Abstract
Introduction
Thrombosis in neonates is a rare but serious event, with an incidence of 2.4 per 1000 neonates in the intensive care unit. 1 Severity ranges from asymptomatic thrombus to fatal events, with mortality ranging from 9% to 18%.1–3 In the neonatal period, the perinatal risk factors associated with the developing hemostatic system increase neonatal susceptibility to thromboembolic events. With the widespread use of central venous catheters in neonates, the survival of preterm infants has increased, but it has been accompanied by an increased risk of thrombotic complications. Other risk factors for thrombosis are high hematocrit levels, increased plasma viscosity, maternal diabetes, sepsis, disseminated intravascular coagulation, congenital heart disease, maternal antiphospholipid syndrome, and inherited prothrombotic disorders. 4
The balance between the fibrinolytic system and the coagulation system determines thrombus formation in response to endothelial injury. In children, these systems exhibit age-dependent differences, including a lower plasminogen concentration than in adults and thus reduced plasmin generation. 5
The varying risks of thrombus formation resulting from the above-described situations highlight the requirement for a patient-specific thrombolytic therapy in neonates and infants. However, in contrast to the extensive experience in adult patients, there are few studies of fibrinolytic treatment in neonates. In recent years, anticoagulation using streptokinase, urokinase, and recombinant tissue plasminogen activator (r-tPA) has become the main treatment modality in the management of thromboembolic events in children and neonates. The fibrin-specific human r-tPA promotes the transformation of plasminogen into plasmin, which then stimulates the fibrinolytic pathway for thrombus resolution. The drug has been used to treat arterial and venous thrombosis and has many advantages over urokinase and streptokinase. 6
Low-molecular-weight heparin (LMWH) may be used both for primary prophylaxis and for the treatment of thromboembolism. Plasma binding of LMWH differs between different age groups, and its anticoagulant effect is reduced in neonates. 6 Enoxaparin is the preferred form of LMWH currently in use.
The literature on neonatal thrombosis consists of case reports and small series. In this report, thrombosis was evaluated in 17 neonates hospitalized in the neonatal intensive care unit. As such, ours is the largest series reported in the literature to date.
Patients
This retrospective analysis was performed on newborn infants with arterial or venous thrombosis who were followed from January 2007 to December 2009 in the neonatal intensive care unit of Zekai Tahir Burak Maternity Teaching Hospital, in Ankara, Turkey. The study was approved by the local ethics committee. Especially, high-risk pregnancies are followed at our hospital, which is also a tertiary neonatology center. Of the 25 000 infants born at the hospital each year, about 5000 are followed at its intensive care unit. As the incidence of premature births is high in pregnant women admitted to our hospital, the newborns frequently require central venous catheters. Among the infants catheterized during the study period, thrombosis was diagnosed in 17 of them.
Methods
In our unit, we perform echocardiography to all newborns that are below 30th gestational week and to the infants that had high risk factors for cardiac pathologies like respiratory distress, sepsis, infants of diabetic mothers, etc. We do not perform echocardiography to all patients that have umblical arterial or venous catheter if the patient is asypmtomatic. All echocardiograms for atrial thrombosis were performed by a stuff pediatric cardiologist. The arterial thrombosis other than atrium or venous thrombosis were investigated only if there were findings like pallor, hyperemia, or cyanosis of the extremities. For arterial or venous thrombosis, doppler ultrasonography was performed by a radiologist. Gestational age, birth weight, gender, prenatal history, diseases such as sepsis, respiratory distress syndrome, polycythemia, dehydration, congenital heart disease, retinopathy of prematurity, catheterization, cranial ultrasound and echocardiography findings, laboratory data, thrombosis panel, and type of thrombolytic therapy were recorded.
All patients were evaluated for clinical, radiological, and biological parameters before receiving thrombolytic therapy. Complete blood cell counts, activated partial thromboplastin time, fibrinogen, prothrombin time, coagulation factors, and thrombosis panel were determined. Contraindications to thrombolytic therapy were thrombocytopenia (< 100 × 109/L), low fibrinogen concentration (<100 mg/dL), and severe coagulation factor deficiencies. 7 Cranial ultrasonography was performed to evaluate intracranial hemorrhage before and after the completion of therapy.
