Abstract
Preeclampsia (PE) is a pregnancy disorder that increases maternal and fetal morbidity and mortality worldwide. High plasma levels of homocysteine (Hcy) are a risk factor for several cardiovascular diseases. Cystathionine β-synthase (CBS) plays an important role in Hcy homeostasis catalyzing the irreversible degradation of Hcy to cystathionine, protecting the endothelium from injury caused by hypoxia. Several mutations and polymorphisms may alter the expression of the CBS gene, resulting in variable levels of Hcy. The purpose of this study was to investigate the association of CBS gene polymorphisms with PE in Mexican women. A case–control study consisting of 129 pregnant women with PE (37 severe and 92 mild) and 173 women with uncomplicated pregnancies was performed. Polymorphisms, such as G797A, C785T, T833C, G919A, T959C, C1105T, and 844ins68 base pair, in the CBS gene were genotyped. The polymorphism G797A was monomorphic in cases with the presence of only G797A-G allele. Allele C785T-T and genotype C785T-C/T were associated with susceptibility in severe and mild PE. Alleles G797A-G and T959C-T were associated with susceptibility only in severe PE. Haplotype TGTWGTC was of susceptibility for severe PE and of protection for mild PE. Haplotypes CGTWGCC and CATWGTC seem to be protective for severe PE, but the latter is related to susceptibility in mild PE. The results suggest that C785T, G797A, and T959C mutations are contributing in different ways in severe and mild PE in our population and could be count as another related factor for this disease.
Introduction
Preeclampsia (PE) is a pregnancy disorder defined as the new onset of sustained hypertension, systolic blood pressure (BP) ≥140 mm Hg and/or diastolic BP ≥90 mm Hg, and proteinuria >300 mg/d arising after 20 weeks of gestation in a previously normotensive woman. In the absence of proteinuria, PE may be associated with any of the following: thrombocytopenia, renal insufficiency, or impaired liver functions. 1 The underlying physiopathology of PE comprises 2-stage process. 2 The first stage is characterized by reduced placental perfusion with impaired trophoblast invasion and inadequate remodeling of the uterine spiral arteries. The second stage refers to the maternal systemic manifestations characterized by inflammatory, metabolic, and thrombotic responses that converge to alter vascular function, resulting in multi-organ damage. 3,4 This disorder represents a primary cause of maternal and fetal morbidity and mortality.
It is widely accepted that early and late PE have different etiologies and hemodynamic states; therefore, they should be considered as different forms of the disease. 5 –7 Early-onset PE (before 34 weeks) appears to be more frequent in patients with lower body mass index (BMI) and is commonly associated with bilateral notching of the uterine artery Doppler, fetal growth restriction, and adverse maternal and neonatal outcomes. 7 –9 In contrast, late-onset PE (after 34 weeks) appears to be more frequent in patients with high BMI and low total vascular resistance and is more associated with a normal or mild uterine resistance index, a low rate of fetal involvement, and most favorable perinatal outcomes. 7,9,10
In the United States, the incidence for PE increased from 3.4% in 1980 to 3.8% in 2010. 11 Prevalence is between 2% and 10% worldwide, with ∼8.5 million women affected each year. 12,13 Until 2007 in Mexico, the PE represented ∼34% of maternal deceases, being the major cause of death during the pregnancy. 14
Endothelial dysfunction in the placental revascularization is distinctive of PE, 15 and altered levels of homocysteine (Hcy) have been involved as a predisposing factor. The Hcy is a sulfur amino acid originated from the metabolism of methionine, which oxidizes easily forming homocysteic acid and produces free radicals of superoxide anion and hydrogen peroxide, leading to damage of vascular endothelial cells. Thus, Hcy is an important indicator reflecting damage of vascular endothelial cells. 16,17 It has been described that there are high levels of Hcy, both in mild and severe PE, which indicates that there is a close relationship between Hcy and hypertensive disorders that complicate pregnancy and the severity of PE, presenting a directly proportional relationship between the level of Hcy and PE. 18,19
The human cystathionine β-synthase (CBS) is a homotetramer consisting of 63-kDa subunits, which binds 2 cofactors, pyridoxal 5′-phosphate (PLP) and heme. The amino acid conformation of each CBS subunit is of 551. The CBS plays a central role in the regulation of plasma Hcy, catalyzing the PLP (B6 vitamin)-dependent conversion of Hcy to cystathionine. 20 It degrades Hcy by condensation with either serine or cysteine. Cysteine is not only required on protein synthesis but also acts as a precursor of glutathione, a potent antioxidant. 21 In the latter reaction, the formation of hydrogen sulfide (H2S) takes place, a gasotransmitter involved in many inflammatory processes, such a sepsis and vascular homeostasis, 22 which protects the endothelium from injury caused by hypoxia. 23 The expression of CBS is tissue-specific, mainly found in the neural and cardiovascular systems, liver, kidney, and muscle. 24 There are evidence indicating that impaired Hcy metabolism is associated with risk of several diseases, such as atherothrombotic stroke, 25 neural tube defects, 26,27 and some cancers. 28,29 High levels of Hcy are also a risk factor for endothelial dysfunction and cardiovascular disorders. 30,31
To date, 164 CBS mutations and polymorphisms have been identified in CBS-deficient patients, of which 68% are missense mutations. 32 Some of these mutant alleles are present in all populations (eg, T833C), but others only occur in specific populations. However, there are also studies in which variants of CBS associated with different diseases such as arterial occlusive disease, primary hypertension, or pregnancy complications have been identified. 33 –36 The aim of this study was to analyze whether there is an association between the development of early or late PE and some CBS mutations/polymorphisms in a case–control study of Mexican women.
