Abstract
No-reflow phenomenon due to cardiac microvascular dysfunction or disturbance aggravates clinic outcomes of a portion of patients with acute myocardial infarction undergoing percutaneous coronary intervention or thrombolytic therapy. Our working hypothesis was that cardiac microthrombosis would play an important role in the pathogenesis. We investigated that cardiac microthrombi were observed by Martius, Scarlet, Blue methocl (MSB) and Masson trichrome staining. Furthermore, we investigated the expression of fibrinogen-like protein 2 (fgl2) in rats with acute myocardial ischemia/reperfusion (MI/R) and its possible pathological and clinical significance. The fgl2 was highly expressed in myocardium of rats with acute MI/R and located at cardiac microvascular walls. We found that the expression of fgl2 in peripheral mononuclear cells of rats with acute MI/R significantly increased correspondingly with its cardiac expression. Expression of cardiac fgl2 was correlated with no-reflow size of rats with acute MI/R, which was detected and calculated by thioflavin S staining. No-reflow size was in line with cardiac diastolic dysfunction of rats with acute MI/R monitored by hemodynamics. Thus, microthrombosis is involved in cardiac microvascular dysfunction or disturbance of rats with acute MI/R as one cause, and fgl2 may emerge as a predictor of the occurrence of no-reflow phenomenon.
Introduction
As a severe consequence of coronary heart disease, acute myocardial infarction (AMI) places the lives of millions of people in great danger all over the world. 1 Reperfusion therapy including thrombolytic or mechanical interventional treatment stands mainstream in the management of AMI. 2 However, in the clinical practice, a portion of patients with AMI who receive reperfusion therapy are still suffering severe myocardial ischemia due to myocardial reperfusion defect, which is termed slow-flow or no-reflow phenomenon. 3 It is observed by coronary angiography that there is no or weak blood flow in distal vessels after the successful interventional operation of obstructive arteries, grading with thrombolysis in myocardial infarction (TIMI) 0 or 1.4,5 Clinical investigations have revealed that no-reflow is a strong independent predictor of poor in-hospital outcomes and 5-year mortality, which is associated with AMI, unstable angina, and cardiogenic shock.5–7 Thus, reperfusion therapy is a double-edged sword, although it is able to benefit infarct salvage, together with cardiomyocyte damage and microvascular dysfunction.
Microvascular dysfunction is attributed greatly to reperfusion injury8–10 and is responsible for no-reflow phenomenon by limiting adequate blood and oxygen supply. Also, no-reflow is defined as inadequate myocardial perfusion through a given segment of the coronary circulation without angiographic evidence of mechanical vessel obstruction. 11 Over the last decade, a novel inducible endothelial cell procoagulant, fibrinogen-like protein 2 (fgl2), 12 was found to be expressed abundantly in microvasular endothelial cells of impaired tissues or organs. It was involved in microvascular dysfunction of various diseases such as viral hepatitis, 13 acute vascular xenograft rejection,14–16 cytokine-induced fetal loss syndrome, 17 severe acute respiratory syndrome, 18 and autoimmune glomerulonephritis. 19 However, less knowledge is available about its expression and possible mean in microvascular dysfunction due to reperfusion injury.
As yet, animal models of mechanical coronary artery ligation and reperfusion are suitable for researching the no-reflow phenomenon.11,20,21 In this study, we examined whether microthrombosis and fgl2 were expressed in microcirculation of a rat model with acute myocardial ischemia/reperfusion (MI/R) and attempted to seek its relevance to no-reflow phenomenon because direct evidence in homo sapiens is not clinically feasible.
Materials and Methods
Animals and Treatment
Adult male Sprague-Dawley (SD) rats (body weight: 230–270 g) were obtained from the animal center of Tongji Medical College of Huazhong University of Science and Technology. During the experiment, animals were given tap water and rodent diet ad libitum and treated in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health in 1996 and Laboratory Animals Regulation Administration published by China National Science and Technology Commission in 2004.
Male SD rats were randomly divided into the sham group and the acute MI/R group. The time points for the acute MI/R group and the sham group were 1, 3, and 7 days. A minimum of 6 successfully constructed rats were used in both the groups per time period in a single method. There were totally 134 rats (108 total samples) in this research for avoiding unsuccessful construction and that the same one could not be used in various detecting methods, such as echocardiography, hemodynamics, myocardial staining, histopathology, and immunohistochemistry.
Acute MI/R Rat Model
The acute MI/R rat model was established as described previously. 22 Anesthetized by intraperitoneal injection of 2% pentobarbital (2 mL/kg), and rats were positive-pressure ventilated by small animal respirator (Harvard Apparatus Rodent Respirator) after tracheal intubation. The exposed heart was prepared by opening left thorax. In the acute MI/R group, ligating left anterior descending (LAD) coronary artery was consecutive for 45 minutes before reperfusion, whereas it was just placed around by string not ligated in the sham group. After closing the chest layer by layer, benzylpenicillin sodium (400 000U/kg) was injected through intraperitoneal once a day for 3 days. Successful acute MI/R was confirmed not only by visual inspection of left ventricle color alteration but also by ST-segment-characterized electrocardiogram.
Evaluation of Echocardiography
Two-dimensional and M-mode echocardiography was performed in both the short- and the long-axis views using a 10S probe connected with Vivid7 echocardiogram machine (GE medical, Milwaukee, WI, USA). Inherent analysis software captured digital images in 3 consecutive cardiac cycles and made average measurements at the time of the study.
Hemodynamics
A sidearm of the aortic cannula insert in right carotid artery connected to a pressure transducer (Millar Instruments, Houston, Texas, USA) and BL-420F organism function experiment system amplified the signal. Through the ascending aorta, the catheter was inserted finally into the left ventricle.
