Abstract
Generic active pharmaceutical ingredients (APIs) have been commonly used in Brazil, since 1999, but most of them are synthetic and small molecules. Recently, a large number of generic enoxaparins were introduced into the market raising concerns related to product-to-product interchangeability, efficiency, and drug counterfeiting. These drugs are produced from biological sources and their production involves complex procedures and purification processes. The present article evaluates several generic enoxaparins, structurally and pharmacologically, and compares them with the branded products. Structural analysis showed that the generic products are, indeed, quite similar to the branded products, however, this similarity cannot be extended to their pharmacological activities. The results showed that generic products must go through extensive structural, pharmacological, and clinical evaluation in order to assess their quality, efficacy and, ultimately, avoid drug counterfeiting before clinical use. Variation was also observed between the branded products, showing that such drugs must be at constant surveillance.
Introduction
Generic drugs are usually produced and distributed without patent protection. They must contain the same active ingredients as their original formulation, meaning that they must be identical or within an acceptable bioequivalent range when compared to the original formulation. The US Food and Drug Administration (FDA) considers generic drugs identical in dose, strength, route of administration, safety, efficacy, and intended use. 1
Enoxaparins are produced by the chemical depolymerization of unfractionated heparin (UFH), leading to a unique agent, which shares structural features with the parent compound and possesses production fingerprints due to the nature of the chemical reaction that it underwent. Therefore, the nature of the parent heparin and the depolymerization reaction are crucial for the quality and efficacy of the final product.2,3
In 2009, US enoxaparin sales were around US$2.5 billions. Due to this tremendous market and due to the fact that the US patent protection has expired, a large number of pharmaceutical industries are trying to obtain FDA approval for generic enoxaparins.
Different from the US market, generic enoxaparins are being used in Brazil for several years. The increase in enoxaparin supply may be beneficial considering treatment costs for hospital and patients. Nevertheless, it also raises concerns about drug safety, efficiency, and counterfeiting, demanding the development of detailed regulatory guidelines for the regulation of such products.
For these reasons, generic enoxaparins currently marketed in Brazil were compared structurally and pharmacologically to the branded products using a variety of analytical and biological assays in order to test whether they should be considered similar and, therefore, interchangeable.
Materials and methods
Heparin and Low-Molecular-Weight Heparins
Generic enoxaparins (∽100 IU/mg) were obtained from Eurofarma Laboratórios Ltda (São Paulo, Brazil), Blausiegel Farmacêutica (Cotia, Brazil), Aspen Pharma (Rio de Janeiro, Brazil); branded enoxaparins (∽100 IU/mg) were obtained from Sanofi-Aventis (São Paulo, Brazil) and UFH (∽180 IU/mg) from Gentium SpA (Villa Guardia Como, Italy). Enoxa 1 and 5 are branded products and Enoxa 2-4 are generic products.
Molecular Weight Determination
The molecular weight (MW) determinations were made using gel permeation chromatography (GPC) and a high-pressure liquid chromatography (HPLC) system (Waters 845, Waters corporation, Milford, MA) equipped with software designed for polymer analysis (Millennium 2000, Waters corporation, Milford, MA). The HPLC system consisted of a computer (Digital, Pentium III, Waters corporation, Milford, MA), an LAC/E interface module, two 510 HPLC pumps, a 712 WISP autoinjector, an R401 differential refractometer, and a 484 tunable absorbance detector (Waters). The ultraviolet (UV) and the refractive index (RI) detectors were linked in series, with the outlet of the joint columns (TSK G3000SW and TSK G2000SW) attached to the UV detector. The MW profile was determined using a method reported elsewhere. 4 Analysis of heparin preparations were carried out by injecting 20 μL aliquots of a sample solution (10 mg/mL in 0.3 mol/L sodium sulfate) into the GPC-HPLC system. The flow rate for the mobile phase (0.3 mol/L sodium sulfate) was 0.5 mL/min and the run time was 65 minutes. UV determination was made at 234 nm at room temperature. Following each run, the elution profile of each sample was analyzed by the narrow range calibration (NRC) method. 5 Calibration of the GPC-HPLC system was performed in a similar manner using narrow range calibrators (10 mg/mL calibrators in 0.3 mol/L sodium sulfate).
Nuclear Magnetic Resonance
For nuclear magnetic resonance (NMR) experiments, the samples were deuterium exchanged by repeated dissolution in D2O and freeze-drying. Spectra were obtained from solutions in D2O at 30°C, using TMSP as standard (δ = 0). All spectra were obtained with a Bruker 400 MHz AVANCE III NMR spectrometer with a 5 mm inverse gradient probe. One-dimensional (1D) and 2D signal assignments were performed using 1 H-(zg and zgpr) and heteronuclear single-quantum coherence (HSQC; hsqcetgpsi) programs. Heteronuclear Single Quantum Coherence spectra were acquired using 8 to 16 scans, respectively, per series of 2 K × 512 W data points with zero filling in F1 (4 K) prior to Fourier transformation. 6
Bioassay and Anticoagulant Activity of Generic Enoxaparins
The agents were supplemented with pooled normal human plasma (NHP) and tested in a concentration range of 0 to 100 μg/mL. Active partial thromboplastin time (APTT), HEPTEST, and thrombin time (TT) were performed to determine the anticoagulant activity of these agents using commercial kits (Helena Laboratories Corp, Beaumont, Texas). Using commercial kits (Hyphen Biomed, Neuville-sur-Oise, France), amidolytic antifactor Xa (FXa) and antifactor IIa (FIIa) activity was also measured in order to determine the antiprotease profile of these agents. For the clotting assays, NHP samples supplemented with enoxaparins were incubated with the specified reagent for each clotting assay at 37°C. The clotting time was measured using an ACL 300Plus. Anti-FXa activity was measured by incubating the supplemented plasma samples for 2 minutes at 37°C with FXa (5.0 μg/mL) and a chromogenic substrate specific for FXa, spectrozyme Xa (2.5 μmol/L) was added and the optical density at 405 nm/min was determined. Similarly, the anti-FIIa was measured by incubating the supplemented plasma samples for 1 minute at 37°C with FIIa (5 U/mL) and a chromogenic substrate specific for thrombin, spectrozyme TH (1.0 μmol/L) was added and the optical density at 405 nm/min was determined. The Anti-Xa and Anti-IIa results were expressed in percentage of inhibition. The neutralization of the anticlotting and antiprotease activities of the enoxaparins was evaluated after incubation with 0.1 U/mL heparinase 7 at 30°C as described. 8 The anticlotting and antiprotease assays were performed in triplicates at different days.
