Abstract
Introduction:
Sticky platelet syndrome (SPS) is most likely a hereditary thrombophilia characterized by platelet hyperaggregation after low concentrations of platelet inducers—adenosine diphosphate and/or epinephrine. We present 9 kindreds with SPS familial occurrence.
Material and Methods:
Familial trait of SPS was looked up in the database of the National Center of Hemostasis and Thrombosis. Families with at least 3 SPS-positive members were studied, described, and presented.
Results:
In the group of 1093 symptomatic patients, SPS was confirmed in 240 cases. Familial occurrence with at least 3 SPS-positive relatives was found in 9 cases.
Conclusion:
The exact pathogenesis of SPS is not sufficiently explained. Our findings seem to support the idea that SPS might have an autosomal dominant hereditary fashion.
Introduction
Sticky platelet syndrome (SPS) was first described by Holliday at The Ninth International Joint Conference on Stroke and Cerebral Circulation in Arizona 1 in 1983. It is considered to be a hereditary thrombophilia characterized by platelet hyperaggregability after stimulation by very low concentrations of platelet inducers—adenosine diphosphate (ADP) and/or epinephrine (EPI), while platelet response to other inducers is normal. 2 Patients may present with angina pectoris, acute myocardial infarction (MI), transient cerebral ischemic attacks (TIAs), stroke, retinal thrombosis, fetal loss syndrome, peripheral arterial occlusion, deep venous thrombosis, sometimes recurrent under oral anticoagulant therapy. 3 –17 Clinical symptoms, especially arterial thrombosis, often present following an emotional stress. 18 According to Mammen, SPS is the second most common hereditary thrombophilia associated with approximately 21% of otherwise unexplained arterial and 13.2% of otherwise unexplained venous thrombosis. 19,20 Sticky platelet syndrome may manifest at any age, particularly up to 50 years, and children can be affected as well. 17,19,21 Depending on the results of aggregometry, 3 types of SPS are distinguished: type I—an increased aggregation induced by both inducers; type II—an increased aggregation induced only by epinephrine, and type III—an increased aggregation induced by ADP. 19,20 The exact cause of SPS has not been yet completely identified. Abnormalities of membrane glycoprotein (GP) receptors (GP IIb/IIIa and GP Ia/IIa) leading to platelet hyperfunction are speculated (assessed by flow cytometry and molecular methods as well), but their relevance remains still obscure. 22 Some authors supposed the role of abnormal GAS-6 protein in the pathogenesis of SPS. The GAS-6 c.834+7G>A polymorphism was investigated but this association was not proved. 23–25 Recently, a significantly higher expression of surface “platelet membrane activation markers” CD62 (P-selectin), CD63, and CD51 was proved in patients with SPS without acute signs of thrombosis compared with healthy controls using flow cytometry. 26 Familial occurrence of SPS is described in the literature 27 ; however, the mode of heredity is still not completely elucidated and seems to be autosomal dominant. The SPS is usually found in a patient with arterial or venous thrombosis through routine thrombophilia testing and subsequently confirmed in the kindred. We present the familial occurrence of SPS in 9 kindreds examined in the National Center of Hemostasis and Thrombosis, Martin, Slovakia. We suppose our findings may contribute to elucidation of SPS transfer in relatives.
Materials and Methods
Familial occurrence of SPS was looked up in the database of the National Center of Hemostasis and Thrombosis, Jessenius Faculty of Medicine, Comenius University (JFM CU) and Martin University Hospital (MUH). In the first step, the patients with arterial or venous thrombosis were investigated using the whole range of thrombophilia tests including ProC global test, factor VIII activity, activity of natural coagulation inhibitors (antithrombin III, protein C, and protein S), screening tests for antiphospholipid syndrome, DNA analysis of gene polymorphisms—factor V Leiden, factor II 20210A, and glycoprotein III.
Diagnostics of SPS
The blood drawn from antecubital vein, collected into tubes prefilled with 3.2% buffered natrium citrate (anticoagulant–blood ratio, 1:9), was used for the testing of platelet aggregation. The samples were processed and analyzed within 2 hours after sampling. We tested platelet aggregation using light transmission aggregometry (PACKS-4 aggregometer, Helena Laboratories, Beaumont, Texas, USA) according to the method introduced by Mammen. 18–20 Each sample was tested with 3 low concentrations of ADP (2.34, 1.17, and 0.58 μmol) and EPI (11.0, 1.1, 0.55 μmol). The criteria of Mammen and Bick were used for the diagnosis and classification of SPS. 18,20 The intra-assay coefficients of variation for platelet aggregation 3.6% (ADP) and 4.5% (EPI) were assessed. The testing was performed while the patient did not receive antiplatelet therapy (discontinuation of acetylsalicylic acid or ADP inhibitors for at least 7 days before testing, omitting the use of other drugs with possible effect on platelet activity, eg, nonsteroidal anti-inflammatory drugs for the same time) and did not have acute thromboembolic event (interval from the last episode at least 3 months). In cases with confirmed SPS, the consanguineous relatives of patients were screened for SPS too. All symptomatic patients and their relatives in whom increased sensitivity to aggregation inducers was found in the first testing were investigated at least twice to confirm this phenomenon was persistent and could be considered hereditary. 28 The period between these tests was 10 days. We looked for thromboses (both arterial and venous) and spontaneous fetal losses in the personal history of family members and considered kindred asymptomatic, if there was no history of thrombotic event. Lifelong antiplatelet therapy/prophylaxis (acetylsalicylic acid [ASA] at the dose 100 mg once daily for adults) was recommended to each individual diagnosed as SPS. The efficiency of ASA was verified by a control aggregometry 1 month after the beginning of treatment and the therapy was modified if necessary. Families with at least 3 SPS-positive members were studied, described, and presented here.
Results
Overall, 1093 symptomatic patients (arterial, venous thrombosis, and miscarriage) were tested for SPS in the National Center of Hemostasis and Thrombosis JFM CU and MUH since 2004. SPS was confirmed in 240 cases. Familial occurrence of SPS with at least 3 SPS-positive relatives was found in 9 cases.