Materials and Methods
Patients and Controls
We performed a case–control study, including 129 pregnant women diagnosed with PE and 173 with uncomplicated pregnancies, paired by age and residence with the cases. Afterward, patients were divided into severe (n = 37) and mild (n = 92) PE with the following inclusion criteria: Mild PE comprised women after 20 weeks of gestation who had repeated hypertension ≥140 mm Hg for systolic BP and ≥90 mm Hg for diastolic BP or proteinuria ≥300 mg/d. Severe PE included women after 20 weeks of gestation with recurrent maximum systolic BP ≥160 mm Hg and/or maximum diastolic BP ≥110 mm Hg or had proteinuria ≥300 mg/d. The exclusion criteria considered obstetric complications other than PE, infections during pregnancy, consumption of medicines that potentially affects the metabolism of lipids and carbohydrates, use of alcohol or other illicit drugs, and preexisting hypertension before pregnancy. The definition of the control group was women who, after 20 weeks of pregnancy, did not present signs of hypertension or any other complication. The recruitment of patients and controls was between 2004 and 2005 at the Department of Obstetrics and Gynecology of the Hospital General “Dr Manuel Gea Gonzalez.”
This work complies with the current health laws of Mexico and approved by the Ethics in Research and Research Committees of the Hospital General “Dr Manuel Gea Gonzalez.” The objective of the study was notified to all participants, who were included only after providing written informed consent.
DNA Extraction and Genotyping
DNA was obtained from 10 mL of EDTA-peripheral blood using proteinase K and phenol/chloroform protocol. 37 The single-nucleotide polymorphisms (SNPs) of CBS analyzed were 2 for exon 7 (C785T [rs149119723, T262M] and G797A [rs121964969, R266K]), 2 for exon 8 (T833C [rs5742905, I278T] and G919A [rs121964962, G307S]), 1 for exon 9 (T959C [rs781567152, V320A]), 1 for exon 10 (C1105T [rs117687681, R369C]), and 1 for the intron 7–exon 8 boundary (844ins68 base pair [bp]). The alleles and genotypes of the polymorphic regions were determined using the primers and conditions described by Kim et al, 38 who was the one who defined the activity of each mutation in a yeast system, with exception of 844ins68 bp. In addition, these mutations were tested to restore the folding and functional activity of the CBS, using solvents or chaperone molecules, presenting different behaviors that depended on the variant involved. 39 –42 Mutation T959C [V320A] was sporadically included in these analysis. All the variants studied were nonsynonymous missense mutations, while 844ins68 bp polymorphism is an insertion. The methodology for the analysis of these polymorphisms was polymerase chain reaction–restriction fragment length polymorphism, with the endonucleases reported by Kim et al. 38
Statistical Analysis
Calculation of allele and genotype frequencies was carried out by direct counting and then it was compared between patients and controls. The bivariate χ2 analysis with the correction of Yates and the calculation of the odds ratio (OR) and 95% confidence intervals (95% CIs) were performed with the SPSS (v21.0) software (IBM Corp, Armonk, New York). Fisher exact test was used when appropriate. The Bonferroni correction was calculated with the SNPSpD (https://genepi.qimr.edu.au/general/daleN/SNPSpD) software, based on the effective number of SNPs. 43 It was determined that the effective number of independent tests was 7; therefore, the level of corrected significance for the SNP association was established at P ≤ .007. We used logistic regression analysis to determine the association of the CBS SNPs with total PE, severe PE, and mild PE. Statistical power analysis and sample size calculation were performed using the G*Power (v3.1.9.2) program. Based on an effective size index of 0.4 (corresponding to a “moderate” gene effect), the sample size used in our study had >95% power at an α error of .05 with 2 tails. 44
Haplotypes and linkage disequilibrium (LD) blocks were generated by CI method using Haploview (v4.2). 45 To establish the most informative model of Mendelian association (dominant, codominant, recessive, overdominant, or log additive), the online program SNPStats (https://www.snpstats.net/start.htm) was used. 46 Genotype frequencies for each SNP were tested for possible deviations from the Hardy-Weinberg equilibrium using χ2 test with 1 degree of freedom.
