Abstract
The post-thrombotic syndrome (PTS) is a frequent and burdensome disease, but no gold standard test exists to diagnose it. The value of patient-reported outcomes (PROs) in recording transient signs/symptoms, thereby possibly improving diagnosis/monitoring and subsequent clinical care in some diseases, is increasingly being recognized. For example, PROs have been successfully used in cancer, rheumatoid arthritis, and endometriosis. In this context, we describe a patient-reported questionnaire (the Patient-Tracked Symptoms questionnaire) that contains questions on the presence of signs/symptoms of PTS. This questionnaire has face validity and was successfully used in the Home-LITE trial. It could prove valuable in monitoring patient symptoms, especially in home-based patients who do not have regular contact with hospital clinicians. Patients who report symptoms can then undergo follow-up assessment by health care professionals at their next clinic visit. The Patient-Tracked Symptoms questionnaire could help clinicians to recognize the PTS early, enabling them to initiate treatment promptly.
Introduction: The Post-Thrombotic Syndrome
The post-thrombotic syndrome (PTS) is a common sequela of deep vein thrombosis (DVT), occurring in 20% to 60% of patients within 2 years following a DVT1–3 and, according to a recent study, often within 3 months after the first episode of symptomatic DVT. 4
The pathophysiology of PTS is not completely understood. Its development is thought to involve valvular reflux, as a consequence of damage to the venous valves by the thrombus itself or accompanying inflammation, as well as persistent venous obstruction, due to incomplete thrombus clearance resulting in impaired venous return. Incomplete resolution of leg symptoms and signs at 1 month after acute symptomatic DVT was shown to strongly predict development of PTS during the next 2 years. 2 Other risk factors for PTS include venous thrombosis of the common femoral or iliac vein, previous ipsilateral venous thrombosis, high body mass index, and older age.4,5
Several measures have been investigated to prevent the development of PTS following DVT, especially in at-risk patients. The use of graduated elastic compression stockings after symptomatic proximal DVT was shown to substantially reduce the risk of PTS (odds ratio: 0.31 95% confidence interval [CI], 0.20–0.48), 6 but these may be challenging to use in real life. Thrombolysis of acute DVT may reduce the occurrence of PTS, although there is only limited evidence to support this approach, and catheter-directed thrombolysis may be invasive and costly. 7 Appropriate prevention strategies to reduce the development of a DVT in higher-risk patients are likely to reduce the development of PTS. Provision of adequate anticoagulation after a DVT has occurred may also reduce the occurrence of PTS by lessening the impact of damage to the valves and the microcirculation as well as reducing inflammatory processes.8,9 In the Home-LITE study, patients treated with tinzaparin for 12 weeks after an objectively diagnosed DVT experienced a significantly lower incidence of patient-reported leg ulcers and symptoms of PTS than those treated with long-term warfarin. 10 Of note, patients in Home-LITE did not object to long-term treatment with tinzaparin; in fact, patients in the long-term tinzaparin group expressed significantly greater treatment satisfaction than those treated with long-term warfarin. 10
PTS has a significant negative impact on quality of life (QoL), limiting daily activities, and interfering with work and social activities 2 as well as being costly, both in terms of direct and indirect health care costs and health resource utilization.11,12 Indeed, Bergqvist et al 13 estimated that the additional long-term health care cost for PTS was approximately 75% of that of the primary DVT. Therefore, it is desirable to identify patients at risk of PTS as early as possible in order to prevent its development, as management options for advanced PTS are limited.
In this article, we describe, within the context of patient-reported outcomes (PROs) generally, a patient-reported questionnaire (the Patient-Tracked Symptoms scale) for identifying PTS. We consider the possible usefulness of this questionnaire, in particular for reporting acute and/or transient symptoms of PTS that may be missed at clinic visits.
How Is PTS Diagnosed?