All patients were monitored in the neonatal intensive care unit. Infants receiving thrombolytic therapy did not undergo invasive procedures like peripheric central catheterization or surgery. The infusion dose of r-tPA was 200 µg/kg per hour over a 6-hour period for 6 days. Plasma fibrinogen was determined before each r-tPA dose. Echocardiography or Doppler ultrasonography was performed at the end of the sixth day of treatment.
All patients except the patient 9 received enoxaparine at a dose of 1.5 mg/kg, q12 hour.
Results
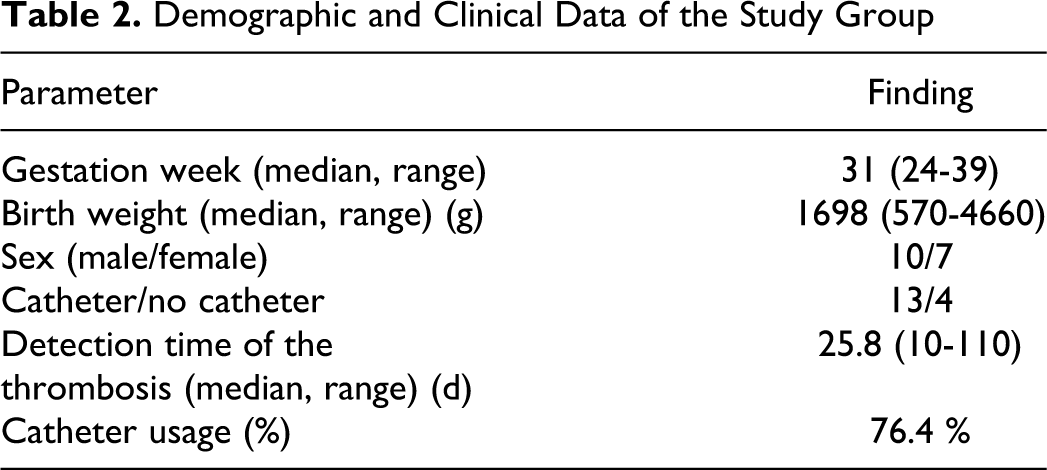
Thrombosis was diagnosed in 17 infants. In 10, the thrombus was located in the atrium and was diagnosed echocardiographically. Two newborns had thrombosis of the femoral vein; in one patient the thrombosis was located in the radial vein and in another the brachial vein. In one neonate, the axillary artery was involved and in another the femoral artery; both of these cases were diagnosed with Doppler ultrasonography. In 1 infant, cranial MRI revealed a thrombus in the sagittal sinus. The clinical characteristics of the infants are listed in Table 1 , and their demographic and clinical features in Table 2.
