Abstract
Objectives:
In the follow-up of patients in a trial of intracoronary sodium nitrite given during primary percutaneous coronary intervention (PCI) after acute myocardial infarction (AMI), we found a reduction in the incidence of major adverse cardiac events (MACEs). Specifically, MACE rates were 5.2% versus 25.0% with placebo at 3 years (P = .013). Such MACE reductions should also be associated with economic benefit. Thus, we assessed the cost utility of sodium nitrite therapy versus standard primary PCI only.
Methods and Results:
We developed a model to simulate costs and quality-adjusted life years (QALYs) over the first 36 months after ST-Segment Elevation Myocardial Infarction (STEMI). Decision tree analysis was used to assess different potential cardiovascular outcomes after STEMI for patients in both treatment groups. Model inputs were derived from the NITRITE-AMI study. Cost of comparative treatments and follow-up in relation to cardiovascular events was calculated from the United Kingdom National Health Service perspective. Higher procedural costs for nitrite treatment were offset by lower costs for repeat revascularization, myocardial infarction, and hospitalization for heart failure compared to primary PCI plus placebo. Nitrite treatment was associated with higher utility values (0.91 ± 0.19 vs 0.82 ± 0.30, P = .041). The calculated incremental cost-effectiveness ratio of £2177 per QALY indicates a cost-effective strategy. Furthermore, positive results were maintained when input parameters varied, indicating the robustness of our model. In fact, based on the difference in utility values, the cost of nitrite could increase by 4-fold (£2006 per vial) and remain cost-effective.
Conclusion:
This first analysis of sodium nitrite as a cardioprotective treatment demonstrates cost-effectiveness. Although more comparative analysis and assessment of longer follow-up times are required, our data indicate the considerable potential of nitrite-mediated cardioprotection.
Introduction
Acute myocardial infarction (AMI) remains a major cause of mortality and morbidity, despite the introduction of early reperfusion strategies such as primary percutaneous coronary intervention (PCI). As such, new treatments are required to protect the heart against acute ischemia/reperfusion (I/R) injury in order to preserve cardiac function and prevent heart failure (HF)—a strategy termed “cardioprotection.” Agents reducing I/R injury should, by decreasing long-term morbidity and mortality, produce significant economic benefit. However, few studies have assessed the cost-effectiveness of cardioprotective agents, mainly due to the limited success of translation of therapies from experimental to clinical application. In a recent phase II clinical trial, sodium nitrite given intracoronary at the time of primary PCI was associated with significant reductions in major adverse cardiac events (MACEs) during 3 years of follow-up (5.2% vs 25.0%, P = .013). 1,2 The extent of economic benefit associated with such MACE reductions is, as yet, unassessed. Therefore, we aimed to evaluate the cost–utility of sodium nitrite as an adjunctive therapy administered prior to primary PCI in patients with ST-segment elevation myocardial infarction (STEMI). Our analysis was conducted from the perspective of the United Kingdom healthcare system.
Methods
Source Trial
The patients included in this economic analysis were participants in a single-center randomized, controlled trial conducted by the Department of Cardiology, Barts Health NHS Trust & Queen Mary University of London, London, United Kingdom. NITRITE-AMI was a double-blind, randomized, single-center, placebo-controlled trial to determine whether the intracoronary injection of sodium nitrite reduces infarct size in patients with acute STEMI undergoing primary PCI. The trial was approved by an independent ethics committee, the Medicines and Healthcare Products Regulatory Agency, registered in approved registries (NCT01584453, EudraCT nr. 2011-000721-77) and was performed in accordance with the Declaration of Helsinki (1996) and the principles of the International Conference on Harmonization Good Clinical Practice guidelines. Full details of the trial protocol and primary and secondary outcomes have been published. 1 –3 All participants gave written informed consent before being included in the study After coronary angiography, patients were randomized (1:1) to a high-dose bolus injection of intracoronary sodium nitrite (1.8 μmol in 10 mL of 0.9% NaCl) or placebo (10 mL of 0.9% NaCl) administered via an over-the-wire balloon catheter just prior to balloon inflation. All study personnel were blind to treatment allocation until the study and all analyses were completed.
