Abstract
Introduction
Venous thromboembolism (VTE), comprising deep venous thrombosis (DVT) and pulmonary embolism (PE), is a major cause of morbidity and mortality. 1 Venous thromboembolism can be classified into VTE caused by transient risk factors (such as surgery), permanent risk factors (such as cancer or paralysis) or without any identifiable risk factors (idiopathic VTE). 2
Guidelines recommend acute treatment of VTE with low-molecular-weight heparin (LMWH) and dose-adjusted warfarin started immediately upon VTE diagnosis. Low-molecular-weight heparin should be discontinued once a therapeutic international normalized ratio (INR) has been achieved, usually defined as 2 back-to-back INR tests within therapeutic range (ie, 2.0-3.0). Warfarin treatment should be continued for at least 3 months, after which the physician should decide whether long-term secondary prevention should continue based on the risk of recurrence, risk of bleeding, and the preference of the patient to continue with warfarin treatment. 3
When first starting warfarin, great effort is exerted to find the correct individual dose and achieve a stable therapeutic INR. As the recommended treatment duration is 3 months, this initiation phase may represent a substantial portion of the total treatment period. International normalized ratio levels outside the therapeutic range are associated with increased risk of VTE recurrence and bleeding. The objectives of this study were to evaluate the pattern of anticoagulation and quality of INR control after VTE in actual clinical practice and to assess the effects these have on VTE recurrence and mortality.
Methods
Source of Data
Information for the study was obtained from the General Practice Research Database (GPRD), which comprises the computerized medical records of general practitioners (GPs) in the UK. General practitioners play a key role in the UK health care system, as they are responsible for primary health care and specialist referrals. Patients are semi-permanently affiliated to a practice, which centralizes the medical information from the GPs, specialist referrals, and hospitalizations. The data recorded in the GPRD include demographic information, prescription details, clinical events, preventive care provided, specialist referrals, hospital admissions, and their major outcomes. Specialists and hospitals are required in the UK to inform the GP about the patient’s medical care, and summary information from specialist care is typically entered into the GPRD by the GP. The General Practice Research Database patients in English practices are now linked individually and anonymously to the national registry of hospital admission (Hospital Episode Statistics [HES]) and to the death certificates (as collected by the Office of National Statistics). For each hospitalized patient, the hospital charts are reviewed. The dates of admission and discharge, and main diagnoses are extracted, coded by coding staff and collated nationally. The death certificates list the date and causes of death. Hospital Episode Statistic data were available from 2001 to 2007 for 200 practices and death certificate data from 2001 to 2008. A high level of validity of VTE recording in GPRD has been reported previously.4,5
Study Population
The study population consisted of patients aged 18 years or older with a diagnosis of VTE on or after January 01, 1995 and during the period of GPRD or HES data collection (until October 30, 2009). The index date was defined as the date of the first record of VTE from either source. Venous thromboembolism was defined as a composite of distal and proximal DVT and PE. Patients with less than 1 year of GPRD data previous to their first VTE event were excluded. The risk of VTE is increased antepartum and 6 weeks postpartum. These events will not be managed with warfarin and will therefore be underrepresented. Patients with a record of pregnancy within 44 weeks before, or those with a delivery record in the 6 weeks after were excluded (2912 patients). Information from the free text area was used to exclude patients with negating or questionable free text (eg, “?” or “ruled out”). Each patient with a VTE event was matched to up to 3 controls, by general practice, gender, and year of birth (within 5 years). The pool of controls consisted of patients who had no record of VTE in GPRD or HES. The study population was followed from the index date until the end of data collection, patient’s death, or transfer out of the GP practice, whichever date came first.
In order to describe the etiology of VTE, patients were classified into the following categories based on data recorded prior to the index date: Patients with a modifiable (transient) strong risk factor for VTE (category A). Hip or leg fracture, hip or knee replacement, major surgery (pelvis, lower leg, knee, feet, veins, arteries, spinal cord, and unspecified region), major trauma (head, neck, spinal cord, trunk, pelvis, lower leg, knee, feet, and unspecified region). Patients with a modifiable (transient) moderate/low risk factor for VTE (category B). Major surgery (head, neck, trunk, and arms/hands), major trauma (arms/hands), pneumonia, chronic obstructive pulmonary disease, hormone therapy, or oral contraceptives. Patients with an “unmodifiable” risk factor for VTE (category C). Active cancer, congestive heart failure, varicose veins. Patients with no obvious risk factor (category D). All the other patients, not falling in the previous categories.
Conditional logistic regression was used to compare the medical history of VTE cases and controls, estimating the odds ratios (ORs) and 95% confidence intervals (95% CIs).
International Normalized Ratio and Percentage of Time Spent Within Therapeutic Range
Anticoagulation treatment requires frequent blood tests to monitor the level of anticoagulation. This is measured by the INR. International normalized ratio values are typically found in the GPRD if the laboratory sends the results electronically to the general practice. The association between the rate of mortality and VTE recurrence and rate of discontinuation associated with the level of anticoagulation was evaluated during the time, with INR measurements evaluating the time spent within therapeutic range. This was done for VTE cases with at least 3 records of INR measurements. For each of these cases, the percentage of time spent within the therapeutic range (2.0-3.0) was calculated using the Rosendaal method of linear interpolation. 6 Patients were censored in this analysis if no repeat INR was measured within 3 months of a previous INR measurement, or at the patient’s date of death, or end of data collection.
