Abstract
Identification of outpatients with high platelet reactivity (HPR) on antiplatelet treatment is an unmet need. The present study was conducted in healthy individuals (n = 50) and in outpatients with coronary artery disease (CAD) at a distance from the acute ischemic episode (aspirin group, n = 71; aspirin/clopidogrel group, n = 106). We studied the feasibility and the precision of whole blood multiple electrode aggregometry (MEA) after triggering platelet aggregation by arachidonic acid or adenosine diphospate (ADP). The MEA can be performed on whole blood within 2 hours after sample venipuncture. The threshold for the diagnosis of HPR is situated at 55 and 50 U for the arachidonic acid and ADP test, respectively. Frequency of HPR was 7% and 20% in aspirin and aspirin/clopidogrel groups, respectively. In 3.8% of patients in aspirin/clopidogrel group, combined HPR on aspirin and clopidogrel was found. In outpatients with CAD, use of MEA is feasible for the diagnosis of HPR.
Keywords
Introduction
Daily treatment with aspirin and clopidogrel is recommended in patients undergoing percutaneous coronary intervention (PCI) for the prevention of early stent thrombosis and recurrence of coronary artery thrombosis. 1–3 However, at least 10% of these patients present a major adverse cardiac event, ischemic stoke, or peripheral artery thrombosis while they receive dual antiplatelet treatment. 4 Failure of antiplatelet treatment to prevent recurrent thrombotic episodes, also called clinical resistance to antiplatelet treatment, is in part associated with biological inefficiency of aspirin or clopidogrel to inhibit platelet activation and aggregation induced by arachidonic acid or ADP, respectively. 5,6 Clinical resistance to antiplatelet drugs used at fixed recommended doses may also be related with a high residual platelet activation after treatment initiation. 7–12 Several laboratory assays, different in their concept, are sensitive to detect high platelet reactivity (HPR) on treatment with aspirin or clopidogrel. 5,6
Multiple electrode aggregometry (MEA) performed in whole blood is a point-of-care computerized assay based on the principle of impedance aggregometry. 13,14 Multiple electrode aggregometry and other assays have been mainly studied in trials that evaluated HPR on aspirin or clopidogrel treatment within the first month after the PCI. Prospective trials have established that HPR diagnosed with MEA is an independent risk factor for early stent thrombosis whereas profound inhibition of platelet aggregation induced by ADP is an independent risk factor for bleeding. 15,16 These studies assessed the predictive value of HPR in clopidogrel-treated patients and used the upper quintile of the area under the curve (AUC) values (calculated by the platelet aggregation curve) in order to establish the cutoff of MEA. 15,16
However, numerous patients while being on antiplatelet treatment manifest thrombotic episodes at a distance from the acute coronary syndrome and PCI. In this population of patients, the performance of assays for the diagnosis of HPR has not been adequately evaluated. Identification of outpatients who are at HPR during antiplatelet treatment by assessing their on-treatment platelet function is an unmet need in daily clinical practice.
The present study evaluated the feasibility and the precision of MEA performed in outpatients with coronary artery disease (CAD) receiving long-term treatment with aspirin and clopidogrel. The present study using whole blood MEA provides evidence for the profile of platelet responsiveness to aspirin and clopidogrel at a distance from the acute coronary episode or the PCI.
Materials and Methods
Control Group
The control group consisted of 50 apparently healthy volunteers who had no personal history of thrombotic or hemorrhagic episodes; they did not suffer from any other evident disease and were not taking any medication for at least 10 days before blood collection. Healthy volunteers were medical students or staff of the laboratory or the clinical departments. Healthy volunteers were recruited throughout the study period.
Groups of Patients
From October 2007 to January 2008, 177 outpatients, who suffered from acute coronary syndrome, were prospectively included in the study after giving informed consent. Patients receiving antiplatelet treatment were eligible for inclusion if hospitalization for acute coronary syndrome or PCI occurred in interval longer than 30 days. Compliance to the antiplatelet treatment was thoroughly ascertained by personal interview before entry in the study. Patients receiving long-term treatment with anti-inflammatory drugs or had taken an occasional anti-inflammatory drug during the last 10 days were excluded from the study. All studied patients had normal hepatic and renal function. Risk factors for atherothrombosis were evaluated with a standardized questionnaire. The study design did not allow to perform MEA before antiplatelet treatment administration.