Myocardial Staining
Myocardial ischemic risk size and no-reflow size were, respectively, observed as described previously. 11 Thioflavin S (4%; 2 mL/kg) was first injected intracordially below left auricle and then 1% Evans Blue (1 mL) was injected. After 5 minutes of intracordial injection, the whole heart was cutoff and transected parallel to the atrioventricular groove at the center of the infarct area.
Histopathology and Immunohistochemistry
Each heart was cut for a transverse section. Each sample was tested by histopathology and immunohistochemistry. For the identification of cardiac microthrombi, MSB staining and Masson trichrome staining were adopted. Masson trichrome staining was also used for the clear discrimination of vascular wall structure. Myocardial pathology was observed by hematoxylin and eosin (HE) staining. Cardiac fgl2 was detected and located by immunohistochemistry using monoclonal mouse anti-rat fgl2 (WH0010875M1; Sigma [St Louis, MO, USA]) following the instruction of the manufacturer.
Western Blot Analysis
The retained heart tissues were located in the anterior wall of left heart below the position of LAD coronary artery ligation. Proteins from the tissues of the posterior wall of left heart or the right heart on day 3 in the acute MI/R were also used as a control.
Equal amounts of protein from each sample (4 mL whole blood or 100 g tissue) were separated with a 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Protein samples were homogenized in a buffer (Sigma). The samples of protein are blocked for 2 hours with 5% defatting milk, after transferring onto nitrocellulose membranes. Subsequently, they were incubated overnight at 4°C with monoclonal mouse anti-rat fgl2 (ab77642; Abcam [Cambridge, MA, USA]) diluted at 1:1000. The membrane was incubated with the corresponding secondary antibody remarked with horseradish peroxidase followed by 3 consecutive cycles of washing. Immunoreactive bands were visualized in dark with the enhanced chemiluminescence kit (Amersham, Sweden). A densitometer analysis system (Quantity One V4.62) was used to measure band intensities. Equal protein loading was confirmed by staining the gel with Ponceau S and probing with β-actin antibody (Abcam).
Statistical Analyses
All values are expressed as mean ± standard deviation. Data were analyzed using SPSS 13.0 software. P < .05 was considered statistically significant. All statistics were analyzed by analysis of variance or Spearman correlation analysis.