Family 1
The patient was a 14-year-old boy tested for thrombophilia due to the cerebral venous thrombosis (occlusion of superior sagittal sinus, transversal sinus, sigmoidal sinus, cavernous sinus, and internal jugular vein). Screening for thrombophilia was completely negative except for the methylenetetrahydrofolate reductase C677T mutation (heterozygote) and SPS type I. The SPS was found also in the patient’s relatives, type I in his brother and mother, type II in his father, all of them were asymptomatic (Figure 1 ). Thrombotic events were found in patient’s family history. Grandfather from father’s branch got double ischemic strokes (the first one at the age of 47 years) and 2 MIs. Grandmother from mother’s side developed ischemic stroke at the age of 55 years. These relatives were not tested for thrombophilia.
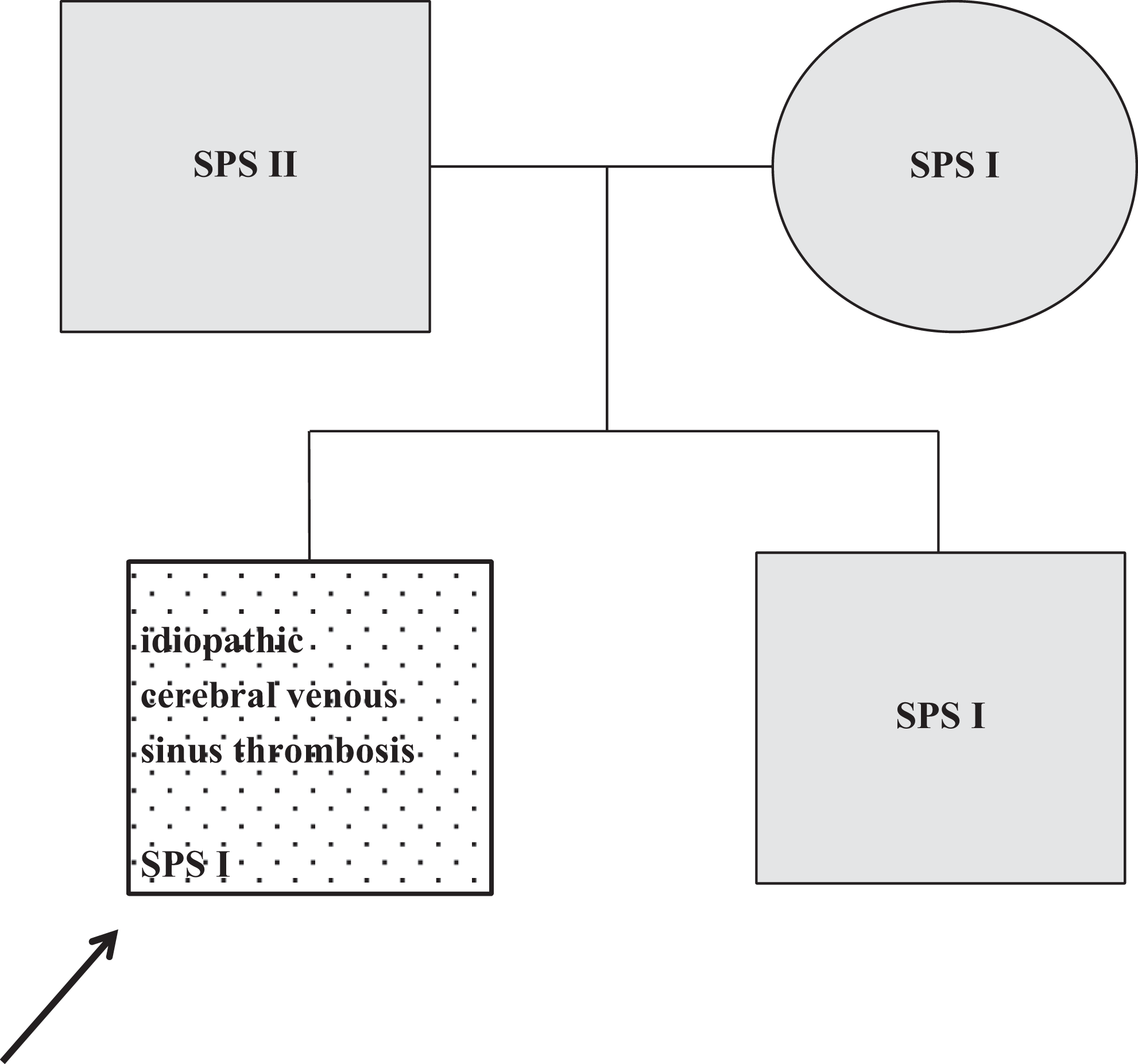
Family 1. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA. SPS indicates sticky platelet syndrome; ASA, acetylsalicylic acid.
Family 2
The patient was a 24-year-old female who developed deep vein thrombosis and pulmonary embolism 1 week after commencement of the oral contraceptives use. The thrombophilia testing showed SPS type II and antiphospholipid syndrome (strong positivity of anticardiolipin antibodies and lupus anticoagulans). Screening of the family members revealed SPS II in patient's sister, brother, and both parents. All these family members were asymptomatic (Figure 2 ).