Results
Molecular Weight and its Distribution
Average MW values are shown in Table 1 . The data revealed that the tested agents possess quite similar average MW values, ranging from 3738 Da up to 4217 Da, all values being within the accepted range for enoxaparin preparations. 9 Interestingly, the sample with the largest MW also showed the highest polydispersity (1.23), and the one with the lowest MW presented the lowest polydispersity (1.16).
Molecular Weight and Polydispersity of Generic Enoxaparins
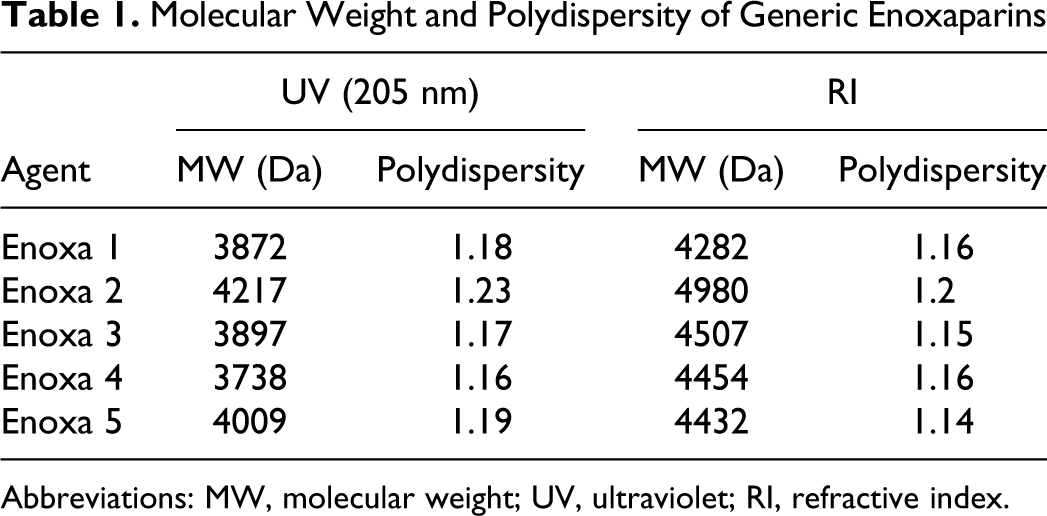
Abbreviations: MW, molecular weight; UV, ultraviolet; RI, refractive index.
Also the MW distribution values are rather alike with the majority of their components on the 2.5 to 5 kDa range (Table 2 ). The fact that Enoxa 2 has components with MW greater than 12 kDa is in agreement with the average MW determination and explains its largest value. According to the enoxaparin monograph, the MW distribution values for all agents are within the acceptable range and percentage.
Molecular Weight Distribution of Generic Enoxaparins
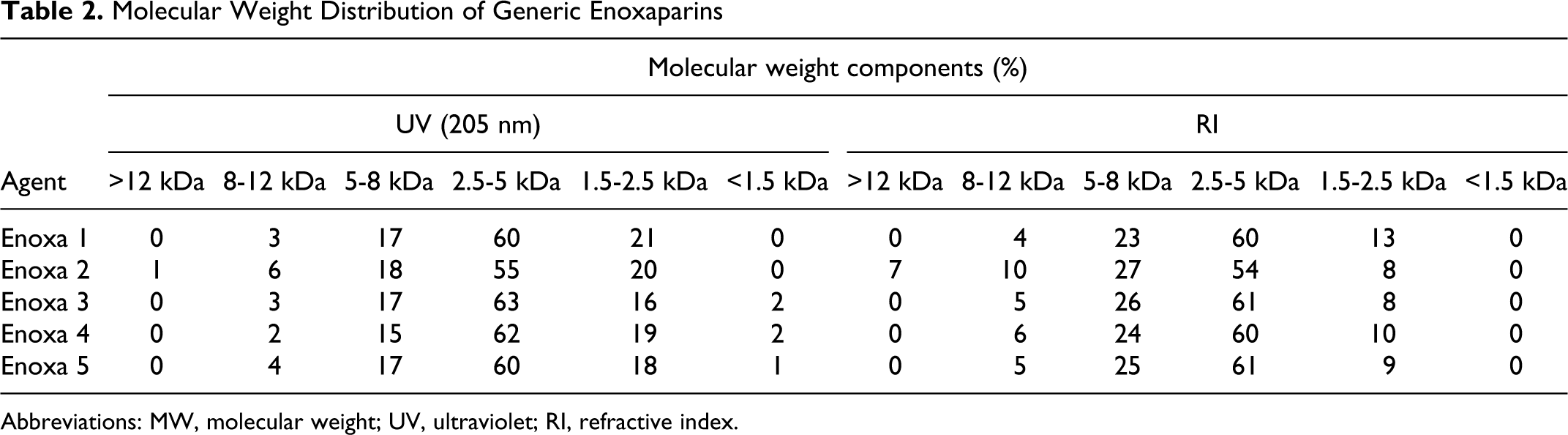
Abbreviations: MW, molecular weight; UV, ultraviolet; RI, refractive index.