Results
Cardiac Diastolic Dysfunction in Rats With Acute MI/R
We examined whether cardiac function changed in a short period after ischemia–reperfusion established in a rat model by echocardiography and hemodynamics (Figure 1 ). There was no significant finding about cardiac systolic function using the parameters left ventricular internal dimension (LVIDs), ejection fraction, fractional shortening, left ventricular systolic pressure, left ventricular end-diastolic pressure, or +dp/dt max in the sham and the acute MI/R group on days 1, 3, and 7 (P > .05). However, given equal heart rates, the parameter reflecting diastolic function showed a decreasing tendency in the acute MI/R group when compared with the sham group at the same time point (Figure 2 ). The −dp/dt max of the acute MI/R group on day 3 was significantly lower than that of the sham group (1.49 ± 0.37 mm Hg/ms vs 2.80 ± 0.70 mm Hg/ms, P < .05), whereas the values on days 1 and 7 showed no significant difference when compared with the sham group (2.16 ± 0.68 mm Hg/ms and 2.46 ± 0.67 mm Hg/ms vs 2.75 ± 0.59 mm Hg/ms and 2.49 ± 0.65 mm Hg/ms, P > .05).
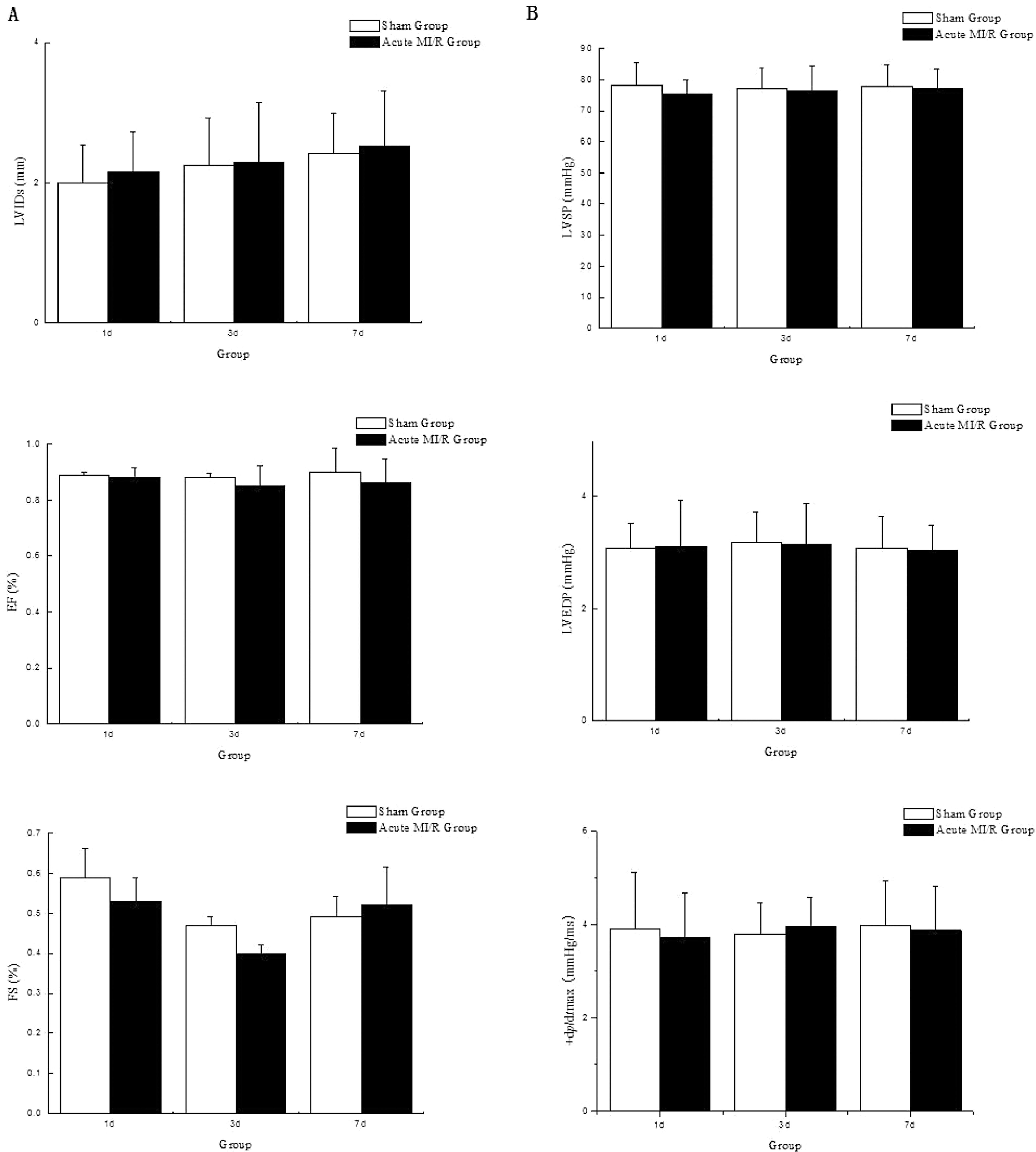
Cardiac systolic function in rats with acute MI/R. There was no significant difference about cardiac systolic function, respectively, by echocardiography and hemodynamic monitoring between the sham and the acute MI/R group on experimental days 1, 3, and 7 (P > .05, n = 6). A, Cardiac systolic function by echocardiography. B, Cardiac systolic function by hemodynamic monitoring. LVIDs indicates systole left ventricular internal dimension; EF, left ventricular ejection fraction; FS, left ventricular fractional shortening; LVSP, left ventricular systolic pressure; LVEDP, left ventricular end-diastolic pressure; +dp/dt max, the maximum change rate of left ventricular pressure rise; MI/R, myocardial ischemia/reperfusion.
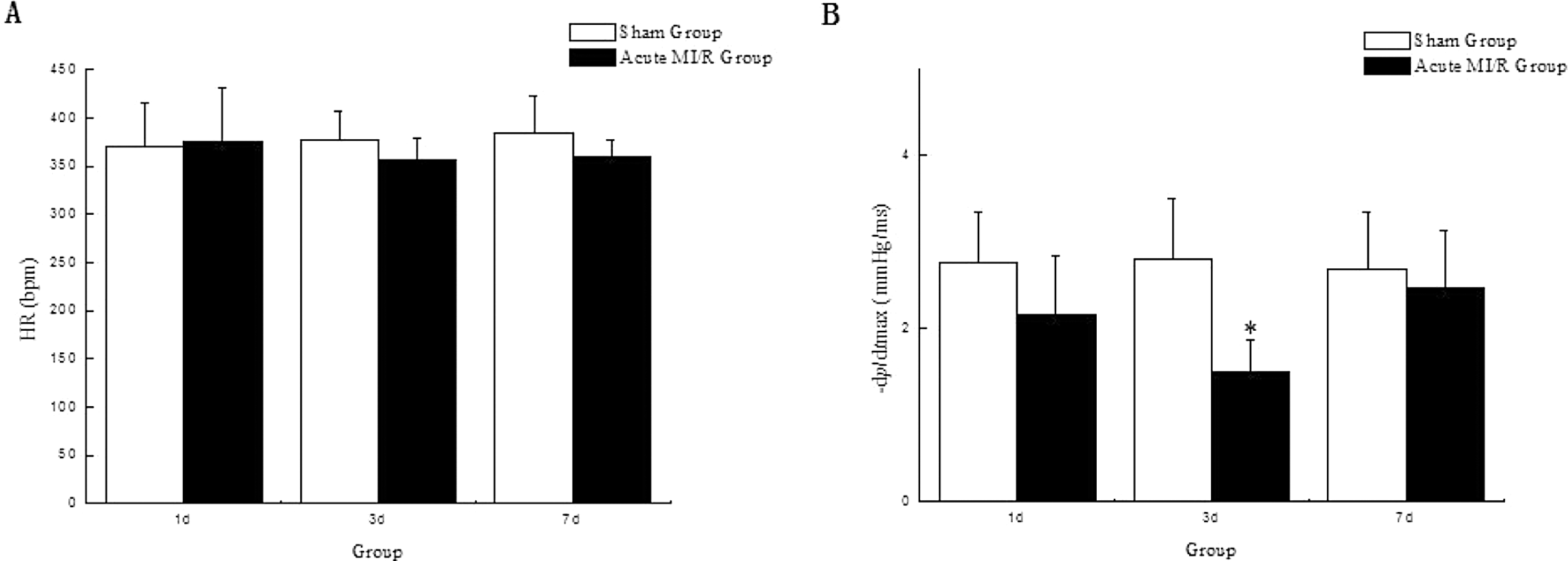
Cardiac diastolic dysfunction by hemodynamic monitoring in rats with acute MI/R. A, Heart rates of the sham group and the acute MI/R group. There was no significant difference between the 2 groups on experimental days 1, 3, and 7(P > .05, n = 6). B, −dp/dt max: the maximum change rate of left ventricular pressure fall. The −dp/dt max of the acute MI/R group on day 3 was significantly lower than that of the sham group on day 3 (*P < .05, n = 6). MI/R indicates myocardial ischemia/reperfusion.
