Abstract
Background:
CYP4A11 (cytochrome P450, family 4, subfamily A, polypeptide 11) converts arachidonic acid to 20-hydroxyeicosatetraenoic acid (20-HETE), which plays a crucial role in the modulation of cardiovascular homeostasis. The aim of the present study was to assess the association between the human CYP4A11 gene and coronary artery disease (CAD).
Methods:
A total of 361 patients with CAD and 315 controls were genotyped for 4 single-nucleotide polymorphisms (SNPs) of the human CYP4A11 gene (rs9332978, rs4660980, rs3890011, and rs1126742). The data were assessed for 3 groups: total participants, men, and women via case–control studies.
Results:
For total participants and men, the distribution of SNP3 (rs3890011) genotypes showed a significant difference between CAD and control participants (P = .030 and P = .013, respectively), the distribution of the recessive model of SNP3 (GG vs CC + GC) was significantly higher in CAD patients than in control participants (P = .011 and P = .014, respectively), the significant difference was retained after adjustment for covariates (for total participants, 95% confidence interval [CI]: 1.137-2.423, P = .009; and for males, 95% CI: 1.173-3.013, P = .009).
Conclusions:
rs3890011 maybe a novel polymorphism of the CYP4A11 gene associated with CAD in a Han Chinese population.
Introduction
Coronary artery disease (CAD) is a complex multifactorial and polygenic disorder thought to result from an interaction between an individual’s genetic makeup and various environments. 1 Various gene variants have been shown to be associated with CAD.2,3 Cytochrome p450 (CYP), a superfamily of cysteinato-heme enzymes, is responsible for not only the metabolism of xenobiotics but also a host of endobiotics whose metabolites have critical roles in the maintenance of cardiovascular health.4,5 Mounting evidence has demonstrated that CYP enzymes are involved in the pathogenesis of CAD. Polymorphisms of CYP genes, for example, CYP1A1, CYP1A2 (which metabolizes polycyclic aromatic hydrocarbons and aromatic amines during smoking),6,7 CYP2C8, CYP2C9, CYP2J2 (epoxyeicosatrienoic acid [EET] synthesis),8–10 CYP8A (prostacyclin synthesis), 11 CYP11B2 (aldosterone synthesis), 12 CYP17, and CYP19 (synthesis of sex hormones), have been demonstrated to have a relationship with CAD. 13 The CYP4A enzymes mainly metabolize arachidonic acid to hydroxyeicosatetraenoic acid (HETE) which has critical roles in the regulation of renal, pulmonary, and cardiac function as well as vascular tone. 5 In humans, CYP4A11 (cytochrome P450, family 4, subfamily A, polypeptide 11) converts arachidonic acid to 20-HETE, which plays a crucial role in the modulation of cardiovascular homeostasis. 4 The relationship between the polymorphisms of the CYP4A11 gene and hypertension as well as cerebral infarction has been established.14–16 The aim of the present case–control study was to assess the association between the human CYP4A11 gene and CAD.
Participants and Methods
All patients gave written informed consent and explicitly provided permission for DNA analyses as well as collection of relevant clinical data. The study protocols were approved by the Ethics Committees of the participating institutes.
Participants
All patients and controls were Han Chinese who had a differential diagnosis for chest pain encountered in the Cardiac Catheterization Laboratory of First Affiliated Hospital of Xinjiang Medical University (Urumqi, China) from 2007 to 2009. The study population involved 361 patients with CAD defined by angiographic means (main coronary artery stenosis of >50%). Control participants (n = 315) did not have coronary vessel stenosis and did not show clinical or electrocardiographic evidence of myocardial infarction (MI) or CAD. Data and information about traditional coronary risk factors (including hypertension, diabetes mellitus [DM], and smoking) were collected from all study participants. The diagnosis of hypertension was established if patients were on antihypertensive medication or if the mean of 3 measurements of systolic blood pressure (SBP) >140 mm Hg or diastolic blood pressure (DBP) >90 mm Hg, respectively. Diabetes mellitus was defined by patient recall and also if patients were taking antidiabetic medication or insulin therapy. “Smoking” was classified as smokers (including current or ex-smokers) or nonsmokers. All patients with impaired renal function, malignancy, connective tissue disease, or chronic inflammatory disease were excluded.