By definition PTS is a syndrome; symptoms and clinical signs vary between individual patients. Typical features of PTS include chronic pain, swelling, heaviness, edema, and skin changes in the affected limb. In severe cases, venous ulcers may develop which may be chronic, painful, slow to heal and recurring, and form the most severe complication of PTS. Data from contemporary prospective studies with more than 12 months of follow-up indicate that between 5% and 10% of DVT patients will develop severe PTS, which may include venous ulcers.1,2 Symptoms can be intermittent or persistent, aggravated by standing or walking, and improving with rest and leg elevation. There is no gold standard laboratory, imaging, or functional test that confirms the diagnosis and hence PTS is diagnosed on clinical grounds, based on the symptoms and signs described above in patients with prior DVT.
Several clinical scales for the diagnosis of PTS have been developed14–17 using the occurrence of symptoms and signs16,18 or clinical signs that represent a progressive gradation of disease severity (“clinical, etiological, anatomical, pathophysiological” or “CEAP” classification).14,15 The Subcommittee on Control of Anticoagulation of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis recommends that the Villalta scale is used to diagnose PTS and grade its severity, as a step toward standardizing measurement of PTS. 19
The Villalta scale rates the severity of PTS symptoms and signs using a clinical scoring system16,19 (Table 1 ) and can be used as a binary, categorical, or continuous measure. A recent review of the scale’s measurement properties shows it to be a valid, reliable indicator of PTS, 20 correlating with relevant health outcomes, including disease-specific as well as generic QoL and known anatomic or physiological correlates of PTS. Notably, the Villalta scale requires assessment of a patient’s symptoms by a health care professional.
aInterpretation: Total ≥5: PTS is present; 5–9: mild PTS; 10–14: moderate PTS; ≥15 or venous ulcer present: severe PTS.
Are Self-Reported Patient Outcomes Reliable?
The value gained from understanding health outcomes from the patient’s perspective has been acknowledged increasingly in recent years. National surveys of patient experience are now a feature of National Health Service regulation in the United Kingdom and the pharmaceutical industry recognizes the use of such outcomes to support claims made about a medical product in its labeling.21,22
The PROs provide the patient’s perspective on the effectiveness of treatment, and for many diseases, the patient is really the only source of health outcome end point data—for example, with respect to pain, range of motion, or the ability to perform usual work or social activities. 23 Measures that focus on test results (eg, hemoglobin A1c levels) or the achievement of intermediary outcomes (eg, blood pressure <140/90 mmHg) are important, but provide only a limited amount of information about patient experience with care, encouraging physicians to focus on the diagnostics rather than on the patient as a whole. Greater emphasis on PROs has the potential to improve clinical care and to facilitate patient–clinician communication on issues that are important to patients. Better communication could be expected to enable shared decision making, consistent and continuous monitoring of disease progression and treatment response, and identification of vulnerable patients and continuous assessment of patient care.24,25 Since patients see and feel symptoms every day, they are in the best position to record them, when educated to do so. Health care professionals who see patients at infrequent intervals may miss transient signs and symptoms, such as acute ulcers in the assessment of PTS. A possible disadvantage with PROs is that they might interfere with doctor–patient communication, and patients may have concerns about who will have access to the data.
The practical impact of PROs in clinical practice has been demonstrated in several systematic reviews. In cancer clinics, feedback of PROs to health care professionals increased the frequency with which doctors and patients discussed symptoms and QoL issues, without extending clinical contact time. 26 Similarly, data from rheumatoid arthritis research suggest greater sensitivity in patient-reported than in physician-reported outcomes, leading to the recommendations that greater emphasis be attributed to PROs. 27 For patients with endometriosis, better self-reporting of symptoms led to a greater chance of an accurate diagnosis, 28 and in patients with chronic obstructive pulmonary disease, PROs accurately reflected health care professionals’ assessments of clinically relevant changes in health-related QoL. 29 In contrast, a review of the use of PROs in cardiovascular trials showed that reports were not well used, even when they could have had a significant impact on clinical decision making. 30
Can Patient-Reported Symptoms be Used to Diagnose/Monitor PTS in Patients With a Preceding DVT?
