Abstract
Patients with end-stage renal disease (ESRD) are known to have an elevation of a variety of abnormal thrombotic and inflammatory markers associated with high cardiovascular mortality. Vascular endothelial growth factor (VEGF) is also dysregulated in ESRD but not much is known about the serum levels of VEGF in patients with ESRD. Published reports suggest that elevated levels of VEGF may be protective to the kidney during periods of acute injury and may maintain local glomerular function. Impaired production of VEGF may lead to proteinuria, hypertension, and thrombotic microangiopathy. However, its role in chronic kidney disease or ESRD remains undefined. In our study, we analyzed blood samples of 52 patients with ESRD on stable hemodialysis regimen and measured predialysis serum levels of VEGF and compared these with blood samples obtained from 50 healthy volunteers in order to study differences between baseline levels of VEGF and also attempted to determine its role in ESRD-related cardiovascular mortality.
Introduction
End-stage renal disease (ESRD) refers to advanced kidney disease in the presence of signs and symptoms of uremia necessitating replacement therapy in the form of dialysis or transplant. 1 The ESRD is associated with increased cardiovascular mortality (5-year survival rate of ~34%) and studies indicate that wide arrays of thrombotic and inflammatory markers are elevated in patients with ESRD.2,3 The cardiovascular morbidity and mortality cannot be explained by the traditional risk factors, and it is hypothesized that dysregulation of these markers of thrombosis and inflammation may partly be contributing to high cardiovascular mortality; however, there are no confirmatory studies or clinical trials. Vascular endothelial growth factor (VEGF) is a regulator of blood vessel growth and is present in higher levels during the formation of glomerulus as demonstrated in animal models. 4 It has also been known to mediate vascular permeability and is considered an endothelial cell mitogen.5,6 The VEGF is a polypeptide made by the gene located on chromosome 6p21.3, and it includes placental growth factor (PLGF) and VEGF A to E.7,8 Alternative splicing of this gene yields 4 major isoform encoding polypeptides of 121, 165, 189, and 206 amino acids. Other isoforms containing 148, 162, and 183 amino acids and an inhibitory isoform, VEGF 165b, are also known.7,9,10 The VEGF is secreted by macrophages, neutrophils, platelets, eosinophils, mast cells, vascular smooth muscle cells, fibroblasts, hepatocytes, epithelial cells, and tumor cells lines. One cell type may produce more than 1 VEGF variant. 11 These VEGF polypeptides bind to their tyrosine kinase receptors and help mediate several important functions including vasodilatation (possibly via nitric oxide synthesis), macrophage recruitment, lymphangiogensis and hemangiogensis via macrophage recruitment, maturation of dendritic cells, and polarization of T cells.11,12
The VEGF production in the kidney has been most consistently seen in the podocytes but has also been known to be expressed in tubular epithelium, collecting ducts, and mesenchymal cells in the developing kidney. 13 The VEGF seems to play an important role in maintaining vascular integrity in the kidney as demonstrated in animal models and its inhibition has been associated with renal thrombotic microangiopathy. Proteinuria, elevated serum creatinine, and hypertension are known to be developed by selective inhibition of VEGF by bevacizumab (monoclonal antibody against VEGF) in humans. 14 This is consistent with available literature showing increased concentration of soluble Flt-1 (VEGF receptor 1) seen in maternal body in preeclampsia. Excess soluble Flt-1 binds to available VEGF in circulation thereby decreasing its concentration leading to proteinuria and hypertension of preeclampsia. 15 Elevated levels of VEGF are seen during acute insult to the kidney and are thought to help with endothelial repair.16,17
Increased expression of VEGF is seen in various injury and inflammation and its effects on vascular integrity and blood pressure, and thus it is possible that abnormal levels of VEGF may be a contributory factor for the markedly increased cardiovascular mortality reported in patients with ESRD. Little or no information is available about serum levels of VEGF in patients with ESRD. In this study, we analyzed predialysis serum samples of 52 patients with ESRD on a stable hemodialysis and compared these with serum levels of VEGF obtained from 50 healthy adults.
Materials and Methods
Blood samples were obtained from 52 patients on a stable hemodialysis regimen at Loyola University Dialysis Unit by venous blood draw, just prior to dialysis, and before any heparin was given. Blood samples were also obtained from 50, healthy, age- and gender-matched controls. Study criteria included patients older than 18 years and on maintenance hemodialysis for more than 3 months prior to the study. Patients who were actively infected or hospitalized at the time of blood draw and those who were not able to give informed consent were excluded. Investigative Review Board of Loyola University Medical Center approved the study protocol. All patients in this study were on high-flux polysulfone hollow fiber dialyzers.
The blood collected was centrifuged within 2 hours at 3000 rpm for 15 minutes and plasma aliquot was frozen at −70°C. Later, these frozen citrated plasma samples were retrospectively analyzed. Similar method was used for healthy volunteers for blood draw and processing of blood for testing.