No-Reflow Evaluation in Rats With Acute MI/R
We sought the reason for cardiac dysfunction by myocardial staining. By Evans blue staining, myocardial ischemic risk area appeared red in contrast with blue-staining nonischemic risk myocardium. By thioflavin S staining, no-reflow size appeared dark under ultraviolet rays, whereas fluorescence-staining area represented good blood supplied myocardium. Myocardial ischemic risk size was described by the percentage of myocardial ischemic size to the whole heart section size (AR/WH%). No-reflow size was described by the percentage of no-reflow size to the whole heart section size (AN/WH%). On days 1, 3, and 7 in the acute MI/R group, myocardial ischemic risk size was, respectively, calculated to be 22.63 ± 0.67(%), 22.64 ± 0.54(%), and 22.47 ± 1.12(%) and showed no significant difference (P > .05). The equal level of myocardial ischemic risk normalized certain reperfusion area at each time point in the acute MI/R group. Furthermore, by thioflavin S staining, no-reflow phenomenon in the reperfusion section was observed and evaluated. In the acute MI/R group on day 3, no-reflow size was markedly or significantly larger (19.30 ± 0.84(%) vs 16.62 ± 0.91(%) on day 1 in the acute MI/R group or 15.32 ± 0.81(%) on day 7 in the acute MI/R group, P < .05), which was in-line with cardiac diastolic dysfunction of rats with acute MI/R (Figure 3 ).
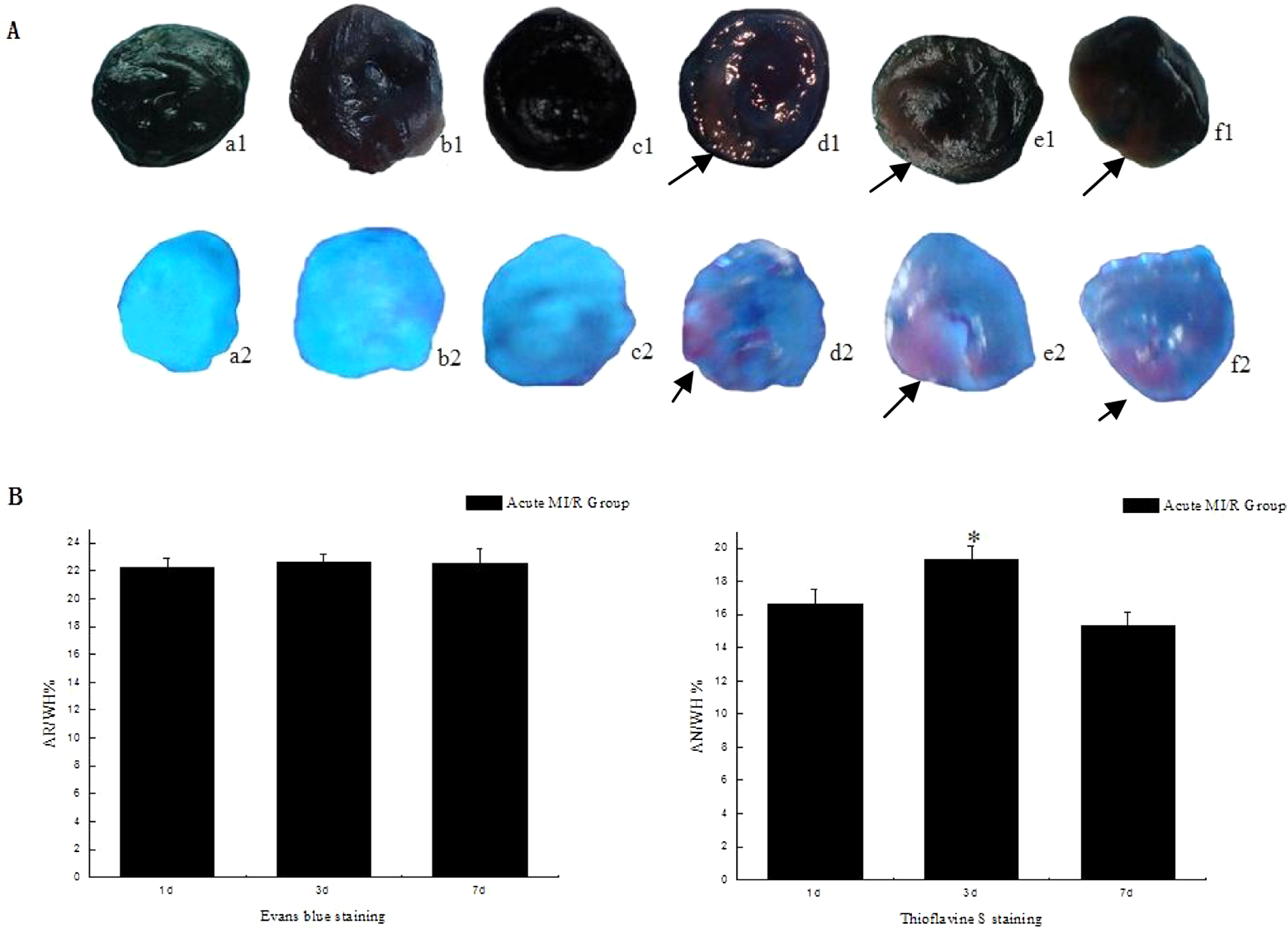
No-reflow evaluation in rats with acute MI/R. A, Myocardial ischemic risk area by Evans blue staining (a1–f1) and no-reflow attacked section by thioflavin S staining (a2–f2) between the sham (a1–c1 and a2–c2) and the acute MI/R group (d1–f1 and d2–f2) on experimental days 1 (a1, a2, d1, d2), 3 (b1, b2, e1, e2), and 7 (c1, c2, f1, f2). B, Percentage of myocardial ischemic risk size (area of risk [AR]/whole heart [WH]%) in the acute MI/R group on days 1, 3, and 7. There was no significant difference about AR/WH% on days 1, 3, and 7 in the acute MI/R group (P > .05, n = 6). C, Percentage of the no-reflow size (area of no-reflow [AN]/WH%) in the acute MI/R group on days 1, 3, and 7. On day 3, AN/WH% was significantly higher than that on days 1 or 7 in the acute MI/R group (*P < .05, n = 6).MI/R indicates myocardial ischemia/reperfusion.