Economic Evaluation
This study is a trial-based cost–utility study comparing the use of a novel cardioprotective agent (sodium nitrite) versus standard treatment from a health-care system perspective. Each parameter such as procedural cost, death, unplanned revascularization, and hospital stay was considered an outcome of interest. We assessed costs using a top-down approach based on the United Kingdom NHS model of care. We developed decision-based model outcomes and probability estimates for treatment options based on the clinical trial outcomes measured 3 years after the initial STEMI.
The primary economic outcome measure was the cost–utility of sodium nitrite after 3 years of follow-up, calculated as the incremental cost-effectiveness ratio (ICER) for nitrite treatment versus standard primary PCI only. This was generated from the difference in medical care costs, quality-of-life (QoL) scores, and MACE-free survival between groups. The economic evaluation was registered with ClinicalTrials.gov, number: NCT01584453.
Decision Tree Model
We developed the decision tree model potential outcomes for STEMI patients in both groups (Figure 1). We employed an intention-to-treat approach. Post discharge states are limited to those relevant to cardiac complications. We assumed no intergroup differences in noncardiac outcomes. We calculated risk ratios for all-cause mortality, combined MACE, nonfatal myocardial infarction (MI), or the need for revascularization and hospitalization for HF (Table 1), based on data obtained from the NITRITE-AMI trial. These data were used to calculate probability estimates for each outcome in the decision tree (Figure 1).
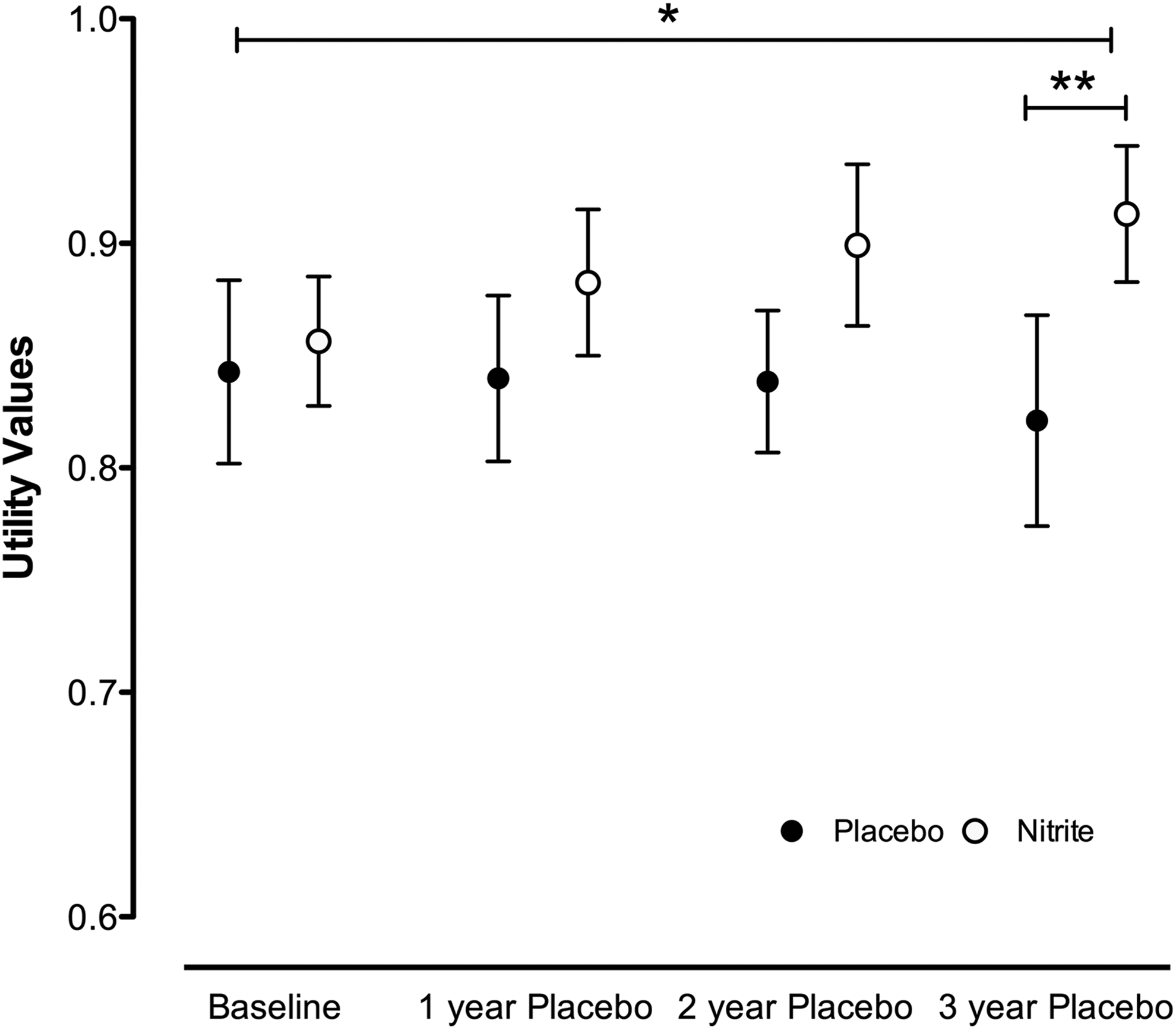
The QALY outcomes measured over time. The EuroQol 5-dimension questionnaire was measured at baseline (post-PCI), 1, 2, and 3 years comparing nitrite-treated versus control is displayed. Data are expressed as mean ± SEM. For comparisons between treatments, statistical significance is shown as * for P < .05 performed using 2-way Analysis of variance (ANOVA) and **P < .01 for Bonferroni posttest comparing specific time points between groups. PCI indicates percutaneous coronary intervention; QALYs, quality-adjusted life years.