Noteworthy, the branded products also presented distributions variation in average MW and MW, showing that the debate on generic low-MW-heparins must include the branded forms already in use for more than 20 years.
1H Nuclear Magnetic Resonance
The major signals on the enoxaparin 1 H NMR spectra arise from the prevalent disaccharide repeating unit of UFHs and enoxaparins: I2S-ANS,6S (α-L-IdoA2SO3-α-(1→4)-D-GlcNSO3,6SO3; Figure 1 ). Minor signals are also observed and the majority of them arise from under- and oversulfated sequences as well as those associated with the depolymerization reaction.
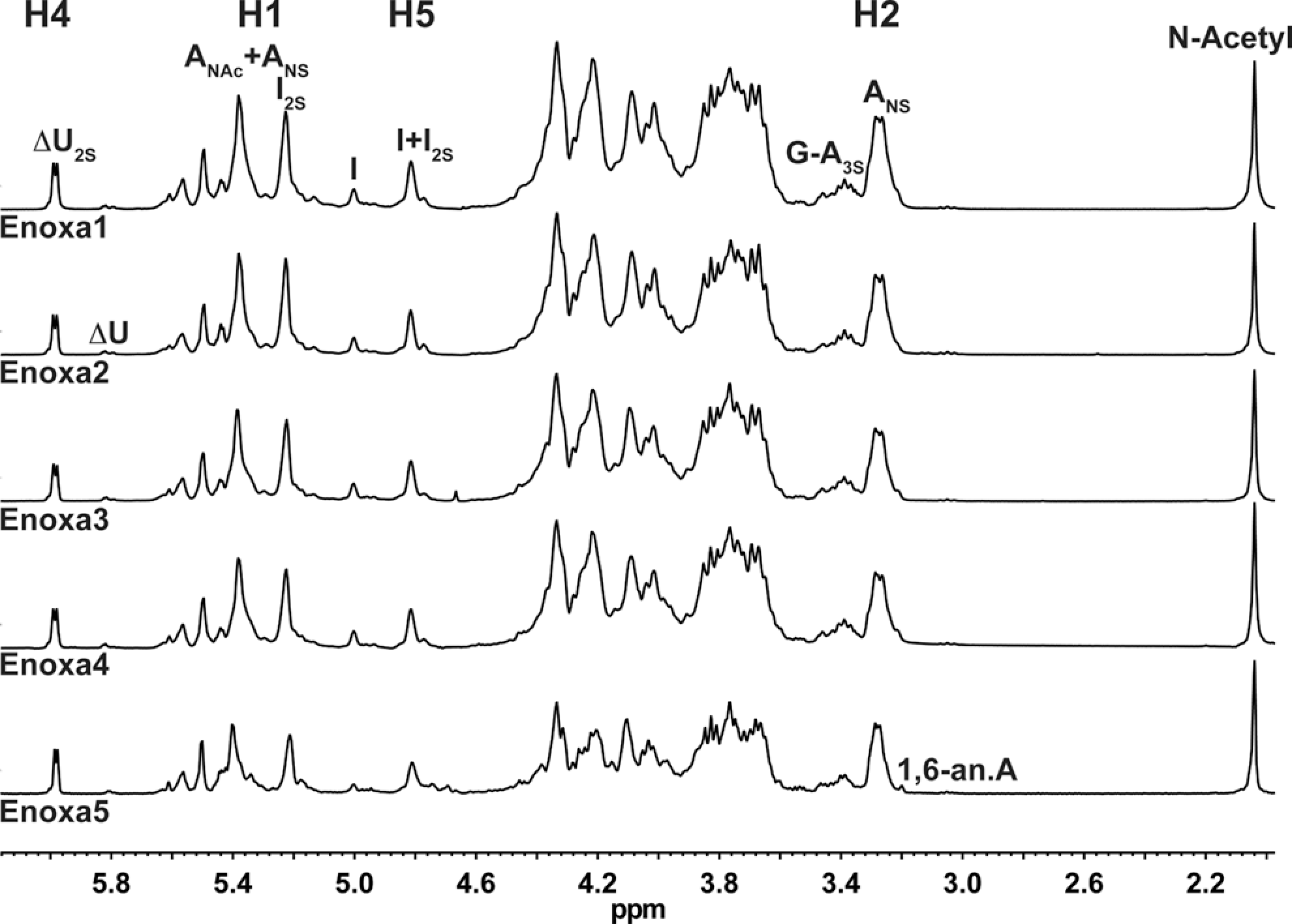
1H NMR of the different enoxaparins. ANS indicates 2-deoxy-2-sulfoamino-
Enoxaparin is produced by chemical β-eliminative reactions which generates, at the nonreducing end, unsaturated at C4-C5, 2-O-sulfated uronic acid (ΔU2S), which is rapidly noticed by a signal around 6 ppm (Figure 1). Also the presence of ΔU and 2-sulfo-amino-1,6-anhydro-2-deoxy-β-D-glucopyranose (1,6-anA) gives rise to signals of around 5.8 ppm and 3.2 ppm, respectively. Together, these facts make the enoxaparin 1 H NMR spectrum more distinct from UFH than any other LMWH. Also, this higher complexity is due to multistep reactions as well as side reactions that occur when fragments are generated during the alkaline treatment. 2
Essentially, the 1 H NMR spectra for all samples are the same, showing that their active pharmaceutical ingredient (API) is, indeed, very similar. The minor differences observed arise from process-related impurities such as sodium acetate, ethanol, methanol, and formic acid and preservatives such as benzyl alcohol (Figure 2).
