Abstract
The blood platelet proteome of hemodialyzed patients with uremia exhibits significant difference in comparison with the blood platelet proteome of healthy individuals. This alteration is manifested by the presence of high concentrations of low-molecular peptides within the whole range of isoelectric points. Increased platelet apoptosis has been put forward as a possible cause of this phenomenon. The aim of the present research was to assess whether blood platelet populations from hemodialyzed patients with uremia exhibit higher binding capacity of Annexin V than control samples from healthy donors. It was found that blood platelets of hemodialyzed patients, in the period between dialyses, bound significantly more Annexin V with no different incorporation of propidium iodide in comparison with platelets of control donors and conservatively treated patients with uremia. The results support the hypothesis that the process of hemodialysis may be at least partially responsible for triggering blood platelet apoptosis and result in increased risk of thrombosis.
Introduction
Despite the significant progress in the treatment of patients with renal failure, there is still a high risk of bleeding during surgery and other invasive procedures. Bleeding in uremia originates from structural and biochemical abnormalities of uremic platelets1–3 as well as from the effect of toxins present in uremic plasma on blood platelets.4,5 Hemodialysis, combined with recombinant human erythropoietin (EPO) administration, is a common treatment during the end-stage renal disease (ESRD) and during the period awaiting renal transplantation. 6 Although hemodialysis normalizes some biochemical parameters of blood and prolongs the patient’s life, the blood platelets remain less reactive in the period between dialyses 7 and the risk of bleeding is still relatively high. Recently, preliminary results of blood platelet proteome analysis in uremic patients, treated both pharmacologically and with hemodialysis, have been described. 8 It was reported that blood platelet proteome of hemodialyzed uremic patients exhibits significant difference in comparison with the blood platelet proteome of healthy individuals. In discussion, it was pointed that blood platelets of hemodialyzed patients, due to the contact with artificial surfaces, can be committed into apoptosis-like process. Indeed, signs of apoptosis, that is, activation of caspases and increased expression of phosphatidylserine (PS) at the outer surface of blood platelets of hemodialyzed patients, have been reported. 9 For proper interpretation of results from flow cytometry experiments, it is necessary to distinguish apoptotic cells from necrotic cells because necrotic cells also expose PS. 10 This can be done by the use of propidium iodide (PI) for necrotic cells staining. 11 The aim of the present research was to assess whether blood platelet populations from hemodialyzed patients with uremia exhibit greater binding of Annexin V to surface-exposed PS, corrected by PI incorporation, than platelet populations of conservatively treated patients and control healthy donors.
Materials and Methods
Twelve stable ESRD patients included in the study (7 men, 5 women; aged 58 ± 16 years, range 22-82 years) were treated with hemodialysis for approximately 4 hours (range 4-4.5 hours) 3 times a week (mean time on dialysis is 33 months, range 3-82 months). Adequacy of hemodialysis measured by Kt/v ratio in all the patients was ≥1.2 (1.225 ± 0.060). All the patients were treated with low-flux dialysis with polysulfone dialyzer membrane, where effective surface area was 1.5 to 2.1 m 2 . All hemodialyzed patients were treated with EPO. The etiology of ESRD in this group of patients was as follows: hypertensive nephropathy (n = 1), chronic glomerulonephritis (n = 5), and unknown etiology (n = 6). Thirteen stable ESRD nondialyzed patients were also included in the study (5 men, 8 women; aged 64 ± 8 years, range 53-79 years). No EPO was used in this group. The etiology of ESRD in conservatively treated patients was hypertensive nephropathy (n = 4), chronic glomerulonephritis (n = 4), polycystic kidney disease (n = 1), and unknown etiology (n = 4). In this study, the nondialyzed patients were included in the fourth stage of ESRD (estimated glomerular filtration rate [eGFR] 15-29 mL/min/1.73 m 2 ); mean eGFR 23.4 range 15.1-28.8 mL/min/1.73 m 2 ). Diabetic patients were excluded as diabetes may alter platelet intracellular mechanisms. Other exclusion criteria included acute infection or blood transfusion in the past 3 months, unstable clinical conditions including vascular and cardiac instability, a positive history for First Use syndrome, unstabilized EPO dosage, a history of stroke, myocardial infarction, thromboembolic complications, and a history of malignancy. None of the patients were known to have a preexisting hemostatic disorder unrelated to uremia and all had been free of medications known to affect platelet function for at least 3 weeks prior to the study. Twelve age- and gender-matched healthy individuals (serum creatinine <1.5 mg/dL) with normal platelet count were included as normal control individuals.