Input Parameters and Utility Values Applied in the Model.a

Abbreviations: CI, confidence interval; HF, heart failure; MI, myocardial infarction; PCI, percutaneous coronary intervention.
Utility Values
In the trial, 3 the EuroQol 5-dimension questionnaire (EQ-5D) was completed by patients. 4 The EQ-5D is a generic health state preference measure. 5 Data were collected for each arm at baseline, 12, 24, and 36 months. We used patient-level EQ-5D data from the trial and generated utility scores for both groups at every follow-up point. The health state preference values (utilities) for EQ-5D profiles were based on time trade-off valuations by members of the United Kingdom general public. 5 Mean imputation was used to manage missing data. As this was a single-center trial, we also performed additional analysis to check/validate trial-reported utility scores using typical utility values from previous studies after primary PCI (repeat MI 0.700, repeat revascularization 0.700, event-free follow-up 0.850, angina symptoms 0.683, and death 0; Table 1). 6,7
Costs
Costs were based on the 2015/2016 NHS England Tariff (Enhanced Tariff Option) and on market research data for current device prices of drug eluting stents, guide catheters, and angioplasty balloons 8 (Table 2). Costs for death were based on a mean value for cardiac arrest with a range from 0 to £4895 for complex cases. Costs for repeat MI and revascularization were taken from standard admissions with ranges incorporating simple to complex cases. The extra costs of the sodium nitrite comprised the cost of the delivery balloon Over the Wire Balloon (OTW) and the cost of the nitrite therapy. Nitrite therapy costs £14.98 per vial (4350 μmol in 10 mL), however, not at the therapeutic dose required for cytoprotection (1.8 μmol in in 10 mL). Therefore, we estimated £500 for generation of a stable deliverable drug. The extra costs of the sodium nitrite comprised the cost of the delivery balloon (OTW) and the cost of the nitrite therapy, meaning a total per patient cost of £650 allowed for the combined cost of drug and delivery.
Cost Data Applied in the Model.
