Abstract
Introduction
Bicuspid aortic valve (BAV) is the most common congenital heart defect, occurring in 1% to 2% of the general population. 1 One third of all patients with BAV will develop one of the serious complications associated with BAV, including aortic stenosis, aortic regurgitation, infective endocarditis, and aortic dissection, during their lifetime. 2 Bicuspid aortic valve is usually associated with dilation of ascending aorta, attributed to either aortic valvular dysfunction or intrinsic aortic wall pathology. 3 The abnormal opening mechanism of bicuspid valves also causes excessive postvalvular recirculation vortices, even in the absence of a significant transvalvular gradient, and these vortices are not only confined to the sinuses of valsalva but also extend into the mid-ascending tract. 4
Studies have indicated that platelet size reflects platelet activity indirectly and that it is measured using mean platelet volume (MPV). 5 Large platelets are more reactive and produce more prothrombotic factors.6,7 It has been shown that platelet activation occurs in patients with aortic stenosis. 8 However, to our knowledge, no study has evaluated platelet activation assessed by MPV in patients with normally functioning BAV. We hypothesized that impaired aortic flow and vascular dysfunction may induce platelet activation in patients with BAV, and MPV might be a sign of this indirect platelet activation. Therefore, the aim of this study was to measure MPV values in patients who had isolated BAV without significant valve dysfunction.
Materials and Methods
Study Population
Thirty-nine patients with isolated BAV and 25 age- and gender-matched healthy participants were included in the study. Patients with aortic valve stenosis and more than mild aortic regurgitation, previous cardiac surgery, concomitant dysfunction (>mild degree) of other heart valves, cardiomyopathy, left ventricular (LV) systolic dysfunction (LV ejection fraction [EF] < 50%), arrhythmias, Marfan syndrome, other congenital heart defects, hypertension, coronary artery disease, diabetes mellitus, a history of smoking, hypercholesterolemia, a history of renal or liver disease, a malignancy, hematological disorders, acute or chronic inflammatory disease, and patients who were using any cardiovascular drugs were excluded from this study. The study was approved by the local ethics committee and informed consents were obtained from the patients.
Echocardiography
Echocardiography was performed using a GE Vivid 7 system (GE Vingmed Ultrasound AS, Norten, Norway) with a 2.5 MHz phased-array transducer. The morphology of the aortic valve was defined in the parasternal short axis view. The 2-dimensional echocardiographic criteria for BAV included the partial or complete fusion of 2 of the aortic valve leaflets resulting in either a partial or complete absence of a functional commissure between the fused leaflets. 1 Left ventricular EF was calculated by the biplane Simpson method. Interventricular septum, posterior wall thickness, LV end-diastolic diameter, and LV end-systolic diameter were also measured. Aortic velocity (m/s) was measured from apical 4 chambers using continuous wave Doppler.
Aortic Dimensions and Elasticity Parameters
Two staff cardiologist made off-line measurements of the ascending aorta for transthoracic echocardiography (M.K. and I.H.T.). The diameter of the ascending aorta was measured on the M-mode tracing at 3 cm above the aortic valve. The systolic diameter was measured at the maximal anterior motion of the aorta, whereas the diastolic diameter was measured at the peak of the QRS complex on the simultaneously recorded electrocardiogram. Aortic dimensions were obtained from inner wall to inner wall.
For aortic elasticity, the following formulas were used
9
: Aortic strain (%) = (aortic systolic diameter − diastolic diameter) × 100/aortic diastolic diameter Aortic stiffness index = ln(systolic blood pressure/diastolic blood pressure)/aortic strain Aortic distensibility (cm2 dyn−1 10−6) = (2 × aortic strain)/(systolic blood pressure − diastolic blood pressure) Aortic elastic modulus (dyn cm−2 106) = (systolic blood pressure − diastolic blood pressure)/aortic strain
Laboratory Measurements
Venous blood samples were obtained from all participants between 8:30 and 10:00
Statistical Analysis
Data analysis was performed using SPSS for Windows 11.5 statistical software (SPSS Inc, Chicago, Illinois). Normal and continuous variables were described by means and standard deviations, and abnormal and continuous variables were described by median; categorical variables, however, were summarized as the number of patients and percentages. Statistical significance was accepted for P < .05. To determine the relationship between 2 variables, Pearson or Spearman correlation analysis was used. Student t test and Mann-Whitney U test were used to compare differences between continues variables. Chi-square test was used to compare differences between categorical variables.