Patients were stratified in to 2 predefined groups, according to antiplatelet treatment. The aspirin group consisted of 71 patients receiving aspirin 100 mg once daily. The aspirin plus clopidogrel group consisted of 106 patients receiving aspirin 100 mg and clopidogrel 75 mg once daily. Patients were prospectively evaluated for the major adverse cardiovascular events (acute coronary syndrome, ischemic stroke, and death from cardiovascular cause) within 90 days after inclusion in the study.
The study protocol was approved by the local investigational review board of Papageorgiou General Hospital, Thessaloniki, Greece, and was performed in compliance with the principles of good clinical practice and in accordance with the Declaration of Helsinki.
Blood Samples
Blood was drawn by venipuncture of an anticubital vein and was then collected into blood collecting tubes containing r-hirudin with a final concentration of 25 μg/mL (obtained by Dynabyte Medical, Munich), as recommended by the manufacturer of Multiplate. The first 5 mL of blood were discarded. Whole blood platelet aggregation with Multiplate Analyser was performed in less than 2 hours after blood collection.
Whole Blood Platelet Aggregation
Whole blood platelet aggregation was studied using the new generation impedance aggregometer Multiplate Analyzer (Dynabyte Medical, Munich, Germany). Briefly, the analysis takes place in a single use test cell, which incorporates 2 independent sensor units. After triggering platelet activation, by adding an agonist, the platelets stick on the sensor wires and enhance the electrical resistance between them. The instrument detects the impedance change of each sensor separately. Platelet aggregation in whole blood was triggered using arachidonic acid 0.5 mmol/L (ASPI test) or adenosine diphospate (ADP test) 6.4 µmol/L and was recorded for 6 minutes. A representative curve of platelet aggregation recorded by Multiplate Analyser is depected in Figure 1. The following parameters are retrieved by the instrument: Area under the aggregation curve quantifies aggregation response plotted against time and can be expressed with 2 different units: AU × minutes (arbitrary aggregation units) or U (units). A 10-AU × minute corresponds to 1 U. Herein, AUC will be expressed in U, as proposed by the manufacturer and referred in the most recent publications.
17,18
Aggregation, expressed in AU, quantifies the maximum height of the aggregation curve. Velocity of platelet aggregation, expressed in AU/min, quantifies the maximum slope of the aggregation curve.
In previous studies, the AUC has been used for the diagnosis of HRP in patients on clopidogrel treatment.
15,16
For conformity with the existing literature among the 3 parameters of MEA, only the AUC will be analyzed.
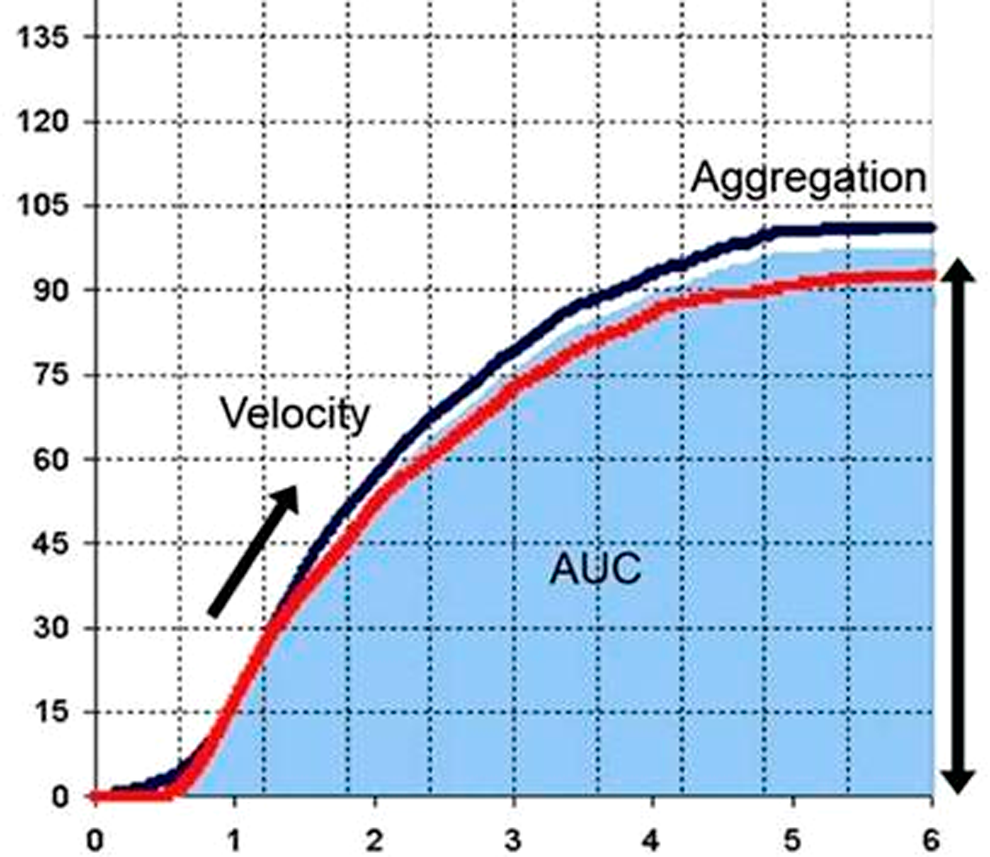
Representative curve of normal platelet aggregation recorded by Multiplate instrument. Parameters of platelet aggregation are depicted. AUC indicates area under the curve.
