Abstract
Background:
Patients with coronary artery disease (CAD) should be treated with statins to attain very low cholesterol levels, in order to reduce cardiovascular adverse events. More than 70% of these patients do not reach the appropriate cholesterol goal despite moderate statin doses. However, it is not known whether therapeutic uptitration with different lipid-lowering strategies has a similar “pleiotropic” effect on atherosclerotic endothelial dysfunction evaluated by measurement of endothelial progenitor cells (EPCs).
Objective:
We sought to compare, in patients with stable CAD and with a low-density lipoprotein cholesterol (LDL-C) >70 mg/dL on treatment with simvastatin 20 mg, the effects on EPCs by increasing simvastatin to 80 mg versus adding ezetimibe 10 mg.
Methods:
Patients (n = 68, 63 ± 9 years, 39% men) were randomly allocated to receive ezetimibe 10/simvastatin 20 mg or simvastatin 80 mg for 6 weeks. Circulating EPCs were measured by flow cytometry before and after the treatment.
Results:
Both strategies presented similar effects on metabolic parameters. The LDLs were equally reduced by ezetimibe 10/simvastatin 20 mg and simvastatin 80 mg (28.9% ± 13% vs 21.1% ± 33%;
Conclusions:
Among stable patients with CAD and with an LDL-C >70 mg/dL on simvastatin 20 mg, increasing simvastatin dose to 80 mg or adding ezetimibe 10 mg promoted similar further cholesterol reduction but did not have incremental effects on circulating EPCs. These data suggest that the effects of simvastatin moderate doses on EPCs are not increased by intensive lipid-lowering strategies (clinicaltrials.gov: NCT00474123).
Introduction
Among patients with coronary artery disease (CAD), a robust evidence base supports the beneficial effects of statin therapy on mortality and other adverse cardiovascular outcomes. 1 Additionally, large trials have demonstrated that very low levels of low-density lipoprotein cholesterol (LDL-C) achieved by statins are associated with a significant cardiovascular risk reduction.2,3 Current guidelines recommend that patients with CAD, considered as a high-risk group, should be treated with statins to attain an optimal LDL-C level < 70 mg/dL. 4 More than 70% of the high-risk patients treated with statins on monotherapy, particularly at starting doses, do not reach this LDL-C goal and need further therapeutic titration. 5 In this scenario, 2 alternative pharmacologic strategies are currently available to promote additional cholesterol reduction: a high-dose statin alone or a combination of ezetimibe plus a moderate-dose statin. 6 It is unknown whether these 2 strategies have different cholesterol-independent effects.
Beyond the benefits of LDL-C reduction, pleiotropic effects of statins on inflammation and coagulation might explain, at least in part, the large benefits demonstrated in randomized trials.7,8 Moreover, statins may also improve endothelial function, increasing the levels of endothelial progenitor cells (EPCs) via the phosphoinositide 3-kinase/Akt pathway. 9 The EPCs contribute to reendothelization and neovascularization particularly in vascular injured sites. 10 In patients with CAD, however, the reduction in EPC levels is related to poor cardiovascular outcomes.11,12
Hence, in patients treated with a moderate statin dose, but still out of the optimal LDL-C goal, the effects of statin uptitration versus combining ezetimibe to the initial statin dose on EPCs were not compared before. Therefore, in this study, we aimed to compare the effects of simvastatin 80 mg versus ezetimibe 10/simvastatin 20 mg on the circulating EPCs of patients with stable CAD and with an LDL-C >70 mg/dL, who were previously treated with simvastatin 20 mg.
Methods
Study Design
A randomized trial testing lipid-lowering drugs was performed from July 2006 to January 2009 in a single tertiary specialized cardiology hospital (clinicaltrials.gov: NCT00474123). 13 From the original group of 83 patients, we performed flow cytometry for measurement of EPCs in 68 patients (63 ± 9 years, 39% men). All the patients met the following inclusion criteria: angiographically documented CAD defined as a coronary obstruction >50%, stable or no angina, simvastatin treatment (20 mg/d) for >4 weeks, LDL-C >70 mg/dL, and age between 18 and 80 years. Exclusion criteria were a history of myocardial infarction or revascularization within the last 3 months, moderate/severe left ventricular systolic dysfunction (ejection fraction <45%), warfarin treatment, malignancy, inflammatory diseases, creatinine >1.5 mg/dL, active liver disease or known liver cirrhosis, and unexplained transaminase increase (>3-fold of normal).
All the patients were randomly assigned in a double-blind fashion and 1:1 ratio to receive either ezetimibe 10/simvastatin 20 mg or simvastatin 80 mg for 6 weeks. The experimental regimen was initiated without a washout period of simvastatin. Fasting venous blood samples, drawn immediately after randomization and at the conclusions of the 6-week study period, were used for the evaluation of metabolic parameters and EPCs. All the samples (collected on baseline and at 6 weeks) were analyzed at the same time.
