Abstract
Keywords
Introduction
In patients with end-stage renal disease (ESRD), a predisposition to bleeding is frequently seen due to the disturbance in platelet adhesion and aggregation. 1 Mucosal, serosal, or cutaneous bleeding may occur in these patients.2,3 Although the main cause of hemostasis disturbance is defective platelet function, anemia, dialysis, and drug accumulation due to a decrease in the clearance and use of heparin during dialysis constitute the other causes.4,5
Various tests have been utilized to evaluate the disturbance of hemostasis in patients with ESRD. Skin bleeding time (SBT) is commonly used as a screening test for assessing primary hemostasis. As an in vivo method, the SBT was first described by Duke in 1910 and subsequently modified and standardized6,7 by Ivy et al 8 in 1941. More recently, the template method was introduced. The template method has further standardized the incision technique using a surgical blade in a holder, which cuts 1 mm deeper than the template is thick. Despite some disadvantages of the SBT (ie, it is a painful procedure and may differ according to the administrator, the method used, the quality and the temperature of the skin as well as the length and direction of the incision), it has some advantages; it is a rapid, easily performed, inexpensive, and specific test that does not require the use of additional equipment. Indeed, there appears to be agreement within the nephrology community that bleeding time is probably the best laboratory test with which to predict clinical bleeding in patients with renal failure.9,10 The bleeding tendency appears to be associated with the duration and severity of the uremic syndrome. 11 Dialysis may partly reverse the disturbance of hemostasis due to uremic toxins, but full reversion is not possible. However, dialysis itself may contribute to bleeding. 4
In this trial, we evaluated the SBT in patients presenting to our hospital with uremic symptoms (pericarditis, metabolic acidosis, uremic encephalopathy, and pulmonary edema) induced by chronic renal failure in whom hemodialysis (HD) was performed for the first time. We also examined the effects of HD and uremia on this test and investigated its effectiveness in predicting the hemorrhagic complications before the implementation of invasive procedures in patients with uremia.
Materials and Methods
This study was conducted in accordance with the Declaration of Helsinki. Twenty-nine consecutive patients with chronic kidney disease (CKD) who presented to the emergency department and outpatient clinic of the nephrology department in the Yuzuncu Yil University (YYU) Medical Faculty with symptoms of uremia (pericarditis, severe metabolic acidosis, encephalopathy, and pulmonary edema) induced by chronic renal failure and treated with HD for the first time (13 men, 16 women; mean age 59.7 ± 18.1) were enrolled in this trial. Bicarbonate HD was performed to all patients using a polysulfone membrane of 1 m2 in the first dialysis and 1.6 m2 in the second dialysis for 2 and 4 hours, respectively. Anticoagulation was not performed in the first dialysis. In the second dialysis, an average of 5000 unit of standard heparin was used for anticoagulation. Patients with a tendency for hemorrhagic diathesis and individuals using drugs that affect platelet functions (such as aspirin and nonsteroidal anti-inflammatory drugs [NSAIDs]) were excluded from this trial. None of the patient needed blood transfusion during the study period.
Blood samples were obtained before the initiation of first dialysis and after the second dialysis; the samples were analyzed for complete blood count, venous blood gases, urea, and creatinine. Serum urea and creatinine levels (spectrophotometric method, Roche Diagnostics, Germany) were measured on a Hitachi Modular P 800 chemistry analyzer (Hitachi, Germany). The hematological analysis was analyzed by Coulter LH 750 hemogram device (Beckman Coulter, Fullerton, California). The SBT test was performed using a disposable Simplate R device (Organon Technica Corporation, Durham, North Carolina) that produces 2 standardized incisions on the volar aspect of the forearm. A sphygmomanometer cuff around the arm was inflated to 40 mm Hg standardized venous pressure. The time required for bleeding to cease was determined by the blotting of blood emerging from the wounds with filter paper. 12 The normal range in our laboratory is 3 to 8 minutes.
Statistical Analysis
All the obtained data were coded and transferred to a computer program. Statistical analysis was performed using the SSPS 16.0 program. The paired and unpaired samples t test and Pearson correlation analysis were used for data comparison. The results were indicated as mean ± standard deviation, and values with P < .05 were regarded as significant.