The study was accepted by the local Bioethical Committee (RNN/160/03/KE of August 23, 2003), and informed consent was obtained from all the patients and controls.
Blood was obtained from hemodialyzed patients with uremia immediately before hemodialysis. At the same time, blood samples from nondialyzed patients with uremia and control healthy donors were also collected. Blood was anticoagulated with sodium citrate and was immediately exposed to fluorescent agents.
Fluorescein-labeled Annexin V and PI were from Roche (Mannheim, Germany). Annexin V, a member of annexin group of cellular proteins, specifically bounds PS, and therefore is useful for surface staining of apoptotic cells,12,13 whereas PI is a cell membrane-impermeant fluorescent agent commonly used for staining of dead cells. 14
PerCP-CD61, PE-CD62, and FITC-PAC-1 antibodies were delivered by BectonDickinson (Becton Dickinson Polska, Warsaw, Poland). PerCP-labeled antibody against CD61 antigen (PerCP-CD61) recognizes platelet integrin β-3 (platelet glycoprotein IIIa [GPIIIa]) and is commonly used in flow cytometry for blood platelets gating. PE-labeled antibody against CD62 antigen (PE-CD62) specifically bounds P-selectin (CD62 antigen), a cell adhesion molecule present on surface of activated blood platelets and endothelial cells. Normally this molecule is stored in intracellular granules of non-activated cells and therefore can serve as an activation marker. The FITC-labeled PAC-1 antibody (FITC-PAC-1) recognizes an epitope on the GPIIb/IIIa (GPαIIbβ3) complex of activated platelets at or near the platelet fibrinogen receptor.
The samples were incubated for 10 minutes in the dark and after that the labeled samples were processed in a BectonDickinson FACScan flow cytofluorimeter. PerCP-labeled CD61 antibody was used for the selection of blood platelet population from the whole blood. The fluorescein–Annexin V probe was used for the detection of blood platelet subpopulation having expressed PS on the surface. Since necrotic cells also expose PS due to the loss of membrane integrity, apoptotic cells had to be differentiated from necrotic cells by secondary labeling used for dead cells identification. For this purpose, PI probe has been selected. Both PE-CD62 and FITC-PAC-1 antibodies were used for the estimation of the level of blood platelet activation. Measurements followed the published protocols. 15 The data were collected for 20 000 platelets for every sample.
Statistical Evaluation
The data are presented as the mean value accompanied by standard deviation. Analysis of variance test with Bonferroni P value correction was used for statistical analysis. P < .05 was considered as significant.
Results
Our interest was focused on binding of Annexin V to blood platelets. This phenomenon reflects cell apoptosis but is also present in necrotic cells. Additional staining with PI was necessary to distinguish apoptotic cells from necrotic cells. Binding of PI to DNA of necrotic cells was found to be lowest for platelets of control donors, although no statistical relevance was obtained. Annexin V binding to platelets exposing PS was highest for hemodialyzed patients with uremia, and the differences observed between the examined groups were significant. Taking away the part of PI-positive cells, the difference becomes very significant (Table 1). No significant difference was found between both nondialyzed and control blood platelets.
Binding of Annexin V and PI to Blood Platelets in the Citrated Whole Blood.