Results
The clinical and echocardiographic characteristics of the 39 patients with BAV and 25 control participants are listed in Table 1 . There were no significant differences between groups with respect to age, gender, heart rate, blood pressure, functional status, and LV EF. Mild aortic regurgitation was present in 21% of the patients and mild mitral regurgitation was present in 7%. None of the patients had aortic velocity >2 m/s.
Basic and Clinical Characteristics of the Study Population
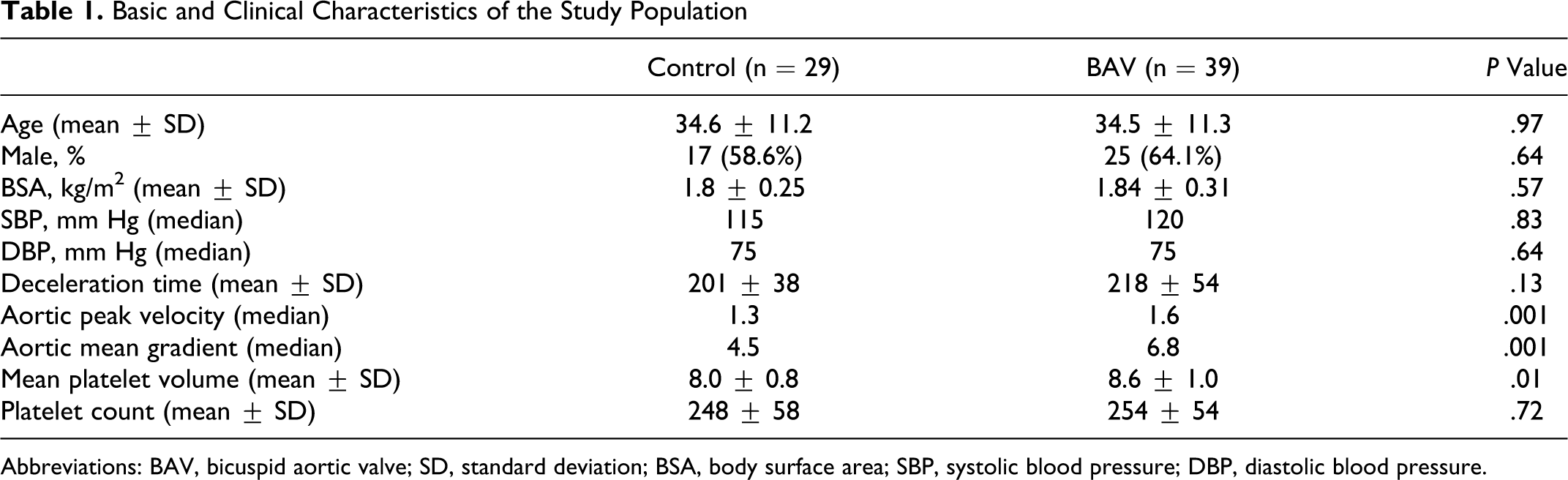
Abbreviations: BAV, bicuspid aortic valve; SD, standard deviation; BSA, body surface area; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Aortic strain and distensibility of the patients with BAV were lower compared to the participants in the control group (aortic strain: 3.52 vs 7.40, respectively, P: .0001; aortic distensibility: 1.13 ± 0.76 vs 2.66 ± 1.53 cm2 dyn−1 10−6, respectively, P: .0001). Aortic stiffness index and aortic modulus of the patients with BAV were greater in comparison to the participants in the control group (aortic stiffness index: 12.81 ± 8.00 vs 6.60 ± 3.13, respectively, P: .001; aortic modulus: 44.48 ± 25.59 vs 23.49 ± 11.57 dyn cm−2 106, respectively, P: .0001; Table 2 ).