Clinical Characteristics of the Patients
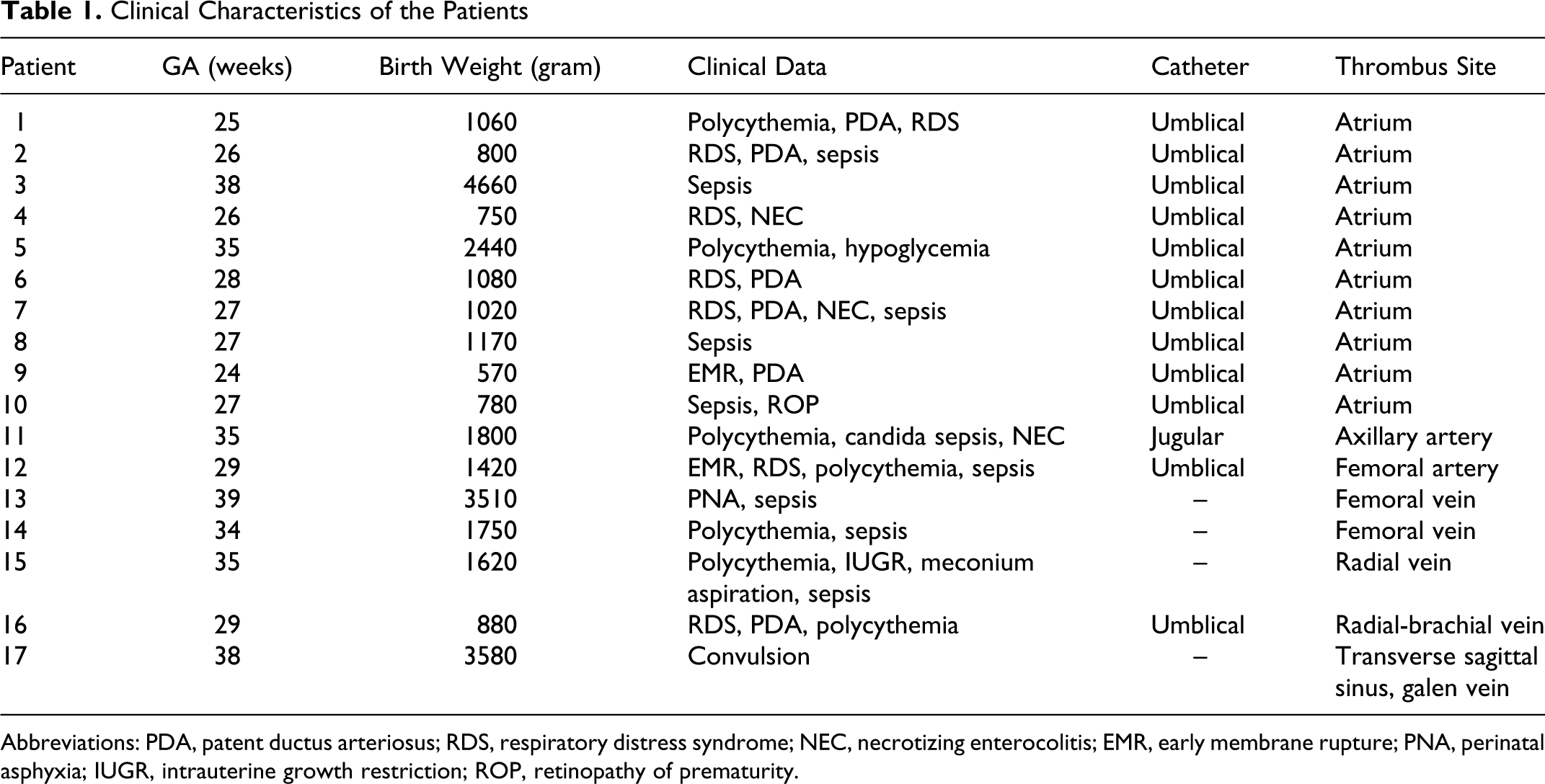
Abbreviations: PDA, patent ductus arteriosus; RDS, respiratory distress syndrome; NEC, necrotizing enterocolitis; EMR, early membrane rupture; PNA, perinatal asphyxia; IUGR, intrauterine growth restriction; ROP, retinopathy of prematurity.