The VEGF quantitation was performed using Randox Evidence Investigator. The evidence investigator VEGF assay is a sandwich chemiluminescent immunoassay for the detection of VEGF in human serum and uses immobilized monoclonal antibody to VEGF as the biochip substrate. The advantages of using Randox Investigator include the requirement of a small sample volume and quicker analysis. The assay provides a range of 0 to 1000 pg/mL. All precautions were taken for proper handling of samples per instructions provided by the manufacturer in order to avoid and minimize technical and procedural errors.
Results
The results from the testing and comparison with healthy volunteers indicate that levels of VEGF are elevated in patients with ESRD (Figures 1 and 2). The control group represented a mean value to 2.2 ± 0.1 pg/mL, whereas the ESRD patients exhibited a mean of 31.4 ± 2.6 pg/mL. The levels determined in healthy volunteers ranged from 0.8 to 4.8 pg/mL, whereas a much wider scatter was observed in the patients with ESRD ranging from 10.43 to 105.6 pg/mL. There was a significant difference between the 2 populations (P < .05).
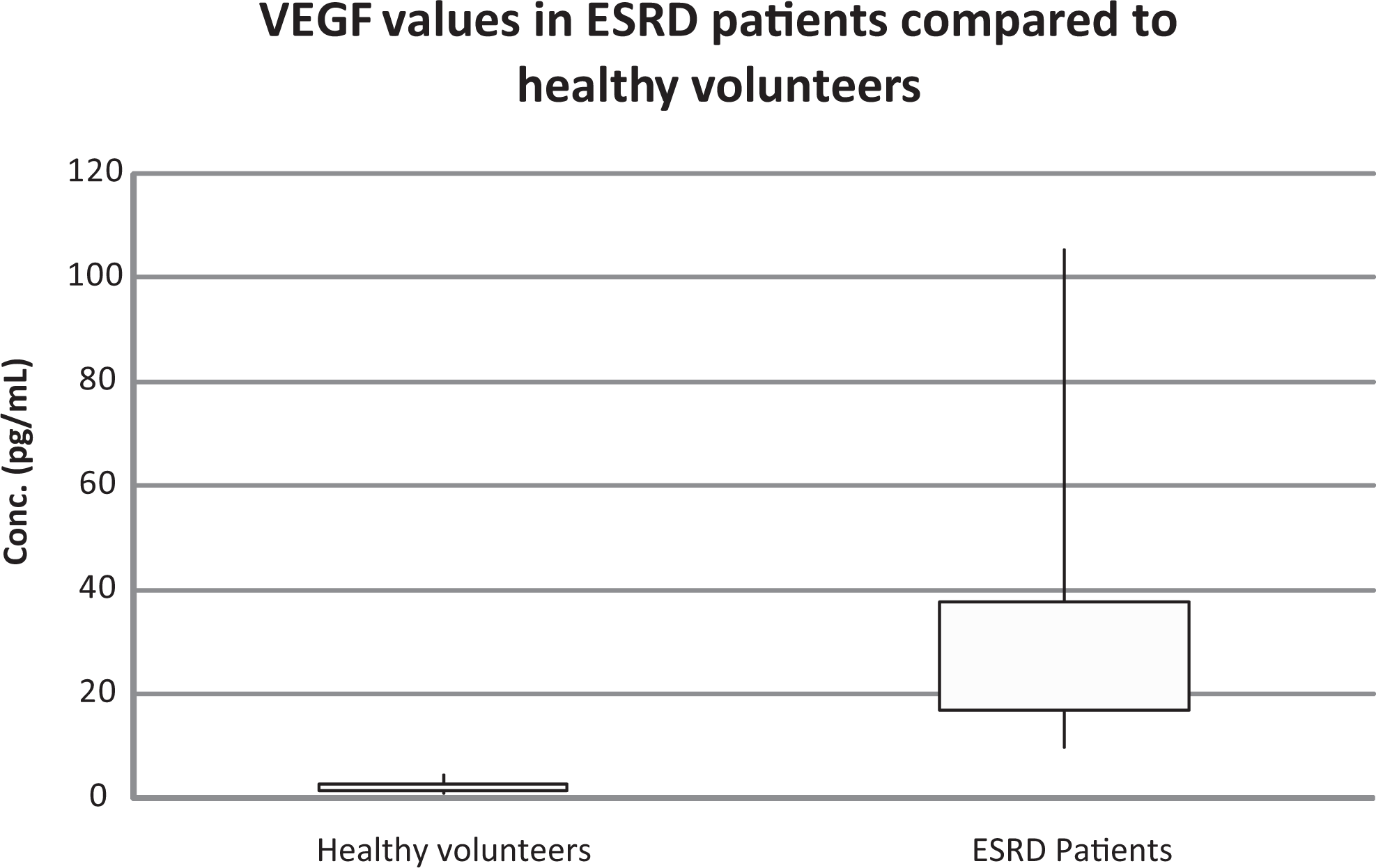
Values of vascular endothelial growth factor (VEGF) in patients with end-stage renal disease (ESRD) compared with healthy volunteers.