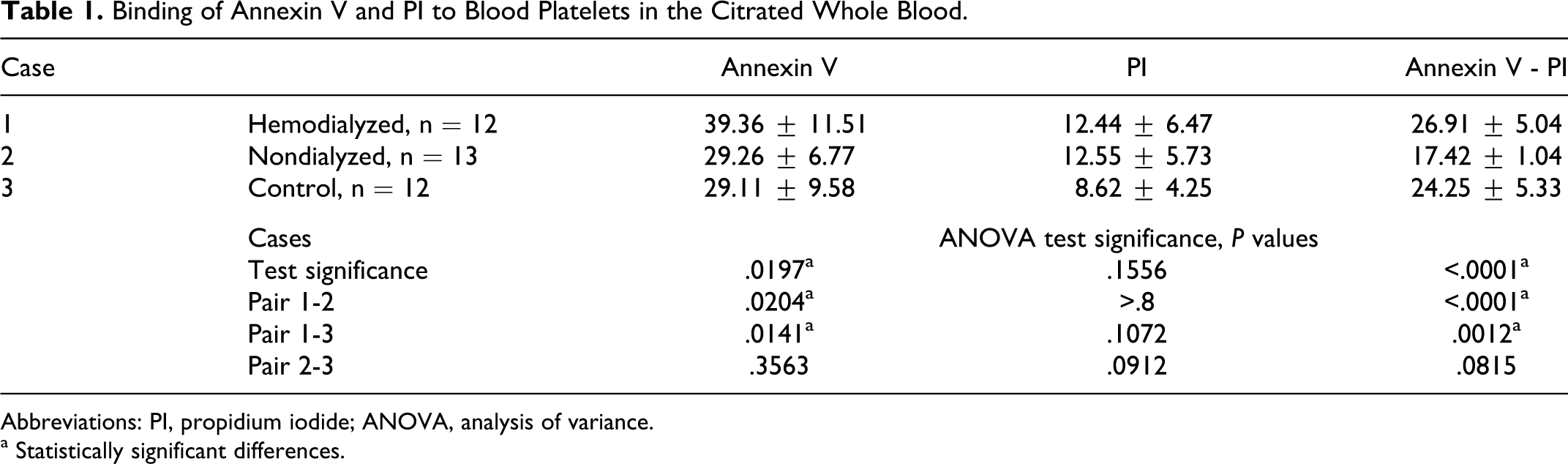
Abbreviations: PI, propidium iodide; ANOVA, analysis of variance.
a Statistically significant differences.
Next observation concerned spontaneous blood platelet activation reflected by the level of expression of well-characterized activation markers—active fibrinogen receptor and surface expressed P-selectin. The level of activation was the same for both control and hemodialyzed patients, but nondialyzed patients exhibited significantly higher level of exposed active fibrinogen receptor molecule recognized by PAC-1 antibody (Table 2). On the other hand, expression of P-selectin on blood platelets of nondialyzed patients was lower, but this difference was not relevant.
Blood Platelet Activation Assessed in the Citrated Whole Blood.
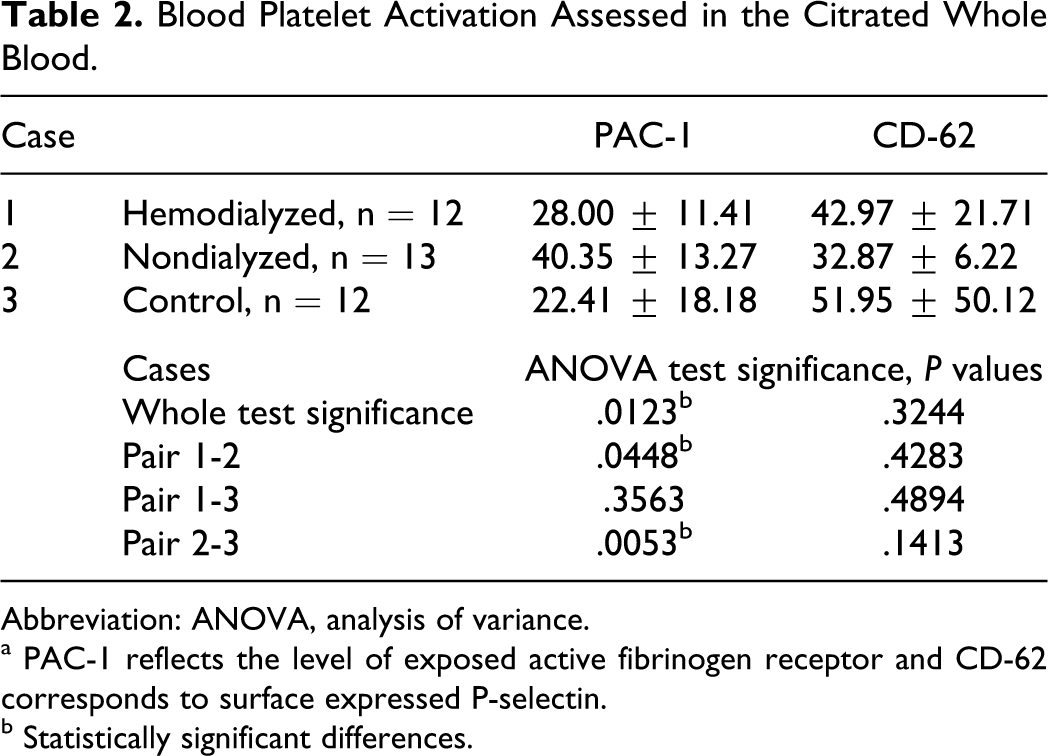
Abbreviation: ANOVA, analysis of variance.