Genotyping
We selected 4 single-nucleotide polymorphisms (SNPs) in the human CYP4A11 gene as markers for assessment of genetic association. We designated these SNPs as SNP1, SNP2, SNP3, and SNP4 (rs9332978, rs4660980, rs3890011, and rs1126742) in order of increasing distance from the 5’ end of the gene (Figure 1 ). The minor allele frequency of each SNP was >10%, which indicated that they should be effective genetic markers. All 4 SNPs were confirmed using database single nucleotide polymorphisms (dbSNP) at the national center for biotechnology information (NCBI) Web site and the Applied Biosystems-Celera Discovery System. SNP4 (rs1126742) is located in exon 11. Thymidine substitutes cytosine at the locus (T8590C) that results in a nonsynonymous phenylalanine→serine (F-to-S) substitution at amino acid residue 434 of CYP4A11. 14 SNP1, SNP2, SNP3 are located in introns.
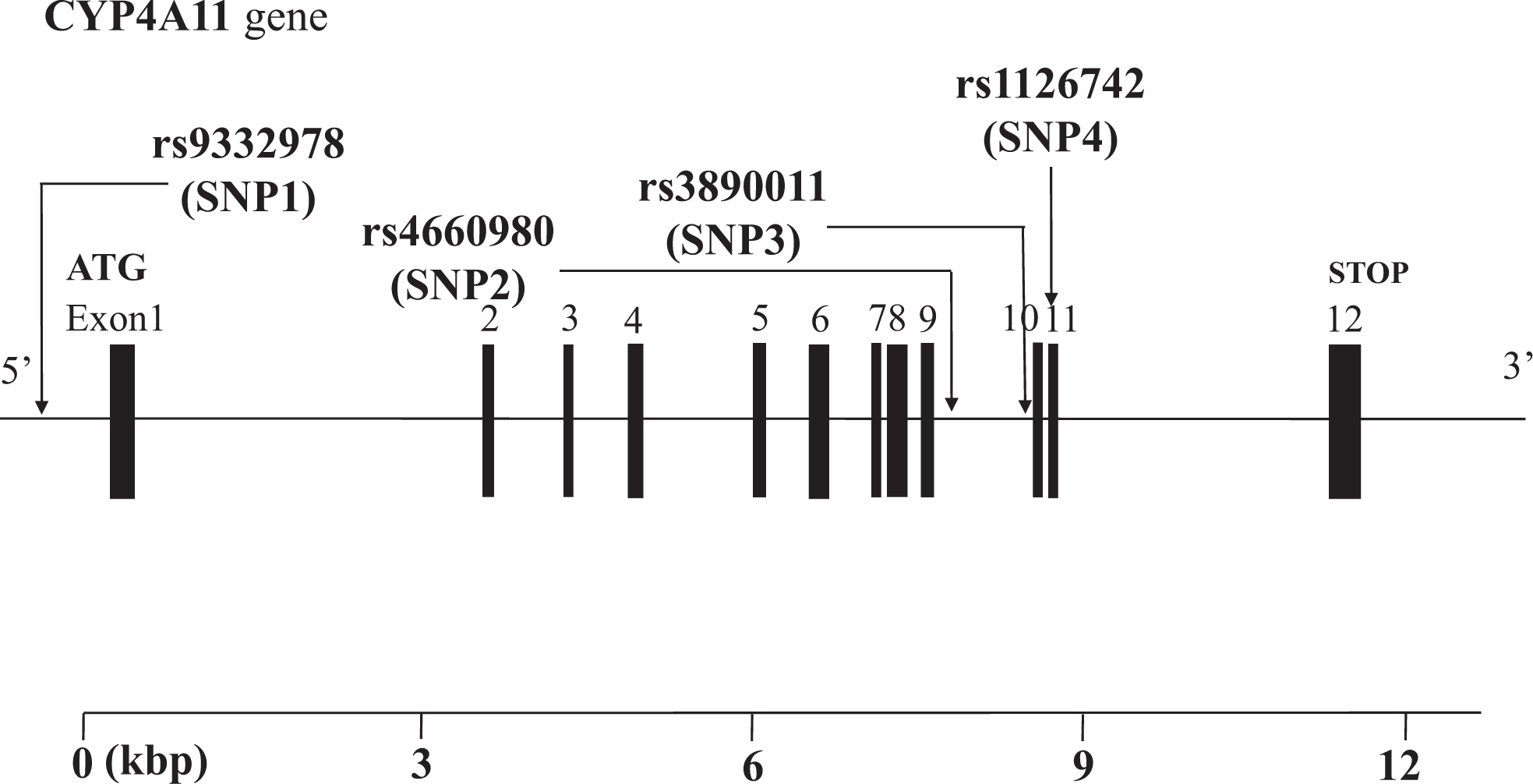
Structure of the human CYP4A11 gene. This gene consists of 12 exons separated by 11 introns. Boxes indicate exons, and lines indicate introns and intergenic regions. Filled boxes indicate coding regions. Arrows mark the locations of polymorphisms.