Treatment Persistence
Treated cases were identified as those with a prescription for coumarins (warfarin, acenocoumarol, or phenindione) or LMWH or at least 2 INR measurements. For patients identified as being hospitalized, the index date was modified to that of the discharge date. For each coumarin prescription, the expected duration of use was taken as 28 days. Episodes of use were calculated by looking for repeat prescriptions in the period of time of expected duration of use plus 30 days.
Treatment persistence (ie, repeat prescribing over time) immediately following VTE diagnosis was examined in those with a first-ever prescription for a coumarin on or within 30 days after the index date. Discontinuation was defined as no repeat prescription within 90 days after the expected end of the treatment course. Cox proportional hazards regression was used to estimate discontinuation with anticoagulant therapy over time. Fully adjusted models included (where appropriate) age, gender, BMI, smoking status, alcohol use, socioeconomic status at the location of the practice, hip, knee or lower leg fracture, hip or knee replacement, major trauma, recent surgery, active cancer, spinal cord injury, heart failure, chronic obstructive pulmonary disease, dementia, falls, oral contraceptive use, use of hormone therapy, or varicose veins.
Outcomes After VTE
The VTE cases were followed for the following 3 outcomes: (I) hospital readmission for VTE after the initial VTE diagnosis (based on HES data), (II) death due to VTE (based on death certificate data), or (III) all-cause death. The study population was followed for these outcomes from the index date to the end of data collection, patient’s death, or transfer out of the practice, whichever date came first. Readmission for VTE was counted if a VTE was recorded at least 10 days after the index VTE event, as either the primary or secondary reason for hospitalization. Death due to VTE was counted if VTE was recorded as one of the causes of death. Cox proportional hazards regression was used to estimate the relative hazard rates (RRs) of outcomes over time. Fully adjusted models included the same variables as listed in the section on treatment persistence. We also evaluated the patterns of the hazard rates (ie, absolute risk) for death (based on the death certificates). The hazard rates were estimated by dividing the follow-up time into 100 periods (over 5 years) and by calculating the absolute rate within each small period (the hazard rate provides the risk of the outcome over a small period of time). These estimates were then smoothed using the methods proposed by Ramlau-Hansen. 7