A striking outcome from the Home-LITE study was the reduced incidence of patient-reported symptoms of PTS in patients who received 12 weeks of therapeutic tinzaparin versus the usual-care group with long-term warfarin, with an overall odds ratio of 0.77 (95% CI, 0.67–0.90; P = .001) in favor of tinzaparin. 10 The Patient-Tracked Symptoms scale used in Home-LITE was a questionnaire completed at the end of the 12-week study by patients, which recorded 8 symptoms or signs whose presence in combination is commonly used to diagnose the presence and severity of PTS (Figure 1 ; Table 2 ).
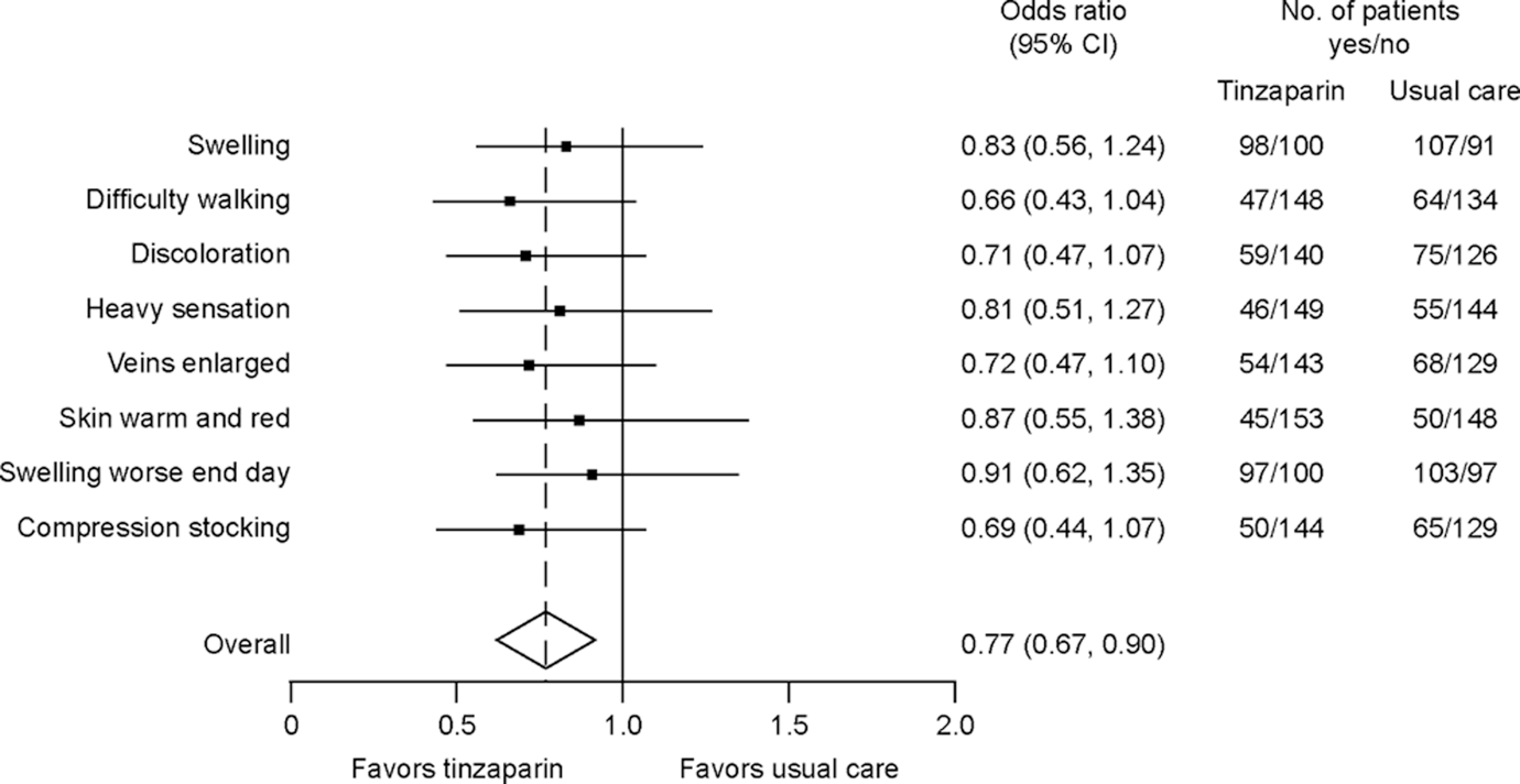
Symptoms/signs of the post-thrombotic syndrome (PTS) as reported by patients using the Patient-Tracked Symptoms questionnaire for PTS, following 12 weeks of therapy with either tinzaparin or usual care in the Home-LITE study. 10 Patients replied “Yes” or “No” concerning the presence of the symptoms/signs listed (the exact wording of the questions is given in Table 1). Odds ratios <1 favor tinzaparin (ie, symptom/sign less likely to be present). Overall odds ratio = 0.77 (P = .001). Reprinted from Hull, et al 10 by permission from Elsevier and the Association of Professors of Medicine.
aPatients responded “Yes” or “No” to each item. Reprinted from Hull, et al 10 by permission of Elsevier and the Association of Professors of Medicine.
Patients were also asked to respond “Yes” or “No” to the statement “I have an ulcer in the skin above my ankle on the affected leg.” This wording was carefully chosen to ensure that the patients reported only genuine leg ulcers and responses could include acute ulcers. In various scoring systems for PTS that have been developed, a venous ulcer is recognized as an indication of severe PTS, irrespective of any other signs and symptoms. 31 During the 12-week study period, 1 patient on tinzaparin reported the presence of a leg ulcer compared with 8 patients in the usual care group, yielding an odds ratio of 0.12 (95% CI, 0.01–0.97; P = .02) for tinzaparin relative to usual care. In each case, the presence of a venous ulcer was confirmed by a health care professional.