Blood samples were collected from all participants. Genomic DNA was extracted from peripheral blood leukocytes using phenol and chloroform. Genotyping was undertaken using the TaqMan SNP Genotyping Assay (Applied Biosystems [ABI], Foster City, California). TaqMan SNP Genotyping Assays were carried out using Taq amplification. The primers and probes used in the TaqMan SNP Genotyping Assays were chosen based on information available at the ABI Web site (http://myscience.appliedbiosystems.com). Thermal cycling was done using the Applied Biosystems 7900HT Fast Real-Time PCR System. Plates were read on the sequence detection systems (SDS) automation controller software v2.3 (ABI).
Biochemical Analyses
We measured the plasma concentration of blood urea nitrogen, creatinine (Cr), glucose (Glu), triglyceride (TG), total cholesterol (TC), high-density lipoprotein (HDL), and low-density lipoprotein (LDL), using standard methods in the Clinical Laboratory Department of First Affiliated Hospital, Xinjiang Medical University.
Statistical Analyses
All continuous variables were expressed as mean ± standard deviation (SD). Differences in continuous variables between patients with CAD and control participants were analyzed using the Mann-Whitney U test. Differences in categorical variables were analyzed using Fisher’s exact test. Differences in distributions of genotypes and alleles between patients with CAD and control participants were analyzed using Fisher exact test. Based on the genotype data of the genetic variations, we undertook linkage disequilibrium (LD) analyses using the expectation maximization (EM) algorithm. Multivariate analyses were conducted with multiple logistic regression methods, and adjusted estimations of conditioned relative risk and 95% confidence intervals (CIs) were calculated. P value <.05 was considered significant. Statistical analyses were carried out using SPSS software for Windows, version 12 (SPSS Inc, Chicago, Illinois).
Results
Table 1 shows the clinical characteristics of the study participants. For total, male, and female participants, there was no significant difference in age between patients with CAD and control participants. That is, the study was an age-matched case–control study. For total participants, the plasma concentration of Cr, Glu, and TC was significantly higher for patients with CAD than for control participants, the prevalence of essential hypertension (EH), DM, and smoking was significantly higher for patients with CAD than for control participants, and the plasma concentration of HDL was significantly lower for patients with CAD than for control participants. For males, the plasma concentration of Cr and TC was significantly higher for patients with CAD than for control participants, the prevalence of EH and DM was significantly higher for patients with CAD than for control participants. For females, the plasma concentration of Cr and Glu was significantly higher for patients with CAD than for control participants, the prevalence of DM was significantly higher for patients with CAD than for control participants, and the plasma concentration of HDL was significantly lower for patients with CAD than for control participants.
