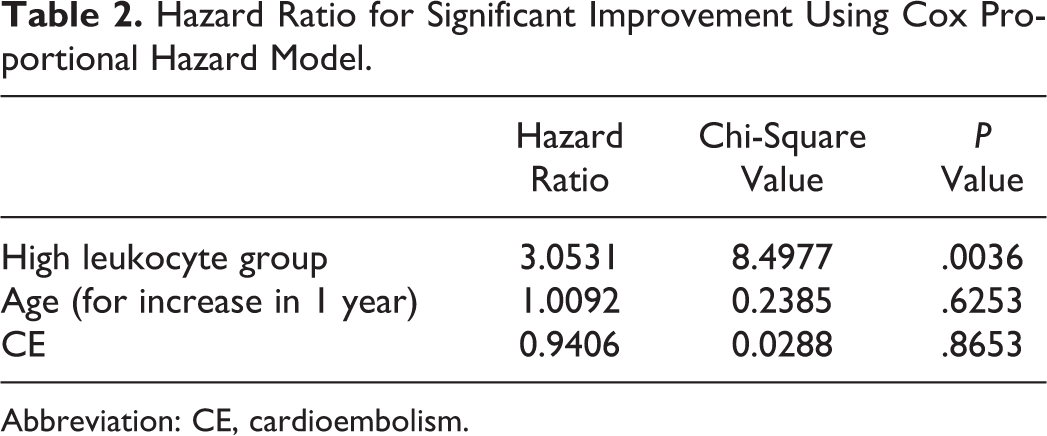
Abbreviation: CE, cardioembolism.
Discussion
In the present study, we found that leukocyte counts on admission were not correlated with initial stroke severity, and the higher values were associated with SI of neurological condition during the 24 hours after intravenous rtPA infusion in patients with acute ischemic stroke, although the effect was wiped out because of DFI.
It is generally believed that leukocytes have deleterious effects on patients with ischemic stroke via inflammation. 14 Recent studies have shown the association between higher leukocyte counts in the peripheral blood and initial stroke severity or poor outcome. 5,6 We previously investigated sequential changes in leukocyte counts in peripheral blood, finding increased values at the acute stage, especially in patients with large-vessel disease. 15 It was also reported that higher leukocyte counts were associated with reocclusion after thrombolysis with intravenous rtPA in ischemic stroke. 16
In addition to inflammation, leukocytes have an important role in fibrinolysis. 17 The most efficient fibrinolytic enzyme is plasmin, and some plasminogen activators like rtPA that enable the formation of plasmin from plasminogen are used in clinical practice as fibrinolytic therapy. Other enzymes such as neutrophil elastase, cathepsin G, and urokinase from leukocytes, which are not probably involved in physiological fibrinolysis, can act as fibrinolytic agents under special conditions. 7 It was reported that leukocytes from peripheral blood in patients with acute cerebral ischemia had smaller fibrinolytic capacity than those of control participants in an in vitro fibrinolysis model. 18 However, in the case of a very limited local area, like an occluded vessel, this may not be true. Because there are a considerable number of leukocytes in human thrombus, 10 fibrinolytic enzymes from leukocytes may work by themselves or promote plasmin-dependent fibrinolysis around the thrombus. Furthermore, apart from this plasmatic fibrinolysis, leukocytes themselves in cooperation with singlet oxygen can consider fibrin to be a pathogen and destroy it. 8,9 Audebert et al investigated the sequential changes of leukocyte counts in patients with ischemic stroke treated with rtPA, showing higher (but not significant) median leukocyte counts before rtPA therapy in patients with significant NIHSS improvement than in those without it. 19 Ribo et al also reported higher (but not significantly higher) mean leukocyte counts in recanalized patients with rtPA after 1 hour compared with those in nonrecanalized patients. 20 From these findings, we speculated that higher leukocyte counts in peripheral blood were linked to more leukocyte rich thrombus, which caused a higher rate of SI of neurological condition in HLG of our study via both plasmatic and cellular fibrinolysis.
However, sustained higher leukocyte counts in patients with acute ischemic stroke probably reflect systemic inflammatory response, leading to worse outcome. 14 In the present study, initial NIHSS scores were correlated with leukocyte counts at the first blood test after rtPA treatment rather than those on admission, and leukocyte counts increased after rtPA treatment in the non-SI group, but not in the SI group. Another study also found that systemic inflammatory response was attenuated by successful thrombosis. 19 Another possible downside of leukocytes is their relationship with severe hemorrhage after rtPA therapy. 21 Metalloproteinase 9 plays important roles in hemorrhagic transformation after ischemic stroke and its release from leukocytes was shown to be promoted by rtPA therapy. 22
Since excessive generation of plasmin after rtPA therapy may also cause hemorrhagic transformation, clinical fibrinolysis may imitate the physiologic selective fibrinolysis that is mainly of cellular origin. 23 Using leukocytes themselves in clinical practice is not recommended owing to their unfavorable effects as mentioned above. However, the use of fibrinolytic enzymes such as neutrophil elastase, which also has the capacity to prolong the lifespan of fibrin-bound plasmin, 24 may enhance the effect of rtPA therapy, avoiding the downsides of leukocytes.
In conclusion, we found that leukocytes might have 2 opposing effects in intravenous rtPA treatment for ischemic stroke. Our study was small in size, and the data were limited to a single center. A larger multicenter study should be performed to confirm our results. Additionally, it is necessary to investigate whether the selective utilization of the fibrinolytic function of leukocytes can enhance the effect of rtPA in patients with acute ischemic stroke.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