An informed consent was obtained from each patient, and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Endothelial Progenitor Cells
The EPCs were characterized according to cell surface antigen expression, following a previous description. 14 In our experiments, the peripheral blood lymphomononuclear cells were isolated by density gradient (Ficoll-Paque). As blood samples were drawn from distinct patients at different time points, the density gradients were performed as each sample was received and the resulting lymphomononuclear cells were frozen at −80°C. Later on the cells were thawed, and phenotyping of all samples was performed at the same time. Briefly, 100 µL of mononuclear cells were incubated with monoclonal antibodies against human CD31/fluorescein isothiocyanate (FITC; BD Biosciences, Frank Lakes, New Jersey); vascular endothelial growth factor receptor (VEGFR)-2/phycoerythrin(PE; R&D Systems, Minneapolis, Minnesota); CD3/PE-cyanine (Cy) dye 5 (BD Pharmingen, San Diego, California); CD19/PE-Cy5 (BD Pharmingen); CD34/PE-Cy7 (BD Pharmingen); CD14/allophycocyanin-Cy-7 (BD Pharmingen); and related isotype controls. After 30 minutes of incubation at room temperature, the cells were washed with phosphate-buffered saline and fixed in 1% paraformaldehyde. At least 70 000 events were acquired per tube by flow cytometry (FACSARIA; BD Biosciences, San Jose, California). Data analysis was performed using FACSDIVA software (BD Biosciences). The cells were gated on a forward-scattered versus side-scattered light plot (P1, Figure 1A). Next, P1-gated cells were further analyzed (Figure 1B) as follows: cells that expressed CD3 or CD19 (PE-Cy5) were excluded (dump channel), and CD31-positive cells (FITC) were selected. The cells were then evaluated for CD14, VEGFR2, and CD34. The CD3 and CD19 antibodies were used to exclude lymphocytes, and the CD14 antibodies were used to eliminate monocytes from the analysis. Thus, the cells negative for CD3, CD19, and CD14 and those positive for CD31, CD34, and VEGFR2 were labeled as EPCs.
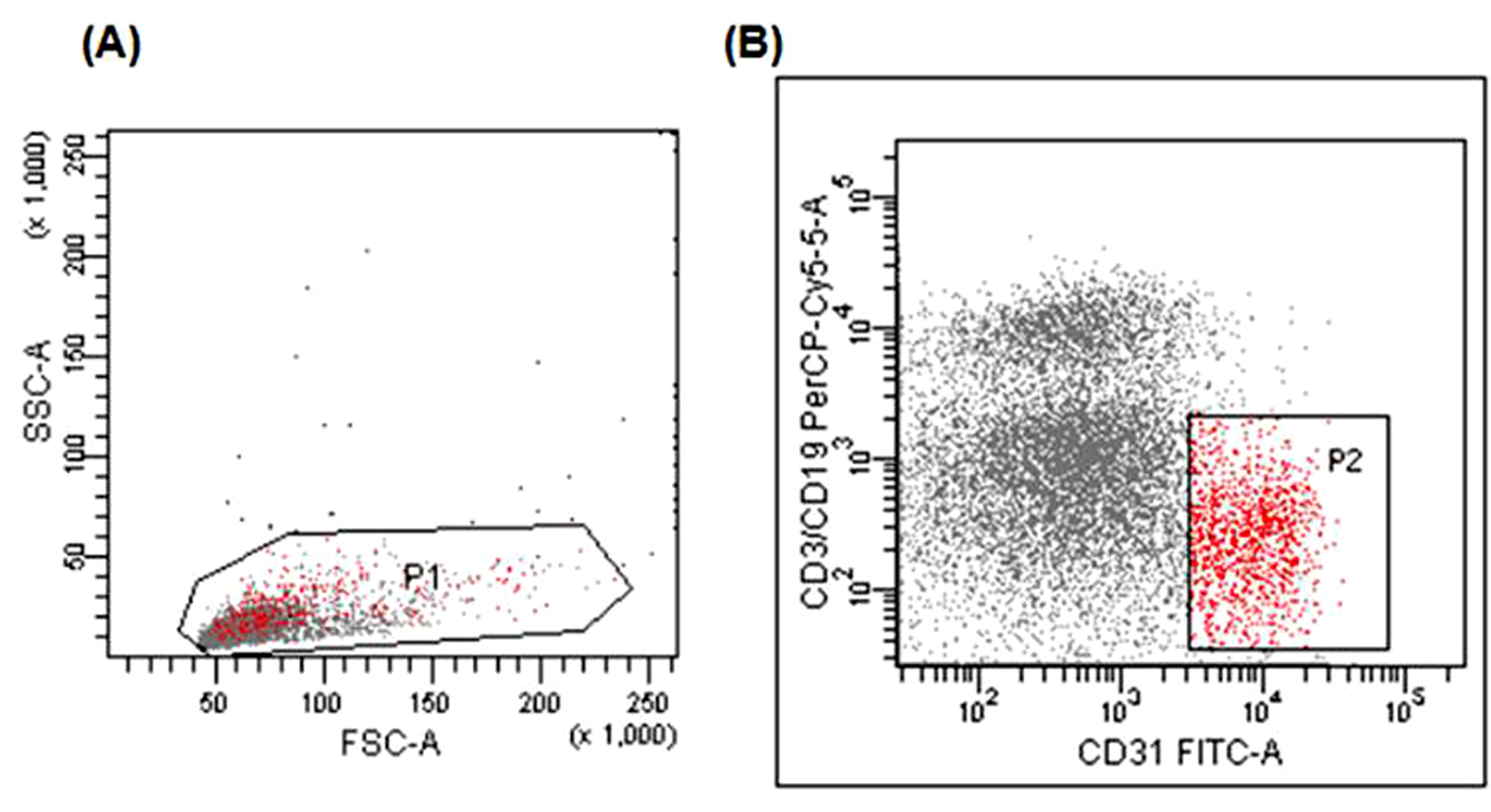
A, Plot representing forward-scattering (FSC) versus side-scattering (SSC)-gated cells (P1). B, Representative plot showing CD31-positive, CD3/CD19-negative cells (P2). Analysis included only P1-gated cells.
To assess reproducibility, the circulating EPCs from each patient (n = 22) were measured twice using 2 independent blood samples. A significantly high correlation was obtained from those readings (
Statistical Analysis