Results
Characteristics of the Groups
Maternal age in the control group was 24 ± 6 years, while overall in the PE group was 25 ± 7 years; in the severe and mild PE groups, the age was 25 ± 7 years and 26 ± 7 years, respectively.
Cystathionine β-Synthase Allele, Genotype, and Haplotype Distribution
Allele and genotype frequency of CBS polymorphisms in all PE cases and controls is shown in Table 1S. Frequency of controls and patients with severe and mild PE is shown in Table 1. The wild alleles were the most frequent in every group; polymorphism G797A was monomorphic in cases with the presence of only G797A-G allele. Allele C785T-T and genotypes C785T-C/T and T959C-T/T were associated with susceptibility in severe and mild PE, while the counterparts were of protection. Alleles G797A-G and T959C-T were associated with susceptibility only in severe PE. The only SNP not in Hardy-Weinberg equilibrium was G797A (<.0001) and the results found for this SNP could be a bias in our analysis. In all genotype comparisons of the SNPs, the codominant model was prevalent. The general success rate for the typing technique was 99.34%.
CBS Allele and Genotype Frequencies in Severe and Mild PE.
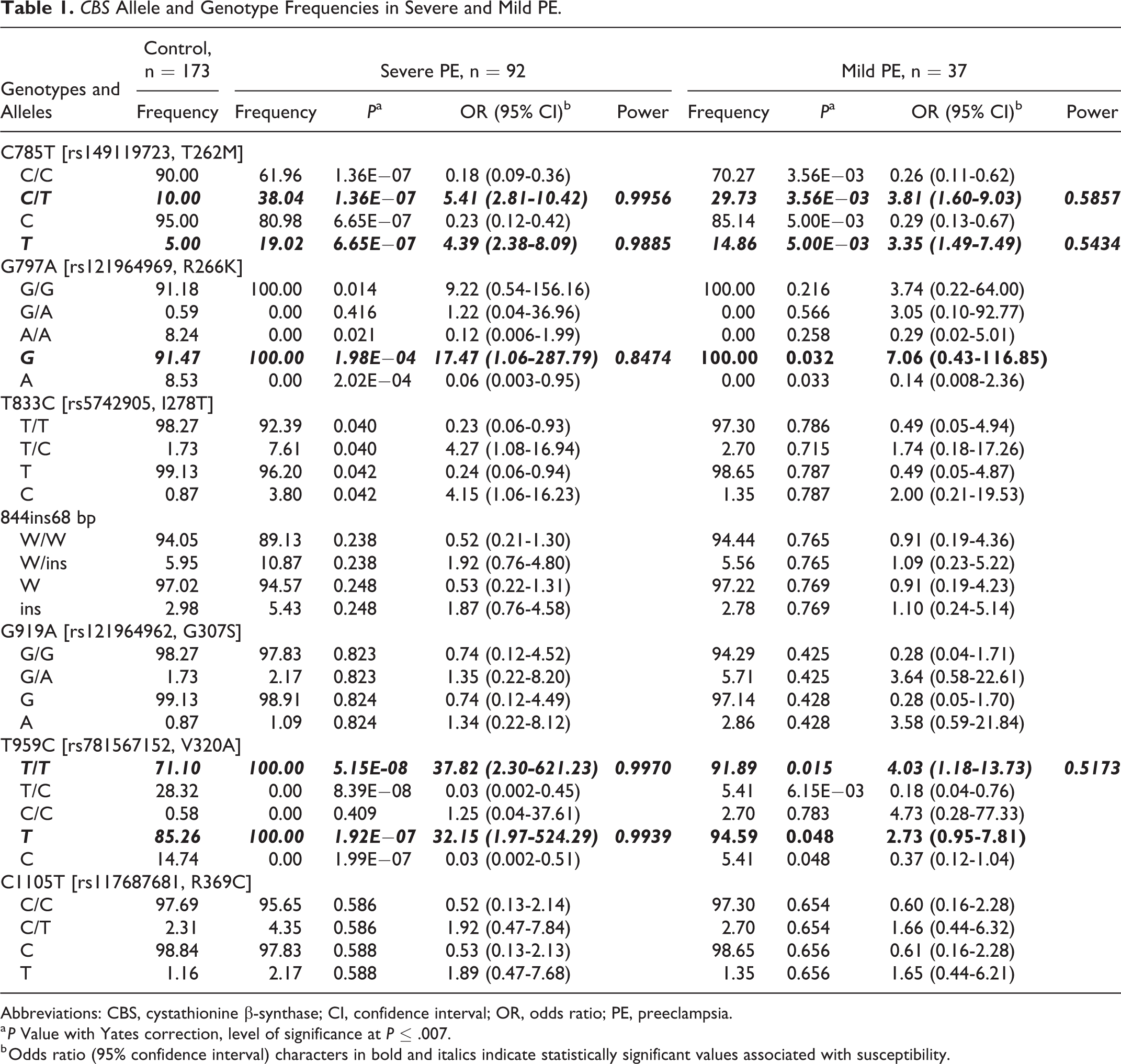
Abbreviations: CBS, cystathionine β-synthase; CI, confidence interval; OR, odds ratio; PE, preeclampsia.
a P Value with Yates correction, level of significance at P ≤ .007.
b Odds ratio (95% confidence interval) characters in bold and italics indicate statistically significant values associated with susceptibility.
The Haploview software established 7 haplotypes for PE, 6 for severe PE, and 5 for mild PE (Tables 2S and 2). When all patients with PE were analyzed, the TGTWGTC haplotype was found to be of susceptibility (P = 4.80E−07; OR [95% CI] = 4.58 [2.42-8.66]), while the CGTWGCC and CATWGTC haplotypes were of protection (Table 2S). However, the TGTWGTC haplotype was of susceptibility for severe PE and of protection for mild PE (Table 2). The CGTWGCC and CATWGTC haplotypes appear to be of protection for severe PE, but the latter is related to susceptibility in mild PE.
CBS Haplotype Frequencies in Severe and Mild PE.a
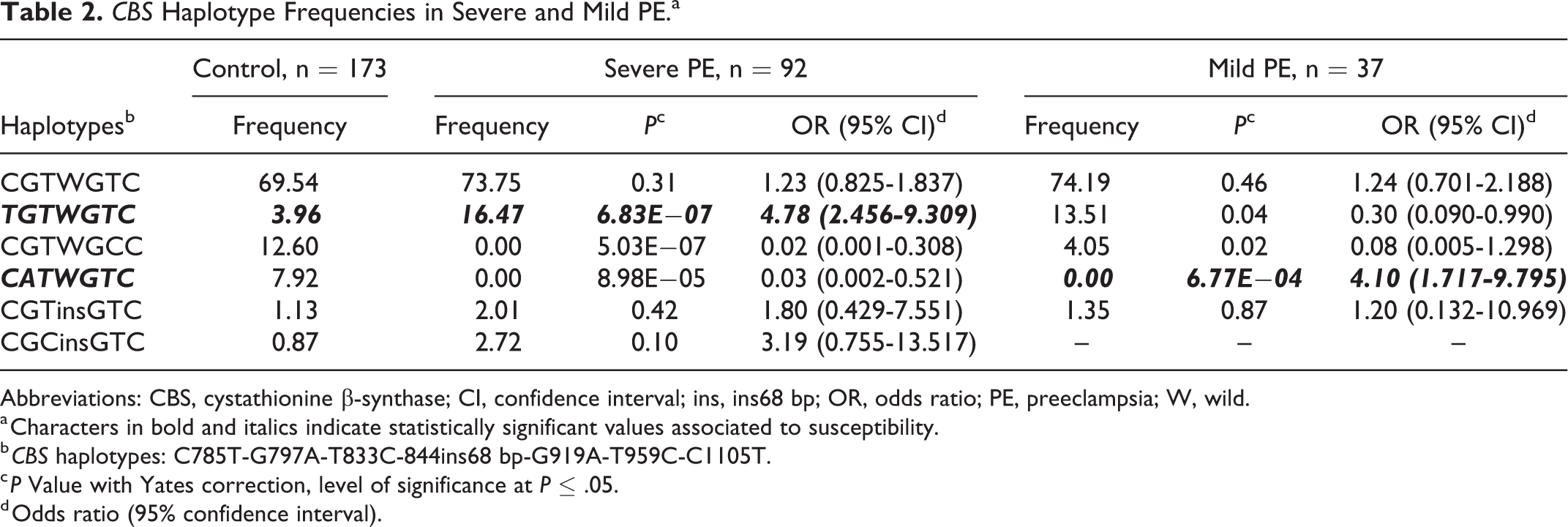
Abbreviations: CBS, cystathionine β-synthase; CI, confidence interval; ins, ins68 bp; OR, odds ratio; PE, preeclampsia; W, wild.