Precision Analysis
The intra-assay variability of whole blood MEA was evaluated by performing arachidonic acid and ADP test 10 times on samples obtained after 1 venipuncture from 1 healthy individual. In total, we studied samples from 10 donors.
The interassay variability of whole blood MEA was evaluated by performing arachidonic acid and ADP test on blood samples from 10 different donors at the inclusion to the study (Day 1), the day after the inclusion (Day 2), and the 7th day after the inclusion (Day 7). The interindividual variability of whole blood MEA was assessed by analyzing results obtained by performing arachidonic acid and ADP test once on the control group (n = 50).
To investigate the impact of the interval between venipuncture and MEA assessment, samples from 5 healthy individuals were obtained and assessed in 30, 60, 90, and 120 minutes after venipuncture. Coefficients of variation (CV) were calculated by the following formula: CV = mean/standard deviation (SD).
Statistical Analysis
Data are presented as mean ± SD. Student t test was used for the comparison of normally distributed values of MEA parameters in the studied groups, and Mann-Whitney test was used when values were non-normally distributed. A 2-sided P < .05 was considered significant. Upper normal limit (UNL) and lower normal limit (LNL) for each parameter of MEA were defined as mean ± 2 SDs of the values obtained by performing MEA in the group of healthy volunteers.
The inhibition of MEA in the groups of patients versus the control group was calculated as follows:
Percentage inhibition of AUC = (1-AUCpatient/AUCcontrol) × 100. The cutoff point for the diagnosis of HPR on aspirin was set at the LNL of the AUC in arachidonic acid triggered MEA. The cutoff point for the diagnosis of HPR on the clopidogrel treatment was the lower value of the AUC observed in the upper quintile of ADP triggered MEA measurements in patients treated with clopidogrel, as established by the prospective study of Sibbing et al. 15 Statistical analysis was performed using the SPSS software for Windows (SPSS Inc, release 10.0.1).
Results
Demographics of the Studied Population
The demographic and clinical characteristics of the studied groups are shown in Table 1. All outpatients included in the study were receiving antiplatelet treatment for an episode of unstable angina. The time elapsed since the acute coronary episode was significantly longer in aspirin group compared with aspirin plus clopidogrel group (52 vs 22 months, respectively; P < .05). Percutaneous coronary intervention with drug eluting stent was performed in 24% of patients in aspirin group and in 71% of patients in aspirin plus clopidogrel group. The frequency of hypertension, lipid disorders, statin use, and smoking was similar in the 2 groups of patients. The incidence of diabetes was significantly higher in aspirin group compared to aspirin plus clopidogrel group (Table 1). The mean age of the control group was significantly lower compared with both the groups of patients. The number of men was significantly higher in aspirin plus clopidogrel group compared with the aspirin group and the control group as well.