Expression of Cardiac fgl2 in Rats With Acute MI/R
The expression of cardiac fgl2 protein was detected by immunohistochemistry and Western blot analysis. In contrast with almost no expression of fgl2 in the sham group, abundant expression of fgl2 was observed in the acute MI/R group and located at microvascular walls (Figure 5 ). On day 3 in the acute MI/R group, expression of fgl2 reached the peak (Figure 4 ). Moreover, by examining the ischemia–reperfusion or nonischemia/reperfusion section in myocardium of rats with acute MI/R, we found that there was the expression of fgl2 only in the ischemia–reperfusion section, which in the experiment was located at the anterior wall dominated by the LAD blood supply (Figure 5), whereas no expression of fgl2 was detected in the nonischemia/reperfusion section including the posterior wall and the right heart. The expression of fgl2 was closely correlated with no-reflow size at each time point in the acute MI/R group (r = .975, P < .05).
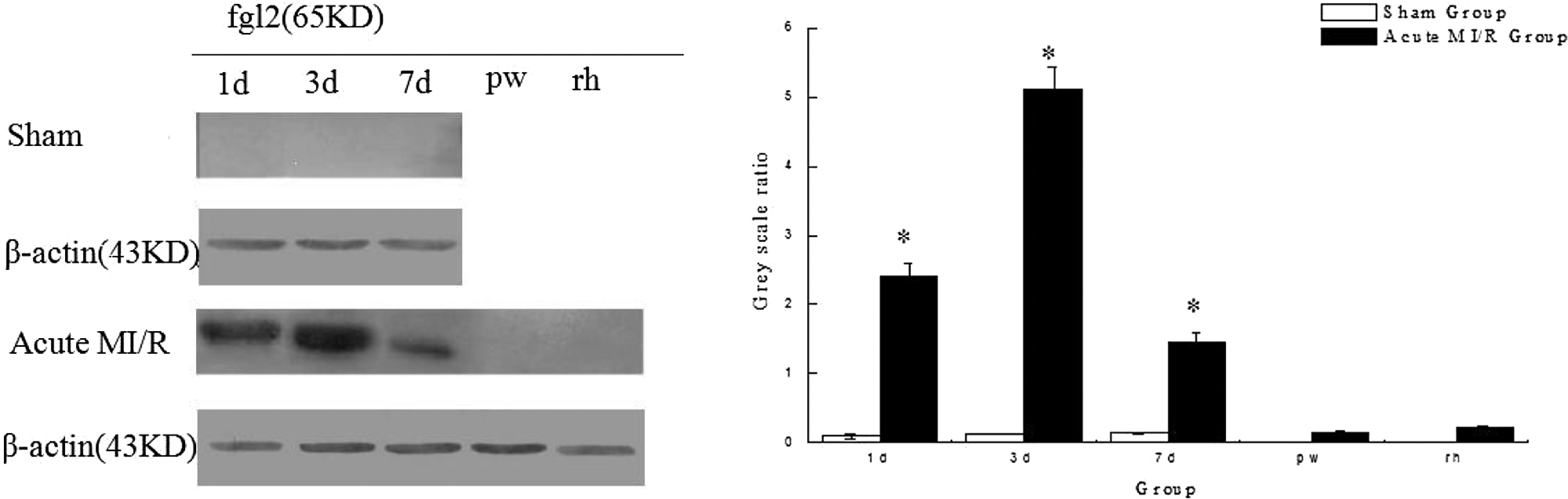
Cardiac expression of fgl2 in rats with acute MI/R. fgl2 was abundantly expressed in the acute MI/R group compared with the sham group on experimental days 1, 3, and 7 (*P < .05, n = 4). There was almost no expression of fgl2 in the posterior wall (pw) or right heart (rh; P > .05, n = 4).MI/R indicates myocardial ischemia/reperfusion.