a Characters in bold and italics indicate statistically significant values associated to susceptibility.
b CBS haplotypes: C785T-G797A-T833C-844ins68 bp-G919A-T959C-C1105T.
c P Value with Yates correction, level of significance at P ≤ .05.
d Odds ratio (95% confidence interval).
The power calculation was performed in the significant associations, finding that the values in PE (Table 1S) and severe PE (Table 1) were above the rejection level of the null hypothesis. The values of power were lower in mild PE, probably due to the sample size.
Linkage Disequilibrium
The pairwise LD (D′) for all 7 SNPs is presented in Figure 1. Noted in green are the 6 SNPs located in coding regions. The LD plot generated in Haploview (v4.2) indicates that SNPs T833C and 844ins68 bp are in strong LD, while C785T, G797A, G919A, T959C, and C1105T seem to be almost independent of one another.
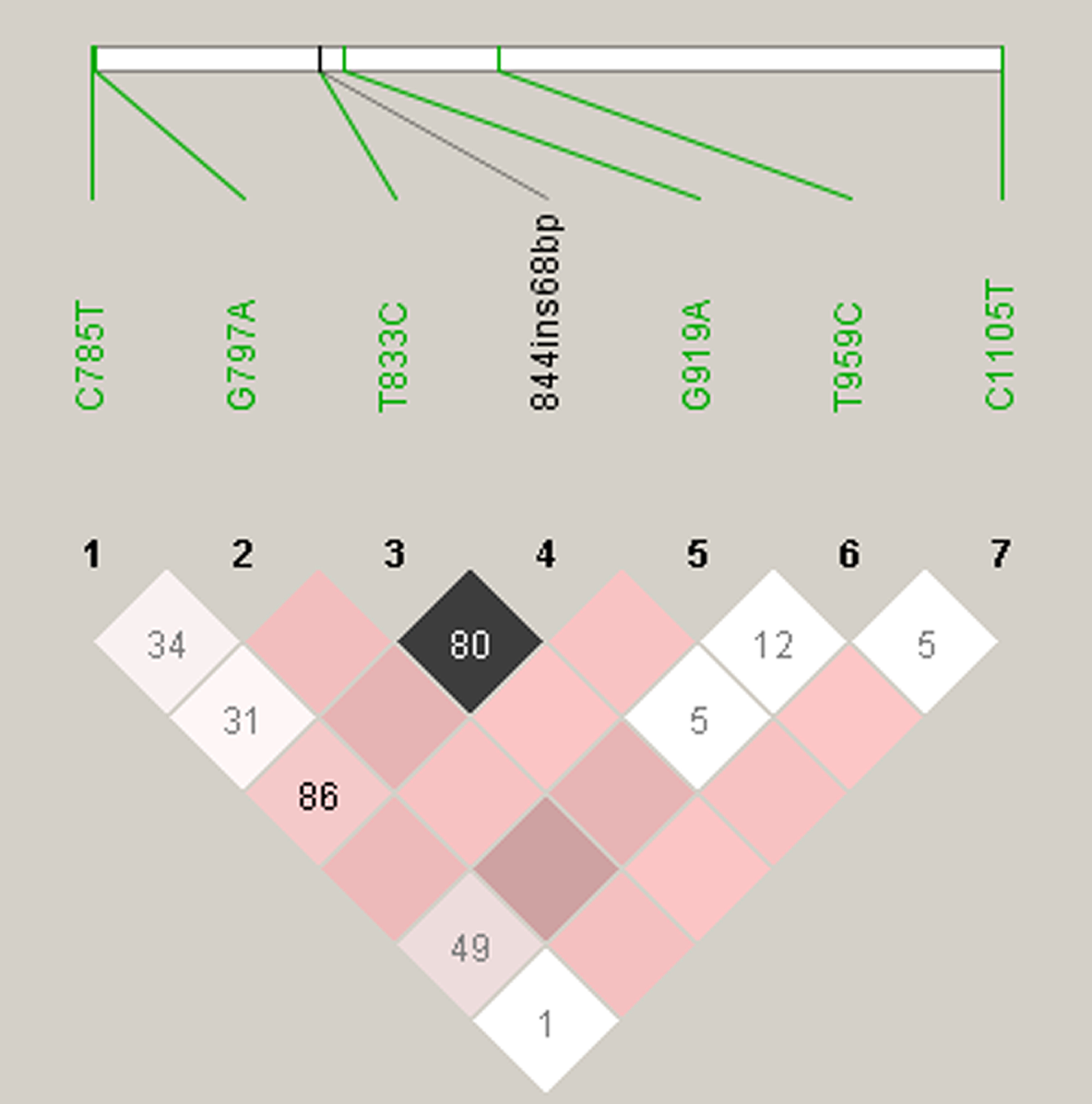
Linkage disequilibrium (LD) among the genotyped CBS SNPs in preeclampsia. The pairwise LD plot was created by HAPLOVIEW 4.2. Within each diamond is presented the standardized coefficient of LD (D′), using the alternate D′/LOD scheme: white represents strong evidence of recombination (low or high LOD and low D′); shades of pink represent uninformative (low LOD and low D′); black represents strong evidence of LD (high LOD and high D′). Intragenic single-nucleotide polymorphisms (SNPs) C785T, G797A, T833C, G919A, T959C, and C1105T are shown in green. A marker pair is said to show moderate or usable LD if |D′| is between 0.33 and 0.5 and strong LD if |D′| is 0.5 or above (ie, at least half the maximum value).
Discussion
Until now, several risk factors related to PE have been reported, such as the increase in the plasma concentration of Hcy, which is an intermediate amino acid in the metabolism of methionine, giving rise to the methionine or cysteine through the transsulfuration pathway, where CBS has the ability to convert Hcy to cysteine, pyruvate, and H2S. 9,47 –50
Therefore, the objective of this study was to test the evidence of association between 7 SNPs in the CBS gene with severe and mild PE. We used a population-based case–control design in a Mexican sample, focusing on the point mutations C785T (T262M), G797A (R266K), T833C (I278T), G919A (G307S), T959C (V302A), C1105T (R369C), and 844ins68 bp, which are located into or near to the heme site and the catalytic domain.
