Abstract
Introduction
Femoral artery puncture and cardiac catheterization are widely used as diagnostic or interventional procedures, 1 and increasingly cardiac catheterization and related procedures are performed safely in an outpatient setting. 2 Cardiac angiography is associated with low but significant risk of complications. Along with major complications, such as coronary artery dissection, thrombus formation, and coronary artery spasm leading to acute occlusion of a coronary vessel, inadequate hemostasis can lead to one or all of the following; significant blood loss, patient’s discomfort, vessel occlusion, thrombosis, formation of arteriovenous fistula, and pseudoaneurysm requiring surgical intervention.3–7 The reported overall vascular complication rates range from 1.5% to 9%8–11 but may be as high as 14% in some cardiac interventional procedures. 12 This potential high prevalence of complications has promoted assessments of different techniques that achieve optimal hemostasis at the time of removal of the arterial catheter and minimize the complications of arterial puncture. In many countries, after femoral sheath removal, manual compression for 15 to 30 minutes followed by applying sandbags and a bed rest for 12 hours has been the conventional practice to achieve arterial hemostasis in patients undergoing cardiac catheterizations via femoral approach. 13 Manual compression, the use of sandbags, and bed rest are associated with patient discomfort, back pain, and longer time of hospitalization. Recently, the efforts to decrease vascular injury and complications have led to the development and use of an array of arterial puncture closure devices (APCDs). Although APCDs have been shown to decrease the time to achieve hemostasis, ambulation, and also faster discharge from hospital compared with manual compression,14–17 they can be associated with higher complication rates than manual compression.18–20 Regarding these problems, using a topical and noninvasive agent in angiography access site that rapidly induces hemostasis may be more efficient in terms of patient comfort due to the elimination of sandbags and earlier hemostasis without the potential risks of vascular injuries and complications. The aim of the present study was to evaluate the effect of using a topical hemostatic agent composed mainly of oxidized regenerated cellulose named ChitoHem® 21 on hemostasis time, re-bleeding, formation of hematoma, the necessity of using sandbags, and patient ambulation, comparing to conventional hemostasis procedure consisting of manual compression and the application of sandbags followed by overnight bed rest.
Methods
The present blind, randomized, controlled clinical trial was carried out in Shahid Rajaee Teaching Hospital, Iran University of Medical Sciences from July 2009 to October 2009. In total 1800 patients were admitted to the hospital for an elective coronary angiography during that period, 124 of whom were enrolled in the study following passing eligibility criteria. Number of samples for this study was determined using G-Power 3.00, an effect size of 0.3 for time to hemostasis (minute), a power of 0.80, and an α of .05. It was established that this study required a sample size of 41 patients per group.
The exclusion criteria of the study were age below 18 years, nonfemoral approach for the procedure, any known bleeding disorder, active bleeding in femoral access site before sheath removal, history of severe peripheral vascular disease, the use of antithrombotic drugs, and refusal from participation in the study. Ethical approval for the study was obtained from the Ethics Committee of Iran University of Medical Sciences. The proposed study was explained to potential participants, and written consent was signed by all patients. The patients were randomly (block randomization) divided into 2 groups of control and treatment. Baseline demographic and clinical characteristics including weight, age, sex, and history of previous cardiac angiography, drug administration before procedure, systolic and diastolic blood pressure, and prothrombin time (PT) were noted. In all the enrolled patients, femoral arterial sheath of 7F size were used and the sheath was removed by a cardiologist after the procedure. In the treatment group before femoral sheath removal, placing a towel over the hands of the nurse, 1 g of ChitoHem® powder was applied on the femoral access site. Prior to the complete removal of the sheath, the powder was pushed back slightly into the puncture site using the cannula itself, the sheath was then removed and the powder was allowed to be mixed with the jetting blood. Manual compression was applied until hemostasis was achieved, sandbags were administered if the time to achieve hemostasis was more than 5 minutes. The patients with no sandbag were advised a bed rest for 2 to 3 hours and following clinical examination by a cardiologist were discharged. Those patients using sandbags stayed in the hospital overnight. In the control group, the femoral sheath was removed by a cardiologist, then placing a towel over the hands of the nurse, hemostasis was achieved by using simple sterile gauze followed by manual compression. Sandbags were placed on the patient if the time to hemostasis was more than 5 minutes. There followed a period of overnight bed rest for patients using sandbags. The patients were discharged from the hospital the next morning following clinical examination by a cardiologist. Time to achieve hemostasis was defined as the time at which no compression was required to control bleeding. The arteriotomy site was monitored to check clot formation and hematoma occurrence after 3, 5, 7, 9, 11, 13, and 15 minutes in both control and treatment groups. The effectiveness of the 2 methods was assessed based on the time to attain hemostasis, re-bleeding rate, the necessity to use sandbags, hematoma formation, and the patients' ambulation during 24 hours after procedure. Data were analyzed using the Statistical Package for Social Science (SPSS, version 16.0, 1999; Chicago, IL). Continuous variables were presented as mean value ± standard deviation (SD) and categorical variables as percentage. The t test was used to determine significant difference for continuous variables and the chi-square test was used to calculate the differences in categorical values between groups. Probability value of <.05 was considered statistically significant.