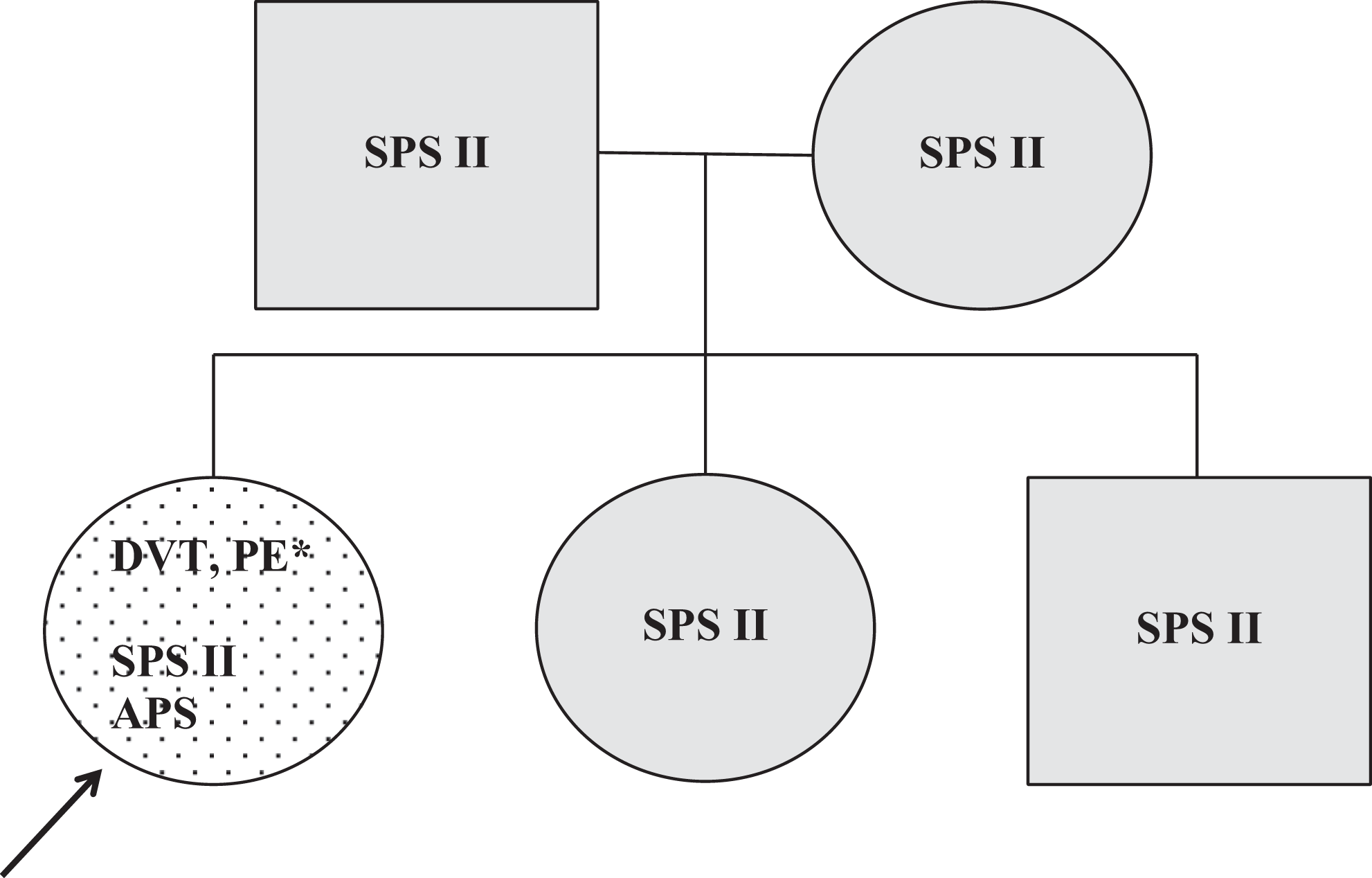
Family 2. Dotted, symptomatic patient; black arrow, the first family member diagnosed as sticky platelet syndrome positive; gray, asymptomatic on ASA; *1 week after the beginning of contraceptive use. DVT indicates deep vein thrombosis; SPS, sticky platelet syndrome; PE, pulmonary embolism; APS, antiphospholipid syndrome.
Family 3
The patient was a 40-year-old female tested for thrombophilia due to an ischemic stroke. The SPS type I was found as the only thrombophilia. In addition, SPS type II was confirmed in her daughter and 4 sisters. All these family members were asymptomatic except for one, who passed one miscarriage and suspected TIA (Figure 3 ). Patient’s mother got a stroke at the age of 50 years, but she was not tested for thrombophilia.
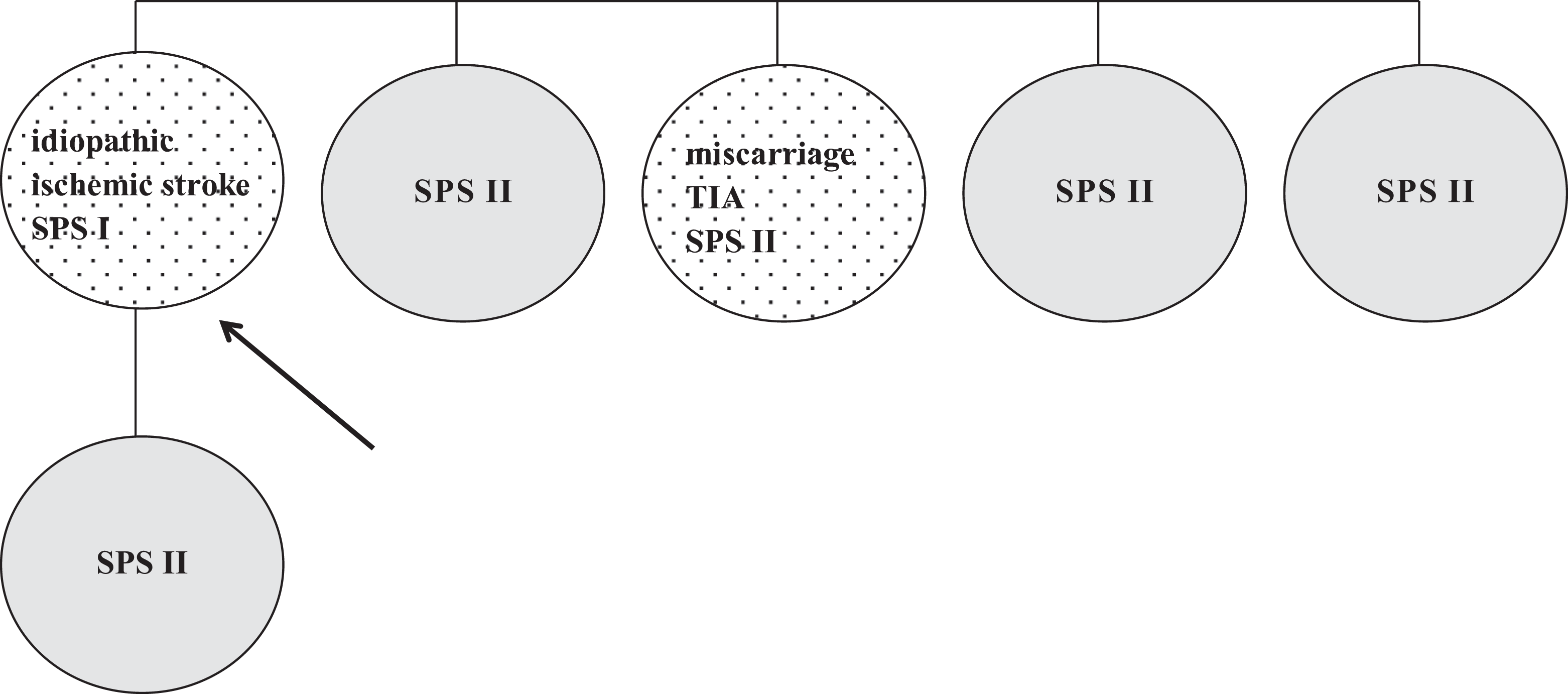
Family 3. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA. TIA indicates transient ischemic attack; ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 4