Demographics and Clinical Data of the Studied Population
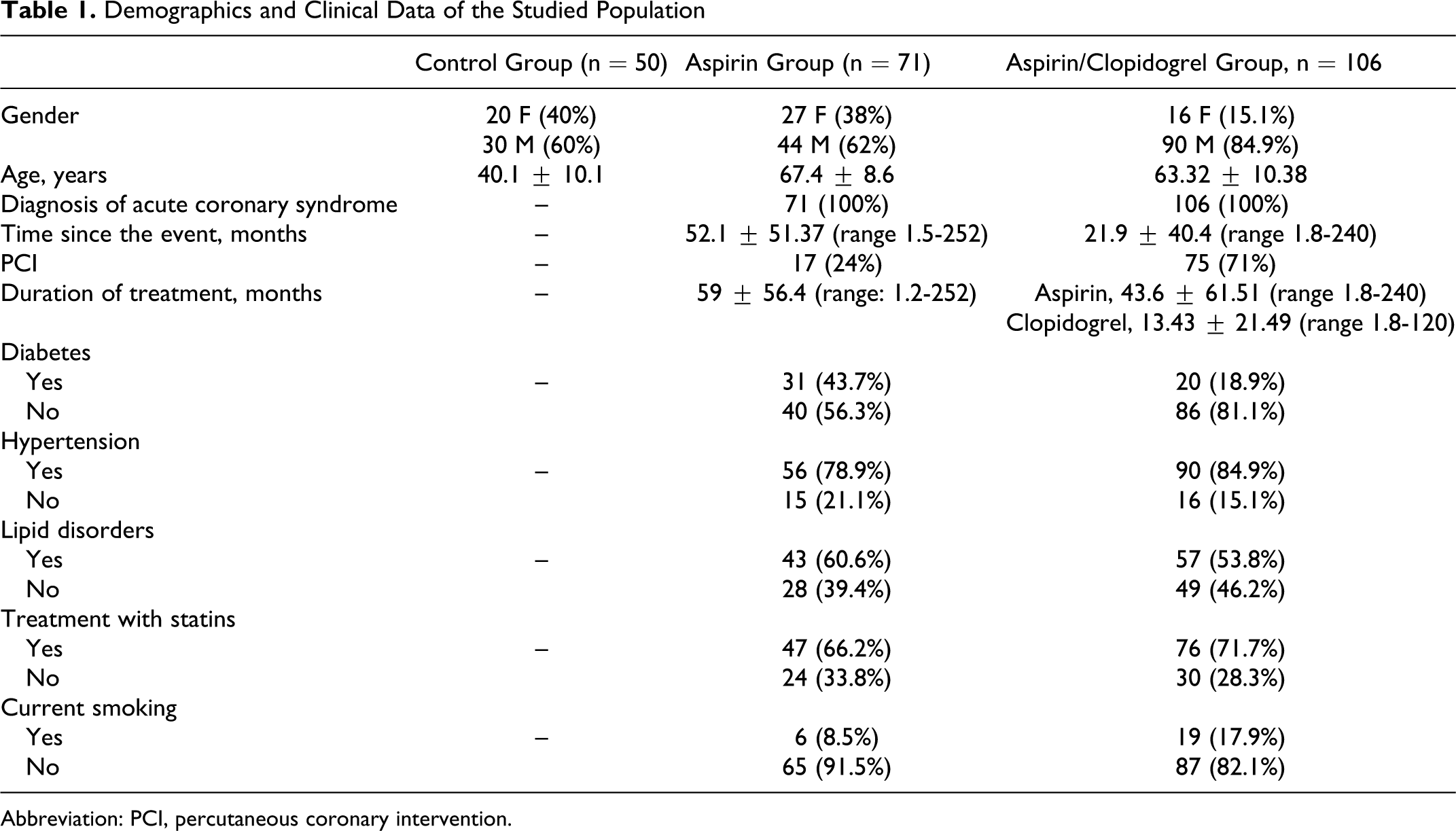
Abbreviation: PCI, percutaneous coronary intervention.
Precision Analysis and Normal Ranges of Whole Blood MEA
As shown in Table 2, values of the AUC of MEA assessed within 2 hours after venipuncture were stable. Assessment of MEA on 3 different days within 1 week showed low variability of the AUC values (Table 2). The intra-assay CV of MEA triggered by arachidonic acid and ADP was 12% and 7%, respectively. The CV of MEA induced by arachidonic acid or ADP performed in the same healthy individuals in 3 different days within 1 week were 7% and 8%, respectively.