Demographic and Clinical Data of the Study Group
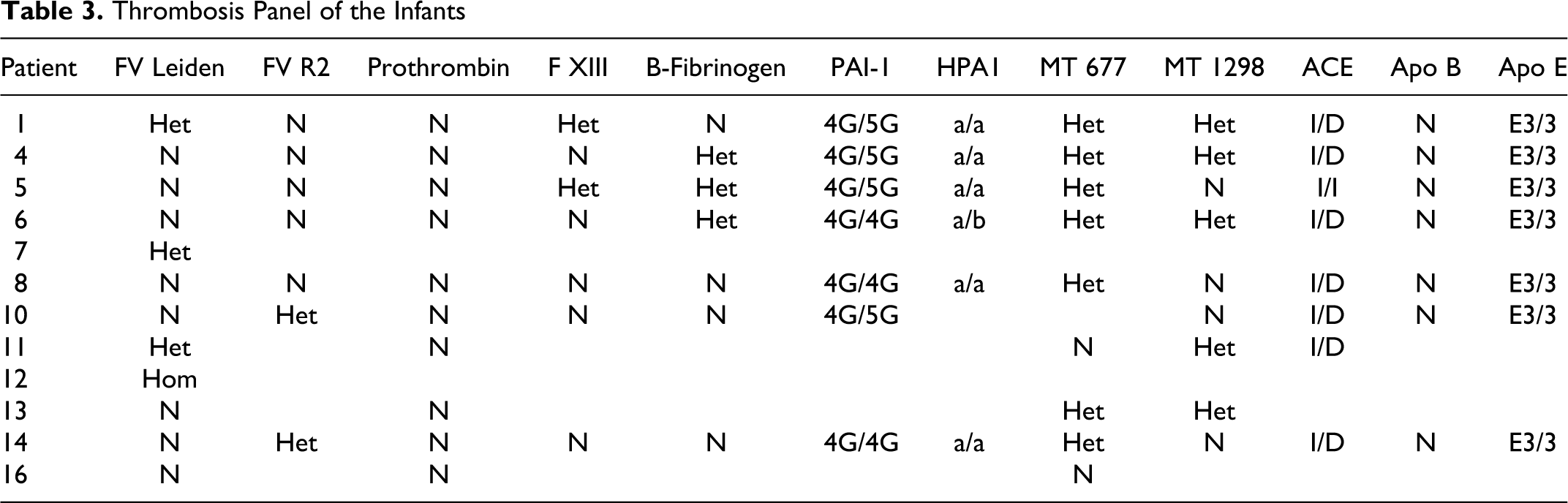
The thrombosis panel could be evaluated in 12 patients (Table 3). We could not evaluate lipoprotein-a concentrations that may be an important thrombophilic marker because of technical impossibility. Nearly all of our patients had heterozygous mutations that increased the risk of thromboembolic disease. Patients 1, 7, and 11 had heterozygous for Factor V leiden mutation. The patient 12 who underwent limb amputation, was found to have homozygous Factor V leiden mutation. The patients 6, 8, and 14 had homozygocity for the 4G polymorphism of the PAI-1 gene and the patients 1, 4, 5, and 10 were 4G/5G for the PAI-1 gene. Methylenetetrahydrofolate reductase (MTHFR) mutations were heterozygous nearly in all patients that were analyzed.
Thrombosis Panel of the Infants
Sixteen patients were treated with enoxaparin (Clexane®), with an initial dose of 1.5 mg/kg, administered twice daily subcutaneously. Ten of these patients also received a total of 11 courses of r-tPA therapy.
One patient died because of sepsis and prematurity at postnatal day 15, which was also the day of thrombus detection; thus, this patient could not be treated (case 9). A neonate with a radial vein thrombosis (case 15) died because of sepsis at postnatal day 26; as this was the end of the first day of treatment, thrombus evaluation was not possible. A patient who was homozygous for a mutation in factor V Leiden had a thrombus in his femoral artery; unfortunately, he did not respond to treatment and amputation of the extremity could not be avoided (case 12). Patients 13 and 14 had femoral vein thrombosis. We used r-TPA in patient 13 who had femoral venous thrombosis because of the presence of thrombus that was larger depending on the Doppler ultrasonography which revealed almost complete occlusion.
None of the patients experienced therapy-related hemorrhagic complications. Fibrinogen counts were low in 4 patients who were therefore given fresh frozen plasma prior to therapy.
Clinical improvement was observed in all patients except the 1 requiring amputation and the 2 patients who died before they could be evaluated. In 1 patient, retreatment was necessary, because of thrombus recurrence at the same site 1.5 months after the first r-tPA injection (case 1). Recurrence was most likely due to the patient’s prothrombotic risk factors. Sixteen patients received LMWH at the dose indicated in Table 4. A complete decrease in thrombus size was achieved in 9 patients, a partial decrease in 5 patients, and 2 patients, as noted above, could not be evaluated.