The patient was a 50-year-old man who had the lower limb arterial occlusion. Standard thrombophilia screening revealed SPS type II. It was also confirmed in his 2 daughters afterward. One of them had paraesthesia of the upper extremity in a stresful situation in her personal history and extremely high platelet aggregation induced by epinephrine. The other 2 patient’s offsprings as well as his brother were SPS negative (Figure 4 ).
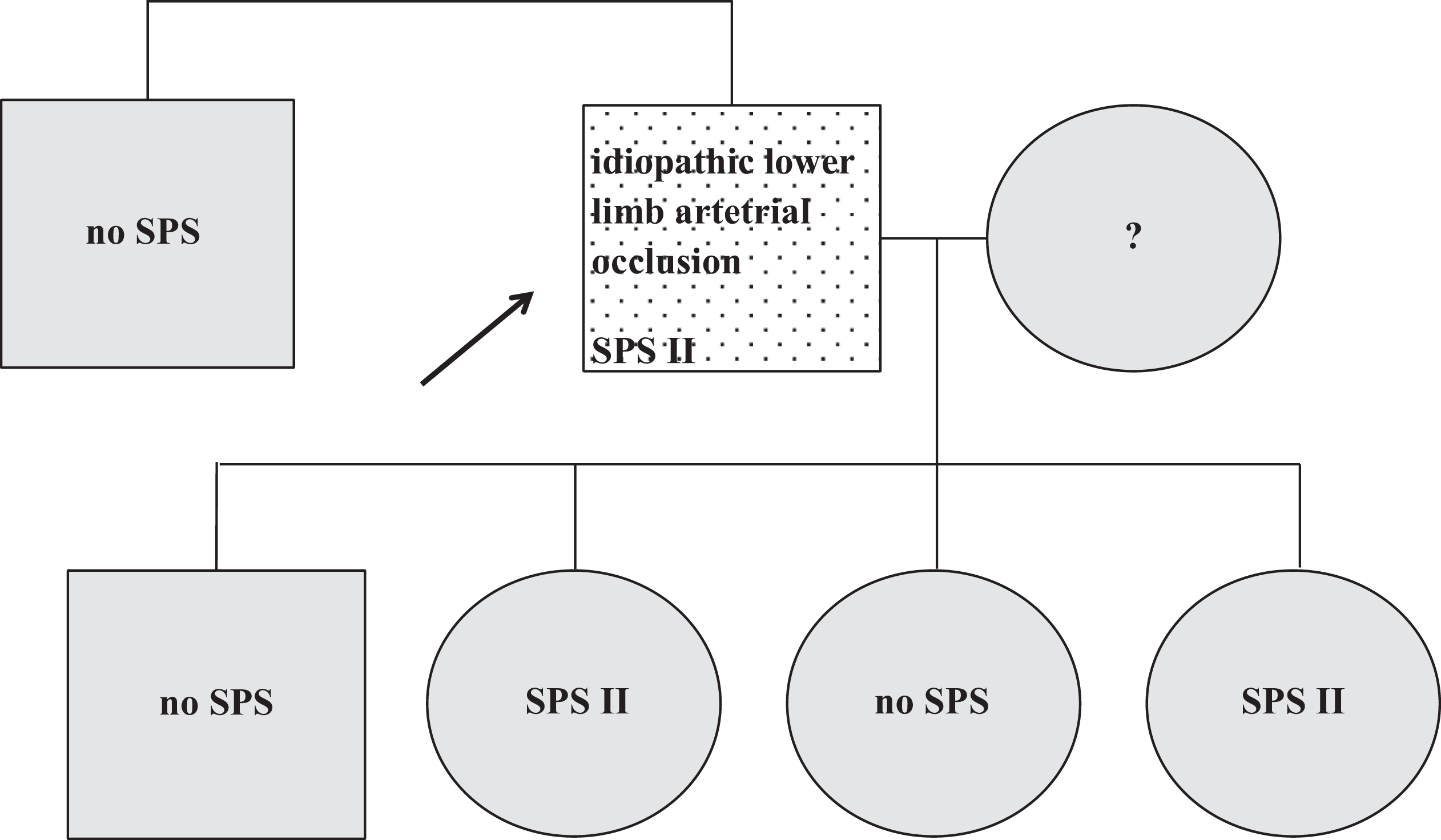
Family 4. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA; no SPS, SPS not confirmed; ?, SPS not tested. ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 5
The patient was a 64-year-old male who passed an ischemic stroke and was screened for thrombophilia afterward. The SPS type II and the GPIIIa C98T mutation (homozygote) were confirmed. We found the SPS in patient’s kindreds (type II in his son and type I in his daughters) all of them were asymptomatic (Figure 5 ). Patient’s father underwent 3 MIs, the first one at the age of 45 years, but he was not tested for thrombophilia.