When PROs are used as a key component in assessment outcomes, it is important that the evaluation uses symptoms that are reliable, valid, and responsive. 32 For example, using rigorous testing with the VEINES-QOL/Sym assessment tool, used to assess the outcomes in chronic venous disorders of the leg (CVDL), was shown to be a practical, scientifically sound, patient-reported measure of QoL, and symptoms for use across the spectrum of CVDL-related conditions. 33 The questionnaire is also quick and easy to administer.
The Patient-Tracked Symptoms scale used in the Home-LITE study has not been formally validated in a test–retest trial or relative to other scales used to diagnose PTS. However, it clearly has face validity, that is, the results are compatible with good clinical sense. The questionnaire was developed based on various practical measures similar to those used in the Villalta scale, while it was in its early development in the 1990s. Moreover, in a recent systematic review and meta-analysis, the effects of long-term treatment of DVT with low molecular weight heparin (LMWH) on recanalization and the PTS were examined. 34 This review showed that the analyses of underlying pathophysiological changes (recanalization and venous competence) reported in various studies using long-term LMWH are consistent with and provide a possible rationale for the lower incidence of PTS and venous ulcers reported by patients in the Home-LITE study.
In another study, a postoperative questionnaire was administered to patients with asymptomatic DVT following total hip or knee arthroplasty. 35 In that study, questions were directed toward residual calf or ankle swelling, persistent edema, pain at rest or during activity, skin dryness, skin discoloration, and the development of ulcers. Patients who reported symptoms were subsequently reexamined. Signs of mild-to-moderate PTS were picked up in 7 of 42 patients in the study. Despite the author-acknowledged shortcomings of their questionnaire, due to the subjective nature of the assessment of signs and symptoms of PTS, they were able to identify patients who developed objective signs of PTS.