Treatment Modality and the Doses of the Therapy
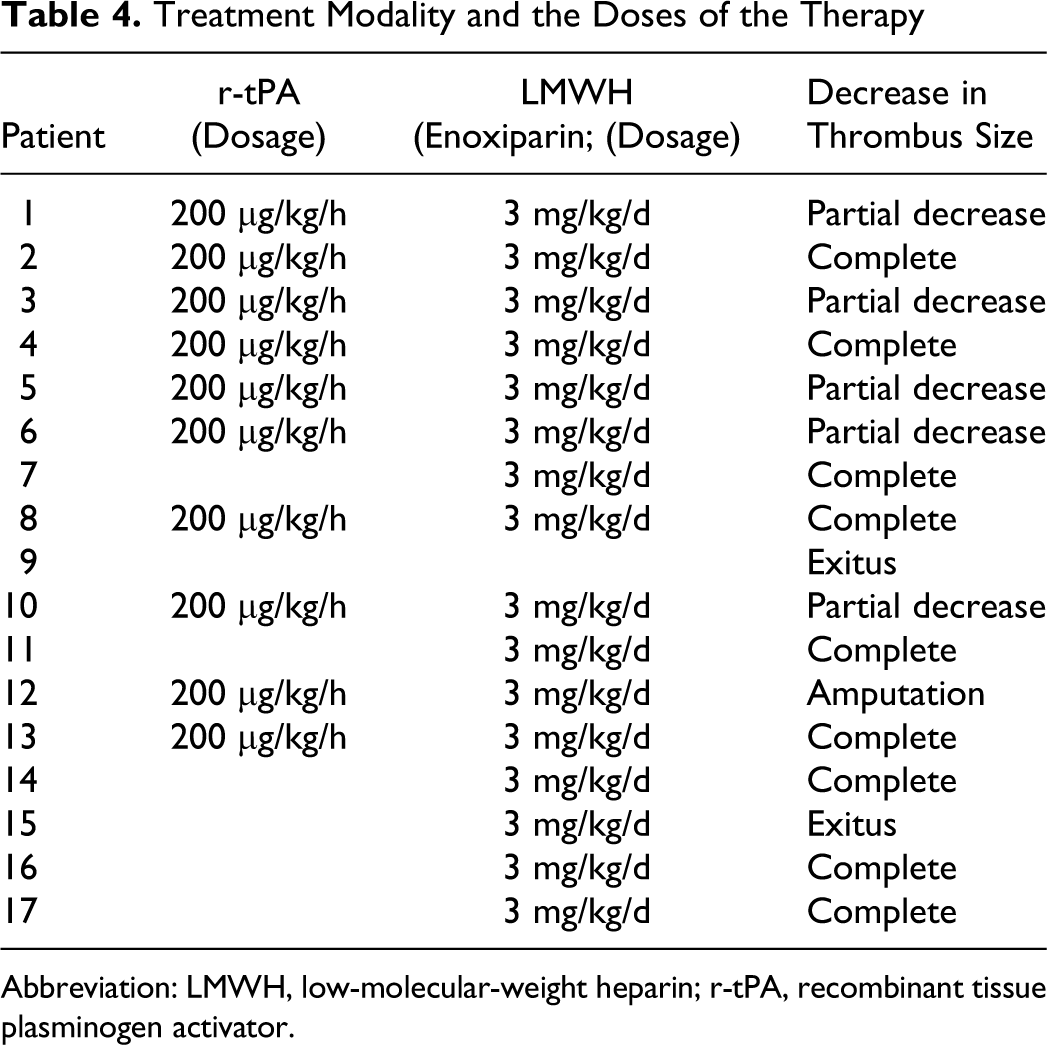
Abbreviation: LMWH, low-molecular-weight heparin; r-tPA, recombinant tissue plasminogen activator.