Results
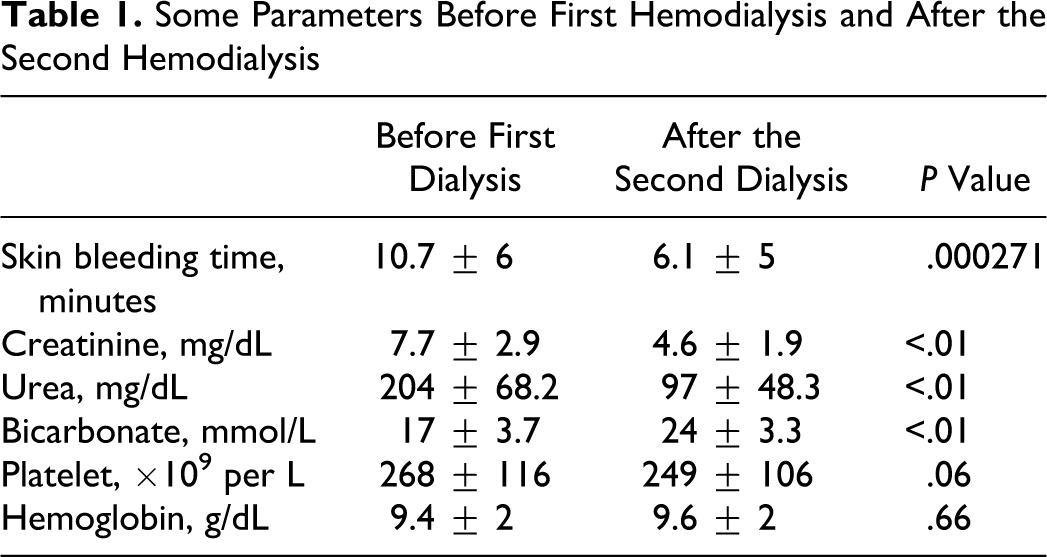
The SBT, measured after the second dialysis, was significantly shorter when compared to predialysis values (P < .05). Skin bleeding time was within the normal range in 15 patients; in the other 15 patients, it was elongated before the first HD. In 13 of 15 patients, whose SBT was elongated, SBT returned to normal limits after the second HD. Skin bleeding time and some biochemical parameters before the first and after the second HD are shown in Table 1. Correlation analysis between the SBT and urea, creatinine, hemoglobin, platelets, and bicarbonate showed no correlation.
Some Parameters Before First Hemodialysis and After the Second Hemodialysis
Discussion
In this trial, we found that the SBT shortened after the second dialysis in patients presenting with symptoms of uremia induced by CKD and in whom dialysis had been performed for the first time. We could not find any correlation between the SBT and urea, creatinine, platelet, hemoglobin, or bicarbonate.
The tendency to bleeding is common in patients with ESRD. Although the cause of uremic bleeding is not completely understood, altered platelet function appears to be of crucial importance. 1 Numerous defects in platelet function have been described in patients with uremia: defective binding of the factor VIII complex to platelets or defective binding of platelets to vessel wall subendothelium by the factor VIII complex, decreased platelet–vessel wall interaction, platelet storage pool deficiency, and defective fibrinogen binding. In addition, this bleeding tendency may depend on platelet inhibition by plasma metabolites (eg, urea, guanidinosuccinic acid, and phenolic acid) and increased levels of PTH. 13 Dialysis may partly reverse these defects. However, complete resolution is not possible and even dialysis itself may contribute to bleeding. 4 Di Minno et al 14 showed that aggregation and secretion defects related to impaired arachidonic acid release from platelet phospholipids as well as a storage pool defect in uremia and the first is improved with dialysis; the second is not. Therefore, they reported that the platelet dysfunction in uremia is multifaceted.
Zupan et al 15 conducted a trial on 34 patients on routine HD program and evaluated the SBT. In vitro bleeding time was measured by PFA, and platelet aggregation was measured using an APACT 2 aggregometer. The authors found that SBT elongated in 20% of patients. They did not find any difference between the patient and control groups in terms of the SBT. In this trial, the effect of HD on SBT was not evaluated.
In a trial on routine patients with HD conducted by Remuzzi et al, 16 the SBT was considerably disturbed when compared to control group. However, no correlation was found between the biochemical parameters and SBT. Besides, no improvement was found in the bleeding time after HD. Therefore, it was stated that certain undialyzable substances play an important role in uremic bleeding. Also in our study, the bleeding time did not correlate with the blood urea nitrogen or creatinine per se. The reason for this is unknown and further studies are needed to clarify the relationship between bleeding time and indices of renal dysfunction.
In another trial performed on 20 prevalent patients with HD, the SBT was significantly longer when compared to values obtained for the control group. Furthermore, a significant correlation was found between creatinine and SBT. However, the authors concluded that SBT poorly correlated with platelet function using tests involving platelet contractile force, clot elastic modulus, and von Willebrand factor antigen. 17
In a trial conducted in 48 patients with stage-4 CKD, it was found that the SBT was lengthened in correlation with the patient age, severity of renal failure, and hematocrit in 25 patients. 18
In the above literature, there are studies evaluating the bleeding time in patients with HD, but these studies are conducted in patients undergoing routine HD. Our trial, on the other hand, was conducted on patients with uremia who were scheduled to undergo dialysis for the first time. Within the limitations of our study, an alternative in vitro test to replicate the SBT has not been identified and there was no control group.
In our trial, another interesting finding was that the bleeding time did not correlate with the hematocrit in patients with uremia induced by CKD and treated with HD for the first time. Our patient’s mean hematocrit was less than the National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative target 19 range of 33% to 36%. Previous studies have demonstrated that the bleeding time begins to normalize once the hematocrit20,21 reaches 30%. Nevertheless, in our study, the mean hematocrit in the dialysis participants was 28%. This finding is not grossly abnormal.
Conclusion
Skin bleeding time could reveal the uremic thrombopathy and beneficial effect of dialysis in the patients who presented with uremic symptoms and were treated with HD for the first time. We suggest that SBT may be an appropriate test for the evaluation of hemostasis disturbance in patients with uremia and prediction of the bleeding risk before invasive procedures, despite its certain disadvantages.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