Results
The study population included 46 335 VTE cases and 138 024 controls. Table 1 shows the baseline characteristics of the VTE cases and their matched controls. The mean age of VTE cases was 65 years and 55.2% of patients were female. Based on data recorded in GPRD, 70.2% of the VTE cases and 86.6% of controls had no obvious risk factors for VTE. The VTE cases were more likely to have a medical history of strong VTE risk factors compared to controls (OR 18.76; 95% CI 17.37-20.27). The occurrence of a recent fracture (6 weeks prior) of the lower extremity, hip or knee replacement, or major trauma were all strongly associated with an increased risk of VTE (ORs of 32.92, 40.26, and 20.56, respectively). Recent use of hormone therapy or oral contraceptives was associated with VTE (OR of 1.42; 95% CI 1.34-1.49). Obese patients were also more likely to suffer VTE (OR of 2.01).
Baseline Characteristics for VTE Cases and Controls
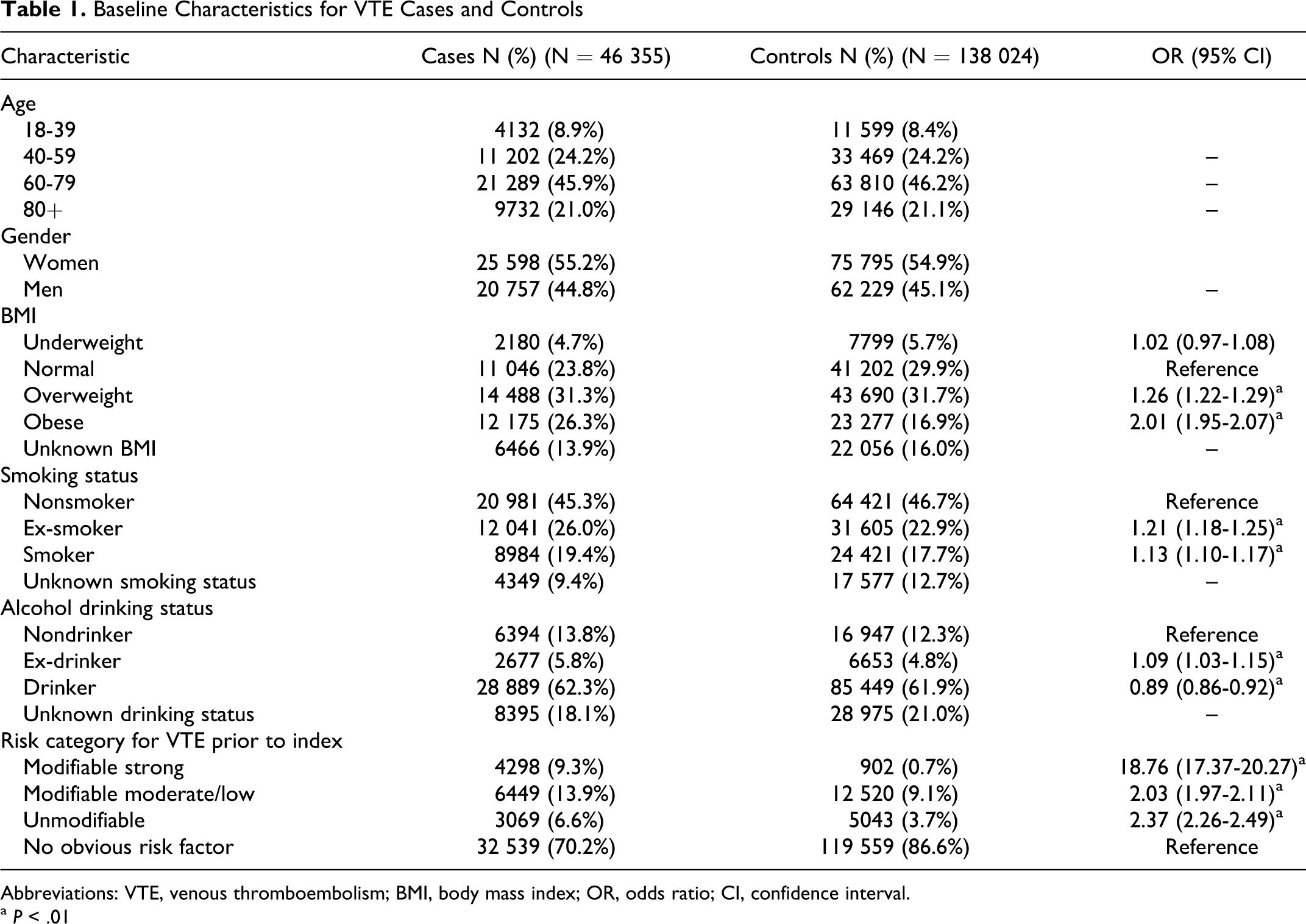
Abbreviations: VTE, venous thromboembolism; BMI, body mass index; OR, odds ratio; CI, confidence interval.
a P < .01
There were 20 090 patients with VTE and 50 535 controls eligible for linkage to HES and 21 796 patients with VTE and 64 673 controls eligible for linkage to the death certificate data. Table 2 shows the mortality rate and causes of death. The death certificate data showed that VTE cases were more likely to die compared to controls (RR of 5.00; 95 CI 4.76-5.24). Frequent causes of death in patients with VTE were neoplasms (RR of 9.37) and diseases of the circulatory system (RR of 3.60). Figure 1 shows the crude RRs of mortality over time after diagnosis in patients with VTE compared to controls. The hazard rate of death was increased substantially around the time of diagnosis (RRs around 21), decreased in the 12 months after VTE diagnosis, and then remained elevated for a further 4 years (RRs around 1.5-2.0).
Mortality Rate and Causes of Death in VTE Cases and Controls
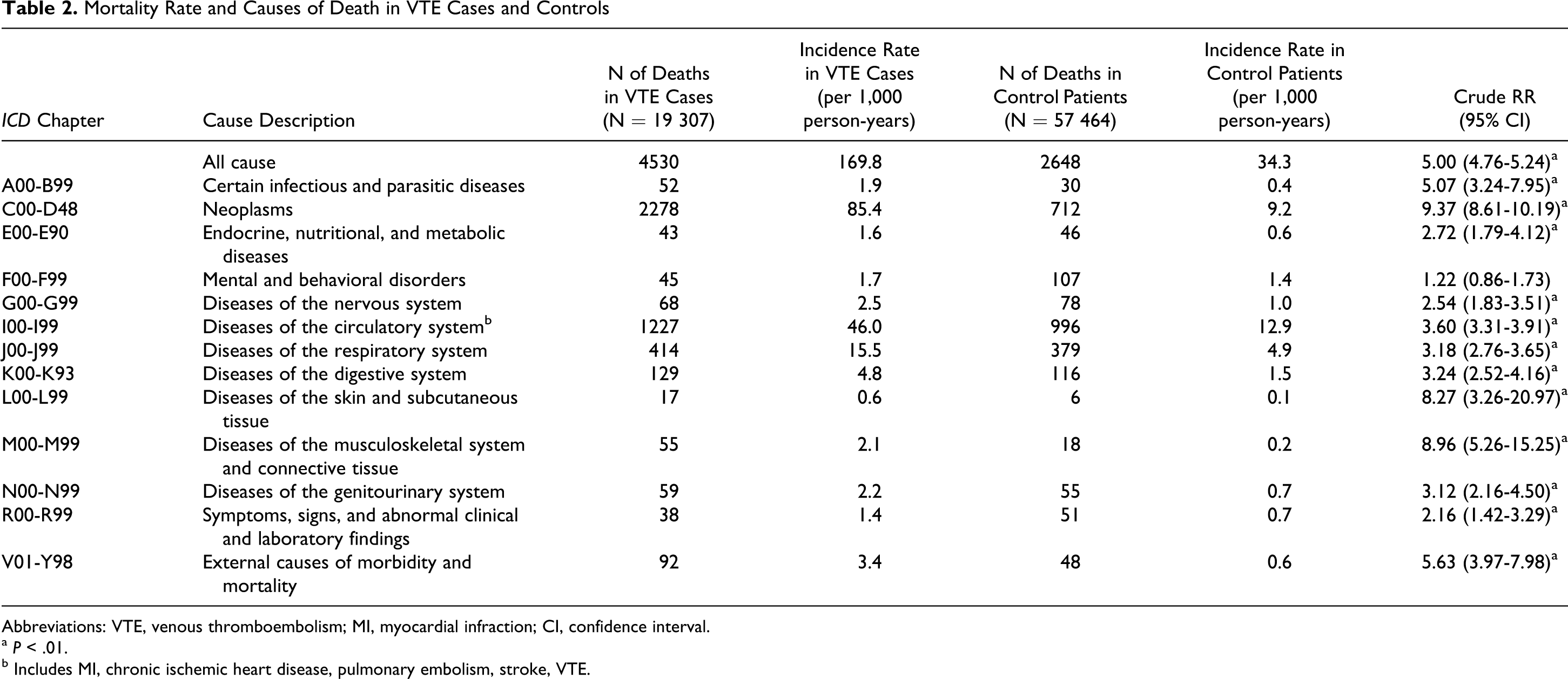
Abbreviations: VTE, venous thromboembolism; MI, myocardial infraction; CI, confidence interval.