To determine the functional behavior of CBS mutations, Kim et al used one growth testing mutations yeast system. They found that strains containing the T833C (I278T), G919A (G307S), and C785T (T262M) mutations were unable to form colonies on standard media without cysteine, indicating a severe affection to CBS enzyme. Cells containing the T959C (V302A) mutation formed significantly smaller colonies than those with the wild-type CBS. Yeast containing the G797A (R266K) and the C1105T (R369C) mutation grew just as well as wild type, indicating that these mutations have less severe effects on enzyme function than the above mentioned. 38
Residues Tyr223 and Gly307 seem to be crucial for substrate specificity, since they are spatially adjacent to the substrate-binding site; afterward, mutation Gly307S has shown <1.7% activity in yeast, with respect to wild type. 51,52 In addition, residue R266 is part of the heme-binding site of CBS, and T262 is in the interface between heme-binding pocket and active site in the α-helix 8, which means that both are adjacent mutations; then, any change in these amino acids will compromise also the protein function. 51 Moreover, clinicians have demonstrated that the patients with these point mutations can be subject of doses of vitamin B6 treatment responding with a better prognosis of the disease. 41 Mutations I278T and V320A are in the active core and have a phenotype with no response for B6 in yeast; however, V320A is inducible by AdoMet and is not behaving like the regulatory domain mutants that have been described. The R369C mutation, located equally in the active core, seems to not affect CBS function in yeast and has only 34% of wild-type activity in Escherichia coli. 38,53
To indicate the position of the mutations along the gene, an amino acid sequence alignment was made (Figure 2), using data from the UniProtKB database for the 2 isoforms (ISO1: P35520 and ISO2: P35520-2) of the human CBS protein and the 7 polymorphisms analyzed in this study. 55 Similarly, Figure 3 was elaborated using the Chimera (v1.11.2) program to show the mutations in the protein tertiary structure, based in the PBPC 4PCU chain A file. 56,57 The residues presented in Figure 3 are in the active core, surrounding the pocket where the PLP junction is and in the heme-binding pocket, then residues V320A and T262M, which are associated with PE, are also close to these structures. The third residue associated with PE is R266K, which is as mentioned previously found at the interface between the heme-binding pocket and the active site. If some of these residues are modified, steric hindrance probably prevents the binding of PLP, altering the function of CBS, which can cause the accumulation of Hcy and the disease.