Our sample size estimation was based on a study comparing the effect of high versus moderate statin doses on EPCs, in patients with acute myocardial infarction. 15 The sample size was determined as 54 patients. This would enable the study to have a power of 80%, with 2-tailed type I error of 0.05, to detect a difference of 4.6 cells/µL between the means of EPCs of the study groups, assuming a standard deviation (SD) of 5.6 cells/µL.
Continuous data are presented as means ± SD, or median (25th, 75th percentiles) when the distribution was nonnormal. For qualitative variables, we presented absolute and relative frequencies. Paired
Results
All the study patients underwent flow cytometry analysis. Baseline metabolic and lipid parameters were similar in both the groups (Tables 1 and 2). The baselines levels of EPCs were also similar between the groups (median [25th, 75th]: 7.0 [2.3; 13.3] vs 6.1 [2.9; 15.2] EPCs/104 mononuclear cells;
Baseline Characteristics of the Patients According to Treatment Group.a

Abbreviations: ASA, acetyl salicylic acid; ACE, angiotensin-converting enzyme inhibitor; AT, angiotensin; AMI, acute myocardial infarction.
aData are expressed as mean ± standard deviation or n (%).
Treatment Effects on Plasma Lipid Levels.a

Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglyceride; Apo, apolipoprotein; ALT, alanine transaminase; AST aspartate aminotransferase; CPK, creatine phosphokinase; CI, confidence interval.
aData are expressed as mean (95% confidence interval) ± standard deviation.
Total cholesterol, LDL-C, and apolipoprotein-B levels were equally reduced by both the treatments (Table 2). Importantly, the LDL-C was markedly reduced by ezetimibe 10/simvastatin 20 mg and simvastatin 80 mg (mean change % [95% confidence interval]: −28.9% ± 13% [−33%; −25%] vs −21.1% ± 33% [−33%; −1%];
The EPC levels were unaffected in those who received ezetimibe 10/simvastatin 20 mg (median [25th, 75th]: pre- vs posttreatment, 7.0 [2.3; 13.3] vs 3.1 [0.1; 13.2] EPCs/104 mononuclear cells;

Baseline (pretreatment) and 6 weeks (posttreatment) levels of endothelial progenitor cells/104 mononuclear cells in both the groups. Data are presented as medians, 10th, 25th, 75th, and 90th percentiles. E10/S20 indicates ezetimibe 10 mg/simvastatin 20 mg; S80, simvastatin 80 mg. Note. The asterisks and empty circles above the box plots represent the outlier patients.
Discussion
In our study, we have found that among stable patients with CAD and with an LDL-C >70 mg/dL previously treated with simvastatin 20 mg, both intensive lipid-lowering strategies, ezetimibe 10/simvastatin 20 mg and simvastatin 80 mg, did not affect the circulating EPCs levels.