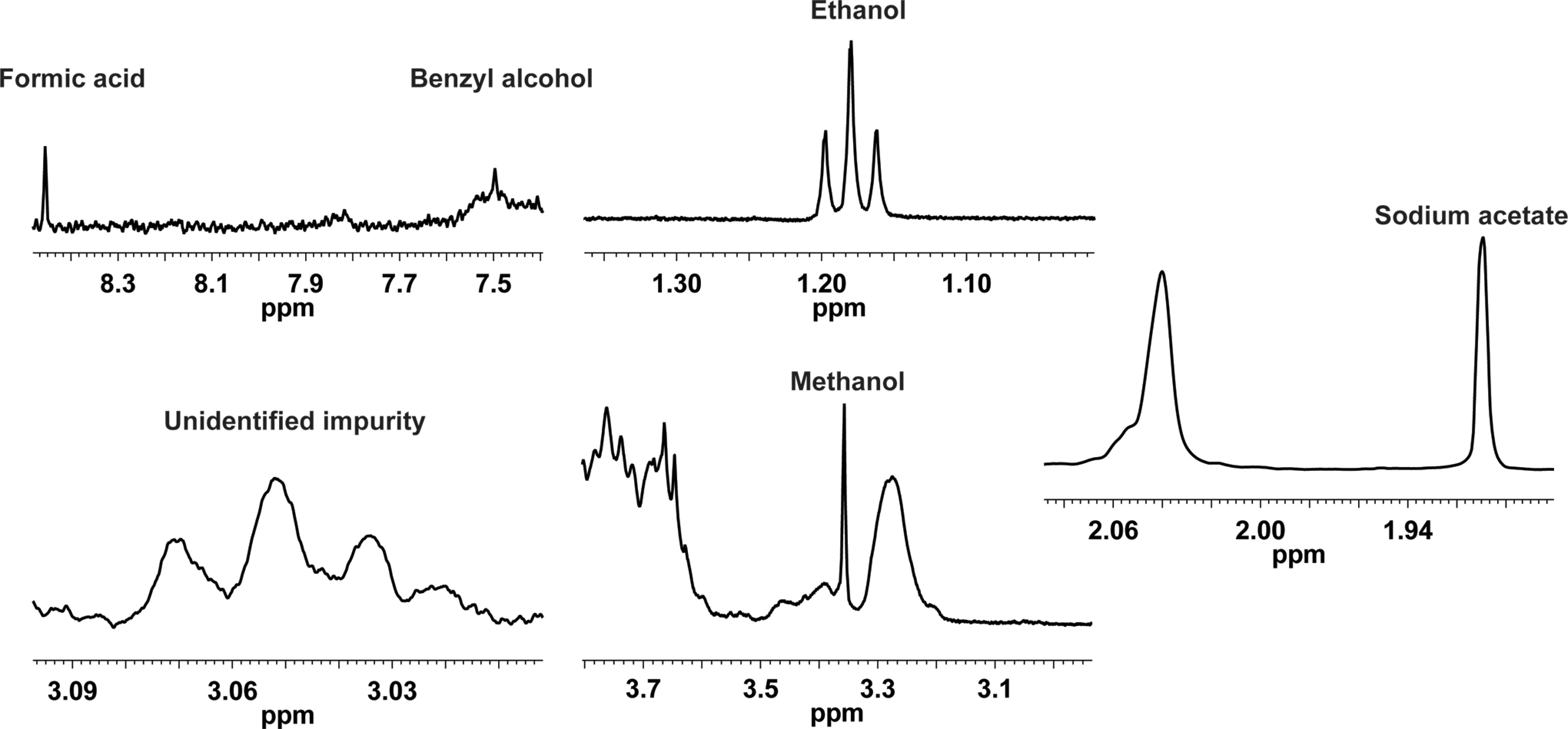
1H spectrum showing the presence of process-related impurities on enoxaparins.
Monosaccharide Composition of Enoxaparins
Monosaccharide composition of heparin preparations can be obtained by the peak integration of monodimensional NMR spectra, but some of these signals are strongly affected by signal overlapping. Also, throughout the depolymerization reaction new structures are generated including extra features on the monodimensional NMR spectra increasing signal overlapping and spectra complexity. Therefore, signals from the HSQC spectra less affected by signal overlapping2,10 were chosen for the monosaccharide composition shown in Figure 3.