Rodger et al 36 demonstrated that physical examination findings of PTS, including those used in the development of the Villalta score, are reliable and reproducible, with the combinations of findings having good-to-excellent interobserver reliability, tending to be more reproducible than individual findings. In Rodger’s study, the assessments were made by study nurses rather than patients themselves; however, the results do strengthen the concept of using a composite score to diagnose PTS.
Possible Limitations of the Patient-Tracked Symptoms Questionnaire
The questionnaire is initially used only once, that is, at the end of a 12-week study. As the signs and symptoms of the PTS are chronic in nature and tend not to fluctuate markedly on a daily basis, 12 weeks is a suitable time point for its initial determination. Any signs/symptoms that develop earlier are unlikely to spontaneously disappear, whereas signs/symptoms may not fully develop before 12 weeks. The questionnaire can be used at subsequent intervals to monitor any changes in severity of the PTS.
To date, the questionnaire has only been completed by patients in Canada whose understanding and English language skills were assessed by their clinicians to be adequate for the task. With any PRO tool, it is important to ensure that patients of different educational and language backgrounds can fill it out consistently. This has not yet been demonstrated for the Patient-Tracked Symptoms questionnaire.
Importance of Home Monitoring of PTS Symptoms
Monitoring symptoms of patients with known DVT over time may offer the best opportunity for the early detection of PTS. Since patients not requiring hospitalization after diagnosis and acute therapy for a DVT are often successfully treated at home under the care of their general practitioner, any monitoring tool must be reasonably easy to use and acceptable to patients for it to be of value. With infrequent physician–patient consultations, and the fluctuating appearance of symptoms, PTS symptoms can easily be missed and not reported during clinic visits. In contrast, patients who see their own legs every day are highly sensitive to changes in skin conditions and particularly in the appearance of ulcers. Patients can then come to the clinic and be questioned by staff and assessed using additional tools, such as the Villalta score. Moreover, as most clinicians in general practice do not deal with the presentation, diagnosis, or treatment of PTS, they are, for the large part, unaware of the suffering of patients and may consequently undervalue the morbidity related to PTS.
Conclusion
As therapeutic options for PTS are extremely limited and results are often disappointing with advanced disease, prevention of the initial or recurrent DVT, recognition of clinical signs or complications, and early treatment of PTS remain the keys to reducing its progression and morbidity.
The questionnaire used by patients in the Home-LITE study to report signs and symptoms of PTS, based on items that are also included in the Villalta scale, provided results with face validity. This Patient-Tracked Symptoms questionnaire provides an effective method of monitoring patient symptoms, especially in patients discharged from hospital for home care who do not have regular contact with hospital clinicians. Patients who report the development of symptoms can then be questioned by staff at clinic visits and assessed using additional tools such as the Villalta scale. The Patient-Tracked Symptoms questionnaire may have a role to play in increasing the recognition of PTS early so that the treatment can be initiated timeously, thus reducing the progression and morbidity of this frequent, burdensome, and costly disease.
Footnotes
Acknowledgments
LEO Pharma supported the preparation of this article with an unrestricted educational grant. Editorial assistance was provided by Grace Townshend and Catherine Jones of Watermeadow Medical.
Authors’ Note
RH had the original idea for the manuscript, provided guidance and input into all drafts, and is the guarantor of the article. PB wrote the first draft of the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RH has been a consultant for Bayer Pharmaceuticals Corp., LEO Pharma, Inc., Pfizer Inc., GlaxoSmithKline, and Wyeth Pharmaceuticals; and sat on advisory boards for Bayer Pharmaceuticals Corp., Pfizer Inc., and Sanofi. PB is an employee of Watermeadow Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LEO Pharma supported the writing of this article with an educational grant. RH has received grants/research support from Bayer Pharmaceuticals Corp., LEO Pharma Inc., and Sanofi. PB received payment from LEO Pharma for work on this manuscript.