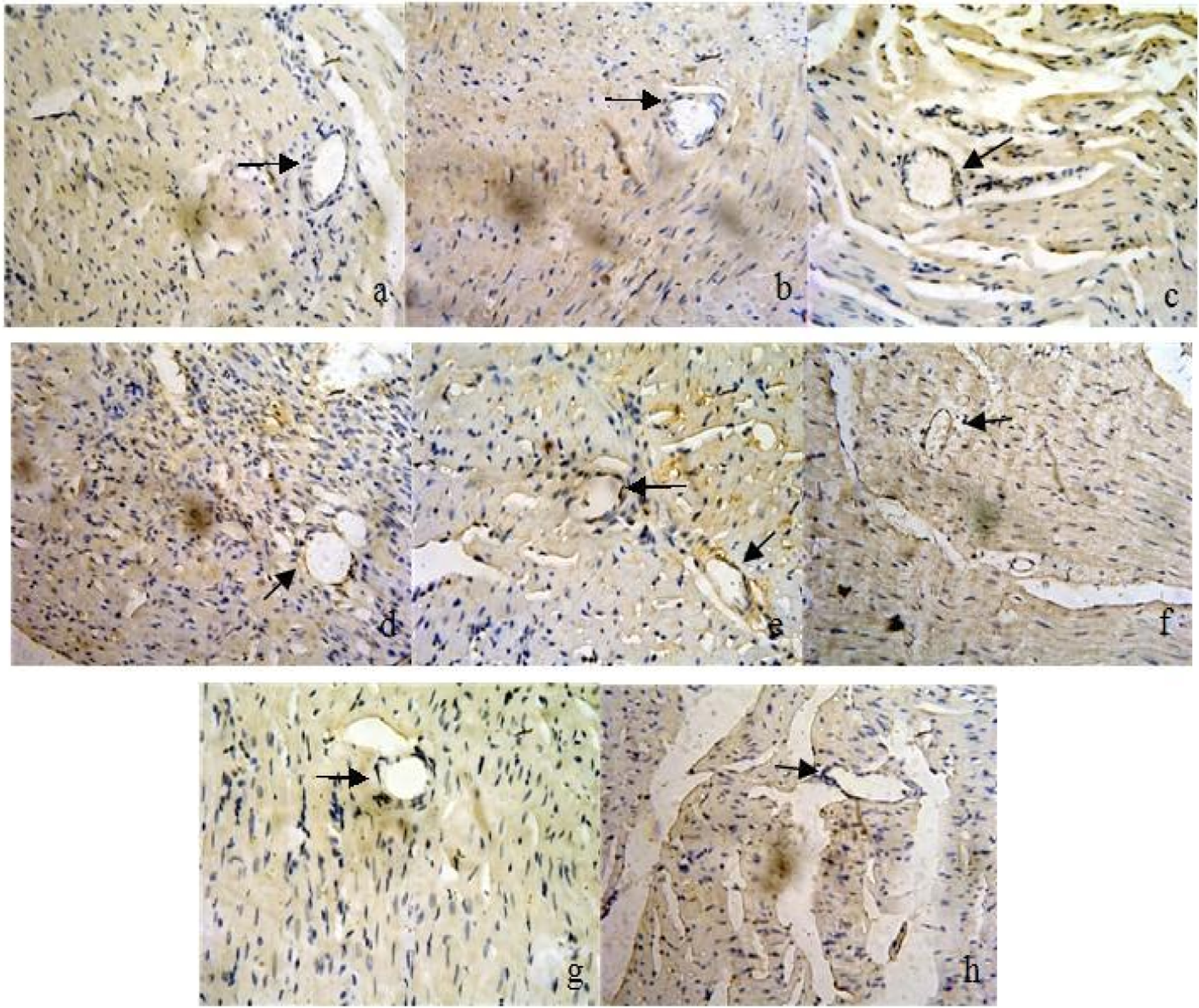
Cardiac expression of fgl2 in rats with acute MI/R by immunohistochemistry (×400). fgl2 was located at cardiac microvessels (diameter <150 μm) in the ischemia–reperfusion section on days 1, 3, and 7 (d–f), but absent in the nonischemia–reperfusion section including the posterior wall (g) and the right heart (h) in rats with acute MI/R on day 3 and the sham group on days 1, 3, and 7 (a–c). MI/R indicates myocardial ischemia/reperfusion.
Cardiac Microthrombosis in Rats With Acute MI/R
Cardiac microthrombosis was observed by Masson staining and MSB staining (Figure 6A). By Masson staining, microvessels or larger arteries could be clearly differentiated (Figure 6B). In cardiac microvessels of rats with acute MI/R, microthrombus-like substances were observed and further defined by MSB staining to be fibrinothrombi, whereas no cardiac microthrombosis was shown in the nonischemia–reperfusion section and the sham group.
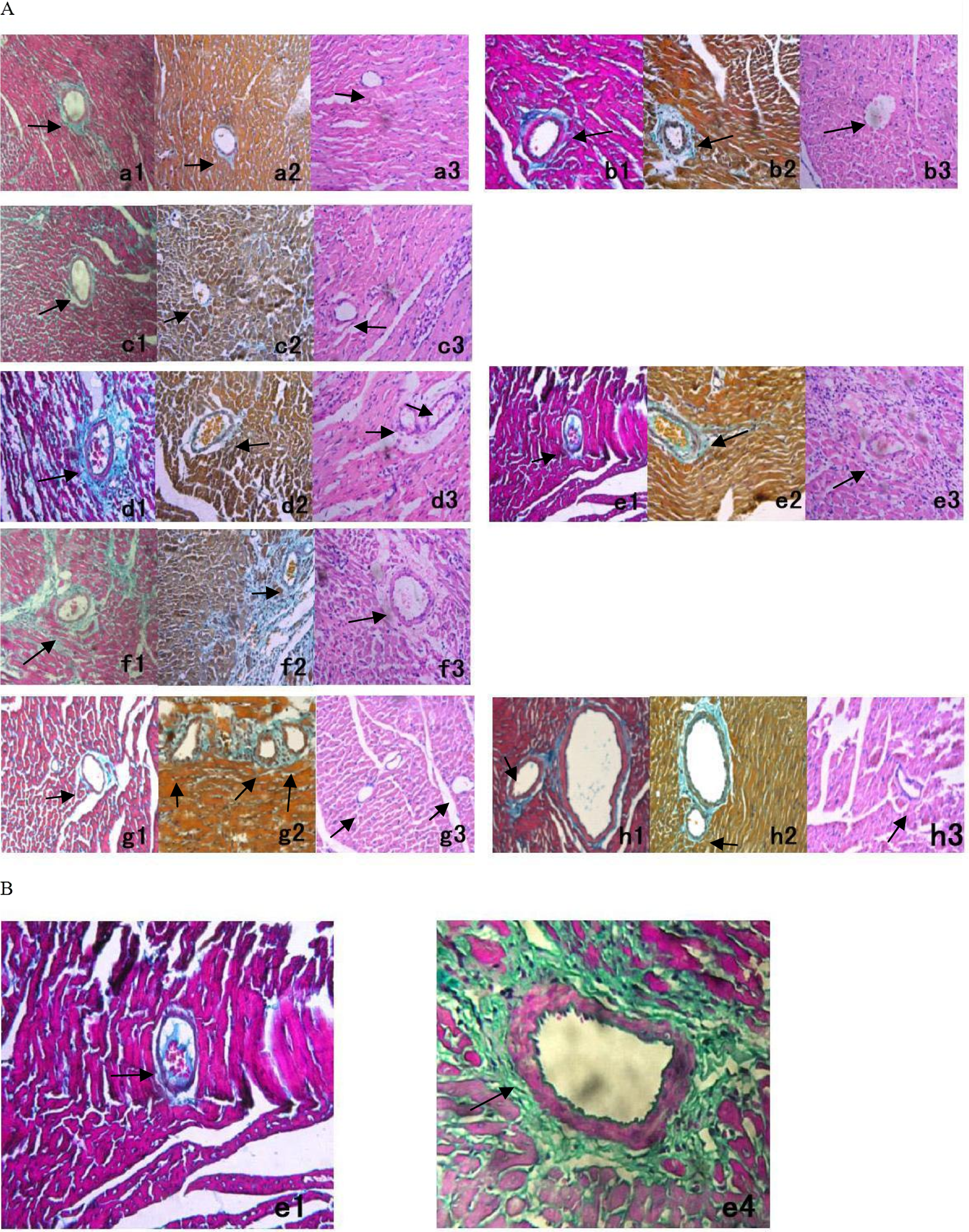
Cardiac microthrombosis in rats with acute MI/R. A, Cardiac microthrombosis was observed by Masson staining (a1–h1), Martius, Scarlet, Blue methocl (MSB) (a2–h2) and by hematoxylin and eosin staining (a3–h3; ×400). Cardiac microthrombi were shown at cardiac microvessels (diameter <150 μm) in the ischemia–reperfusion section on days 1, 3, and 7 (d1–f1, d2–f2, and d3–f3), but absent in the nonischemia–reperfusion section including the posterior wall (g1–g3) and the right heart (h1–h3), and in the sham group on days 1, 3, and 7 (a1–a3, b1–b3, and c1~c3). B, Masson staining (×400) indicated the difference between cardiac microvessels and larger arteries. Thrombi were clearly shown at microvessels (e1), but no thrombosis in a larger artery (e4) in rats with acute MI/R on day 3.MI/R indicates myocardial ischemia/reperfusion.
Slices of the ischemia–reperfusion section stained by HE indicated myocardial pathological alterations. There was myocardial structural disorder to some extent, whereas no cardiomyocyte hypertrophy on days 1, 3, and 7 in the acute MI/R group (Figure 6).
Expression of fgl2 in Mononuclear Cells in Rats With Acute MI/R
It was found that fgl2 was also present in mononuclear cells of rats with acute MI/R on days 1, 3, and 7, whereas absent in the sham group at the same time point. On days 1, 3, and 7, expression of fgl2 in mononuclear cells of rats with acute MI/R was, respectively, 46, 100, and 47 times as much as that in the sham group (Figure 7 ). The expression of fgl2 pattern in peripheral mononuclear cells varied in line with that in cardiac microcirculation (r = .900, P < .05).