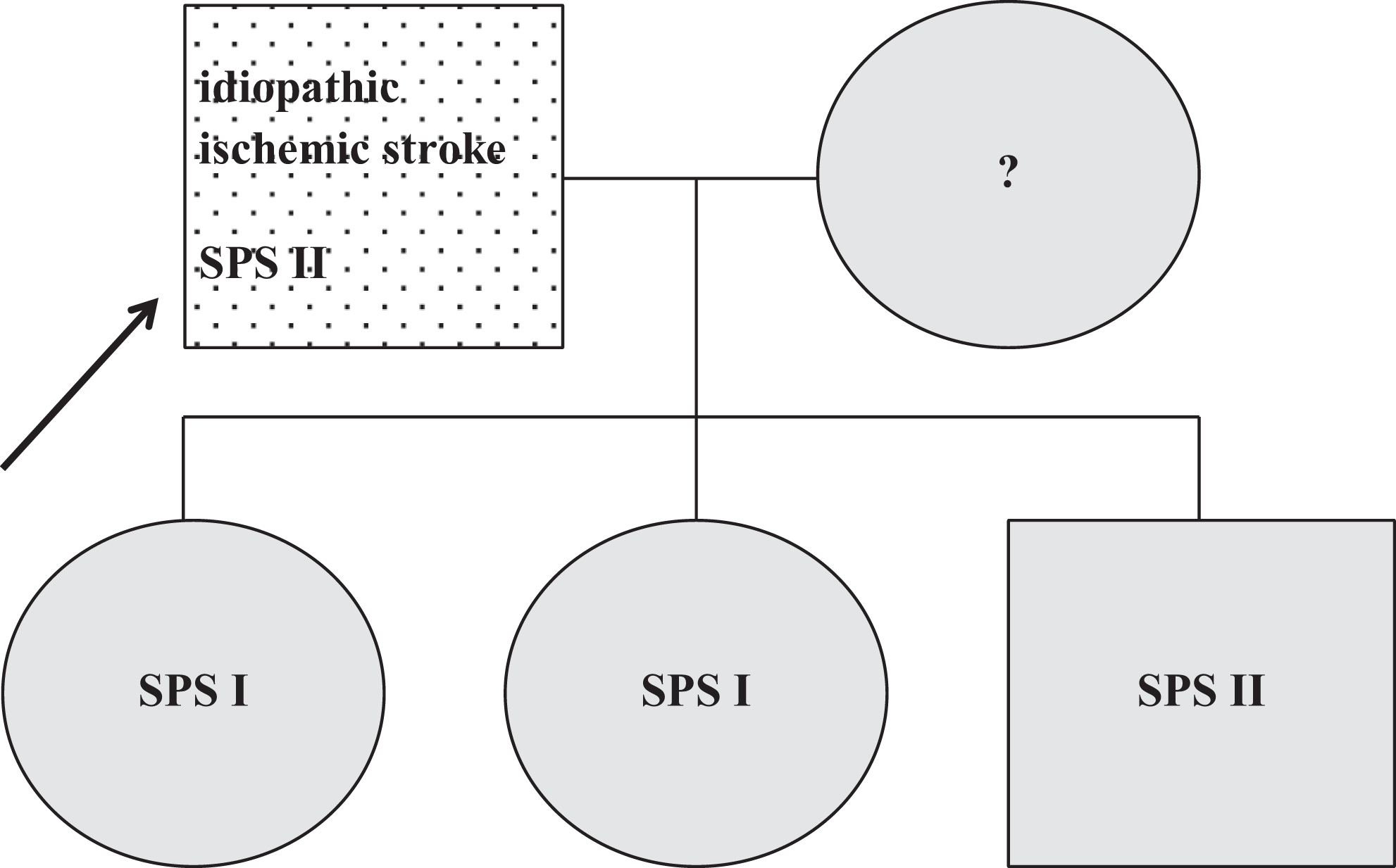
Family 5. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA; ?, SPS not tested. ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 6
The patient was a 46-year-old man who passed an ischemic stroke and died of MI which occurred during a car accident. He had factor V Leiden mutation (heterozygote) and SPS type II. Thrombophilia testing in the family showed factor V Leiden mutation (heterozygote) and SPS type II in his brother, sister, and daughter, one brother had the SPS type I. All these family members were asymptomatic (Figure 6 ).