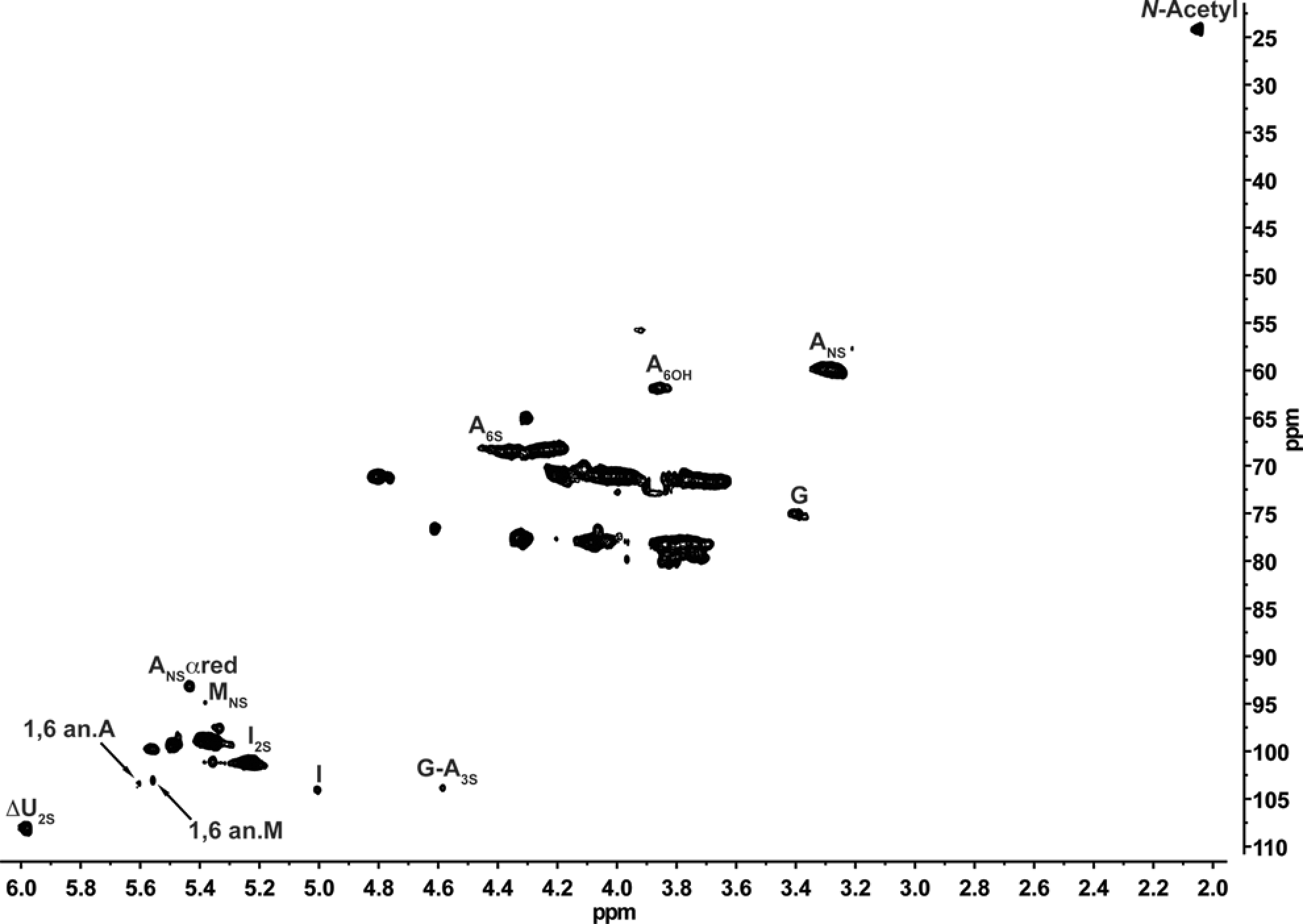
Heteronuclear single-quantum coherence (HSQC) spectra showing the signals used for monosaccharide composition. ANS indicates 2-deoxy-2-sulfoamino-d-glucopyranose; I2S, 2-O-sulfo-iduronic acid; G, glucuronic acid; A3S, 2-deoxy-3-O-sulfo-2-amino-
Monosaccharide composition is shown in Table 3 . As observed for MW, the monosaccharide composition values for all samples are quite the same. The percentage of monosaccharides that arise from the depolymerization reaction and those shared with their parent heparin are very comparable, leading to the conclusion that the starting material and chemical reaction used for their production is extremely similar proving, once again, that the tested agents have highly comparable APIs.