Aortic Elastic Parameters Between Groups
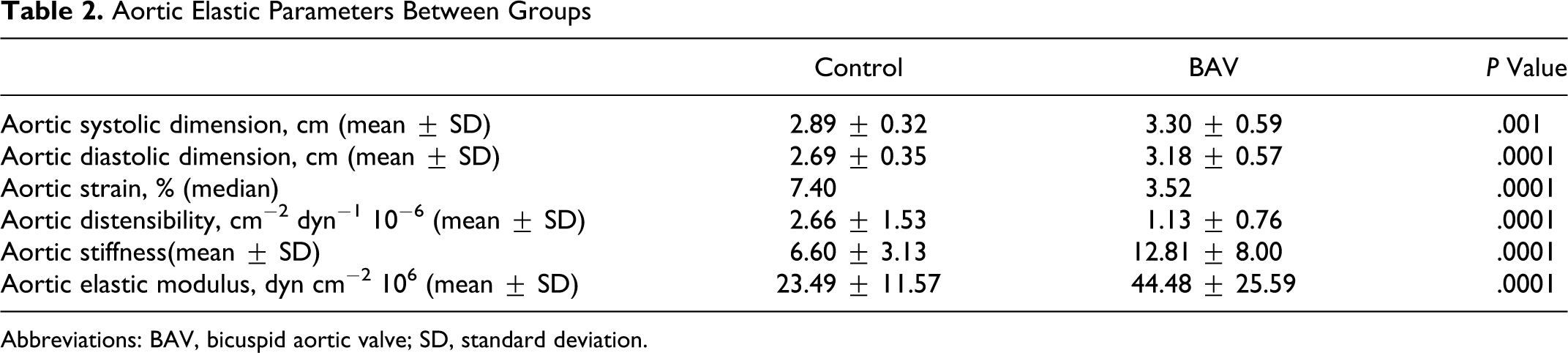
Abbreviations: BAV, bicuspid aortic valve; SD, standard deviation.
Compared to the control group, MPV value was significantly higher in patients with BAV (8.6 ± 1.0 vs 8.0 ± 0.8, P: .01; Table 1 and Figure 1 ). In the correlation analysis, while there was a significant correlation between aortic stiffness (Figure 2 ) and aortic elastic modulus (Figure 3 ) with MPV measures (r: .32 and P: .01; r: .37 and P: .003, respectively), there were no correlation with aortic strain and distensibility (r: −.17 and −.14, respectively).
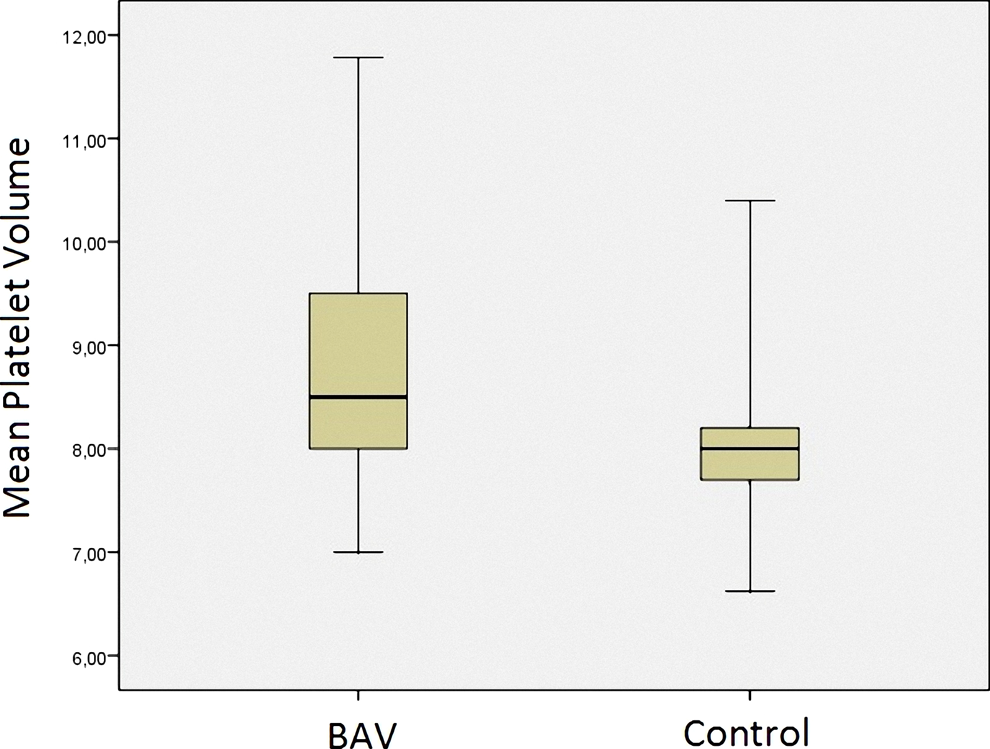
Box plot for mean platelet volume between patients with bicuspid aortic valve and healthy participants.