Precision Analysis and Normal Ranges of the AUC of Whole Blood Platelet Aggregation Parameters Assessed With Multiplate in Healthy Individuals
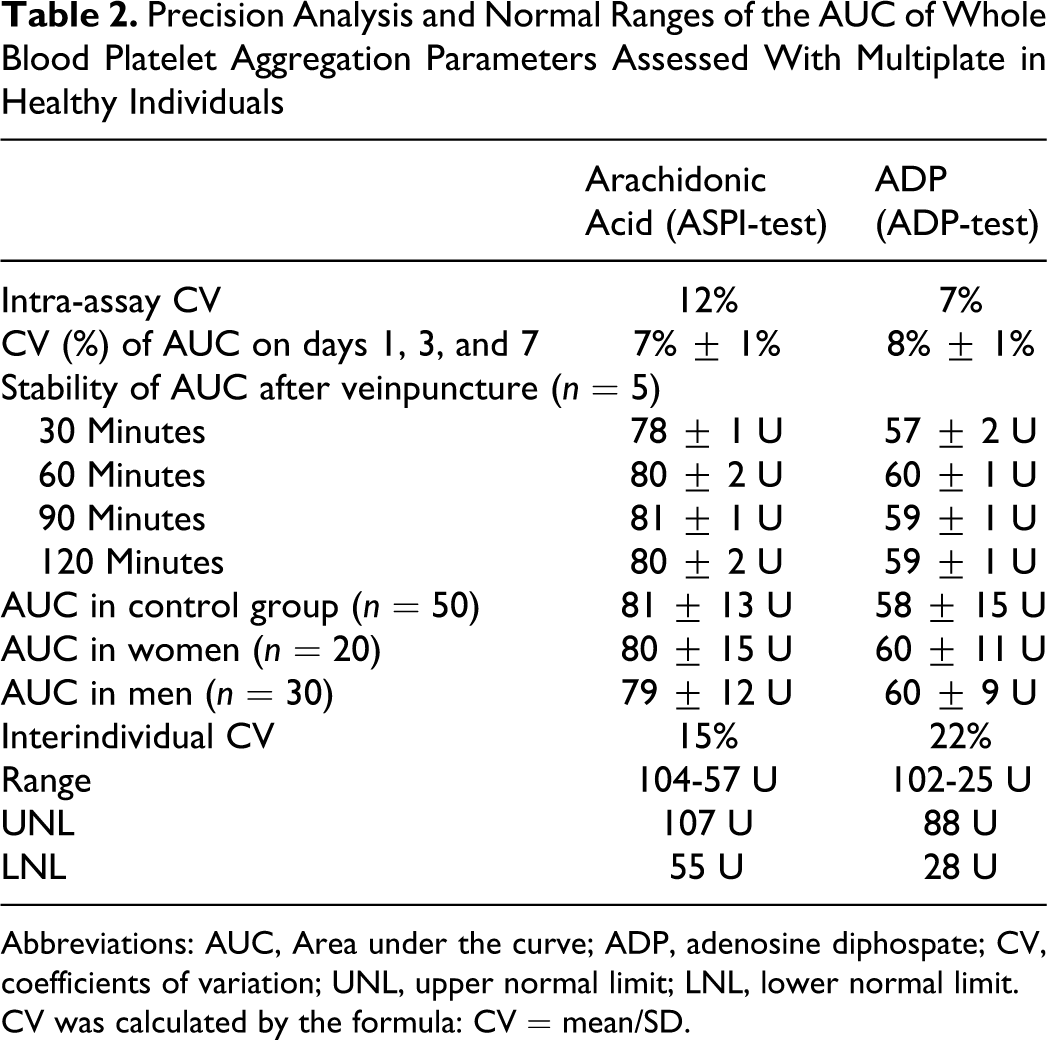
Abbreviations: AUC, Area under the curve; ADP, adenosine diphospate; CV, coefficients of variation; UNL, upper normal limit; LNL, lower normal limit.
CV was calculated by the formula: CV = mean/SD.
Normal values of MEA were determined in the group of healthy volunteers and are shown in Table 2. The interindividual variability of the AUC of MEA triggered by arachidonic acid or by ADP was 15% and 22%, respectively.
No significant correlation was found between the AUC of MEA and age (r = .1; P > .05). Area under the curve showed no statistically difference between men and women (Table 2). The UNLs and LNLs for the AUC of MEA induced by arachidonic acid or ADP are depicted in Table 2.
Whole Blood MEA of Patients Treated With Antiplatelet Agents
Profile of MEA Triggered by Arachidonic Acid
In aspirin group, the AUC was 80% lower compared with the control group (P < .001). In aspirin plus clopidogrel group also, the AUC was 80% lower compared with the control group (P < .005). Moreover, the AUC was 50% lower than that of the aspirin group (P < .005). None of the patients in aspirin plus clopidogrel group showed AUC values higher than the UNL (Figure 2, frame A).