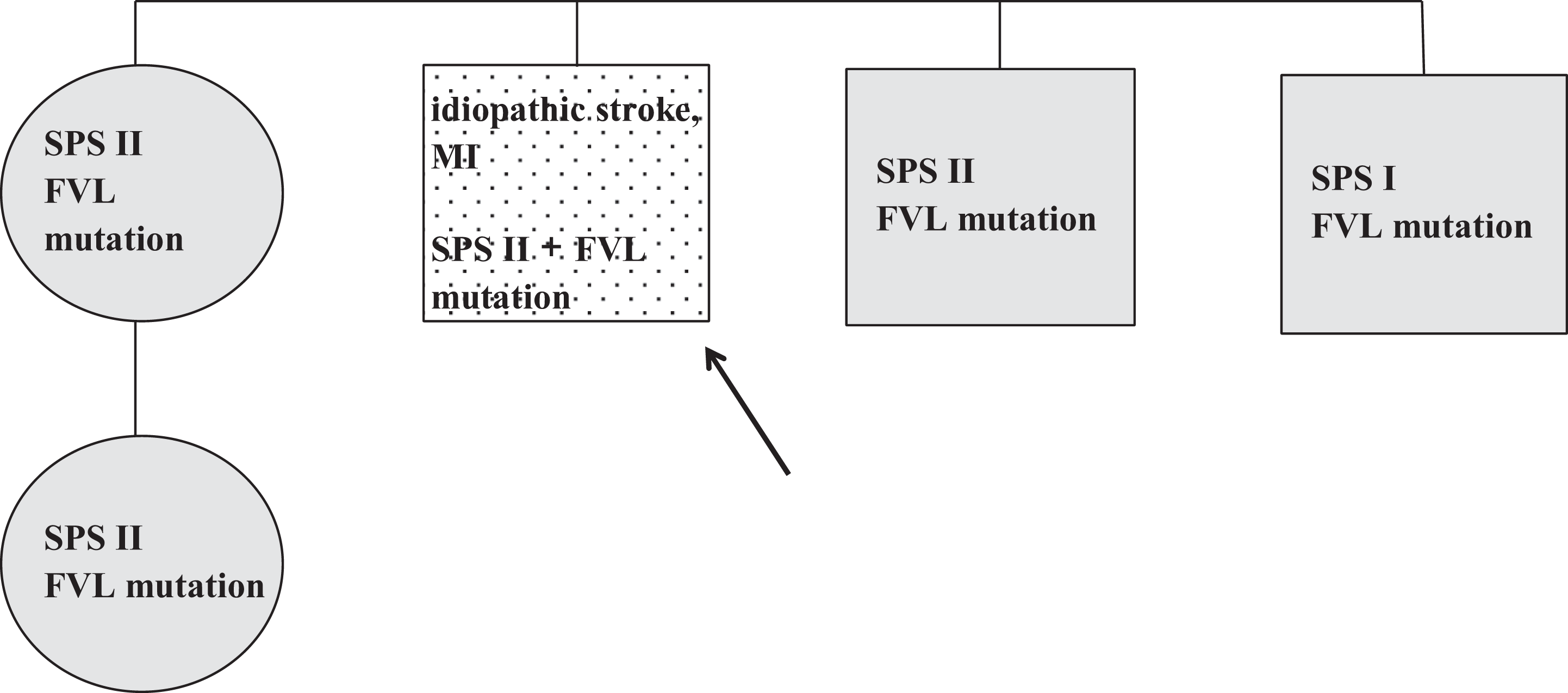
Family 6. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA. MI, myocardial infarction; FVL, factor V Leiden); ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 7
The patient was a 41-year-old male who underwent multiple ischemic strokes. The thrombophilia screening revealed an increased platelet aggregation induced by epinephrine (SPS type II). The same thrombophilic abnormality was confirmed in his brother, daughter (both asymptomatic), and mother who had TIAs (Figure 7 ).
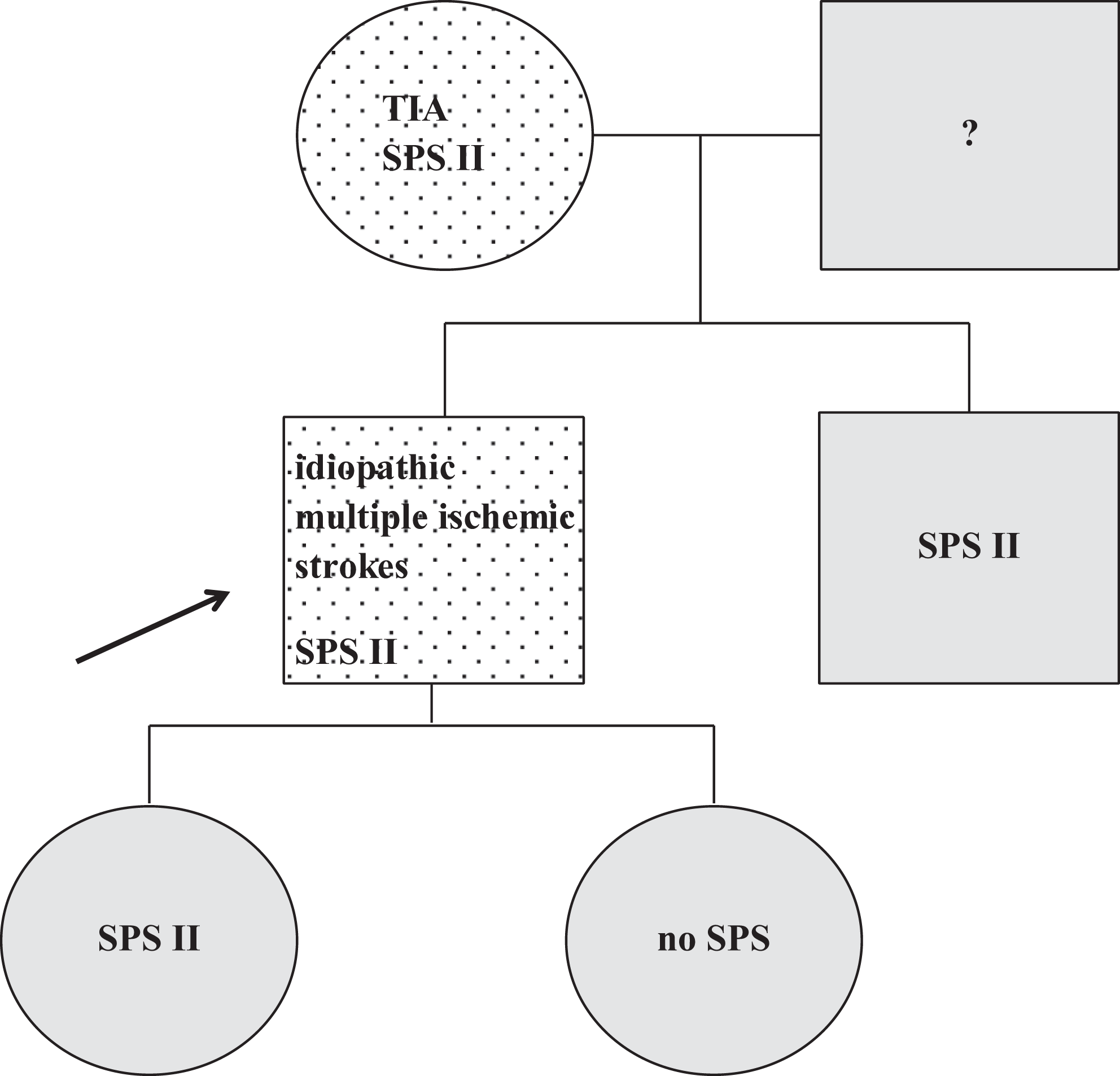
Family 7. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA; no SPS, SPS not confirmed; ?, SPS not tested. TIA indicates transient ischemic attack; ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 8
The patient was a 2.5-year-old boy who developed an ischemic stroke (the thrombosis of right middle cerebral artery) with central lesion of the left facial nerve and left central hemiparesis. He underwent varicella infection 1 month before the thrombotic event. Laboratory tests showed SPS type I. We diagnosed the same type of SPS in patient’s mother, the SPS type II was detected in his sister—both were asymptomatic (Figure 8 ).