Characteristics of Study Participantsa
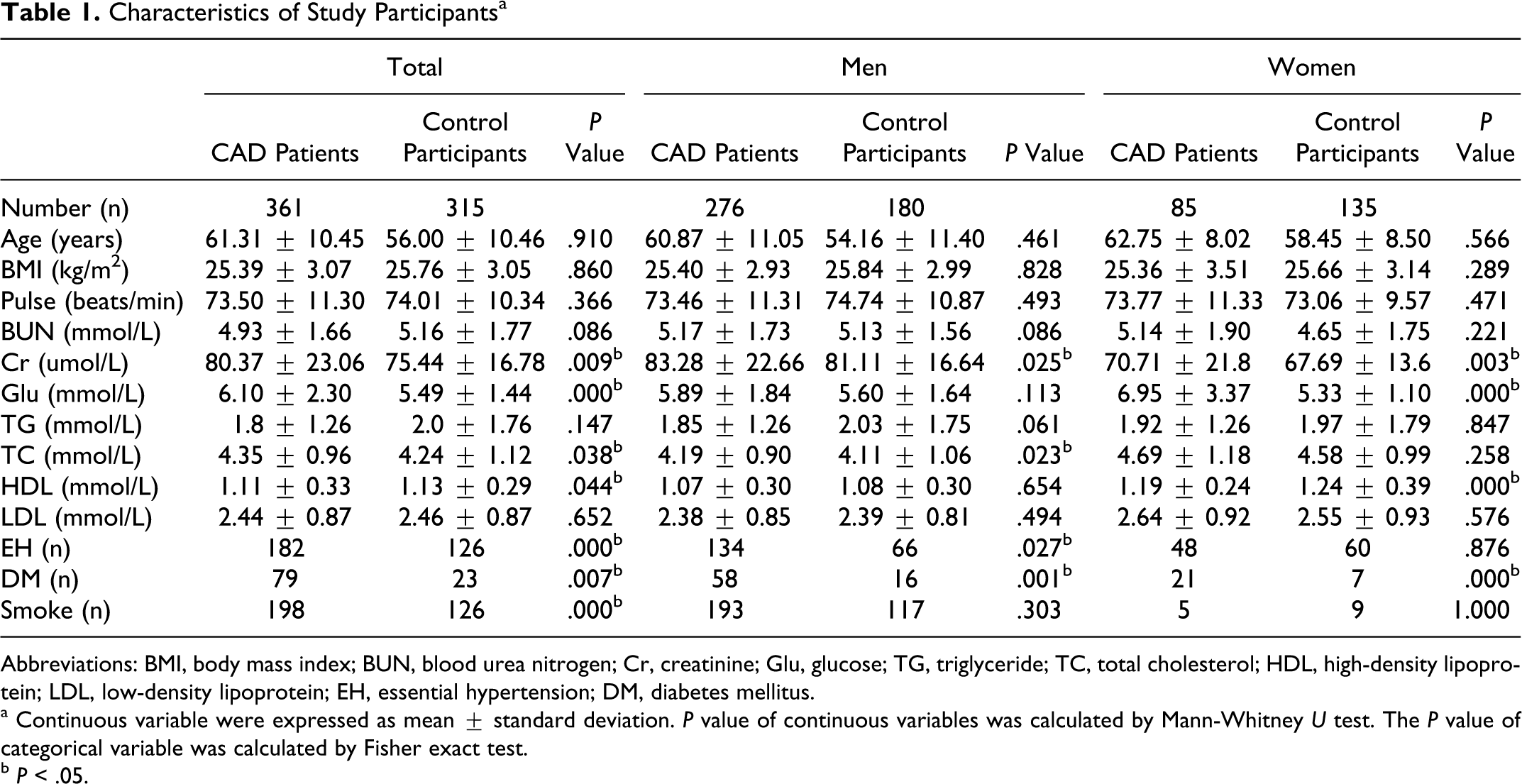
Abbreviations: BMI, body mass index; BUN, blood urea nitrogen; Cr, creatinine; Glu, glucose; TG, triglyceride; TC, total cholesterol; HDL, high-density lipoprotein; LDL, low-density lipoprotein; EH, essential hypertension; DM, diabetes mellitus.
a Continuous variable were expressed as mean ± standard deviation. P value of continuous variables was calculated by Mann-Whitney U test. The P value of categorical variable was calculated by Fisher exact test.
b P < .05.