a P < .01.
b Includes MI, chronic ischemic heart disease, pulmonary embolism, stroke, VTE.
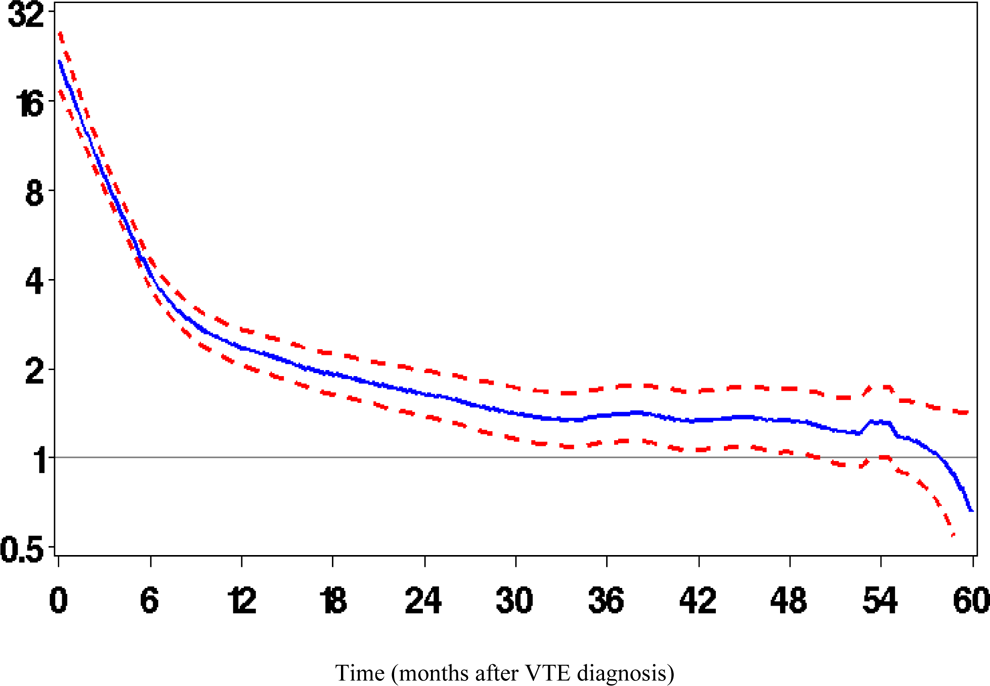
Crude RRs of mortality over time after diagnosis in VTE cases compared to controls (dotted lines indicate the 95% CI). X-axis: Time (months after VTE diagnosis). Y-axis: RR of mortality in VTE cases compared to controls. VTE indicates venous thromboembolism; RR, relative hazard rate.
There were 5162 patients with VTE who completed the first 3 months of treatment. The large majority of patients with VTE discontinued coumarin treatment in the next 3 months. Only 13% of the VTE cases (95% CI 12-14%) were found to have continued coumarin treatment for at least 9 months and 3% (95% CI 3-4%) for at least 2 years.
There were 10 381 patients with VTE with at least 3 INR measurements recorded in GPRD (on average 23 measurements per patient, standard deviation 16). As shown in Table 3 , the mean percentage of time spent within therapeutic range was 57.0%. Patients with VTE (aged 60-79 years) had the highest percentage spent within therapeutic range (59.2%), while youngest patients had the lowest (48.7% in patients aged <40 years). There was no substantive difference between women and men in the percentage of time spent within therapeutic range (58.0% and 56.0%, respectively).
