Abstract
Introduction:
Crimean–Congo hemorrhagic fever (CCHF) is a viral tick-borne zoonosis, which is a severe illness, causing hemorrhages in humans. Mean platelet volume (MPV) is used as a surrogate marker of platelet function and has been shown to be a sign of inflammation. The objective of the present study is to examine the association between MPV and CCHF. We also aimed to investigate the association between MPV and coagulopathy markers in the mortality rates and prognosis of patients with CCHF.
Patients and Methods:
Ninety-three patients with CCHF were enrolled retrospectively into the study and 15 of them were excluded according to the exclusion criteria. Twenty-five healthy individuals were included as a control group which was age and gender matched with CCHF patients. We compared the levels of MPV between the patient and the control groups. We also compared the coagulopathy markers of fatal CCHF patients (n = 9) with nonfatal cases (n = 69).
Results:
Platelet counts were significantly lower in the CCHF group. Levels of international normalized ratio (INR), prothrombin time (PT), and activated partial thromboplastin time (aPTT) were significantly higher in CCHF group (P < .001, <.001, <.001, respectively). A statistically significant increase in MPV was observed in patients with CCHF compared with healthy controls (8.63 ± 1.23 fL vs 7.65 ± 0.42 fL, P < .001). Receiver–operating characteristic curve analysis suggested that the optimum MPV level cutoff points for patients with CCHF was 8.15 fL, with a sensitivity and specificity of 65% and 88%, respectively. The MPV levels were not significantly different between group 2 (nonsurvivor) and group 1 (survivor). However, platelet count,
Conclusion:
In conclusion, MPV may be a beneficial marker in the diagnosis of CCHF, especially in cases with thrombocytopenia, the MPV levels are high. We also conclude that MPV may independently predict the prognosis of patients with CCHF.
Introduction
Crimean–Congo hemorrhagic fever (CCHF) is a tick-borne zoonotic infection caused by Crimean–Congo hemorrhagic fever virus (CCHFV). The CCHFV is classified within the Nairovirus genus in the Bunyaviridae family. 1 Crimean–Congo hemorrhagic fever characteristically occurs in humans via a tick bite or from contact with the blood or tissues of infected livestocks. 2 Health care employees can be infected through direct contact with damaged cutaneous or mucous membranes exposed to blood and body fluids or from percutaneous needle stick injuries. The incubation period of the virus is 1 to 7 days. The onset of the disease can often be sudden. The symptoms of the disease are high fever, headache, fatigue, myalgia, abdominal pain, nausea, vomiting, diarrhea, and mucosal hemorrhagic lesions. Hemorrhage is associated with thrombocytopenia, 3 and high mortality rates (20%-30%) are reported in different countries. 4,5
Mean platelet volume (MPV) is a parameter generated by full blood count analyzers as a part of the routine complete blood count (CBC) test cycle which is generally ignored by the doctors. 6 Recently, MPV has been studied in a group of patients and it was found to be increased in immune thrombocytopenic purpura (ITP), myocardial infarction, and cerebrovascular disease. 7,8 The MPV is widely used as a surrogate marker of platelet function and has been shown to be a sign of inflammation in ulcerative colitis, Crohn disease, and rheumatoid arthritis. 9,10 An association between CCHF and thrombocyte count and coagulation parameters has been established in many studies previously. 11 –13 In some of these studies, the correlation between MPV and the diseases, such as myocardial infarction, acute ischemic cerebrovascular events, septicemia, diabetes, and congestive heart failure, showed a poor clinical outcome. 14 –16 However, there is no report in the literature about the predictive value or usefulness of MPV in patients with CCHF.