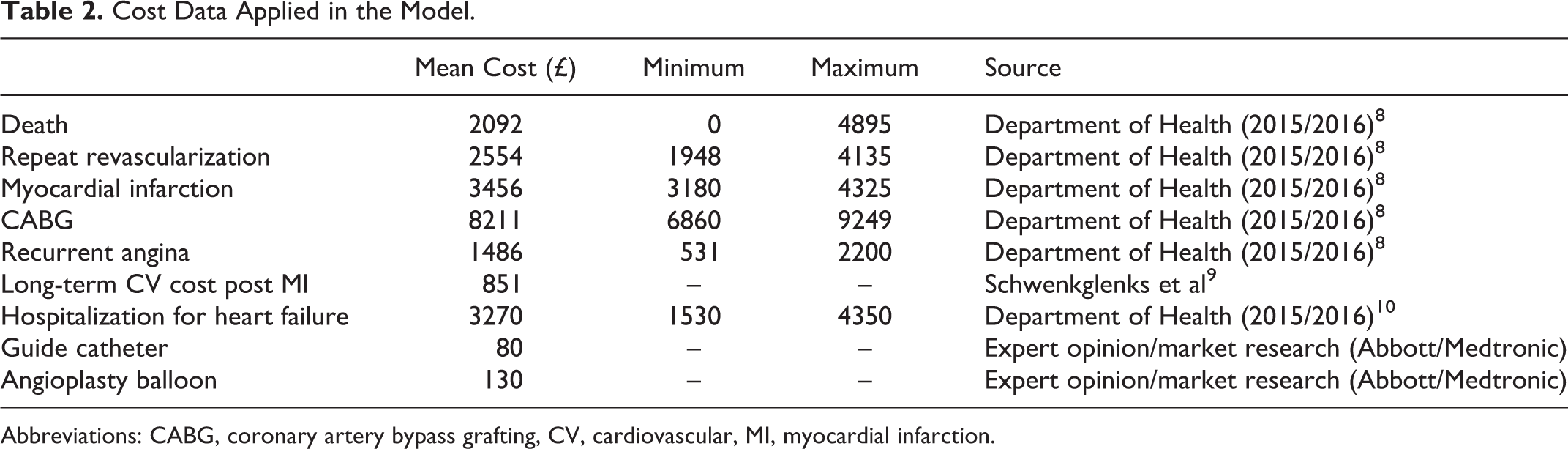
Abbreviations: CABG, coronary artery bypass grafting, CV, cardiovascular, MI, myocardial infarction.
Cost-Effectiveness
Using the methods identified above, total costs and quality-adjusted life years (QALYs) figures were calculated for all patients. For cost-effectiveness analysis, we identified the differences between costs and QALYs for both groups and divided the former by the latter to compute an ICER. When compared against the marginal trade-off for the NHS as a whole—the cost-effectiveness threshold—the ICER provides a broad indication of whether or not nitrite therapy is an efficient use of resources.
Hospital-Free Days
Hospital-free days within 3 years of PCI were calculated by subtracting hospital length of stay (LOS), readmission days, and days spent in rehabilitation and nursing facilities from 1095 (3 × 365).
Statistics and Sensitivity Analysis
Descriptive statistics were reported as mean and standard deviation for normally distributed continuous variables or median and interquartile range for non-normal distributions; normality was tested by Kolmogorov-Smirnov tests. Differences between means were compared using t tests. A Mann-Whitney nonparametric test was used to compare non-Gaussian variables. Differences in proportions were compared using χ2 tests. For QoL data, comparisons between nitrite and placebo-treated patients were performed using Analysis of Variance (ANOVA). Post hoc tests were run only if F achieved P < .05 and there was no significant variance inhomogeneity assessed using GraphPad Prism version 5.0 (GraphPad Software, San Diego, CA, USA). Bonferroni posttests were conducted for comparisons between groups at specific time points within treatment groups. One-way sensitivity analysis was conducted to explore the impact of model inputs and assumptions on the results. The parameters varied in the sensitivity analysis; they included the outcomes based on 99% confidence interval (CI), time horizon, cost of nitrite, QoL ranges, and the costs associated with MACEs. A tornado diagram was used to depict these data.
Results
The patient flowchart is presented in Figure 2. A total of 82 patients with STEMI undergoing primary PCI were randomized to receive intracoronary nitrite therapy as adjunct to primary PCI or placebo (control group). Two patients were subsequently excluded from analyses because they met exclusion criteria. Clinical characteristics, medication use, and procedural data were similar in the 2 randomly assigned groups except for ischemia time, which was significantly longer in nitrite-treated patients (207.1 ± 76.3 minutes vs 171.6 ± 67·7 minutes, P = .03). 1

Tornado diagram. Univariate sensitivity analyses presented as a tornado diagram. The x-axis represents the ICER for nitrite treatment versus control. The diagram is stacked in order of decreasing width, whereby variations in inputs near the top have the greatest impact on the ICER. The black line reflects the base case of £2178, and the dotted black line represents the conventional The National Institute for Health and Care Excellence (NICE) threshold for cost-effectiveness. MACE assumptions reflect variation in the 99% CI of MACE events, QALY (trial); the 99% CI of trial-treated QALY data, MACE cost; variation in cost of MACE events, nitrite cost; variation in the cost of nitrite and QALY literature; ICER calculated from literature data and not trial. CI indicates confidence interval; ICER, incremental cost-effectiveness ratio; MACE, major adverse cardiac events; QALY, quality-adjusted life years.