Table 2 shows the distribution of genotypes and alleles of SNP1, SNP2, SNP3, and SNP4 for CYP4A11 gene. For total participants and males, the distribution of SNP3 (rs3890011) genotypes showed a significant difference between patients with CAD and control participants (P = .030 and P = .013, respectively), and the distribution of the recessive model of SNP3 (GG vs CC + GC) was significantly higher in patients with CAD than in control participants (P = .011 and P = .014, respectively). Dominant and recessive models were defined by their frequency among total participants. The distribution of the genotype of SNP1, SNP2, SNP3, and SNP4 in patients with CAD and controls was compatible with the Hardy-Weinberg equilibrium.
Genotype and Allele Distributions in Patients With CAD and Control Participantsa
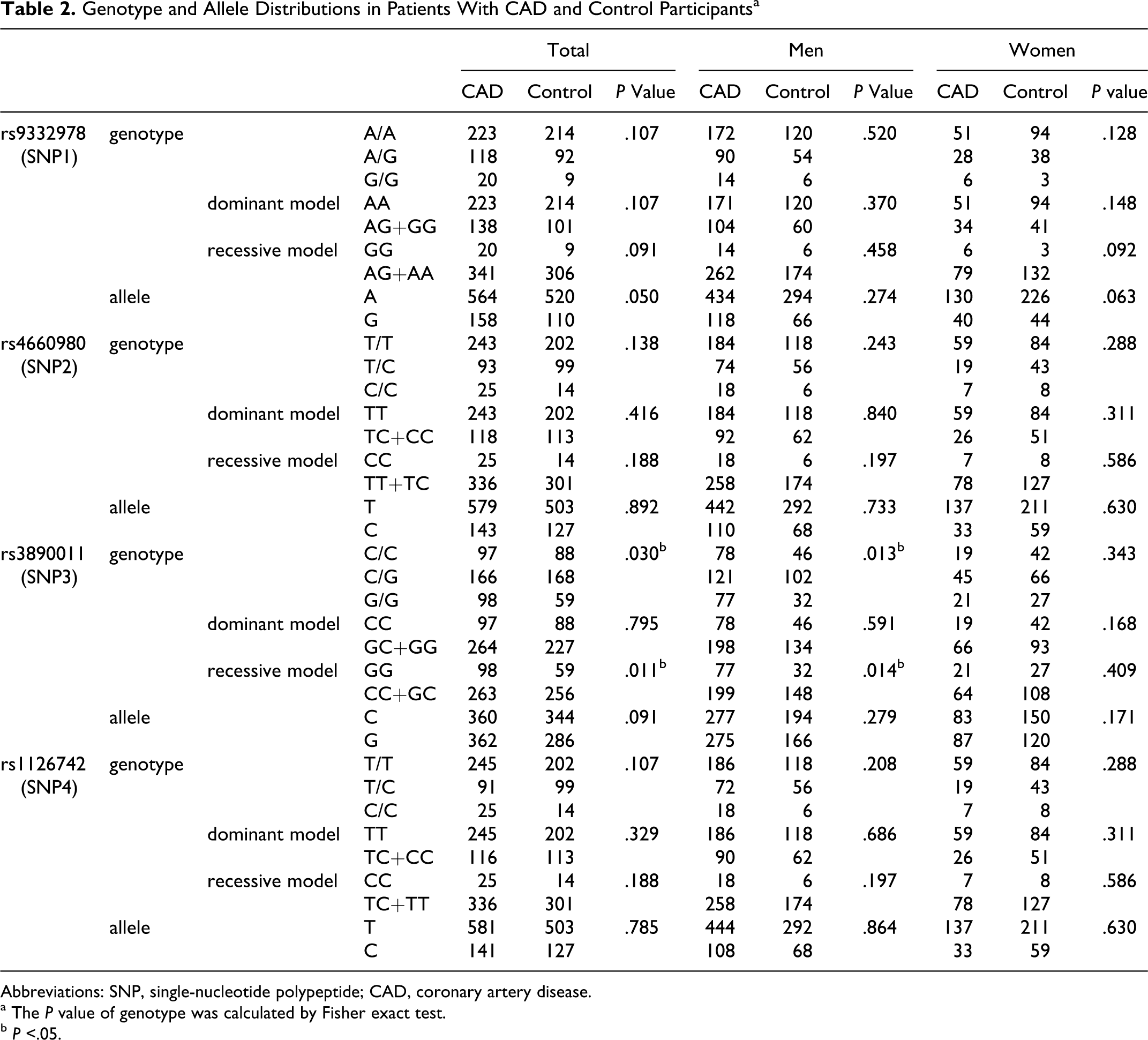
Abbreviations: SNP, single-nucleotide polypeptide; CAD, coronary artery disease.