Results
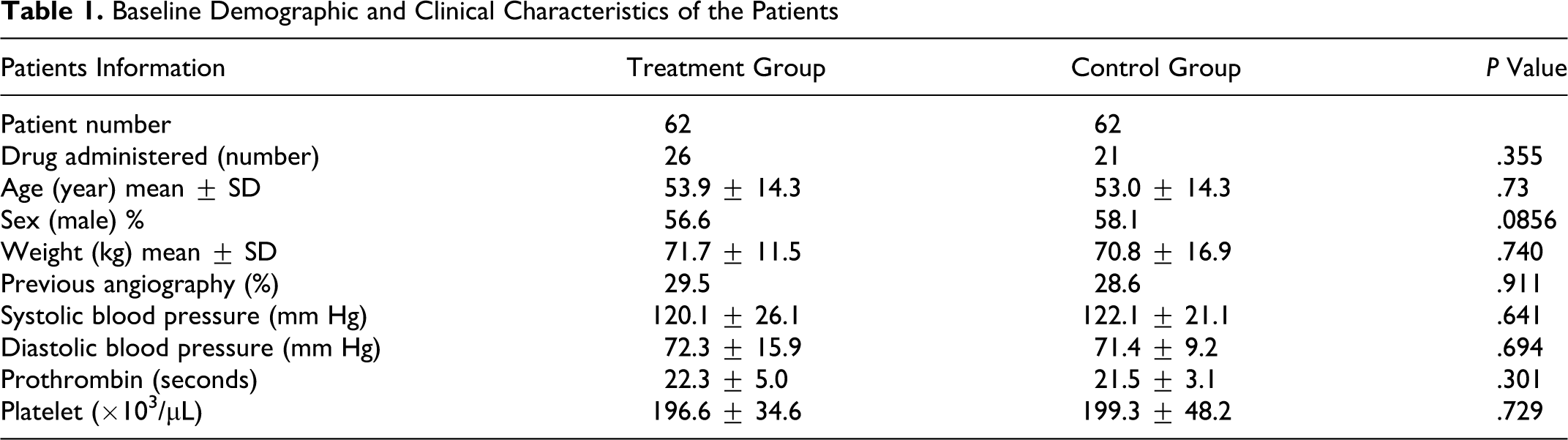
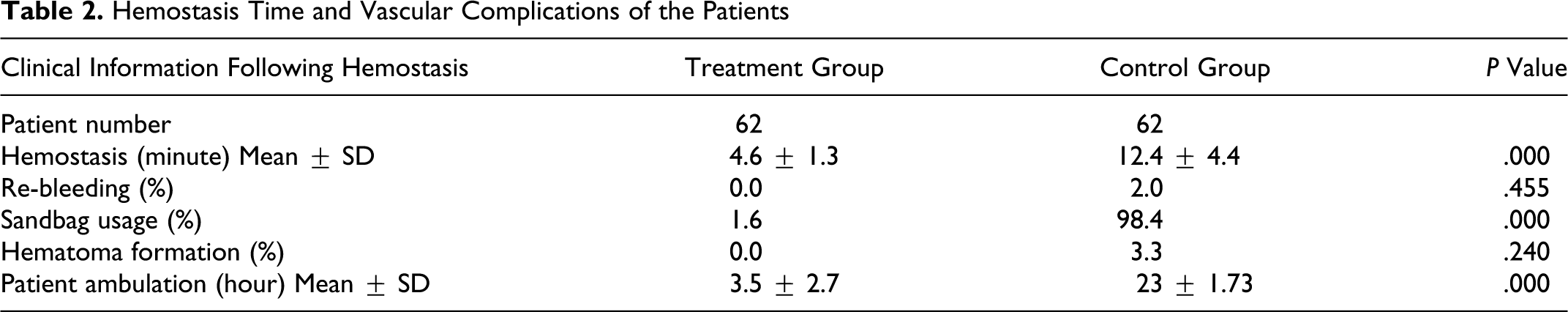
From July 2009 to October 2009, 124 patients undergoing diagnostic coronary angiography met the inclusion criteria and were enrolled in the trial. The patients were randomized into 2 groups of 62 using conventional hemostasis procedure by manual compression and or using ChitoHem® topical hemostasis powder. Table 1 summarizes and compares the patients' baseline demographic characteristics and preoperative parameters in both groups. There were no statistically significant differences between groups regarding age, gender, body weight, anticoagulation drugs used before angiography and the history of cardiac angiography (P > .05). Also the patients did not differ significantly in systolic and diastolic blood pressure, platelet count, and prothrombin time. In the treatment group, using hemostatic ChitoHem® powder, time to hemostasis was shorter compared with the control group (4.6 ± 1.3 in the treatment vs 12.4 ± 4.4 minutes in the control group; P < .05), and time of ambulation was significantly shorter than the control group (3.5 ± 2.7 hours in the treatment vs 23.0 ± 1.73 hours in the control group; P < .05). There was no significant difference in re-bleeding rate and hematoma formation percentages found between the 2 groups. In 1 patient (1.6%) in the treatment group sandbag was used while nearly all patients (98.4%) in the control group had to use 2 sandbags for 7.8 hours. Detailed comparison of the outcomes in 2 groups is shown in Table 2 .