The study population included patients who received moderate statin therapy (20 mg) but still had not achieved the optimal LDL-C therapeutic goal, according to the current guidelines. Our study was specifically designed to evaluate the pleiotropic effect in this setting of patients. As expected from prior studies, ezetimibe 10/simvastatin 20 mg and simvastatin 80 mg have similar incremental effects on cholesterol compared to simvastatin 20 mg. 6 Additionally, ezetimibe reduces cholesterol by a direct effect on intestinal absorption but has no effects on the mevalonate cascade, and consequently, no potential pleiotropic effect based on isoprenoid inhibition. Therefore, we hypothesized that any differences observed between the levels of EPCs in these 2 strategies would not be related to LDL-C’s further decrease, but to specific effects related to simvastatin dosing, 80 mg vs 20 mg.
In the past years, controversial evidence regarding the effects of statins on EPCs has emerged. In vitro studies have revealed that statins may increase proliferation, mobilization, and improve the survival of EPCs. 16 Subsequently, Vasa et al 17 have demonstrated that short-term treatment with atorvastatin was associated with a 50% increase in circulating EPC levels in patients with stable angina. In patients with acute myocardial infarction, 2 small trials demonstrated an increase in EPCs with atorvastatin or rosuvastatin.15,18 However, in a larger trial, Hristov et al 19 found that EPCs remained unaffected after 1 month of treatment with simvastatin 10 mg in patients with CAD. Surprisingly, in the same trial, after 3 months of treatment with simvastatin 40 mg, the EPC levels were significantly reduced. Increase in EPCs by statins demonstrated in previous studies has been restricted to short-term treatments. Thus, the initial increase in EPCs may be transitory and limited by neoendothelial incorporation and depletion of the EPC pool. 20 Notably, our baseline levels of circulating EPCs were comparable to previous studies in patients with CAD.9,19 However, the transient effect of statins on EPCs may be one of the reasons that we could not detect variations in both treatment strategies after 6 weeks.
Previously, the effects of ezetimibe on EPCs were poorly investigated. Similar to statin trials, combined treatment of ezetimibe/statin was tested in small short-term studies. Landmesser et al 21 found that treatment with simvastatin 10 mg (4 weeks), but not ezetimibe 10 mg, increased the EPC levels in patients with heart failure. Later Westerweel et al 22 demonstrated that treatment with simvastatin 80 mg and ezetimibe 10/simvastatin 20 mg (for 6 weeks) was equally effective in increasing the EPC levels among statin-naive patients with metabolic syndrome. On the other hand, patients already receiving simvastatin 20 mg but still with an LDL-C >70 mg/dL, we have found that a 4-fold increase in simvastatin dose or the combination of ezetimibe/simvastatin did not have any further effect on EPCs despite an additional 20% reduction in cholesterol levels.
In fact, some of the pleiotropic effects of statins may be concentrated in moderate doses and are not improved by more intensive strategies. In our original trial, simvastatin 80 mg and ezetimibe 10/simvastatin 20 mg showed a modest impact on inflammatory markers compared to simvastatin 20 mg. 13 Blanco-Colio et al 23 demonstrated that atorvastatin 10 mg reduced the levels of the intercellular adhesion molecule 1, but the dose increment to 80 mg did not produce any additional reduction. Similarly, in the Aggrastat to Zocor (A to Z trial), 24 minimal difference were observed in monocyte chemotactic protein 1 levels between patients receiving simvastatin 80 or 20 mg. Interestingly, the SEARCH trial 25 could not demonstrate advantages in the outcomes in more than 6000 patients receiving simvastatin 80 mg compared to 20 mg. That could be related to the small differences in cholesterol reduction between groups or possibly due to the absence of incremental pleiotropic effects by higher doses.
Therefore, in patients with CAD on previous treatment with simvastatin 20 mg and LDL-C >70 mg/dL, these data do not suggest differences in the effects on EPCs between ezetimibe 10/simvastatin 20 mg and simvastatin 80 mg.
Study Limitations
Our study has some limitations. The effects of statins on EPCs may be time dependent. Our treatment duration of 6 weeks may not be enough to detect the late effects of statins on EPCs. Additionally, the previous use of simvastatin 20 mg for at least 4 weeks and the absence of a washout period before randomization may have influenced our findings. Nevertheless, we believe our study design was justified as the screened patients were already on 20 mg simvastatin. We intentionally chose to avoid a washout phase, considering the high-risk nature of our population (CAD, diabetes, previous infarction), and we aimed to perform a real-world study, including patients already treated with a moderate statin dose but still out of the optimal cholesterol level therapeutic goal.
Conclusion
Among stable patients with CAD on simvastatin 20 mg and with an LDL-C >70 mg/dL, increasing simvastatin dose to 80 mg or adding ezetimibe 10 mg caused similar further decrease in cholesterol but did not have incremental effects on the circulating EPCs. These data suggest that the effects of moderate doses of simvastatin on EPCs are not increased by intensive lipid-lowering strategies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the São Paulo Research Foundation, Brazil (FAPESP/05/57710-3). No industry support was provided for this study.