a The P value of genotype was calculated by Fisher exact test.
b P <.05.
Table 3 shows that multiple logistic regression analysis done with or without EH, DM, and smoking because these 3 variables were the major confounding factors for CAD (Table 1). The significance of the recessive model of SNP3 (GG vs CC + GC) between patients with CAD and control participants was retained after adjustment for EH, DM, and smoking (for total participants, 95% CI: 1.137-2.423, P = .009; for males, 95% CI: 1.173-3.013, P = .009).
Multiple Logistic Regression Analysis of Patients With CAD and Control Participants
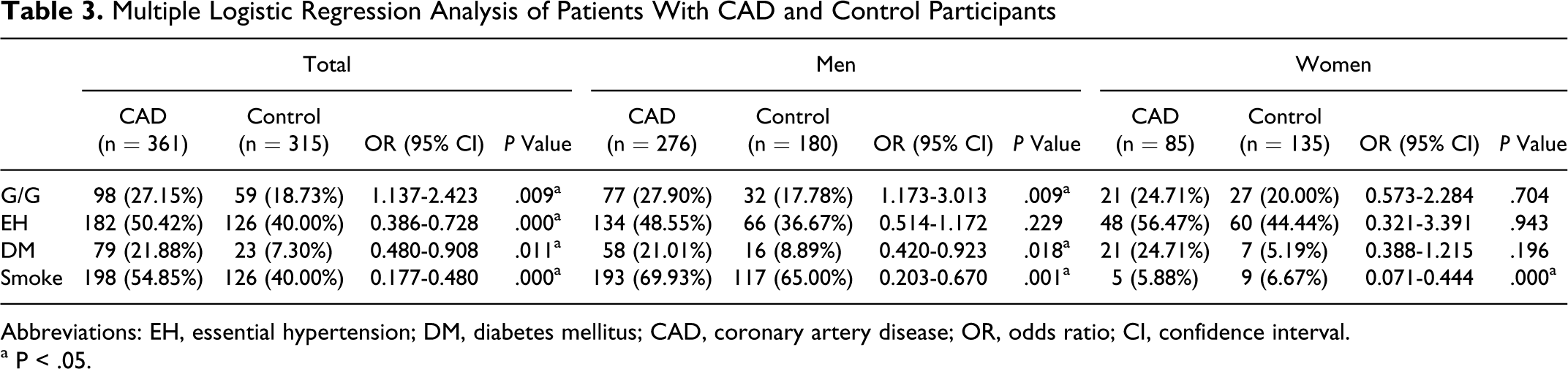
Abbreviations: EH, essential hypertension; DM, diabetes mellitus; CAD, coronary artery disease; OR, odds ratio; CI, confidence interval. a P < .05.