a PAC-1 reflects the level of exposed active fibrinogen receptor and CD-62 corresponds to surface expressed P-selectin.
b Statistically significant differences.
Discussion
Blood platelets are anucleated fragments of parental bone marrow cells, such as megakaryocytes. Platelets are short-lived elements of blood playing a crucial role in thrombosis and hemostasis. Platelet activation and senescence are associated with apoptosis-like processes similar to those observed in nucleated cells during apoptosis. It includes loss of mitochondrial membrane potential, caspase activation, PS externalization, cell shrinkage, and finally cell fragmentation to microparticles. Recently, it was shown that ABT-737, a potent antagonist of B-cell lymphoma 2 protein family in nucleated cells, induces apoptosis-like process in blood platelets with all characteristic signs of apoptosis, although this agent is ineffective in platelet activation. 16 Another recent article describes the role of calmodulin antagonists, well known as potent apoptosis inducers in various tumor cells, in receptor-mediated platelet apoptosis. 17 Incubation of blood platelets with these antagonists also resulted in impaired receptor-related platelet function, although this agent did not cause platelet activation manifested by elevated expression of P-selectin or binding of PAC-1 antibody. It was also shown that dibucaine, acting as calpain activator able to induce apoptosis in several types of nucleated cells, can trigger changes related to apoptosis in blood platelets without platelet activation. 18 Moreover, resveratrol, a component of red wine, exhibiting diverse biological activities including inhibition of platelet aggregation, also markedly stimulates apoptosis in washed blood platelets. 19
Double labeling of blood platelets using Annexin V and PI allows to distinguish apoptosis from necrosis. We observed significantly elevated level of Annexin V binding to blood platelets of hemodialyzed patients, with no increased number of necrotic blood platelets (Table 1). This finding very strongly suggests the presence of apoptosis-like process in blood platelets belonging to these patients, and it is consistent with earlier published report. 9 This observation is also in good accordance with the above-discussed events concerning independence of blood platelet activation and aggregation from apoptosis. In our experiments, platelets of hemodialyzed patients were not different from control platelets in respect of binding of PAC-1 antibody and P-selectin exposure (Table 2) confirming independence of apoptosis-like process from platelet activation in hemodialyzed patients. Significantly increased level of PAC-1 antibody binding to platelets of nondialyzed patients can be explained by intensified binding of RGDS-containing fibrinogen fragments, present in a high concentration in uremic plasma, 4 to inactive fibrinogen receptor and converting this receptor to the active form blocked by bound fibrinogen fragment.
The final stage of cell apoptosis is cell fragmentation and microparticles release. Circulating platelet-derived microparticles exhibit procoagulant activity and are suspected as a main reason for thrombosis in patients with uremia. 20 Thus, reported herein the elevated binding of Annexin V to blood platelets of hemodialyzed patients with uremia indicates increased level of platelet apoptosis and corresponds with increased risk of thrombosis, observed for these patients, induced by procoagulant activity of blood platelet-derived apoptotic fragments.
Concluding, our results support hypothesis that hemodialysis treatment may be at least partially responsible for triggering blood platelet apoptosis and resulting increased risk of thrombosis in hemodialyzed patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by project No. N N401 227434.