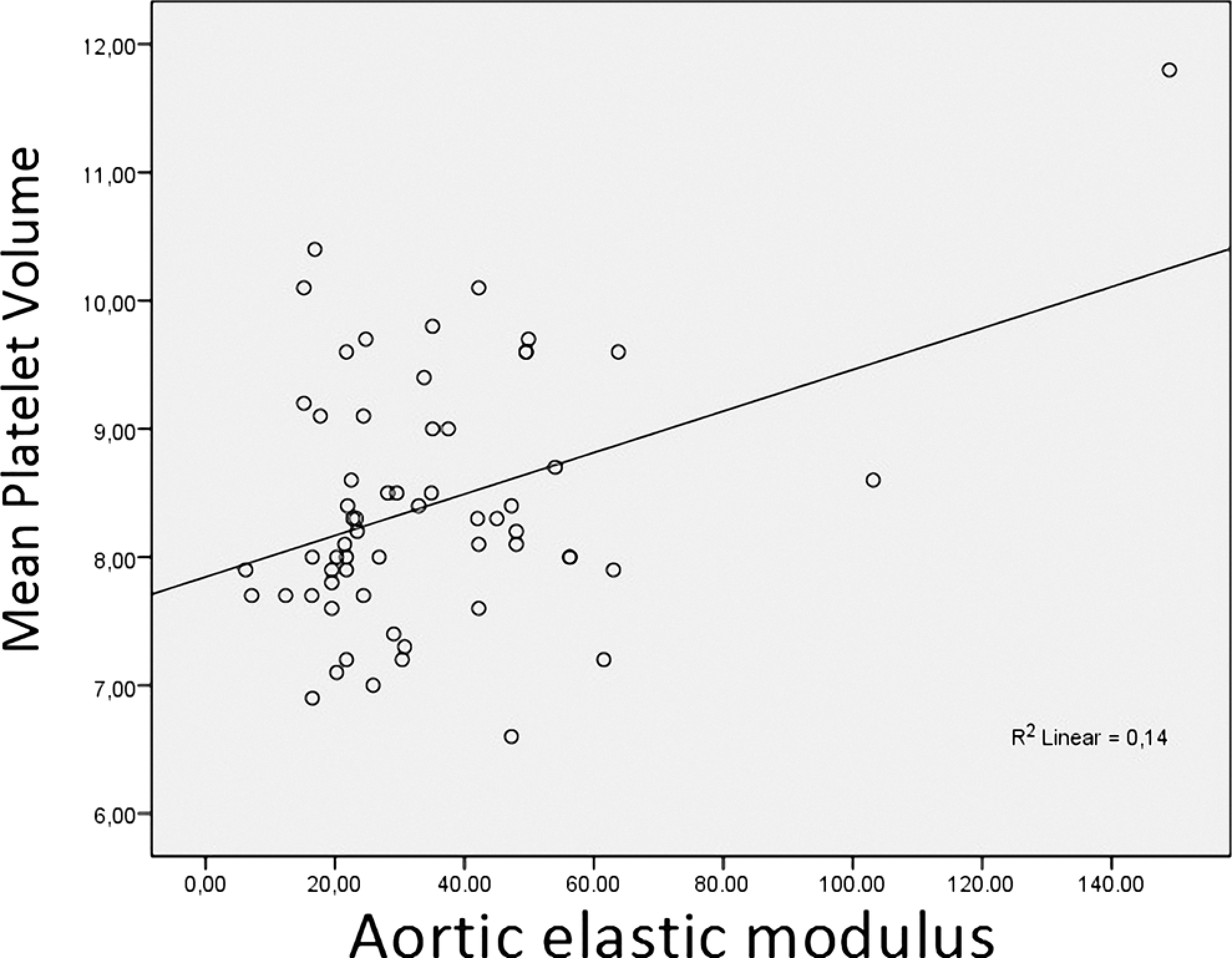
Correlation plot for mean platelet volume and aortic elastic modulus.