Monosaccharide Compostion of Biosimilar Enoxaparins
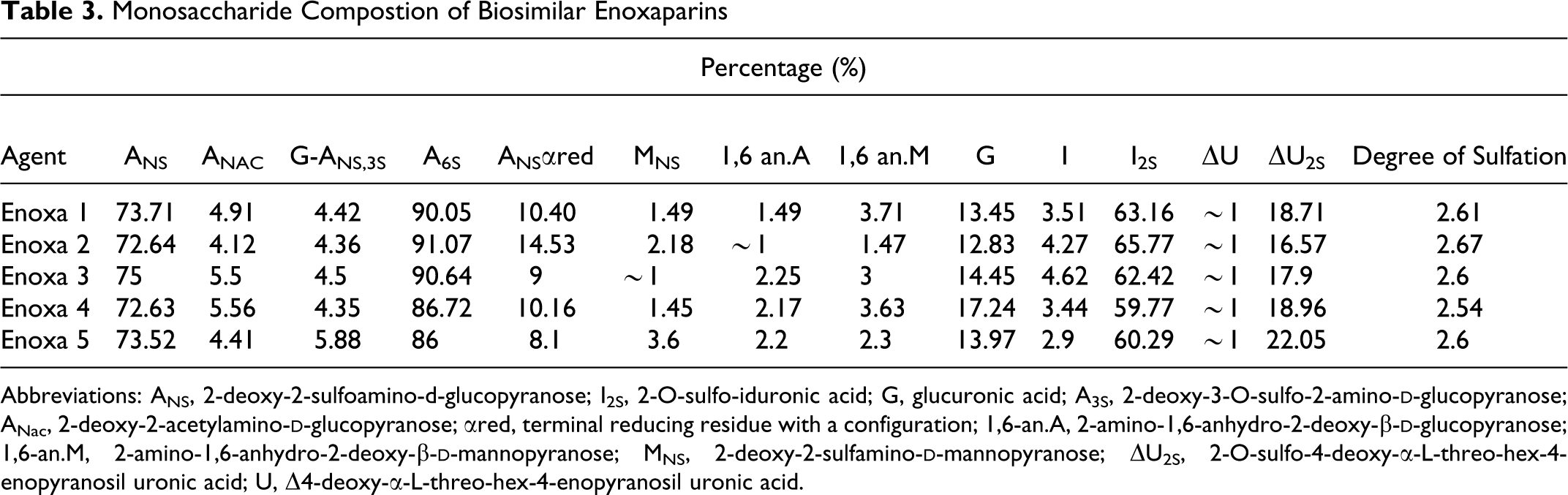
Abbreviations: ANS, 2-deoxy-2-sulfoamino-d-glucopyranose; I2S, 2-O-sulfo-iduronic acid; G, glucuronic acid; A3S, 2-deoxy-3-O-sulfo-2-amino-
Clotting Time
The APTT, HEPTEST, and TT (Figure 4A, C, and E) were performed in order to evaluate the effect of the different generic enoxaparins on clotting time. Important differences were observed for the various enoxaparins among the different clotting assays. On the APTT test, all agents presented similar results, suggesting similar modulation of the intrinsic coagulation cascade.
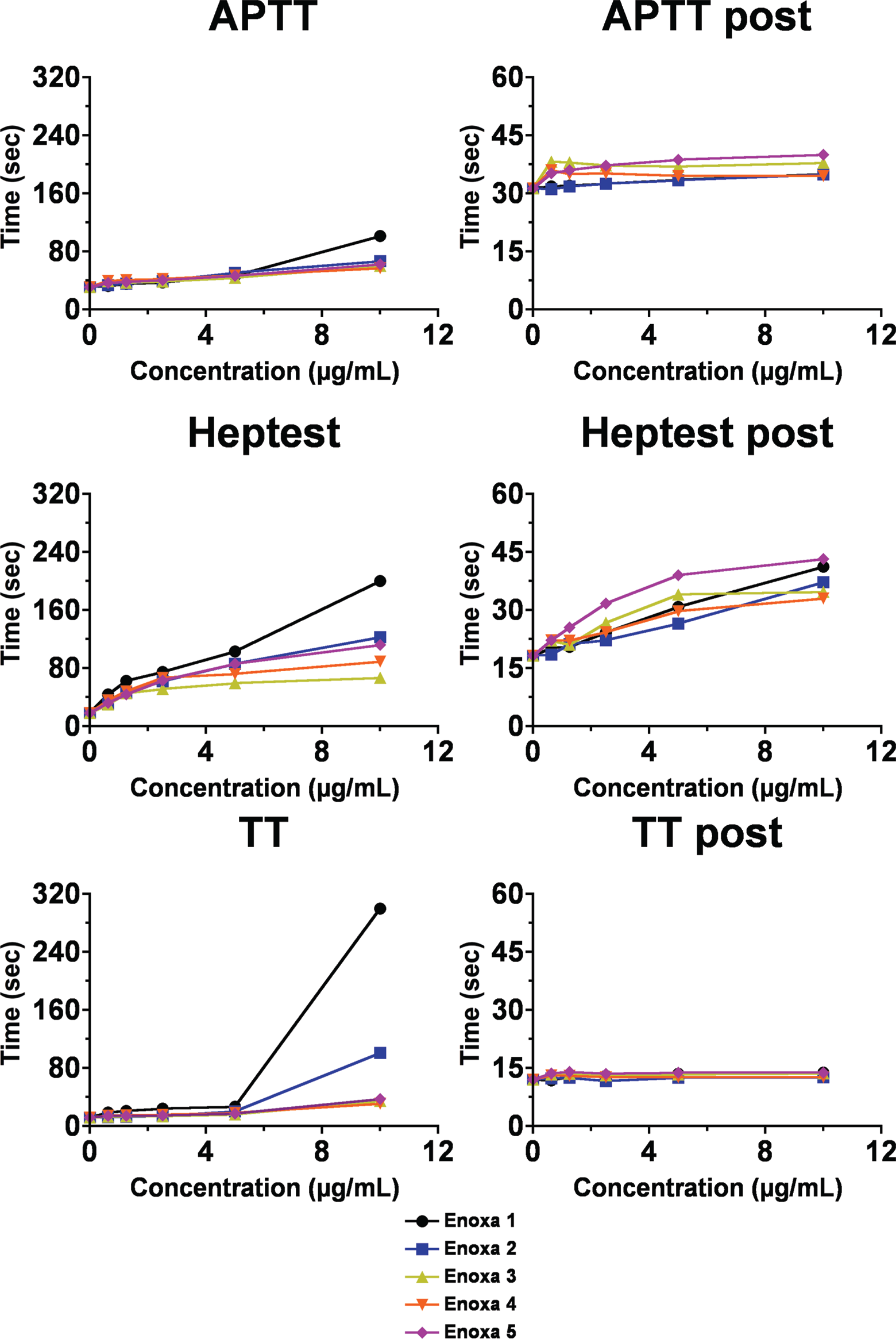
Clotting time assays for the different enoxaparins (A, C, and E) clotting time for the different enoxaparins. (B, D, and F) Clotting time for the different enoxaparins post heparinase I digestion.