Aminoacid sequence alignements among 2 CBS protein isoforms and a generated sequence with the 7 polymorphisms analyzed in the present study. M indicates metal-binding sites 1 and 2; iron, heme axial ligand; B, binding sites 1 and 2; R, pyridoxal phosphate binding region; D, CBS domain; V, natural variants in the protein. V1 indicates VAR_008072. T → M. In CBS deficiency; moderate form. Corresponds to variant dbSNP: rs149119723. V2 indicates VAR_008074. R → K. In CBS deficiency; mild form; decreased cystathionine β-synthase activity; decreased homotetramer formation; no effect on heme-binding; decreased stability. Corresponds to variant dbSNP: rs121964969. V3 indicates VAR_002184. I → T. In CBS deficiency; mild-to-severe form; common mutation; decreased expression; loss of cystathionine β-synthase activity; impaired stimulation by AdoMet and AdoHcy; severely affects homotetramer formation by promoting formation of larger aggregates. Corresponds to variant dbSNP: rs5742905. V4 indicates VAR_002186. G → S. In CBS deficiency; moderate-to-severe form; linked with D-534; common mutation; loss of cystathionine β-synthase activity; impaired stimulation by AdoMet and AdoHcy; no effect on homotetramer formation. Corresponds to variant dbSNP: rs121964962. V5 indicates VAR_008078. V → A. In CBS deficiency; has 36% of wild-type enzyme activity. Corresponds to variant dbSNP: rs781567152. V6 indicates VAR_008084. R → C. In CBS deficiency; when linked with C-491 severe form; decreased cystathionine β-synthase activity; decreased homotetramer formation. Corresponds to variant dbSNP: rs117687681. V7 indicates 844ins68. Insertion of the following sequence: HPGGAFAGLEP*SRALCRSIGWIP. * Stop codon. Data obtained from UniProtKB-P35520 (CBS_HUMAN). 54
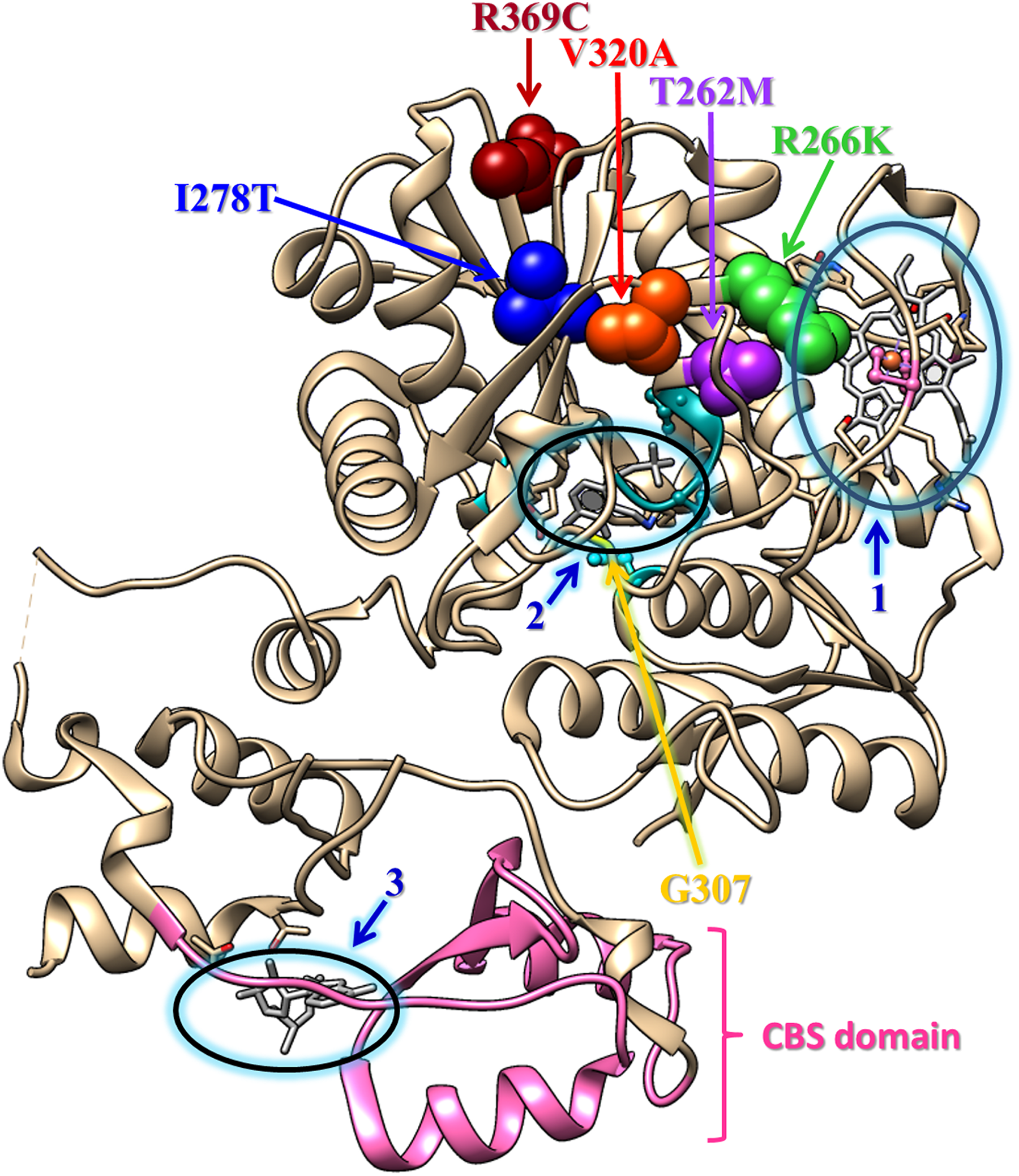
Schematic representation of the tertiary fold of a monomer of CBS (from 4PCU chain A). The molecule is colored in tan. In ball-and-stick representation, colored in dark gray, and surrounded by an ellipse are (1) heme, (2) active site PLP (pyridoxal 5′-phosphate), and (3) AdoMet (S-adenosyl-
Although the SNP T833C (I278T) is the prevalent mutation in several populations, in this study it was not associated with PE, which may be due to differences in the ethnic characteristics of the Mexican population. Regarding the 844ins68 mutation of CBS, Tsai et al found that male patients with coronary artery disease in the upper Midwestern regions of the United States showed significantly lower blood Hcy levels compared to fasting blood samples when they consume methionine orally. 58,59 Our findings demonstrated that this polymorphism does not increase the risk of any form of PE, as was reported by Kim et al and Also-Rallo et al for Caucasian populations, in the United States and Spain, respectively. 35,60