The objective of the present study is to investigate the association between MPV levels and CCHF. We also aimed to examine the association between MPV and coagulopathy markers (platelet count, activated partial thromboplastin time [aPTT], prothrombin time [PT], international normalized ratio [INR] and
Patients and Methods
Ninety-three patients with CCHF were enrolled into the study, retrospectively. Exclusion criteria for patients with CCHF and control individuals were asthma, chronic obstructive pulmonary disease, peripheral and cerebral vascular disease, other hematological disorders, cirrhosis, portal hypertension, and malignancies. Fifteen patients were excluded according to these exclusion criteria. Twenty-five healthy individuals were included as a control group which was age and gender matched with CCHF patients. The following data were obtained from the computerized patient registry database: alanine aminotransferase, aspartate aminotransferase, γ-glutamyl transpeptidase, alkaline phosphatase, bilirubin, INR, PT, aPTT, white blood cell count, platelet count, hemoglobin, and MPV. Two groups of patients were compared and defined as group 1 survivors and group 2 nonsurvivors. Platelet count and MPV determinations were performed on the Siemens Healthcare Diagnostics Item ADVIA 2120i. Normal value of MPV in our laboratory is 7.0 to 11.1 fL.
Statistical Analysis
Data were analyzed with the SPSS software (SPSS 13.0, Chicago, Illinois). Data were expressed as mean ± standard deviation. The independent t test was used for group comparisons. Categorical variables were compared with the chi-square test. Receiver–operating characteristic (ROC) curve analysis was used to identify optimal cutoff values of the MPV level to identify the maximum sensitivity and specificity for diagnosis of CCHF. Logistic regression analysis was performed in order to evaluate the association between mortality and MPV, diabetes mellitus (DM), coronary artery disease (CAD), and hypertension. Statistical significance was set at a P value of <.05.
Results
Demographic features and laboratory tests of patient and control groups are showed in Table 1 . Age and gender were similar in the patient and control groups. Platelet counts were significantly lower in the CCHF group. Levels of INR, PT, and aPTT were significantly higher in CCHF group (P <.001, <.001, <.001, respectively).
Characteristics of Individuals in the Study and Control Groups.
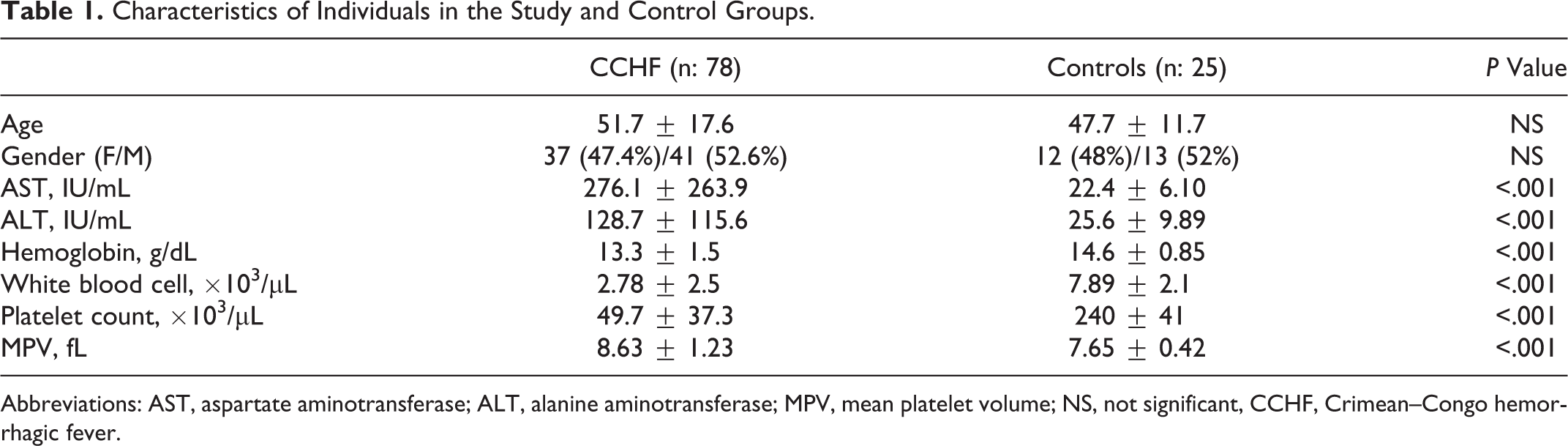
Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; MPV, mean platelet volume; NS, not significant, CCHF, Crimean–Congo hemorrhagic fever.
A statistically significant increase in MPV was observed in patients with CCHF compared with healthy controls (8.63 ± 1.23 fL vs 7.65 ± 0.42 fL, P < .001). The ROC curve analysis suggested that the optimum MPV level cutoff points for patients with CCHF was 8.15 fL, with a sensitivity and specificity of 65% and 88%, respectively.