On the other hand, Enoxa 1, branded product, displayed the most potent effect in all tests, the difference being on the TT assay that measures the amount of fibrin formed due to the presence of excess thrombin (Figure 4C), the most aberrant when the clotting time shows a ∽3-fold increase when compared to the others agents. This observation suggests that Enoxa 1 is much more capable of inhibiting thrombin activity mediated by AT and heparin cofactor II. The Heptest assay—measures the ability of heparin to catalyze the inactivation of exogenous bovine FXa by AT in the presence of naturally occurring plasma antagonist/antagonists —also revealed that Enoxa 1 is, indeed, more capable of modulating clot formation mediated by antithrombin (AT).
Interestingly, the branded products, Enoxa 1 and Enoxa 5, presented a quite different profile on the various assays, showing, once again, that the discussion about enoxaparins efficiency needs to include all of their forms, not only the generic ones.
The neutralization of the enoxaparin effect on the clotting assays after treatment with heparinase I showed an overall decrease of 50% for the HEPTEST (Figure 4B, D, and F). Apparently, no changes in the APTT were observed by neutralization with heparinase, except for Enoxa 1. Also, for the TT, the effect of enoxaparins 1 and 2 is completely abolished by the enzymatic treatment. Thus, heparinase is capable of neutralizing the effect elicited by enoxaparins in clotting assays.
Anti-Xa and Anti-IIa Inhibition
The inhibition ability of the different enoxaparins toward FXa and FIIa was also analyzed (Figure 5A and C). Differently than the clotting time, the overall inhibition profile for the tested agents was somehow similar. For the inhibition of FXa, 2 distinct groups could be easily observed, that is, Enoxa 1 and 2 presented the strongest inhibition potency and Enoxa 3, 4, and 5 formed the group with less potency. Another interesting finding extracted from this test is that fact that Enoxa 1 displayed a more potent inhibition activity than Enoxa 3, 4, and 5 even at half of the concentration, supporting the results from the clotting assays which showed that Enoxa 1 is far more capable on modulating clot formation mediated by AT.
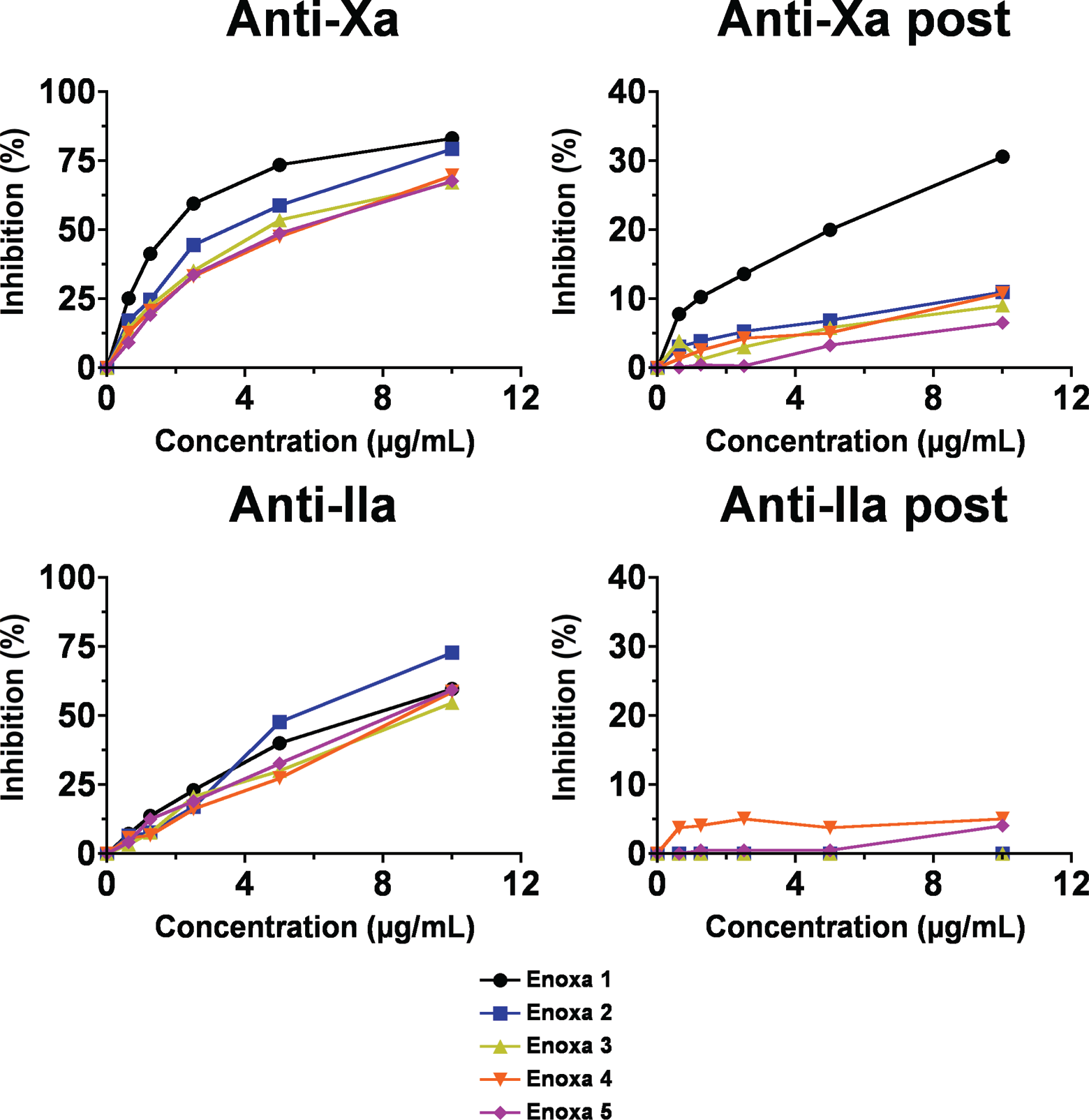
Anti-Xa and Anti-IIa activities for the different enoxaparins. (A and C) Antiprotease activity for the various enoxaparins. (B and D) Antiprotease activity for the different enoxaparins post heparinase I digestion.