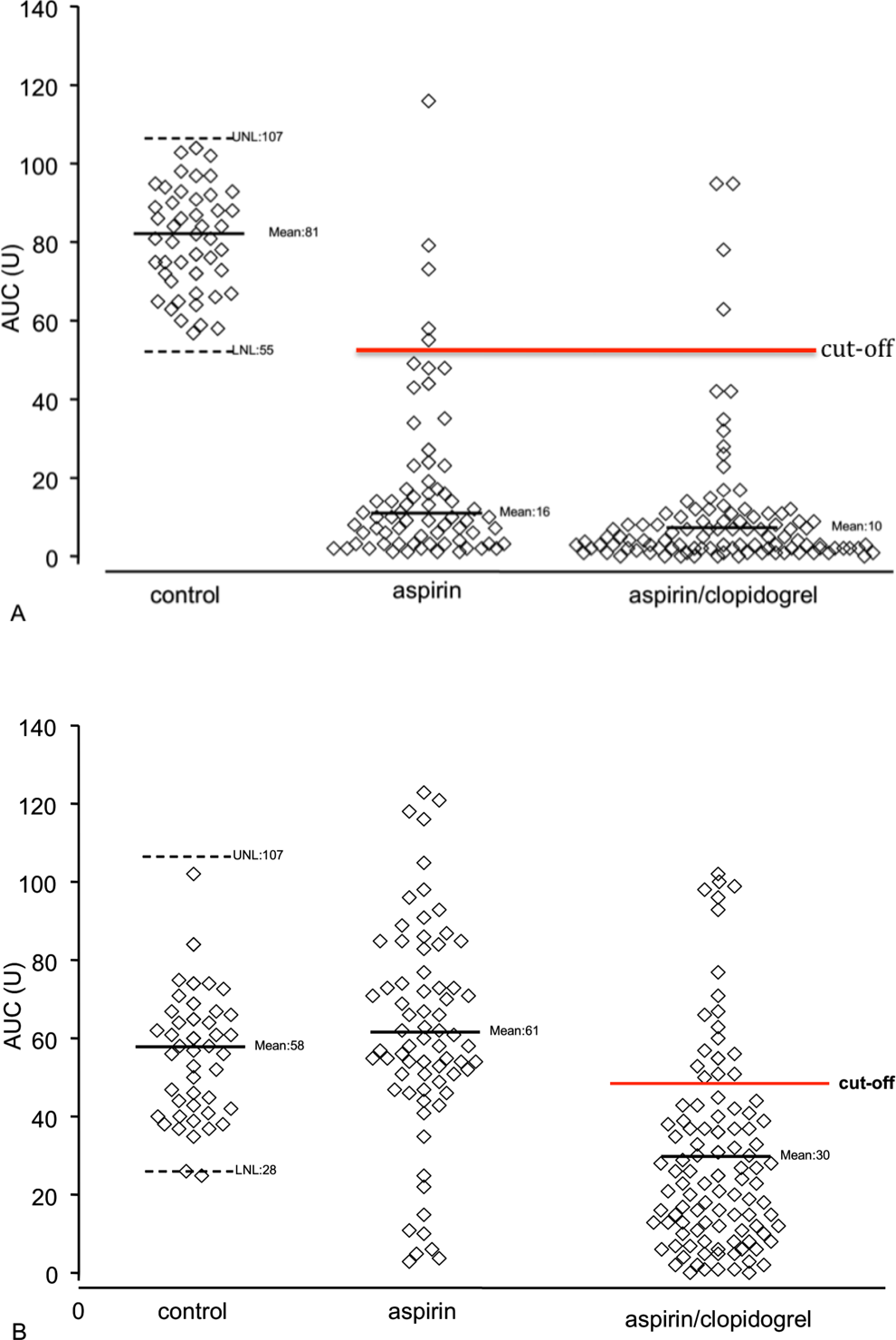
Distribution of the values of AUC after triggering whole blood platelet aggregation by aracidonic acid (frame A) or ADP (frame B) in healthy individuals and in patients treated with aspirin or asprin and clopidogrel. AUC indicates area under the curve; ADP, adenosine diphospate; UNL, upper normal limit; LNL, lower normal limit; Cutoff, 55 U is the cutoff value for the diagnosis of high platelet reactivity (HPR) on aspirin treatment and 50 U is the cutoff value for the diagnosis of HPR on clopidogrel treatment.
Profile of MEA Triggered by ADP
In aspirin group, the AUC was not significantly different compared to the control group. However, 4.8% of patients showed AUC values higher than the UNL. In aspirin plus clopidogrel group, the AUC was 49% lower compared to the control and to aspirin group (P < .001). None of the patients in the aspirin plus clopidogrel group showed AUC values higher than the UNL (Figure 2, frame B).
High Platelet Reactivity on Antiplatelet Treatment
High Platelet Reactivity on Aspirin Treatment
The cutoff for diagnosis of HPR on aspirin treatment was set at the LNL of the AUC in arachidonic acid MEA (55 U). One patient (1.4%) had AUC higher than the UNL. The majority of patients receiving aspirin showed more than 80% inhibition of arachidonic acid triggered MEA. High platelet reactivity on aspirin treatment was significantly more frequent in aspirin group than in aspirin plus clopidogrel group (7% vs 3.7%; P < .05).