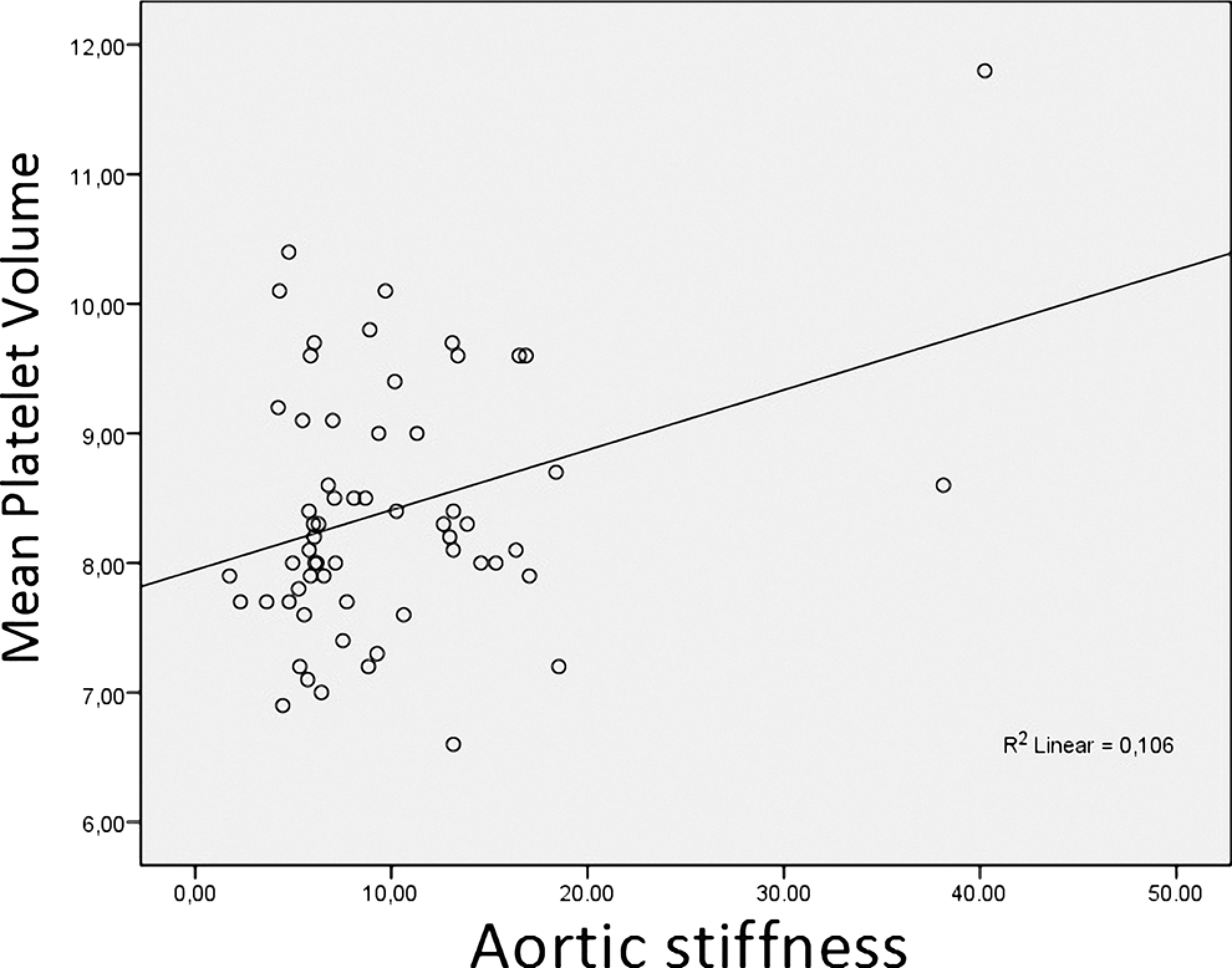
Correlation plot for mean platelet volume and aortic stiffness.
Discussion
To the best of our knowledge, the present study is the first to assess MPV values in BAV. Our results indicate that patients with BAV tend to have an increased platelet activation indirectly assessed by MPV. Bicuspid aortic valve may account for considerable rates of morbidity and mortality compared with other congenital cardiac abnormalities. The pathogenesis of BAV is unclear. Genetic abnormalities, including neural crest abnormalities, deficiencies in endothelial-derived nitric oxide synthase, fibrillin-1 deficiencies, increased matrix metalloproteinase (MMP) levels, and enhanced hemodynamic stress on the ascending aortic wall as a result of turbulent flow over the malformed valve, are implicated in the development of BAV disease and associated aortic abnormalities. 3 Recent studies have demonstrated that the abnormal opening mechanism of bicuspid valves causes excessive postvalvular recirculation vortices, even in the normal functioning valve. 4 It has been reported that shear stresses in turbulent flow as a result of stenotic valves induce platelet activation. However, no study has investigated the relationship between BAV without aortic stenosis and platelet activation.