Looking at the inhibition of FIIa, Enoxa 2 stands out when compared to the others, the fact that is in agreement with its higher average MW that enhances its ability to bind AT and thrombin simultaneously.
Despite the similarity among their API, the differences in activity could be explained by the presence, in different levels, of process-related impurities (Figure 2). Also, the fact that branded products presented different profiles on both tests reinforces the importance of deep discussions on the efficiency of all enoxaparins.
The agents were also treated with heparinase being their anti-Xa and anti-IIa activities measured. When neutralized, the anti-Xa and IIa inhibition decreased to baseline values (Figure 5B and D). The neutralization pattern of the antiprotease followed the pattern of the anticoagulant effects showing a MW dependency.
Discussion
Due to their higher anti-FXa/anti-FIIa activity ratios, with decreased hemorrhagic risk during prolonged treatments, 11 enoxaparins are globally regarded as drugs of choice for treatment and prophylaxis of thrombotic events. Several generic forms of enoxaparins have received approval and are in use in South America and Asia and, more recently, in the United States.
The approval of generic enoxaparins may be beneficial since it would have a great deal of impact on treatment costs for hospitals and patients. 12 Besides the countries regulated by the FDA and The European Medicines Agency (EMEA), no specific guidelines for the manufacture, preclinical, and clinical evaluation are available. They are just being approved based on the perhaps unproven assumption that exact copies having the same active ingredients as their reference drug counterparts can be manufactured, 13 and that they will possess the same efficiency and safety for clinical use as their reference drug.
As shown in this article, the APIs for all of the tested agents are very comparable, indeed, the variation that occurred are in agreement with what is defined as enoxaparin. 9 The types and quantities of monosaccharides generated by the depolymerization reaction provide a fingerprint of what enoxaparin is and its manufacturing procedure. All tested agents showed comparable values for the specific monosaccharides generated by the benzylation followed by alkaline hydrolysis. Together with the fact that the monosaccharides shared with the parent heparin also possess similar values, our results lead to the conclusion that the starting material and chemical reaction used for their production is extremely similar proving, once again, that the agents tested have highly comparable APIs.
On the other hand, the clotting and antiprotease assays showed significant differences among the tested agents and not only between branded and generic forms; variance between the two branded products tested was also observed. Despite their comparable APIs, Enoxa 1—branded product—showed significant higher values on the biological assays especially on those where AT is the direct mediator (anti-Xa, TT, and HEPTEST assays).
The observed differences on the biological assays could be explained by the presence of several process-related impurities found on the different tested agents (Figure 2). Impurities such as sodium acetate, ethanol, methanol, and formic acid and preservatives such as benzyl alcohol were found in different amounts. Once our tests were performed based on dry mass rather than activity (IU), the presence of process-related impurities could strongly affect their activity especially for those with significant amounts of sodium acetate.
It is reasonable to speculate that the presence of such impurities affected the biological tests that the agents undergo and may affect their properties when used in humans. Supporting the fact that such impurities may alter the biological assays, no impurities were found on Enoxa 1, the agent that possessed the strongest activity on all tested assays. Also, of particular interest is the presence of benzyl alcohol, whose harmful effect has been proven in vivo and in vitro14–17; and various manufactures state that their agents are preservative-free, yet, such compound was found on their preparation.
Owing the natural heparin complexity and the quite complicated process involved in the production and purification of enoxaparins, the results presented here provided valuable information on the issue of how similar generic enoxaparins, already in use, are. However, one may say that chemical characterization and in vitro studies are not enough to prove how different these drugs are. Nevertheless, this form of approach is, for certain, the first step for the development of specific guidelines that, hopefully, one day will be used throughout the globe to regulate generic biologics such enoxaparins.
Footnotes
Acknowledgments
The authors would like to thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Authors’ Note
Marcelo A. Lima, and Eduardo H. C. de Farias contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared following financial support for the research, authorship, and/or publication of this article: Fundaçãoo de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), however they had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