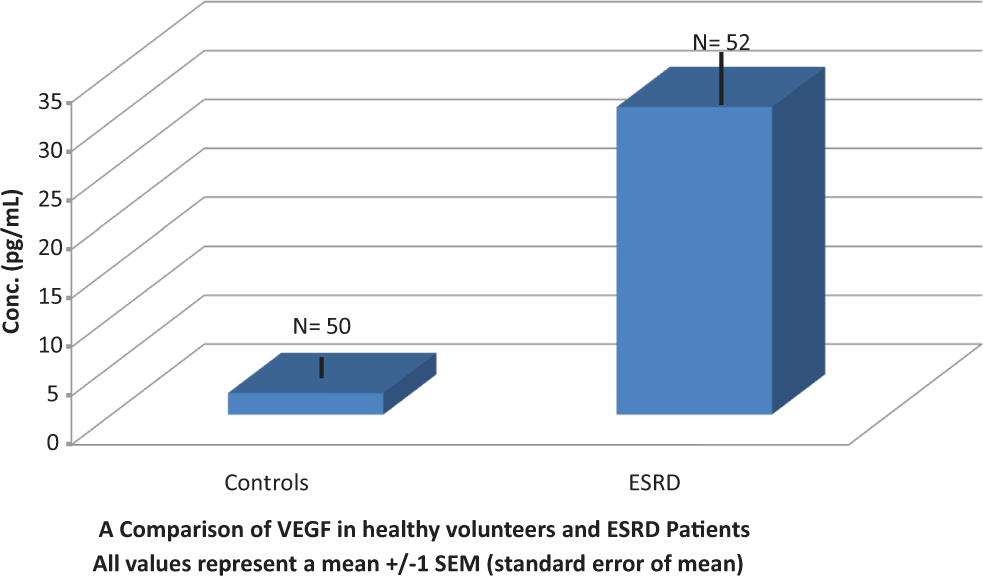
A comparison of vascular endothelial growth factor (VEGF) in healthy volunteers and in patients with end-stage renal disease (ESRD). All values represent a mean ± 1 standard error of mean.
Discussion
The results of our analysis suggest that the levels of VEGF are elevated in patients with ESRD. The VEGF in normal individuals is believed to have a protective effect on glomerular microvasculature. 14 Expression of VEGF is stimulated by hypoxia and is regulated by a wide variety of hormones, growth factors, and cytokines. 18 Moreover, VEGF may have a proinflammatory effect since it is a known chemoattractant for monocytes. In animal models, VEGF has been known to cause destabilization of atherosclerotic plaque. 19 Levels of VEGF are elevated in patients with diabetes, coronary artery disease, and peripheral vascular disease.20–22 Elevated levels of VEGF in these diseases provide evidence of the cellular response to inflammation and injury that leads to an increase in the expression of VEGF. It may be speculated that the elevated levels of VEGF may contribute to the cardiovascular mortality in patients with ESRD, as ESRD is known to be a proinflammatory and prothrombotic state with increase in the expression of other inflammatory chemokines and increase in the levels of markers of thrombosis. 3 Levels of VEGF have been found to be in increased concentration in septic patients with correlation of levels with severity of symptoms including shock. Higher levels were found in patients with shock. 23 Since septicemia represents an inflammatory state, elevated levels of VEGF may also represent the degree of inflammation in patients with ESRD. Infection may be more prone in hemodialysis patients mostly related to access; however, care was taken to exclude any patients with infection.
Increased cardiovascular mortality in ESRD may be due to a complex interplay of all these markers rather than a direct cause and effect of a single protein abnormal expression. Irrespective of the cause for increase in the levels of VEGF, it is possible that a correlation exists in the levels and clinical outcomes since chemokine expression may represent the ultimate response to the offending stimulus (inflammation or injury). Our study had a relatively small sample size but did show a range of VEGF elevation. Clinical outcomes were not measured because the study was mainly focused on evaluation levels of VEGF in patients with ESRD and the sample size is too small to attempt to evaluate any clinical outcome. The wide scatter in ESRD population could represent a multitude of reasons including multiple comorbidities or various reasons for inflammation. This study, however, emphasizes the presence of abnormally high levels of VEGF that may play a role in increased cardiovascular disease incidence present in patients with ESRD. To establish any correlation, a much larger study with long-term follow-up would be necessary.
Conclusion
The VEGF is known to play an important role in the embryological development of kidney, and elevated levels of VEGF are seen in acute injury to the kidney and thought to help with endothelial repair. Our study shows that levels of VEGF are elevated in patients with ESRD on stable hemodialysis regimen. This elevation of VEGF may be due to the inflammatory state that is known to be associated in patients with ESRD and a correlation of levels of these markers with cardiovascular mortality may be possible. Additional studies to prove this concept are suggested.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