Mean platelet volume is a morphological parameter of platelets for showing newly produced platelets. It demonstrates the function of platelets indirectly and assessed easily by a routine hematological analysis. Increase of MPV may occur as a result of the synthesis of prothrombotic and proinflammatory agents in platelets, degranulation of α-granules, and release of reactive platelets from stores. Mean platelet volume is a surrogate marker of platelet function and is associated with a variety of prothrombotic and proinflammatory disease states.10,11 Gasparyan etal 12 showed that high values of MPV were more frequent with hypertension in patients with rheumatoid arthritis, suggesting that MPV reflects enhanced vascular risk. In previous studies, increased platelet activation has been demonstrated in rheumatic mitral valve disease. Yetkin etal 13 have shown that the patients with severe mitral stenosis and sinus rhythm have elevated platelet activation and endothelial dysfunction, measured by plasma b-thromboglobulin, P-selectin and von Willebrand factor (vWF), and percutaneous mitral balloon valvuloplasty results in decreased platelet activity. It has been suggested that increased valvular pressure enhances endothelial synthesis or the release of vWF, which could contribute to the thrombotic tendency in patients with altered hemodynamics. Recently, Varol etal 8 reported that MPV was elevated in patients with aortic stenosis. The etiology was calcific and rheumatic aortic stenosis. In a recent study, MPV and platelet distribution weight were assessed, and these parameters were found to increase in patients who had aortic valve sclerosis without significant valve dysfunction. 14 Additionally, Wang etal 15 found that the elevated MPV was positively correlated to arterial stiffness in the apparently healthy population that had participated in the general health examination. Our study was different from these studies in that it enrolled patients who had pure BAV without valve dysfunction and it excluded those who had coronary artery risk factors that could affect platelet activity, such as hypercholesterolemia, hypertension, obesity, diabetes mellitus, and history of smoking. Several mechanisms might be responsible for increased MPV in our patients. One of them is the abnormal opening mechanism of the bicuspid valves, which causes excessive postvalvular recirculation vortices, even in the absence of significant transvalvular gradient. 4 Also, several studies have demonstrated that shear stress in turbulent flow induces platelet activation. The second mechanism contributing to increased platelet activity might be endothelial dysfunction. It was shown that young men with BAV and dilated proximal aorta manifested systemic endothelial dysfunction, increased carotid-femoral pulse wave velocity, and higher plasma levels of MMP2, suggesting generalized vascular and humoral abnormalities. 16 Systemic endothelial dysfunction results in increased activation of platelets. 17 Additionally, Phillippi etal 18 found dysregulated metallothionein genes in BAV-associated tissue. Metallothionein is a cysteine-rich protein that binds zinc and an array of other metal ions and is a major antioxidant. Increased oxidative stress contributes to the pathophysiology of inflammation. Yamasaki etal 19 reported that arterial stiffness was associated with platelet activation by measuring platelet surface expression of P-selectin and platelet–neutrophil complexes. In our study, the correlation between MPV and aortic stiffness in patients with BAV is compatible with these results. It has been shown that increased MMP activity due to activated platelets results in a reduction in arterial elasticity by degradation of elastic fiber. 20 So these insults might have a direct role in the increase of MPV in our patient group.
This study had some limitations due to the small sample of patients with BAV. However, this BAV group was exceptional in that none of the patients had valvular stenosis and no more than mild regurgitation. This was a cross-sectional study, and we were not able to do any follow-up; therefore, we were not able to know whether increased MPV predicts thromboembolic events in our patient group. Since, in our study, we aimed to investigate MPV values in isolated patients with BAV, we did not extend our blood sampling to other plasma vascular markers and platelet activation markers.
As a conclusion, these findings indicated that BAV is associated with elevated MPV values. The mechanisms may be due to intrinsic aortic wall pathology in the thoracic aorta, systemic endothelial dysfunction, and abnormal opening mechanism of bicuspid valves. Further studies are needed to establish the clinical significance of increased MPV in patients with BAV.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