Procedural Costs
Nitrite treatment was characterized by higher initial procedural costs versus primary PCI only because of greater resource use, an average of £500 per patient. We found no difference in LOS or increase in procedure duration between groups.
Hospital-Free Days
Over the 3-year follow-up, nitrite-treated patients had fewer hospital admissions (5 vs 15, P = .018) and spent less time in hospital (median: 0.2 [2-3] vs 5 [1-7] days, P < .0001) versus placebo-treated patients. Consequently, hospital-free days (HFDs) were higher in the nitrite-treated patients over the follow-up period (1090 [1088-1092] compared to the placebo-treated group 1093 [1092-1093], P < .0001).
Quality of Life Values
Utility scores at baseline were complete for 80 patients. Two patients died during the study; therefore, a utility value of 0 was assigned. We used imputed utility values for 6 patients with incomplete data at 36 months. Quality-adjusted life years were available for 80 patients, 40 in each group. No differences in utility values were seen at baseline. However, significant differences were seen over the follow-up period (Figure 1) between the nitrite-treated and primary PCI-only groups (P = .035), with the greatest difference seen at 36 months (0.92 ± 0.19 vs 0.82 ± 0.30).
Incremental Cost-Effectiveness Ratio
Over the 3-year follow-up period, MACE was lower in the nitrite group than the control group, yielding a hazard ratio of 0.25 (95% CI, 0.13-0.74; P = .013). A more detailed presentation of the MACE results has been previously published. 2 While nitrite therapy is expected to cost more following primary PCI, it appears to provide a difference in QoL. Here, the incremental ICER for nitrite is £2177 per QALY (£196/0.09 QALY) (Table 3).
Total Costs, Health Effects, and Incremental Cost-Effectiveness Ratio.

Abbreviations: ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life years.
Sensitivity Analysis
Sensitivity analysis on change in MACE probabilities (95% CI) and varying costs (mean, minimum, and maximum) was also carried out, and it yielded results with ICERs ranging from £833 to 18 173. The largest variation was obtained by varying the MACE assumptions after the end of follow-up. However, we found no circumstances in which these variations were large enough to change the conclusions, that is, nitrite therapy always appeared to be cost-effective. This was further corroborated by additional analysis validating the trial-reported utility scores using typical utility values obtained from previous studies (listed in Methods) in the literature after primary PCI. These studies gave improved QALY with nitrite treatment (0.8422 compared to 0.7628 = difference of 0.079) and an ICER of £2481. Assuming a QoL improvement with nitrite treatment of 0.09 QALY, as found in the NITRITE-AMI trial, nitrite treatment could cost up to £2000 per patient and remain cost-effective. Our analysis implies that whatever the costs of care are in any given jurisdiction for these subsets of patients, the ICER will be in favor of cost-effectiveness when there is a reduction in MACE rates with nitrite therapy.
Discussion
We found that the use of adjunctive intracoronary sodium nitrite treatment delivered at the time of primary PCI was cost-effective when compared to current standard treatment. In addition, the calculated ICER was well below the threshold required by regulatory and decision-making authorities. This conclusion was robust to sensitivity analyses with all values below the key threshold. Considering the competitive market and ongoing pressure for cost savings, due to the economic challenges faced by the NHS, our analysis shows that sodium nitrite treatment as an adjunct to primary PCI could provide opportunities for considerable savings.
Comparison With Published Studies
This is the first cost-effectiveness analysis of sodium nitrite treatment as an adjunct to primary PCI in patients with STEMI, and consequently, no comparative studies are available. In fact, there are very little data on potential cost benefits of any cardioprotective agent or therapy because results from clinical trials have been disappointing. One study that assessed the cost-effectiveness of remote ischemic conditioning (performed on CONDI study participants) suggested reduction in medical costs during follow-up for those who received remote conditioning (€12 065 vs €14 828) versus patients who did not, which was consistent with cost-effectiveness. 11 Although promising and providing some evidence of benefit, QoL data were not recorded and no ICER was calculated. A hypothetical analysis determined that any cardioprotective agent that reduces the risk of HF and mortality after PCI is likely to be cost-effective; however, the likelihood will depend on the price of the agent, age of the study cohort, and the relative risk of HF after PCI. 12 To be cost-effective, the authors calculated that the maximum cost of the therapy should be less than £2500 and the reduction in HF incidence and mortality should be at least 20%. 12 These values are consistent with our data. Specifically, we showed that if MACE reduction found in our study with sodium nitrite is replicated in a larger study, then the therapy could cost up to £2006 and remain cost-effective.