Variability of Response to Clopidogrel
The cutoff point of the AUC of MEA for the diagnosis HPR on clopidogrel treatment was set to 50 U, which corresponds to the upper quintile of MEA as defined in Materials and Methods section. With this definition, 20% of patients were classified as HPR on clopidogrel treatment. Three patients from the aspirin plus clopidogrel group (3.8%) showed HPR on aspirin and clopidogrel.
In aspirin plus clopidogrel group, 21% of patients had residual MEA less than 10 U. After a 90-day follow-up, none of the patients presented any major adverse cardiovascular event or bleeding episode requiring hospitalization.
Discussion
In the present study, we used MEA to assess platelet response to aspirin or clopidogrel treatment in a cohort of outpatients with CAD that suffered from acute coronary syndrome or underwent PCI in an interval longer than 1 month from inclusion. A recently published meta-analysis showed that the studies assessing responsiveness to antiplatelet treatment focus on the acute phase of the coronary event and PCI by evaluating residual platelet reactivity in the first day of the event. 19 The same study demonstrated a significant association between increased residual platelet reactivity and recurrent cardiovascular events within an average follow-up of 4 months. In patients undergoing PCI, the incidence of major adverse cardiovascular events raises up to 15% within 1 year from angioplasty and is even higher either when follow-up is longer or in high risk patients such as diabetics. 20–23 High on-treatment platelet reactivity, as measured by aggregometry, is significantly related to the risk of ischemic events. 24 Limited number of studies have focused on the response to antiplatelet treatment in outpatients at a distance from the acute episode. 25,26 The available methods for the assessment of response to antiplatelet treatment have not been evaluated in outpatients.
In the first part of the present work, we studied the performance and the precision of whole blood MEA and we established reference values for the assessment of platelet reactivity and response to aspirin or clopidogrel treatment in outpatients with stable CAD. Then, we evaluated the profile of platelet response to arachidonic acid and ADP in outpatients treated with aspirin or dual treatment with aspirin and clopidogrel.
Assessment of platelet aggregation in whole blood does not require any particular preanalytical steps after blood sample collection. Multielectrode aggregation assay on a user-friendly instrument can be performed in whole blood within 30 minutes to 2 hours after venipuncture. Performing whole blood MEA within 2 hours from blood collection warrants stability of the samples and feasibility of the test, being in accordance with previous studies, which used the same method. 27 Whole blood MEA is appropriate for routine use in the outpatient setting. Multiple electrode aggregometry presents low intra-assay and interassay variability of the AUC, which is the most widely used parameter of platelet aggregation, for both reagents used (ASPI test and ADP test). These methodological characteristics of MEA allow laboratories not specialized in platelet studies to evaluate residual platelet reactivity in patients being on treatment with aspirin and clopidogrel.
Common demographic variables such as age and sex did not significantly modify whole blood MEA. In healthy individuals, the AUC of MEA remained stable within 1 week of observation. The interindividual variability of MEA ranged from 15% to 26%, whatever may be the trigger of platelet aggregation. Interindividual variability of a similar magnitude has also been reported for thrombin generation assays in platelet-rich or platelet-poor plasma as well as in minimal tissue factor triggered whole blood thromboelastography. 28–31 It seems that individual characteristics of platelet function and interactions with red blood cells and leukocytes influence platelet aggregation in whole blood. 32
The UNLs and LNLs of the AUC were calculated from the values observed in the control group in order to establish the cutoff point for the diagnosis of HPR on aspirin. This threshold was set at 55 U in arachodonic acid-induced MEA. The threshold for the diagnosis of HPR on clopidogrel was setup by the values in the higher quintile of ACU values in ADP triggered MEA in patients treated with clopidogrel and it was situated at 50 U. The cutoff values for the diagnosis of HPR was different for arachidonic acid- and ADP-induced MEA in patients treated with aspirin or aspirin and clopidogrel. Each agonists, arachidonic acid, or ADP induces a different degree of platelet aggregation, which is inhibited in a different extent by the specific antiplatelet agent (aspirin and clopidogrel, respectively). In more than 80% of aspirin-treated patients, platelet aggregation triggered by arachidonic acid was almost completely inhibited, whereas this was not the case in ADP-induced MEA for patients treated with clopidogrel.