Baseline Demographic and Clinical Characteristics of the Patients
Hemostasis Time and Vascular Complications of the Patients
Discussion
The most important outcome after coronary angiography via femoral artery is to achieve fast and effective hemostasis. ChitoHem® is a superabsorbent topical hemostasis powder, mainly composed of micro spherical oxidized regenerated cellulose. The mechanism of action of this product is 2-fold. Due to the superabsorbent nature of this powder, when applied to the puncture site, following the removal of the catheter the serum of the jetting blood is absorbed by the powder. As a result the rate of blood flow from the puncture site is reduced which in return accelerates the initiation of clot formation. On the other hand, the positive charges available in ChitoHem® topical hemostatic agent will attract the negative charges of the platelets and other blood elements and the rate of agglutination is accelerated too. As exhibited in Table 2, the application of ChitoHem® topical hemostatic powder on the patients has reduced the time to hemostasis significantly compared to the conventional manual compression (4.6 ± 1.3 in the treatment vs 12.4 ± 4.4 minutes in the control group; P < .05). Also in treatment group, the time of ambulation was significantly shorter compared with the control group (3.5 ± 2.7 hours vs 23.0 ± 1.73 hours; P < .05). The Koreny et al 3 published a systematic review about the effect of APCDs. Their analysis included only randomized, controlled trials evaluating APCDs and reporting vascular complications at the puncture site. The analysis of other studies (those that included allocation concealment, blinded outcome assessment, and intention-to-treat analysis) showed that the time to hemostasis and duration of bed rest was shorter in the group with APCDs compared with standard compression but the risks of hematoma and pseudoaneurysm were significantly increased in the APCD group compared with the manual compression group. Some other studies have indicated that these devices may be associated with increased risk of complications compared with manual compression, such as bleeding, ecchymosis, the need for surgical repair, acute femoral closure, and transfusion due to groin complications, readmission for groin complications and infection.18–20, 22–27 In using ChitoHem® topical hemostatic agent, no statistically significant difference was observed between treatment versus control group regarding vascular complications namely re-bleeding and hematoma formation. In a study by Martin et al 14 the authors evaluated a topical hemostatic dressing containing poly-N-acetyl glucosamine (p-Glena) post percutaneous coronary intervention (PCI) in fully anticoagulated patients. Application of the p-Glena dressing after arterial sheath removal resulted in a mean time to hemostasis of 15.5 minutes. This time is longer than the time to hemostasis in our study, but our studied population consisted of patients undergoing diagnostic coronary angiography that is less invasive than PCI. In a study by Mlekusch et al 28 the efficacy of a novel hemostatic wound dressing (Clo-Sur P.A.D) was assessed in 104 patients comparing conventional manual compression. Average time to hemostasis in the treatment group compared to the conventional group was 13.6 vs 20.3 minutes; P < .001.
One of the parameters in the present study was the use of sandbags where in nearly all patients (98.4%) with manual compression in the control group, the use of sandbags were necessary, while in only 1.6% of patients, using ChitoHem® topical hemostasis agent, the sandbag was used. Some studies have reported that sandbag was not effective in reducing vascular complications and bleeding incidence.29,30 Generally sandbags increased discomfort, and it was less tolerated by patients. They suggested the bed rest duration, hospital stay time, and health care costs were decreased, however the patient discomfort increased.29,30 In another study by Yilmaz et al, patients with a sandbag had an increased back pain and discomfort. Furthermore, sandbag did not reduce vascular complications rate. 31
Conclusion
ChitoHem® topical hemostasis powder used in treatment group was statistically superior at reducing the time to hemostasis and ambulation as well as the use of sand bags compared with manual compression in control group. ChitoHem® is simple and safe to be used on patients undergoing diagnostic coronary angiography and has European Community (CE) mark.
Footnotes
Authors’ Note
I am writing to you as the first author of this study. I am an academic member of staff in the Department of Medical Engineering, at the AmirKabir University of Technology, Tehran, Iran. At the same time, I am the Managing Director of ChitoTech Inc, the manufacturer of ChitoHem® hemostatic powder used in this study. I have participated in the conceiving, designing, and obtaining necessary funding for this project. Also I have had the overall managerial responsibility of the study. However, I am not a member of the institution where the study is carried, and other authors have been responsible for the present study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval for research
This is to confirm that the study entitled “a randomized controlled trial on the hemostasis of femoral artery using topical hemostatic agent” has been approved by the ethics committee of Shahid Rajaee teaching hospital, Iran University of Medical Science.