In a study in which several CBS gene polymorphisms are studied to see their association with early- and late-onset PE, Holwerda et al found that women with the minor allele of rs11203172 have a reduced risk of early-onset PE. They described that normotensive pregnant women with the minor allele of rs11203172 and rs234713 have lower cysteine levels than those who do not, while women with the minor allele of rs1789953 have increased levels of cysteine and cystathionine. 36 However, although these results are interesting and support the idea that polymorphisms and mutations of CBS are involved in PE, they cannot be compared with the results of this article, since the polymorphisms analyzed are different.
Our results revealed that CBS SNPs C785T, G797A, and T959C were associated with susceptibility to PE, both severe and mild, in particular the C785T-T allele and the C785T-C/T genotype. These results suggest that CBS SNPs C785T, G797A, and T959C may be influencing the development of PE, as a predisposing factor of the disease, since the statistical power was greater than 0.80 in total PE and in severe PE; however, the sample size of mild PE did not allow to obtain enough power to reject the null hypothesis. Therefore, more studies are needed to confirm these associations, including gene–gene interaction approaches.
Despite the international controversy between the influence of high levels of Hcy and the development of PE, reports continue to emerge supporting this concept, for example, an association of high plasma levels of Hcy and severe PE has been demonstrated in Mexican women, which supports the study of the enzymes involved in the production and consumption of Hcy. 61
Additionally, it should be considered that CBS variants, dietary habits, dietary supplements, and the ethnicity of each population could generate a gradient of the transsulfuration pathway intermediaries, or other pathways involved in vascular protection, affecting the placental microenvironment and producing endothelial dysfunction and impaired placentation. Further studies are necessary to confirm this hypothesis. 54
Supplemental Material
Supplemental Material, CBS-CATH-Table_1S,_2S - Association of Cystathionine β-Synthase Gene Polymorphisms With Preeclampsia
Supplemental Material, CBS-CATH-Table_1S,_2S for Association of Cystathionine β-Synthase Gene Polymorphisms With Preeclampsia by Mercedes Piedad de León Bautista, Mirza Romero-Valdovinos, Beatriz Zavaleta-Villa, Arony Martínez-Flores, and Angélica Olivo-Díaz in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors wish to thank Rocío Jimenez-Lucio and David Sierra-Barrera for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