In the United Kingdom, the annual economic burden resulting from AMI to the NHS is substantial and estimated to be £125 to £181 million, with a further £27 million for nursing home costs. 13 Cost-effectiveness studies of potential cardioprotective strategies are important because they can influence the coverage, reimbursement, and use of many health technologies. Quality-adjusted life years are a widely used metric in these studies that capture the morbidity and mortality gains of an intervention. The metric allows choices between treatments to be based on quantitative evaluation, that is, how much interventions prolong lives and improve patient health. Therefore, cost-effectiveness studies that incorporate QALYs provide a useful indication of whether new medical interventions represent good value for money. Our analysis is based upon up-to-date outcome data from a randomized controlled control (gold standard) with current costs relevant to the modern-day NHS. We suggest that cost-effectiveness analyses should be incorporated into all trials investigating potential cardioprotective therapies. This is particularly important in large studies where the effect size is smaller than expected, but perhaps the cost-effectiveness may still deliver economic benefits.
Hospital-Free Days
Hospital-free days are a novel, patient-centered, composite end point that quantifies the true amount of time patients spend away from home in health-care facilities. There is growing acceptance that outcome measures used in clinical trials should be determined in partnership by patients and physician-researchers and aim to identify outcomes important to patients. 14 Length of hospital stay is often used when therapies are evaluated and compared. However, LOS fails to reflect the positive value of time not spent in hospital. Hospital-free days have been shown to be a readily quantifiable and patient-centered outcome measure in some chronic cardiovascular conditions such as HF 15 and atrial fibrillation. 16 The use of HFDs was recently recommended by a consensus group because it is a composite measure that provides additional data on the outcomes of survivors compared to traditional LOS measures, which can be heavily affected by mortality. 17 Our use of this measure further supports the improved QoL reported in the sodium nitrite–treated patients and provides additional evidence of its cost-effectiveness.
Strengths and Limitations
Our analysis is based on a clinical trial, and therefore, issues concerning interpretation of the study results also apply to the economic analysis. One limitation of the model is the possibility that the study cohort is not representative of the general population of patients with STEMI undergoing primary PCI; more than 400 patients were screened to enroll 82 patients. Indirect costs were not included in the analysis. The inclusion or exclusion of such costs, which may arise during life-years gained because of the treatment, but are unrelated to the treatment, is controversial. 10 Indirect costs can have a substantial effect on the ICER, especially in cardiovascular disease. For example, prevention of MI-associated mortality may lead to later costs because unrelated disease, such as cancer, subsequently occurs. 18 Nevertheless, in our study, there was no mortality advantage with nitrite treatment; because of the low mortality rates after standard primary PCI, it is unlikely any new therapy would do so.
Conclusion
This first analysis of sodium nitrite as a cardioprotective treatment demonstrates cost-effectiveness. Although more comparative analysis and additional follow-up are required, our data indicate the considerable potential of nitrite-mediated cardioprotection. This could have important implications for health-care systems beyond the United Kingdom and for policy makers in the current difficult financial climate.
Footnotes
Authors’ Note
Dr D Jones, K Rathod, A Mathur and A Ahluwalia is also affiliated to entre of Clinical Pharmacology, William Harvey Research Institute, Barts & The London Medical School, Queen Mary University of London, United Kingdom.
Author Contributions
Daniel A. Jones contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Krishnaraj S. Rathod contributed to analysis and interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Peter Whittaker contributed to acquisition, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Amy J. Richards contributed to acquisition, contributed to analysis and interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Mervyn Andiapen contributed to acquisition, contributed to analysis and interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Sotiris Antoniou contributed to conception and design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Anthony Mathur contributed to conception and design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Amrita Ahluwalia contributed to conception, contributed to interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute of Health Research (NIHR)