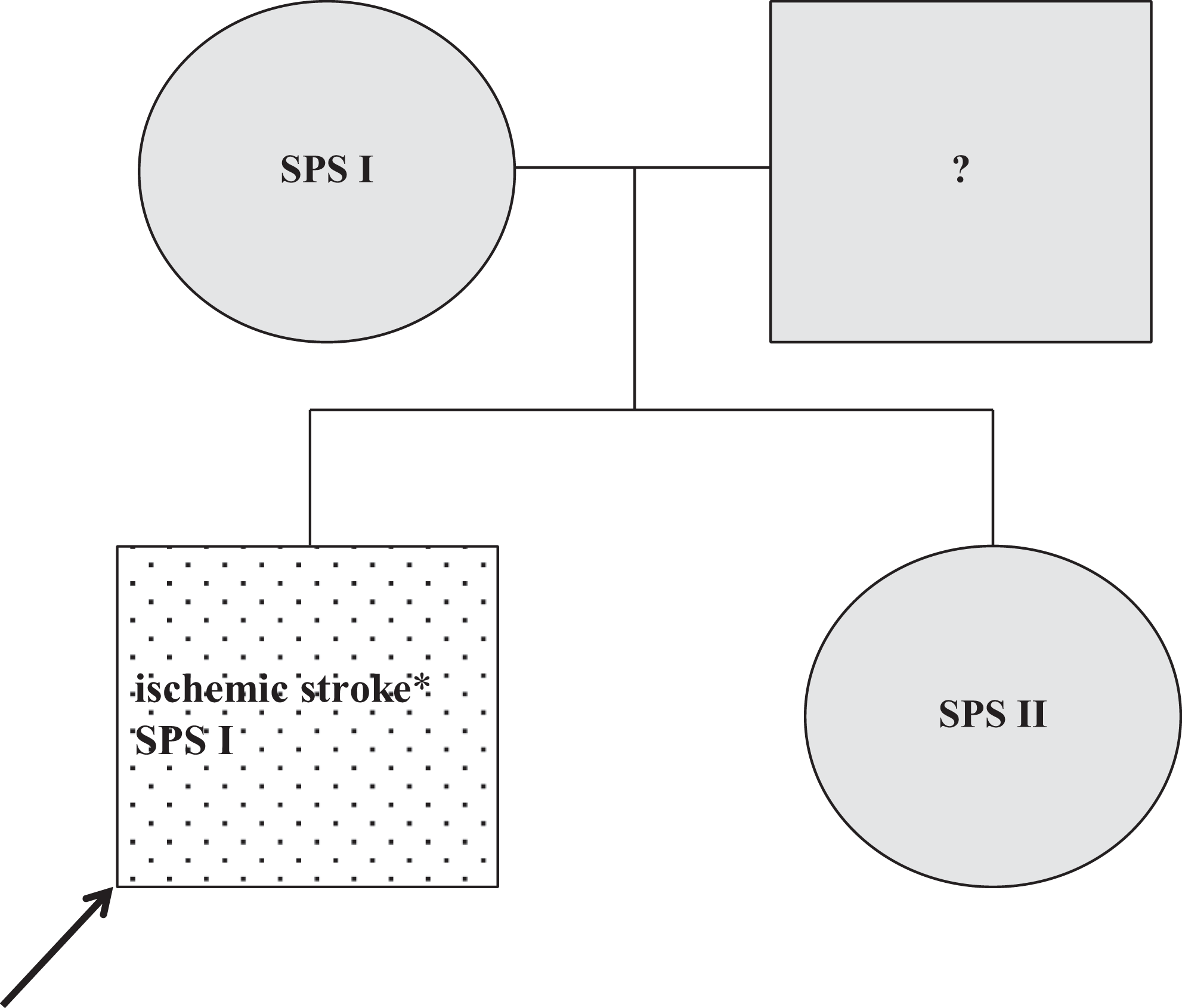
Family 8. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA; ?, SPS not tested; *1 month after varicella infection. ASA indicates acetylsalicylic acid; SPS, sticky platelet syndrome.
Family 9
The patient was a 38-year-old woman with fetal loss syndrome and deep vein thrombosis associated with pulmonary embolism. She started using oral contraceptives 6 weeks before thrombosis onset. The thrombophila screening revealed platelet hyperaggregation induced by epinephrine—SPS type II. The same type of SPS was found in the patient’s daughter, sister, and 2 brothers, another brother was SPS negative. Three children of patient’s siblings were diagnosed as having SPS type I. All kindreds were asymptomatic (Figure 9 ).
