Abstract
Introduction:
The sticky platelet syndrome (SPS) seems to be a common cause of thrombosis, although no molecular substrate to explain platelet hyperaggregability has been found.
Objective:
To analyze an association between the SPS phenotype and the platelet glycoprotein (GP) IIIa PLA1/A2 (human platelet antigen [HPA]-1a/b) gene polymorphism.
Methods:
Along an 18-month period, Mexican mestizo thrombophilic patients were prospectively accrued. The SPS phenotype was assessed by aggregometry, whereas a tetra-primer amplification refractory mutation system (ARMS) polymerase chain reaction analysis was used to detect the PLA1 and PLA2 alleles.
Results:
A total of 95 individuals with SPS and 127 healthy donors were studied; in 11 of the donors and 16 of the patients with SPS the A2 allele of the GP IIb/IIIA was found, yielding a weak and nonsignificant association (odds ratio 2.14, 95% CI 0.94-4.85).
Conclusion:
In Mexican mestizo patients, the platelet GP IIIa PLA1/A2 gene polymorphism does not lead to the SPS phenotype.
Introduction
The sticky platelet syndrome (SPS) was first described in 1983 1 ; however, not until later did its prevalence receive significant recognition in the medical literature. 2–31 The SPS seems to be a rather common cause of arterial and venous thrombosis 2–6 since it accounts for about 20% of otherwise unexplained arterial events and 13% of unexplained venous events. 2–8 Three forms of the SPS have been identified: type I is marked by platelet hyperaggregability with adenosine diphosphate (ADP) and epinephrine, whereas type II is marked by hyperaggregability only with epinephrine and type III only with ADP. 3–6 The platelet abnormality seems to be congenital and the precise nature of the defect is at present not known 4 ; it is supposed that glycoprotein (GP) receptors on the platelet surface membrane may be involved, its abnormality leading to platelet hyperfunction. 7,8 Up to now, no molecular substrate to explain the platelet hyperaggregability has been found in the SPS phenotype, this being the reason why only few research groups have accepted this entity as a true thrombophilic condition. 29 Only few attempts to correlate the SPS phenotype with molecular markers of platelet hyperaggregability have been described: the GP IIIa PLA1/A2 (HPA-1a/b ITGB3:c.196T>C) polymorphism 7 and the growth arrest–specific gene 6 (Gas6; Gas6:c.834+7G >A) polymorphisms. 8
The GP IIb/IIIa is a heterodimeric platelet surface receptor consisting of the αIIb (CD41) and the β3 (CD61) subunits, which serves as the fibrinogen receptor; it is highly polymorphic and the role of the polymorphisms in the pathogenesis of thromboembolism has been studied for several years 7,32 ; some authors have observed an association between the PLA1/A2 polymorphism of the GP IIIa gene and acute coronary thrombosis. 32
In a small study of 9 patients with the SPS phenotype, Kubisz et al 7 were unable to find an association between the SPS phenotype and the polymorphism of the GP IIIa gene. In an effort to further clarify the causes of the thrombophilia of Mexican Mestizos, we have now assessed prospectively the GP IIIa PLA1/A2 polymorphism in a group of 95 Mexican mestizo patients with SPS.
Materials and Methods
Patients
Along an 18-month period, all consecutive Mexican Mestizo patients referred to our clinic by physicians from different parts of the country were prospectively accrued in the study if, in addition to displaying the SPS phenotype, they had one of the following clinical markers associated with a primary hypercoagulable state26,27: a) Venous or arterial thrombosis at age below 40 years; (b) family history of thrombosis; (c) recurrent thrombosis without apparent precipitating factors; (d) thrombosis at unusual anatomic sites; and (e) resistance to conventional antithrombotic therapy, they had SPS phenotype. Individuals with overt malignancy, pregnancy, puerperium, oral contraceptives, or other conditions associated with secondary thrombophilia were excluded from the study.
Analytical Methods
Assessment of the SPS phenotype
In an effort to make the results comparable and consistent with previous studies, the method described by Mammen et al
2,4
was used by following exactly the same conditions, agents, and dilutions: blood was drawn, usually between 8:30 and 10:30
Investigation of the GPIIIa PLA1/A2 polymorphism
A tetra-primer ARMS polymerase chain reaction (PCR)-based analysis was used for the detection of the PLA1 and PLA2 alleles 34 . Briefly, 20 ng genomic DNA was amplified with 0.2 µmol/L common primers GP3F 5′ AGGGCCTGCAGGAGGTAGAGAGTCGCCA 3′ and GP3R 5′ GCTGTCTCCAGAGCCCTTGTCGCTGAGG 3′ and 0.1 µmol/L allele-specific primers GP3T 5′ GGCTCCTGTCTTACAGGCCCTGCCGCT 3′ and GP3C 5′ TCCTTCAGGTCACAGCGAGGTGAGCACG 3′. Five µL of the amplification products were analyzed by 4.5% polyacrylamide gel electrophoresis. The internal control yields a 215 pb product, while the PLA1 and PLA2 alleles were detected as 160 and 109 bp fragments, respectively.
Statistical Analysis
Genotype and allele frequencies were computed for the patients and the healthy control group; normal individuals with the SPS phenotype were not included in the analysis. The genotypic odds ratio for carrying the A2 allele and the allelic odds ratio as well as the corresponding confidence intervals for α = 0.05 were calculated according to Bland and Altman 35 for the complete patient group or subdivided according to the SPS phenotype subtype.