Discussion
Several CYP enzyme families have been identified in the heart, endothelium, and smooth muscle of blood vessels. Increasing evidence points to the role of endogenous CYP metabolites such as EETs, HETEs, prostacyclin (PGI2), aldosterone, and sex hormones indicate the maintenance of cardiovascular health. A link between the expression and activity of CYP and cardiovascular disease (CVD) such as hypertension, CAD, heart failure, stroke, and cardiomyopathy and arrhythmias has been established. 5 In humans, CYP4A11 acts mainly by converting arachidonic acid to 20-HETE. The latter has been shown to have 5 physiological functions. First, 20-HETE increases thymidine incorporation in cells and mediates growth responses in vascular smooth muscle cells (VSMCs) in response to norepinephrine and angiotensin II.17–19 Second, 20-HETE increases the intracellular calcium concentration and mediates the vasoconstrictor responses of angiotensin II, vasopressin, and norepinephrine.20–22 Third, 20-HETE inhibits Na+-K+-ATPase activity and sodium transport in the proximal tubule and is a key mediator of long-term control of arterial pressure.23–25 Fourth, 20-HETE inhibits platelet aggregation and formation of thromboxaneA2 (TxA2) during platelet activation. 26 Fifth, 20-HETE is a significant contributor to ischemia–reperfusion injury, and exogenous administration of 20-HETE significantly increases the infarct size.27,28 Polymorphisms of the CYP4A11 gene can affect the metabolism of arachidonic acid, resulting in an altered generating capacity of 20-HETE. We hypothesized that variability in the gene might also affect the risk of CAD. We genotyped 4 SNPs of the CYP4A11 gene in a Han Chinese population and assessed the association between the CYP4A11 gene and CAD using case–control analyses. For the total participants and the male group, there was a significant difference in the genotypic distribution of SNP3 (rs3890011) between patients with CAD and control participants, and the distribution of the recessive model of SNP3 (GG vs CC + GC) was significantly higher in patients with CAD than in control participants. The significant difference in the recessive model of SNP3 (GG vs CC + GC) between patients with CAD and control participants was retained after adjustment for covariates such as EH, DM, and smoking. This indicated that the risk of CAD may be increased in men with the GG genotype of rs3890011. Although rs3890011 is located in the introns and has no function, it is tightly linked to neighboring functional SNPs and constructs different haplotypes, potentially affecting the structure and/or catalytic activity of the enzyme, 29 we speculated that participants with the GG genotype of rs3890011 may have a high ability of synthesizing 20-HETE. 20-HETE increases the intracellular concentration of calcium and mediates the action of vasoconstrictors. Coronary artery disease is a multifactorial disorder, and this SNP could be a genetic marker for CAD risk.
SNP4 (rs1126742) is one of the most important genetic variants of CYP4A11. The thymidine→cytosine substitution at nucleotide 8590 in the CYP4A11 gene results in a phenylalanine→serine substitution at amino acid 434 which significantly reduces the catalytic activity of the CYP4A11 enzyme through a loss-of-function mechanism in vitro. 14 Several studies recently focused on the association between rs1126742 and hypertension. Gainer et al, 14 Mayer et al, 15 Laffer at al, 30 Fava et al, 31 and Williams et al 32 showed a clear association between the 8590C allele and hypertension, whereas Ward et al 33 and Sugimoto et al 34 showed no association between the 8590C allele and hypertension. These contradictory results may arise from the difference in ethnic background. Few studies focus on the association between rs1126742 and CAD. Although Hermann et al demonstrated that the 8590C allele increased coronary vasoconstriction, 35 Mayer et al did not show an association between the 8590C allele and MI. 36 Fava et al did not show an association between the 8590C allele and CAD or stroke either. 31 We also did not find an association between the 8590C allele and CAD in the present study. Sugimoto et al demonstrated that rs9332978 is associated with hypertension, 34 whereas we found that it was not associated with CAD. Zhang et al showed SNP3 (rs3890011) not to be associated with hypertension, 37 whereas we demonstrated that it is associated with CAD. The different results can be explained by the different pathogenesis of hypertension and CAD, both of which are complex multifactorial and polygenic disorders, so it is hard to interpret them clearly only with respect to genetic makeup.
In conclusion, rs3890011 maybe a novel polymorphism of the CYP4A11 gene associated with CAD. The GG genotype of rs3890011 appears to be a genetic marker of CAD for men in a Han Chinese population. Further studies are needed to isolate functional mutations in the CYP4A11 gene related to CAD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number: 81060020).