Time Spent Within Therapeutic Range in Venous Thromboembolism (VTE) Cases Using Coumarins
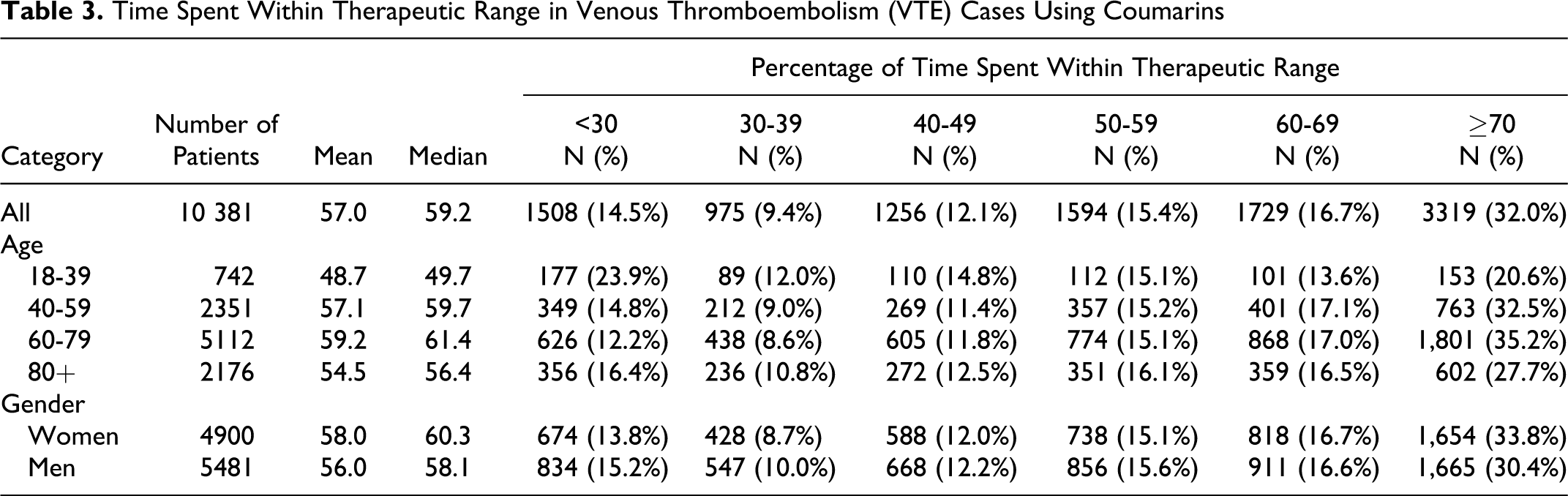
Patients with VTE with a high percentage of time spent within therapeutic range were less likely to discontinue coumarin treatment compared to those with a low percentage (Table 4 ). The adjusted RR in VTE cases with ≥70% within therapeutic range was 0.53 (95% CI 0.48-0.59) compared to those with <30%.
Discontinuation Rate With Coumarins Over the First 2 Years After VTE Diagnosis Stratified by the Percentage of Time Spent Within Therapeutic Range (N = 4337)
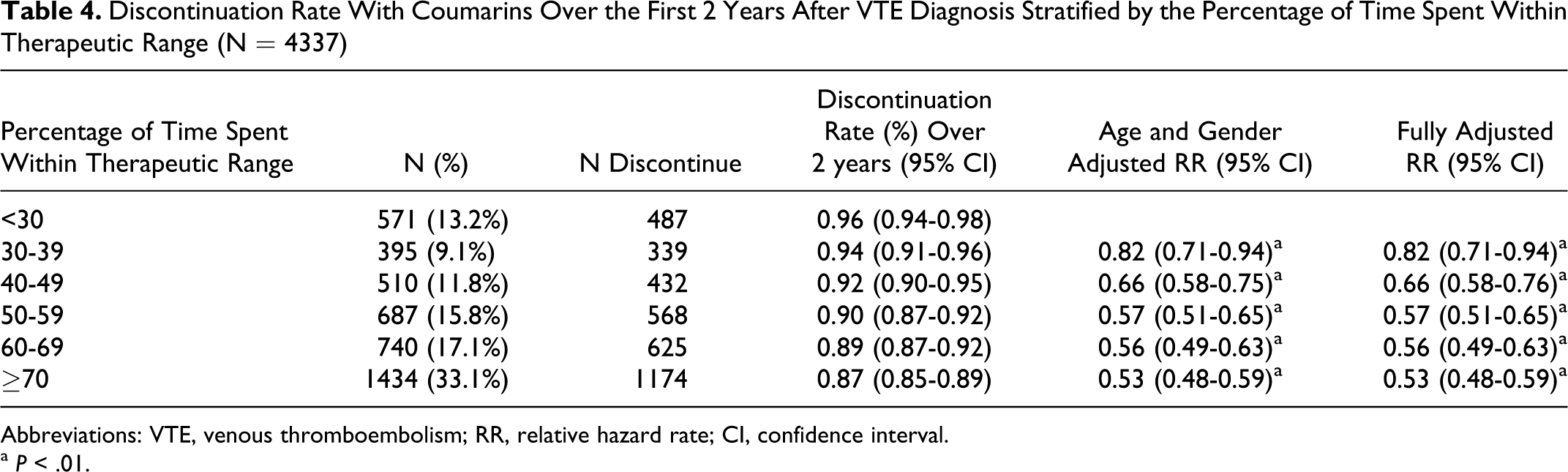
Abbreviations: VTE, venous thromboembolism; RR, relative hazard rate; CI, confidence interval.
a P < .01.