Results
Patients
In a group of 160 consecutive patients with a clinical marker of primary thrombophilia, 95 patients were identified with SPS phenotype (43 males and 52 females). Median age was 42 years and range 1 to 82 years. All patients fulfilled the criteria of a primary hypercoagulable state (vide supra).
The SPS phenotype
Of the 95 patients, 61 had type I SPS, 6 had type II SPS, and 28 had type III SPS. Of the 95 patients, 85 (89%) showed hyperaggregability at the highest dilution with ADP, whereas 46 (48%) of 95 showed hyperaggregability at the highest dilution with epinephrine. All patients showed hyperaggregability at the highest dilution with either ADP or epinephrine.
Glycoprotein IIIa PLA1/A2 polymorphism
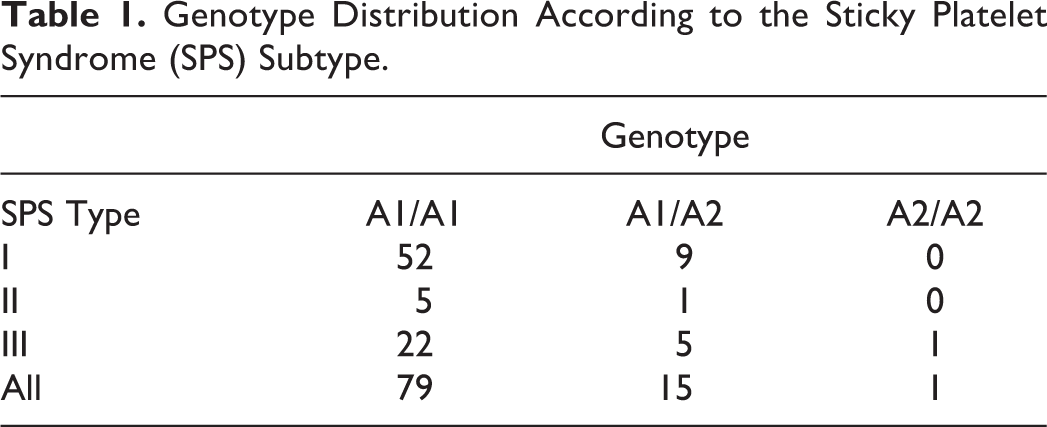
Of the 95 patients, 79 (83%) were found to have the PLA1/A1 genotype, 15 (16%) displayed the PLA1/A2 genotype and 1 (1%) showed the PLA2/A2 genotype (genotype frequencies: 0.83, 0.16, and 0.01, respectively). In contrast, in a population of 127 healthy blood donors, the observed genotype frequencies were 0.91, 0.07, and 0.02. The odds ratio for carrying the A2 allele in the studied populations was 2.14 (95% CI: 0.94-4.85). Table 1 shows the PLA1/A2 genotype distribution according to the SPS type: it is clear that there is no significant association between mutations in the PLA1/A2 gene and the SPS phenotype, irrespective of its subtype.
Genotype Distribution According to the Sticky Platelet Syndrome (SPS) Subtype.
Discussion
There is clinical and experimental evidence that changes in the hemostasis system can lead to a hypercoagulable or thrombogenic state in the circulation that can foster thrombus formation. In the last years, we have been interested in analyzing the changes in the hemostatic system of Mexican Mestizos, which can result in thrombophilia, and accordingly we have found different abnormalities in the natural antithrombotic mechanisms. 6,26,27,36 –39 In these studies, it has been made clear that the SPS is the second most frequent thrombophilic condition identified in Mexican Mestizos with a clinical marker of thrombophilia 6,26,27 only surpassed by the 677C>T mutation in the 5,10-methylenetetrahydrofolate reductase (MTHFR), which by itself is not a fully recognized thrombophilic condition; the consensus seems to be emerging that the MTHFR 677C>T variant is not a significant risk factor for venous thrombosis, per se but probably when associated with other thrombophilic conditions. 26,27 In Mexico, we 6,26,27 and others 22,25 have found that approximately 50% of Mexican Mestizo patients with a clinical maker of thrombophilia display the SPS phenotype. Most patients with the SPS display other thrombosis-prone conditions, but there were also instances of the SPS identified as the single thrombophilia marker 6,26,27 ; accordingly, it is possible that this platelet abnormality may contribute to the so-called multifactorial thrombophilia. 26
The platelet abnormality in the SPS seems to be congenital, but the nature of the defect is at present unknown 4 ; it is supposed that GP receptors on the platelet surface membrane may be involved in its abnormality, leading to platelet hyperfunction. 7,8 Since no molecular substrate to explain the platelet hyperaggregability has been found in the SPS phenotype, there is no consensus about the true existence of this entity. 29,40 It has to be mentioned that there is an extensive list of diseases in which an association of arterial thrombosis with platelet hyperaggregability may be found and that in this study other causes of platelet hyperaggregability were not investigated.
In this prospective case–control study, we have found that the odds ratio for carrying the A2 allele of the GP IIIa gene was 2.14 (95% CI: 0.94-4.85), the association being weak and nonsignificant. This information is consonant with that published previously by Kubisz et al, 7 in a small group of 9 patients with the SPS. These data suggest that the mutations in the GP IIIa gene may not play a role in the expression of the phenotype.
The phenotype and genotype studies of thrombophilia accomplished to date indicate that, similar to most diseases, hereditary thrombophilia is genetically complex. A careful search for additional thrombophilic conditions is needed to address the question of allelic and locus heterogeneity, and population studies are needed to address genotype–phenotype relationships. Our data may be useful to add information to the understanding of the SPS phenotype.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