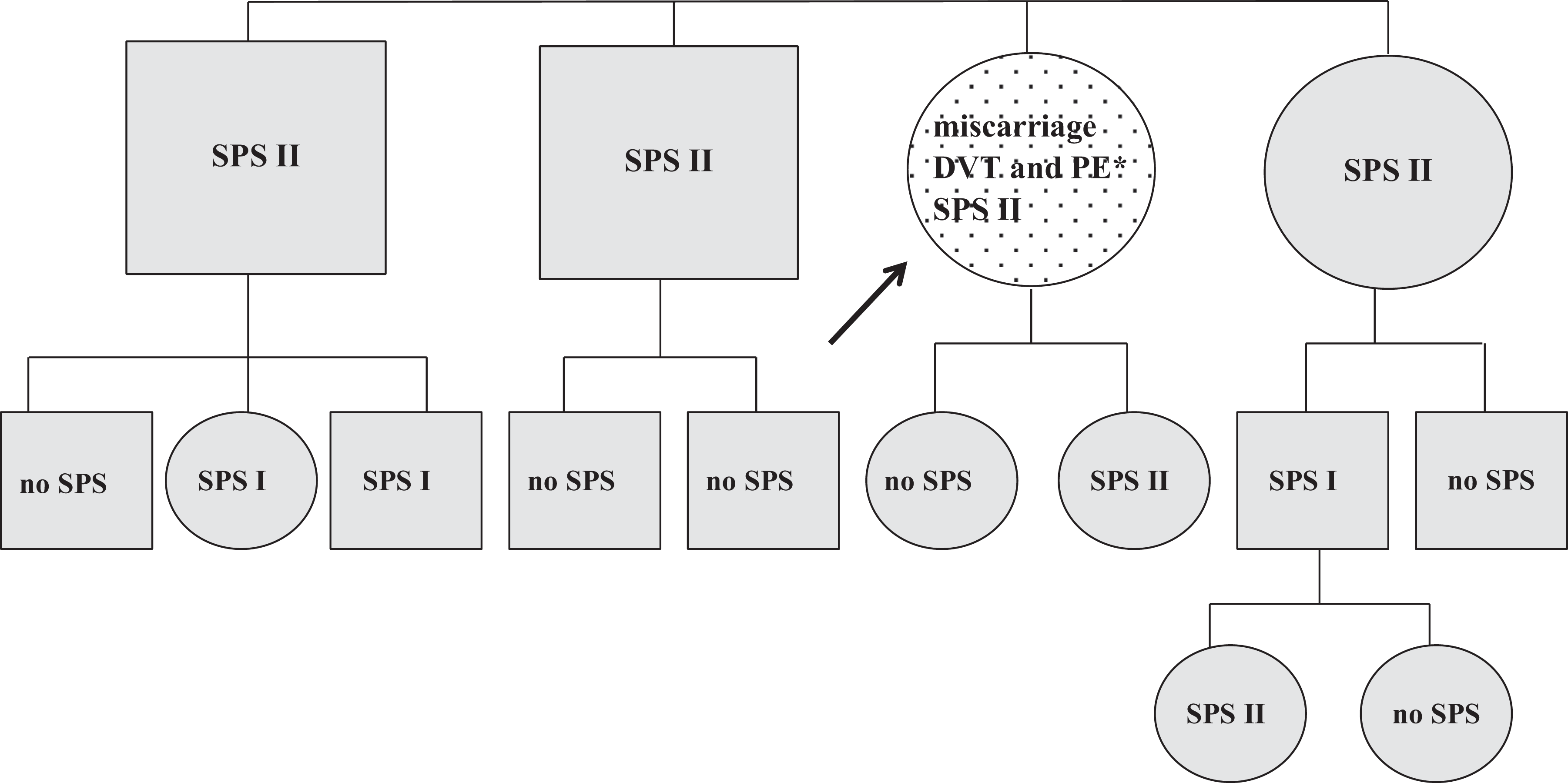
Family 9. Dotted, symptomatic patient; black arrow, the first family member diagnosed as SPS positive; gray, asymptomatic on ASA; no SPS, SPS not confirmed; *6 weeks after the beginning of oral contraceptive use. DVT indicates deep vein thrombosis; PE, pulmonary embolism; ASA, acetylsalicylic acid; SPS, sticky platelet syndrome.
Discussion
Genetic background of the SPS is still not sufficiently explained; however, its familial occurrence is described in the literature. 27 We studied the SPS phenotype in kindreds of symptomatic SPS patients and present 9 families with at least 3 SPS-positive members. According to our findings, SPS seems to be an autosomal dominant thrombophilia and may predispose to venous and arterial thromboses and spontaneous fetal loss. We also present 2 cases of thrombotic episodes in childhood. Our findings are in good agreement with the literature. 3 –17,21 Recently, a very interesting case report of pregnant woman with previous recurrent first-trimester miscarriages who developed extensive deep vein thrombosis and subsequently arterial thromboses and fetal demise at 11th week of gestation was described. The SPS was confirmed as the only thrombophilia afterward. 29 This and many other studies support current SPS position among the other thrombophilias. 3 –21
The SPS seems to be inherited and manifested independently of its type. We observed both the most frequent types (I and II) in almost all kindreds and only 2 families with the same type of SPS.
According to our and other studies, 12 –14,18 –20,29 the SPS can predispose to thrombotic event by itself or sometimes in a cooperation with additional risk factor (stress, oral contraceptives, infection, or another thrombophilic state—factor V Leiden mutation and antiphospholipid syndrome). It is also important to mention that every individual diagnosed as SPS positive (confirmed with double testing) was subsequently treated with ASA in a loading dose of 100 mg once a day (for adults), with its efficiency validated by control aggregometry 1 month after the beginning of the treatment. If necessary, ASA dose was escalated or inhibitors of ADP receptors were used. Thus, every asymptomatic SPS-positive individual in our kindreds has to be considered as asymptomatic at the time of diagnosis and then “asymptomatic on ASA treatment.” It seems that screening of asymptomatic family members might be important for primary prevention of thrombosis (low-dose treatment with acetylsalicylic acid) and could lead to the reduction of cardiovascular morbidity and mortality.
Since pathogenesis of SPS is still not adequately clarified, we hope our study demonstrates the familial occurrence of this syndrome. However, many further studies are needed to discover the exact cause of SPS and its heredity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the project “Center of Excellence for Perinatology Research” (project CEPV II) (ITMS:26220120036) which is co-financed from EC sources and by grants of the Scientific Grant Agency (VEGA) of the Ministry of Education, Science, Research and Sport of the Slovak Republic (MSVVaS SR) and Slovak Scientific Academy (SAV) 1/0029/11, 1/0018/10.