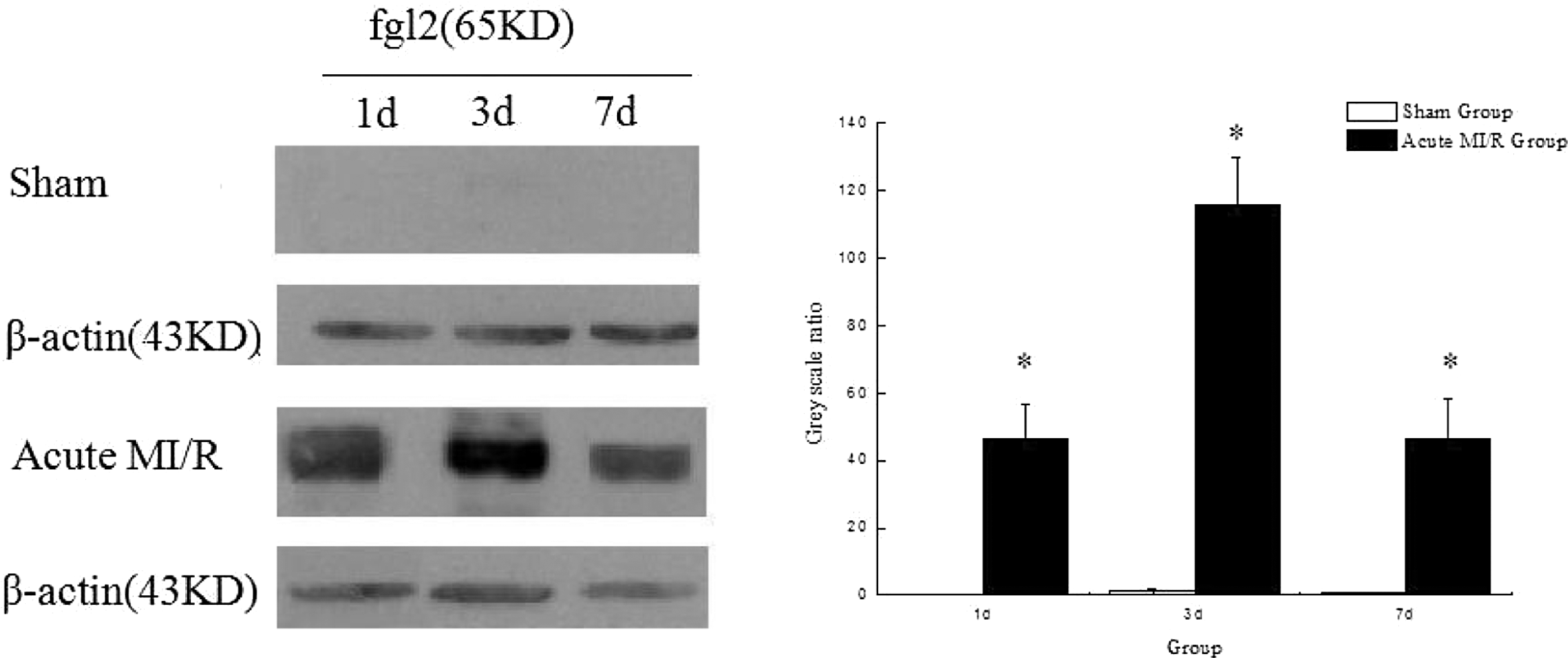
Expression of fgl2 in mononuclear cells of rats with acute MI/R. Level of fgl2 in peripheral mononuclear cells of rats with acute MI/R was markedly elevated on days 1, 3, and 7 (vs the sham group at the same time point, *P < .05, n = 4). MI/R indicates myocardial ischemia/reperfusion.
Discussion
The treatment of no-reflow phenomenon remains challenging, for its underlying mechanism that needs to be explored, although various theories have been proposed. In addition to myocardial interstitial edema, endothelial cell blebbing, neutrophil infiltration, myocardial stunning, and microvascular dysfunction or disturbance have been increasingly demonstrated to contribute greatly to the pathogenesis of no-reflow phenomenon.22–25 In rats with acute MI/R, we provide evidence that microthrombi in situ were present in cardiac microcirculation, which may explain the occurrence of no-reflow phenomenon as one cause.
The established acute MI/R animal model was ever used to evaluate cardiomyocyte damage. Thioflavin S is a fluorescent dye for endothelial cell with blood flow. Using thioflavin S staining, no-reflow phenomenon can be observed and no-reflow size is calculated in rats with acute MI/R. 11 At different time points of the experiment, no-reflow was detected more or less in rats with acute MI/R. It indicated that cardiac microvascular dysfunction or disturbance existing in rats with acute MI/R and no-reflow would be involved in the pathogenesis of cardiac reperfusion injuries. Paralleling with no-reflow size, only cardiac diastolic function of rats with acute MI/R was aggravated to some extent when compared with no alterations of cardiac systolic function, which is typical of early myocardial ischemia. On day 3, no-reflow size was significantly correlated with the impairment of cardiac diastolic function, which suggests that no-reflow would contribute greatly to myocardial injuries of rats with acute MI/R in an acute period. Given that there was no stenosis or obstruction in larger coronary vessels of rats with acute MI/R, myocardial ischemia was properly attributed to no-reflow due to microvascular dysfunction or disturbance as an upstream factor superior to cardiomyocyte damage.