Table 5 shows the outcomes after the VTE diagnosis. The VTE recurrence was found to be higher in elderly patients with VTE compared to younger cases (RR of 1.53 for recurrent hospitalization for VTE and RR of 11.01 for death due to VTE). The VTE recurrence was lower in VTE cases with strong modifiable (transient) risk factors compared to those with idiopathic VTE (RR of 0.84 for recurrent hospitalization for VTE). The lowest rate of VTE recurrence occurred in patients with ≥70% time spent within therapeutic range (RR of 0.50, 95% CI 0.39-0.63 compared to those with <30% time in range).
Outcomes After VTE (Recurrence and Death) Stratified by Age, Gender, Presence of VTE Risk Factors, and Percentage of Time Spent Within Therapeutic Range
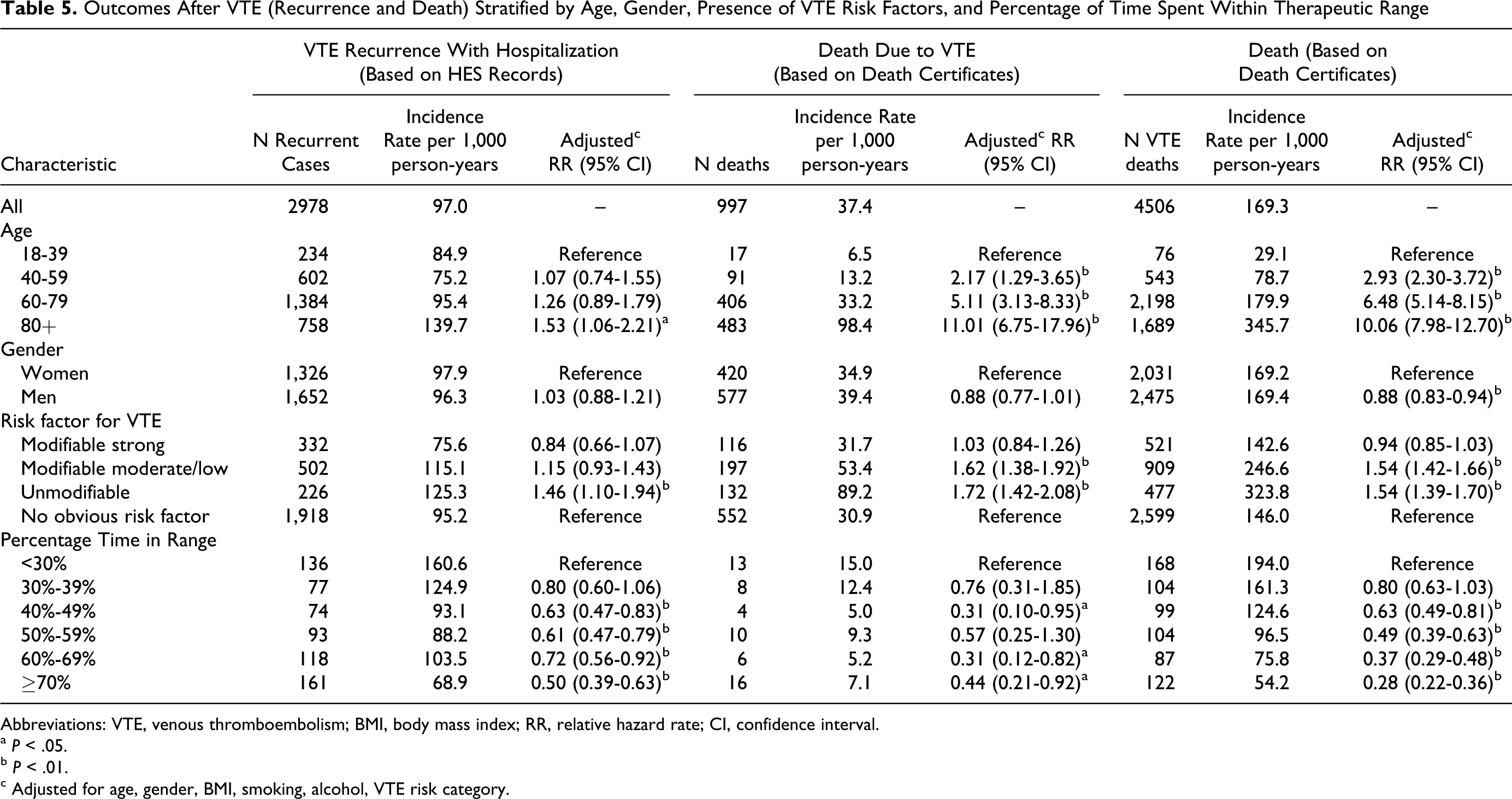
Abbreviations: VTE, venous thromboembolism; BMI, body mass index; RR, relative hazard rate; CI, confidence interval.
a P < .05.
b P < .01.
c Adjusted for age, gender, BMI, smoking, alcohol, VTE risk category.