Discussion
The incidence of neonatal thrombosis is low; however, because it leads to serious complications, urgent treatment is necessary. One of the major causes of thrombosis in hospitalized neonates is the extensive use of catheters in this population. The neonatal hemostatic system differs from that of adults such that neonates are more vulnerable to thrombosis. Neonatal plasminogen concentrations are about 50% of those of adult values and they increase only gradually during the first 6 months of life. 8 Recently, it was shown that plasmin generation induced by r-tPA in vitro was lower in newborn than in adult plasma, which could explain the clinical finding that the elimination of thrombi in newborns is more challenging than in adults. 5
During the neonatal period, thrombin formation, endothelial activation, and procoagulant activity are increased. In our patients, catheter usage was 76.4%, but pathologies that increase the risk of thrombosis and lead to clinical deterioration, such as sepsis, hypoxia, polycythemia, and mutations in thrombosis markers, were also present.
Treatment strategies differ between different centers. Although there is no consensus regarding the dose of r-tPA, 2 therapeutic strategies are frequently used: high-dose and low-dose regimens. In the high-dose regimen, 0.5 to 0.6 mg r-tPA/kg per hour is typically given as a 6-hour infusion, after which the thrombus is reevaluated. If thrombus resolution is limited, treatment is repeated at the same dose 24 hours later. 9 This method should be used when rapid resolution of thrombus is life-saving. However, the high-dose regimen is associated with an increased risk of hemorrhage. 10 In the low-dose regimen, 0.03 to 0.06 mg r-tPA/kg per hour is administered; this regimen can be continued for 48 to 96 hours and hematological parameters should be followed closely. 7 Although in the low-dose regimen thrombus resolution is slower, it is the preferred approach for the treatment of venous thromboembolic events.
There is no clear recommendation regarding the dose of r-tPA in newborns. In our study, 0.2 mg r-tPA/kg per hour was administered as a 6-hour infusion each day for 6 days.11–15 Farnoux et al reported on r-tPA usage for arterial or venous thrombosis in 16 neonates. 16 An initial r-tPA bolus of 0.1 mg/kg was followed by the infusion of 1 mg/kg over a 3-hour period. Two patients in the series died, but clinical improvement was observed in 14. One of the patients who died had been treated despite severe thrombocytopenia and developed massive and fatal intracranial hemorrhage. No recurrence of the thrombosis was observed in the successfully treated patients.11,12 Marks et al used r-tPA at a dose of 0.2 mg/kg per hour, infused for 6 hours, to resolve vegetative intracardiac lesions in infective endocarditis caused by catheters. 17 In the series of Michelson et al, 0.1 to 0.6 mg/kg per hour was administered without bolus for 6 hours. 15 Giuffrè et al used r-tPA to treat catheter-related intracardiac thrombi in 2 premature infants. Thrombus dissolution was observed in both and no adverse effect was reported. 18 Van Overmeire et al treated an intracardiac thrombus in a neonate with r-tPA; complete resolution and clinical improvement were observed. 19 Andersen et al used a low dose of r-tPA (0.05 mg/kg per hour), infusing the drug directly into a thrombus that had developed secondary to a central catheter. This approach was successful in 3 of the 4 patients thus treated; the fourth neonate suffered an intracranial hemorrhage. 20 Torres-Valdivieso et al, in a review of all reports of r-tPA use in neonates between 1970 and 2002, found an overall patency rate of 90% (70% for complete clot dissolution and 20% for partial clot dissolution). 21
The advantages of r-tPA are that it is nonantigenic, specific for fibrin, has a short half-life, and can be produced by recombinant DNA techniques. The drug’s main complication is severe hemorrhage, particularly at puncture or recent catheterization sites (10.4%), intraventricularly (2.7%), in the lungs (0.6%), and in the gastrointestinal tract (0.6%). 4 Additionally, r-tPA is also used for restoring occluded central venous catheters. Soylu et al reported that local instillation of r-tPA successfully restored function to occluded central venous catheters, without subsequent complications. 22
In neonatal intensive care units, enoxaparin, an LMWH, has been used in anticoagulant therapy. Malowany et al reviewed the literature from 1980 to 2007; the mean maintenance dose of enoxaparin ranged from 1.48 to 2.27 mg/kg q12 hours for all infants but was higher for preterm neonates at 1.9-2.27 mg/kg q12 hour. They recommended starting doses of enoxaparin of 1.7 mg/kg, administered over 12 hours, for term neonates and 2.0 mg/kg, administered over 12 hours, for preterm neonates if there is no considerable bleeding risk. In patients with a bleeding risk, 1.5 mg/kg per 12 hours was recommended. 11 In this report, we used enoxaparin at a dose of 1.5 mg/kg q12 hours, depending on this review. We thought that our patients had increased risk of bleeding as we used r-tPA associated with enoxaparin in most patients and most of them had complications of prematurity that may increase the risk of bleeding such as sepsis. Many side effects of enoxaparin such as local adverse effects (induration, bruises, hematomas), osteopenia, scleral hemorrhage, gastrointestinal tract bleeding, thrombocytopenia, thrombocytosis, allergic reactions, transient elvations in liver function tests were reported. None of those side effects were observed in our patients.23–25
Moreover, none of the 10 patients treated with r-tPA and LMWH concomitantly had therapy-related hemorrhagic complications. Thus, combined antithrombotic and anticoagulation therapy was used safely in our study group, with a much lower incidence of hemorrhagic complications than reported in many other studies.