By Masson trichrome staining, thrombus-like substances were detected in cardiac microvessels of no-reflow attacked sections. By contrast, no thrombi were detected in larger cardiac vessels. The microthrombi were further demonstrated including fibrin by the method of MSB staining. Therefore, microthrombi in situ were likely to be involved in the pathogenesis of no-reflow phenomenon.
In the experiment, microthrombi were not originated from larger coronary atherosclerotic or thrombotic debris because SD rats are resistant to atherosclerosis. By immunohistochemistry, fgl2 prothrombinase was shown to be expressed and located in cardiac microvascular endothelial cells of rats with acute MI/R. Microthrombosis related to fgl2 was demonstrated to cause ischemic injuries leading to severe microvascular dysfunction or disturbance. We compared expression of fgl2 and no-reflow size in situ in different heart sections of rats with acute MI/R. Microthrombi in situ and concomitant expression of fgl2 were observed exclusively in no-reflow attacked sections, which was located at the anterior wall of left ventricle, while not in non-no-reflow attacked sections, which included the posterior wall and the right ventricle. The coincidence between the expression of fgl2 and no-reflow size suggests its role in cardiac microvascular dysfunction or disturbance that is responsible for no-reflow phenomenon.
To date, some imaging and biochemical techniques have been applied in the diagnosis of no-reflow phenomenon with different levels of ability.26–28 There is still no proven, well-documented single biomarker. In the experiment, accompanied by the expression of fgl2 in cardiac microcirculation of rats with acute MI/R, expression of fgl2 was markedly elevated in peripheral mononuclear cells. The result suggests that it would be of great value in future to predict the occurrence of no-reflow phenomenon by analyzing the expression of fgl2 in peripheral mononuclear cells, which is more economical, convenient, and sensitive.
In conclusion, this study demonstrates that microthrombosis is involved in no-reflow phenomenon and fgl2 should be a potential predictor of the occurrence of no-reflow phenomenon.
Footnotes
Authors’ Note
L.S. and D.W. contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by National Key Basic Research Program of China (No. 2007CB512000 and Sub-Project No. 2007CB512005) and scientific and technological project of Wuhan city (No. 201161038340-08).