Figure 2 shows the life-table for VTE recurrence (based on recurrent hospitalization for VTE). Readmission rates for VTE were high, especially in those patients with less time spent within therapeutic range. The incidence of VTE recurrence over 2 years was 11% in those with ≥70% and 21% in those with <30% spent within therapeutic range. The largest risks for VTE recurrence occurred within the first 3 months after the first VTE.
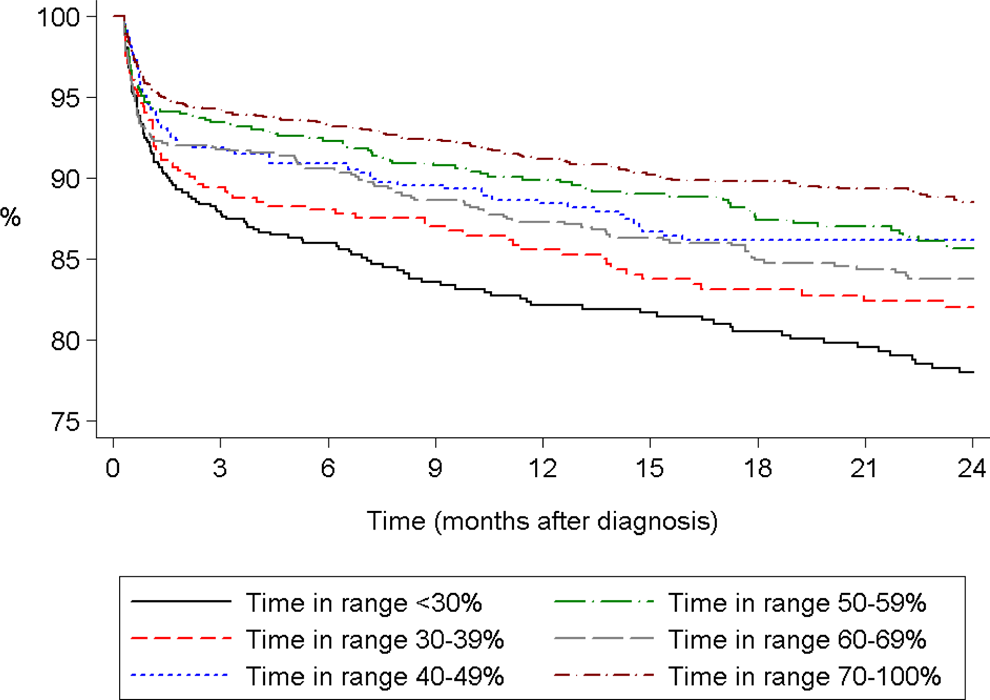
Percentage of VTE cases without recurrence of VTE (based on hospitalization data in HES) stratified by percentage of time spent within therapeutic range. X-axis: Time (months after VTE diagnosis). Y-axis: Percentage of VTE cases without recurrence of VTE. VTE indicates venous thromboembolism; HES, Hospital Episode Statistics.
Discussion
This study found that the risk of VTE was substantially increased in patients with a recent lower extremity fracture, hip or knee replacement, or major trauma. The risk of mortality was substantially higher in VTE cases compared to controls. Recurrence of VTE (as measured by readmission to the hospital) was high and inversely related to the time spent within therapeutic range. Most patients with VTE stopped coumarin treatment within 6 months in line with treatment guidelines and a clear relationship exists between treatment discontinuation and time spent within therapeutic range.
Various studies in the literature have evaluated the risk factors for the development and recurrence of VTE. The results in this study on risk factors are broadly consistent with the body of evidence in the literature. This study confirmed the strong associations between the development of VTE and various transient risk factors (such as major trauma and surgery). 2 Obese patients in GPRD also had an increased risk of developing VTE similar to that previously reported. 8 However, some of our results on VTE recurrence differed with those of various published studies. Several studies have reported considerably higher risks of VTE recurrence in patients with cancer compared to those with transient risk factors.9,10 While we did find a higher risk in patients with cancer, the excess risk was small. This study also found no major difference in VTE recurrence between men and women. A recent meta-analysis found that men had a 50% higher risk of VTE recurrence compared to women. 11 There may be several reasons for these discrepancies (other than bias). One reason may be differences in characteristics and representativeness of the study populations. Many of the studies reported in literature were conducted in specialist centers, while this study was conducted in routine clinical practice.
High recurrence rates of VTE have been reported in other observational studies conducted in nonspecialist settings.9,12,13 The largest study to date, including 2090 VTE cases as recorded in a US claims database, found a recurrence rate of 10.9% after 6 months. 12 However, lower rates of VTE recurrence have been reported in studies conducted in specialist anticoagulation centers.14,15 Lower rates of VTE rates have also been reported in randomized clinical studies. A recent large clinical study comparing dabigatran and warfarin found a VTE recurrence rate of about 2% over 6 months. 16 There has been limited research on the possible reasons for these differences in VTE recurrence rates and whether these differences may be explained by varying patterns of anticoagulation. There may be multiple and heterogeneous reasons for suboptimal anticoagulation with warfarin. These include, among others, co-prescribing of drugs that can interact with warfarin. The list of drugs with a potential to interact with warfarin is continuously increasing. Furthermore, complex dosage instructions may adversely affect patient compliance. Hopefully, the next generation of oral anticoagulants that do not require anticoagulation monitoring may help to improve patient compliance.