In 1956, Jordan and Nandorff described a familial tendency in thromboembolic diseases. 26 In 1965, antithrombin deficiency was described as an inherited defect. 27 Later, reduced levels of proteins C and S were determined to cause familial thrombosis. 28,29 In the 1990s, protein C resistance, factor V Leiden, and prothrombin allele 20210 mutations, all of which led to enhanced coagulation, were discovered.30,31 Nonetheless, identification of a thrombophilic marker does not influence the acute management of these patients, except in those with a deficiency in proteins S or C or antithrombin, in which case replacement therapy is life-saving. 32
Elevated plasma prothrombin and fibrinogen levels resulting from mutations/polymorphisms in factor II (FII 20210A) and fibrinogen genes have been considered to be associated with an increased risk of thrombosis. 33 Clinical studies showed that factor V Leiden mutation is associated with increased risks for venous thromboembolism. 34 The prevalence of factor V Leiden mutation and factor II prothrombin gene mutations was reported at about 5% to 10% and 2%, and homozygocity with a prevalence of 0.02% and 0.014%, respectively. 35 Factor V Leiden mutation prevalence is 10% in Turkey. 36 Three of our patients had heterozygous mutations of factor V Leiden and the patient 12, who underwent limb amputation, was found to have homozygous factor V leiden mutation.
Sartori et al 37 reported a positive association between venous thrombosis and 4G/4G polymorphism of the PAI-1 gene. However, Stegnar et al 38 reported that the 4G/5G mutation was not a major risk factor for thromboembolism. The patients 6, 8, and 14 had homozygocity for the 4G polymorphism of the PAI-1 gene and the patients 1, 4, 5, and 10 were 4G/5G for the PAI-1 gene.
Cadroy indicated that clot retraction is enhanced by the involvement of platelets with the HPA-1b/1b genotype and that thrombi formed from HPA-1b/1b platelets are more resistant to thrombolysis than the thrombi formed from HPA-1a/1a platelets. 39 The HPA-1a/1b polymorphism of the human platelet antigen 1 (HPA-1), localized on the platelet glycoprotein GPIIIa, has also been identified as an inherited risk factor for atherothrombosis, enhancing platelet activation. 40
Associations of neonatal thrombosis and other thrombophilic defects such as the methylenetetrahydrofolate reductase (MTHFR) T677T genotype have recently been reported. 41 Methylenetetrahydrofolate reductase mutations were heterozygous nearly in all patients that were analyzed. It should be kept in mind that thrombosis is often a multifactorial problem in which acquired risk factors are more important than inherited ones, especially in the neonatal period.
In conclusion, neonatal thrombosis may be a very serious event, requiring rapid intervention. Our study of thrombosis in the neonatal period and the efficacy of r-tPA and anticoagulant therapy was based on 17 patients and is the largest series reported to date. The therapeutic approach evaluated was shown to be a safe and effective method for clot dissolution in neonates.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