We compared platelet count,
Association Between Laboratory Parameters and Mortality Rates in the Patient Groups.

Abbreviations: INR, international normalized ratio; aPTT, activated partial thromboplastin time; MPV, mean platelet volume; NS, not significant.
Discussion
In the present study, we found that patients with CCHF have significantly higher MPV values compared with the controls. Similar with the reported studies, we have found a positive correlation between decreased platelet count, elevated
The CCHF is a viral tick-borne zoonosis that causes severe illness and hemorrhages in humans. The virus is transmitted to humans generally through infected tick bites and in rare cases from direct contact with the tissues and fluids of viremic animals or humans. The disease generally appears in Asia, the Middle East, Africa, and southeastern Europe. 17 At present, Turkey is experiencing an increase in outbreaks of the disease, especially in Central Anatolia. The majority of the cases had a history of receiving a tick bite. A few cases were infected through nosocomial accidents. 18
The early diagnosis of CCHF is fundamental in preventing the outbreaks and in decreasing mortality rates. The diagnosis can be made by the detection of the specific IgM antibody or the elevation of IgG antibody level in sera with an enzyme-linked immunosorbent assay. Another option is to detect the viral DNA, where the real-time polymerase chain reaction can be used for this purpose. 19 However, these laboratory tests can only be done in reference laboratories which are not routinely available in most areas. Furthermore, these tests are expensive and determination of the tests is quite hard.
The MPV is routinely determined by CBC analyzers which show the platelet size. The MPV is also a marker of platelet function and activation. It can be influenced by the inflammation. 6 Increased MPV levels have been reported in patients with metabolic syndromes such as myocardial infarction, acute ischemic stroke, diabetes, and nonalcoholic fatty liver disease. 14 –16 The MPV has also been identified as being a sign of inflammation in ulcerative colitis, hepatitis B, Crohn disease, and rheumatoid arthritis. 9,10,20 To the best of our knowledge, an association between CCHF and MPV levels has not been reported before. This study is the first to show the significant increase in MPV levels in patients with CCHF.
Patients with thrombocytopenia were classified as hyperdestructive and hypoproductive thrombocytopenia in the literature. 21 Generally, hyperdestructive thrombocytopenia is a result of extramedullary destruction of the platelets, while bone marrow (BM) production of them is normal or increased; this is seen in patients with condition such as ITP, secondary ITP, and disseminated intravascular coagulopathy (DIC). Hypoproductive thrombocytopenia is commonly secondary to the decreased BM production of the platelets in diseases such as aplastic anemia, acute leukemia, megaloblastic anemia, myelodysplastic syndrome, and amegakaryocytic thrombocytopenic purpura. In the recent studies, it has been shown that patients with hyperdestructive thrombocytopenia are likely to have higher MPV levels. 22,23 Endothelial damage has an important role in the pathogenesis of CCHF. It contributes to hemostatic failure by stimulating platelet aggregation, degranulation, and activation of the intrinsic coagulation cascade which results in DIC. 18 In this study, we have also shown that patients with CCHF have significantly higher MPV levels compared with controls as it was observed in patients with ITP. 8
Hemophagocytosis was reported to be in the pathogenesis of CCHF. The activation of monocytes secondary to the stimulation by high levels of cytokines, such as interleukin (IL)-1 and IL-6, was reported to be a possible mechanism of hemophagocytic lymphohistiocytosis. 18,24 Hemophagocytosis is thought to play a role in the pathogenesis of pancytopenia observed in CCHF. 25 –27 In addition, increased IL-6 levels are responsible for inducing platelet production. The IL-6 can induce the size of platelets by interfering with megakaryopoiesis with the subsequent release of big size platelets from the BM. 28 Therefore, increased MPV levels may be due to increased young platelets.
In conclusion, MPV may be a beneficial marker in the diagnosis of CCHF, especially in cases where thrombocytopenia and MPV levels are high. We also conclude that MPV may be an independent predictor of prognosis in patients with CCHF. This test alone should not be considered as a determining factor for this purpose because it does not have specificity among the other diseases. Therefore, further studies are needed to establish the mechanism of increased MPV levels in CCHF.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