Patients treated with coumarins are regularly monitored in order to maintain INR values within the range of 2.0 to 3.0. A recent US study that reviewed medical charts of VTE cases found that 37.7% of time was spent within therapeutic range. 12 We found that VTE cases treated in the UK with coumarins and with at least 3 INR measurements spent on average 57.0% of time within the therapeutic range. Direct comparisons between studies may be difficult, given the differences in data collection and methods. Furthermore, the present study found that time spent within therapeutic range was a strong predictor of the discontinuation rate with coumarins.This could mean a “positive survivor bias” and a artificial high value in our study. Unfortunately, duration of treatment with coumarins was not reported in the US study. Time spent within therapeutic range was strongly associated with risk of death due to VTE and VTE recurrence. Lower risks were found in those with ≥70% spent within therapeutic range. The strong association between level of anticoagulation with coumarins and adverse outcomes (stroke and mortality) has also been found in patients with atrial fibrillation. 17 A recent study found that patients have substantial difficulties maintaining adequate adherence with coumarins, and that this poor adherence has a significant effect on anticoagulation control. 18 In the present study, we did not have information on the reasons for poor adherence in some of treated VTE cases.
The strengths of this study were that the study population was representative of actual clinical practice. This study is the first VTE study using the GPRD with episodic hospital data linked to longitudinal data from general practice. However, there are also several important limitations of this study. We did not have information on the diagnostic criteria used for the VTE diagnosis, however the GPRD has been used in previous studies evaluating VTE and a high level of validity in the recording of VTE by GPs has been reported.4,5 Also, we did not have the reasons for VTE recurrence. We may have overestimated the rate of recurrence by assuming that further VTE records after 10 days constituted a new event. It is possible that some of these events were preexisting diagnoses being re-recorded, which might inflate recurrence rates. Another limitation is that we did not have information on all risk factors for VTE, such as frailty. Also, like in any epidemiological study, the comparison groups in this study were not randomized. It could not be established whether unstable INR measurements caused the increased risk of mortality and VTE recurrence or whether instability in INR measurements was a symptom of intercurrent illness. We did not have information on INR values for all patients as the monitoring may have been performed elsewhere and not all anticoagulation clinics report INR results electronically to the GPs. It may be that results falling outside of therapeutic range are more likely to be transmitted in this way. Moreover, applying the Rosendaal method to patients with only 3 INR measures has the potential to give a biased picture of INR control compared to patients with a larger number to test results. For example, a patient with 3 consecutive INR tests taken on the first day of the month over 3 months would have a percentage TTR of zero, whereas if the second test was in range the percentage TTR would be 50%. Potentially, distal DVT can be managed in the outpatient setting which could have led to an underestimation of the recurrent VTE rate since our analysis only included recurrent events that required hospitalization. Another limitation concerned the use of death certificates for classification for cause of death. One study found discrepancies between causes of death as reported on death certificates and those found at necropsy. 19
In conclusion, most VTE cases studied were treated with coumarins for less than 6 months and the overall time spent within therapeutic range was relatively low. Higher time spent within therapeutic range was associated with lower risks of VTE recurrence, lower discontinuation rates, and death due to VTE. The challenge remains to improve the time spent within the recommend INR range, but this may be demanding since the time required to achieve a stable therapeutic INR represents a substantial portion of the typical duration of treatment.
Footnotes
Appendix A
International Classification of Diseases, Tenth Revision (ICD-10) Codes Used to Define Venous Thromboembolism (VTE)
ICD-10 Code
ICD-10 Term
I26.0
Pulmonary embolism with mention of acute cor pulmonale
I26.9
Pulmonary embolism without mention of acute cor pulmonale
I80.1
Phlebitis and thrombophlebitis of femoral vein
I80.2
Phlebitis/thrombophlebitis oth deep vessels low extremities
I82.2
Embolism and thrombosis of vena cava
I82.3
Embolism and thrombosis of renal vein
I82.8
Embolism and thrombosis of other specified veins
I82.9
Embolism and thrombosis of unspecified vein
Authors’ Note
GPRD is owned by the UK Department of Health and operates within the Medicines and Healthcare products Regulatory Agency (MHRA). GPRD is funded by the MHRA, Medical Research Council, various universities, contract research organizations and pharmaceutical companies. The department of Pharmacoepidemiology & Pharmacotherapy, Utrecht Institute for Pharmaceutical Sciences has received unrestricted funding for pharmacoepidemiological research from GlaxoSmithKline, Novo Nordisk, the private–public funded Top Institute Pharma (![]() , includes co-funding from universities, government, and industry), the Dutch Medicines Evaluation Board, and the Dutch Ministry of Health.
, includes co-funding from universities, government, and industry), the Dutch Medicines Evaluation Board, and the Dutch Ministry of Health.
Acknowledgments
The views expressed in this article are those of the authors and do not reflect the official policy or position of the Medicines and Healthcare products Regulatory Agency, UK.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bastian Haß and Andreas Clemens are paid employees of Boehringer Ingelheim. At the time of the study Jonathan M. Plumb was a paid employee of Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Boehringer Ingelheim International GmbH (BI). BI has a compound in clinical development for the treatment of VTE.