High platelet reactivity was observed in 7% of patients treated only with aspirin. The incidence of HPR was almost halved in patients receiving both aspirin and clopidogrel. Clopidogrel-induced enhancement of the effect of aspirin might offer a biological explanation for the improved clinical outcome in patients with atherothrombosis who receive dual antiplatelet treatment. 33
The data presented herein show that in outpatients with CAD, the response to treatment with clopidogrel is characterized by a significant interindividual variability. The profile of response to aspirin and clopidogrel in the studied cohort of outpatients is similar to that observed in patients assessed shortly after PCI. 11,16,34,35 Sofi et al recently published a meta-analysis of studies investigating the resistance to clopidogrel. 19 This meta-analysis included 4564 patients with CAD who underwent PCI and were treated with clopidogrel. About 20% of patients had high residual platelet reactivity after triggering platelet aggregation by ADP. In 4 months follow-up, the risk of major adverse cardiovascular event was 5.6-fold higher in patients with HPR on clopidogrel treatment as compared to those classified as responders to clopidogrel. 19
In the present study, we found that applying the methodology described above, the threshold value of the AUC in ADP-induced MEA in outpatients with CAD at a distance from the acute event and PCI is situated at 50 U, being higher to that reported in patients tested near to the acute event. 15 Indeed, Sibbing et al assessed MEA before PCI in 1608 patients who received 600 mg loading dose of clopidogrel and used the upper quintile of the observed values to define the cutoff for the diagnosis of HPR. Prospective evaluation and receiver operating characteristic analysis defined the 46.8 U of the AUC as threshold for the diagnosis of HPR in patients who received loading dose of clopidogrel. 15 These findings allow to propose that the threshold for the diagnosis of HPR using ADP-induced MEA is influenced by the interval between the acute coronary syndrome or PCI. A recent post hoc analysis of the gauging responsiveness with VerifyNow P2Y12 assay—impact on thrombosis and safety (GRAVITAS) trial, which assessed (using the VerifyNow P2Y12 assay) the residual platelet reactivity within 12 to 24 hours after administration of clopidogrel loading dose and PCI, showed that 9% reduction of the threshold—compared to that used in the initial trial—resulted in improved association with lower risk of cardiovascular events. 36 Platelet reactivity is a dynamic phenomenon being markedly increased when assessed closely to the acute coronary event and is linked to inflammation and activation of blood coagulation. 37,38 Thus, for the optimal use of functional assays, the threshold for the diagnosis of HPR in patients on treatment with antiplatelet drugs should be contextualized and the interval from the acute coronary event is taken in consideration.
In outpatients with stable CAD, we distinguished 2 particular profiles of HPR. One was observed in 1 patient on aspirin monotherapy (1.4%) who showed on-treatment platelet aggregation higher than the UNL established for healthy volunteers. The other was observed in 3.8% of the aspirin plus clopidogrel group and was characterized by combined HPR after arachidonic acid- and ADP-induced MEA. The possible association of these 2 profiles with a higher thrombotic risk should be explored in a prospective study.
Of the 5 outpatients, 1 treated with dual antiplatelet treatment showed almost complete inhibition of ADP-induced MEA. Low residual platelet aggregation assessed with MEA has been shown to be associated with 2.5-fold increase of the risk of in-hospital bleeding in patients with CAD who undergo PCI. 16 The clinical relevance of very low platelet aggregation in outpatients on antiplatelet treatment is not known. A future study could explore whether this profile of inhibition of platelet reactivity is associated with premature interruption or reduced compliance to the treatment.
The present study was not powered to evaluate prospectively the predictive value of the threshold of ADP-induced MEA assay to evaluate the risk of ischemic episodes in outpatients with CAD. Noteworthy, no major cardiovascular or bleeding events occurred during a 90-day follow-up. This underlies the need for serial monitoring of platelet reactivity in patients diagnosed with high residual platelet reactivity, while they receive antiplatelet treatment.
In conclusion, evaluation of response to antiplatelet treatment in outpatients with CAD is a feasible strategy for the diagnosis of HPR on antiplatelet treatment. Whole blood MEA is a user-friendly and accurate method, not influenced by common demographic characteristics such as age and sex, and can be performed by laboratories nonspecialized in platelet studies. Moreover, ADP-induced MEA performed in outpatients is sensitive to detect very high or very low response to clopidogrel. The threshold of MEA for the diagnosis of HPR in the outpatients with stable CAD is higher to that used when the assay is performed closely to the acute coronary event and PCI. Whole blood MEA can be a useful tool for individualized optimization of antiplatelet treatment in patients with CAD.
Footnotes
Authors’ Note
The sponsor had no role in the design, the conduct of the study, and the analysis and the interpretation of the data. The manuscript was not prepared, reviewed, or approved by the sponsor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Multiplate device and reagents were kindly offered by Dr A. Kalatzis (Dynabyte).
