Abstract
Prothrombin time/international normalized ratio (PT/INR) is related to both antithrombotic effect and risk of bleeding. Its role in the prediction of venous thromboembolism (VTE) recurrence and bleeding for patients with acute VTE who undergo direct oral anticoagulants (DOACs) treatment is unclear, despite previous studies revealed some association between them. The predictive efficiency of INR for VTE recurrence and bleeding were analyzed in a retrospective cohort with VTE patients who underwent DOACs treatment. Then its predictive efficiency for VTE recurrence and bleeding were validated in a prospective cohort with the acquired cutoffs range, and compared with anti-Xa level, DASH and VTE-BLEED scores. In the retrospective cohort (n = 1083), the sensitivity and specificity of INR for the prediction of VTE recurrence were 79.4% and 92.8%, respectively. The area under the curve (AUC) was 0.881 (0.803-0.960)(P = .025). The cutoff value of INR was 0.9. The sensitivity and specificity of INR for the prediction of bleeding were 85.7% and 77.9%, respectively. The AUC was 0.876 (0.786-0.967)(P < .001). The cutoff value of INR was 2.1. In the prospective cohort (n = 202), the calibration showed that there were 4 (50%) patients with VTE recurrence, 156 (97.5%) patients with non-recurrence and bleeding (non-R&B), and 20 (58.8%) patients with bleeding in the low (INR < 0.9)(n = 8), intermediate (0.9 ≤ INR ≤ 2.1)(n = 160), and high (INR > 2.1)(n = 34) groups, respectively. The baseline PT/INR value at the initiation of DOACs treatment is an independent predictor for VTE recurrence and bleeding in patients with acute VTE who undergo DOACs treatment.
Keywords
Introduction
Venous thromboembolism (VTE) is a common and potentially fatal disease with considerable morbidity and mortality. Approximately 10 million people suffer from VTE globally every year. Broadly it comprises pulmonary embolism (PE), deep vein thrombosis (DVT), superficial vein thrombosis, and splanchnic vein thrombosis, whereas narrowly it comprises PE and DVT.1–3 For patients with established acute VTE, anticoagulant therapy for 3 or 6 months is recommended by authoritative guidelines. In recent years, the use of vitamin K antagonist (VKA) which is represented by warfarin has been gradually replaced by the use of direct oral anticoagulants (DOACs) for the anticoagulation of VTE.4–6 Despite standard and effective anticoagulation, recurrence still occurs in partial patients with acute VTE, and accounts for considerable morbidity and mortality. Meanwhile, bleeding is a major adverse event after the initiation of anticoagulation, and is also associated with morbidity and mortality. Predicting VTE recurrence and bleeding is crucial for VTE patients.1,2,4,5–7
Prothrombin time (PT) is a measurement of the integrity of extrinsic and final common pathways of coagulation cascade. It represents the time in seconds for the plasma to clot in patients after the addition of calcium and thromboplastin which is an activator of extrinsic pathway. 8 Prothrombin time/international normalized ratio (PT/INR, hereinafter referred to as INR) is the result of mathematical calculation using a PT ratio which is the PT of patients over a mean normal PT as further adjusted with a correction factor termed international sensitivity index.9,10 INR is related to both anticoagulant effect and the risk of bleeding. A high INR denotes hemorrhagic tendency, whereas a low one denotes thrombogenic tendency. It is usually used to guide the dose adjustment of VKA instead of DOACs, by maintaining its level in the range of 2.0 to 3.0.4–6,9,11,12
However, in a prospective comparative study that investigated whether the PT-INR level on the third postoperative day could predict the incidence of DVT in 286 patients undergoing total knee arthroplasty (TKA) and thromboprophylaxis of oral administration of 15 mg edoxaban once daily for 14 days, the INR levels were significantly higher in the patients without DVT compared with those with DVT. The INR on the third postoperative day was an independent factor of the incidence of DVT. It suggests that INR level can be used as a useful marker in determining whether 15 mg edoxaban thromboprophylaxis can prevent DVT after TKA. 13 In a prospective observational study involving 80 hospitalized patients treated with apixaban or rivaroxaban for the prevention and treatment of VTE or non-valvular atrial fibrillation, there was a strong correlation between anti-Xa concentrations and INR in patients treated with rivaroxaban, whereas the correlation was relatively weak in patients treated with apixaban. A formula by using INR values that estimated the anticoagulant activity of rivaroxaban was developed and then validated. 14 These studies have confirmed that INR has a predictive role for the anticoagulant efficacy in patients who receive DOACs anticoagulation. As such, we hypothesized that maybe INR had certain role in the prediction of VTE recurrence and bleeding for patients with acute VTE who undergo anticoagulation with DOACs. Accordingly, the present study was launched to address this topic.
Methods
Study Design
Patients hospitalized due to acute VTE who had undergone VTE reexamination upon the completion of 3 or 6 months of standard DOACs anticoagulation according to the recommendations in the guidelines5,6 were retrospectively incorporated into the current study. For patients with active cancer, hypercoagulability, or unprovoked VTE with previous VTE history, 6 months of anticoagulation was usually administrated. 15 DOACs included dabigatran, edoxaban, and rivaroxaban. The application of lead-in low molecular weight heparin (LMWH) for 5 to 10 days were administrated for patients with hemodynamic stability, prior to the treatment of dabigatran, edoxaban as well as rivaroxaban in the present study.1,2,5,6,16 The application of elastic compression stockings was implemented when indicated.1,2,4–6 All specific treatment measures were at the discretion of patients’ attending physicians, after patients’ benefits and risks being weighed, based on the abovementioned guidelines and the specific condition of patients. The baseline was defined as the moment of the initiation of DOACs treatment. The outcomes were the incidence of VTE recurrence by using imaging tests at the follow-up visit which was routinely performed at the completion of 3 or 6 months of DOACs treatment for all patients, and the clinically relevant bleeding events during the DOACs anticoagulation period. Then the predictive efficiency of INR was validated in a prospective cohort with hospitalized patients totally different from those in the retrospective cohort, simultaneously compared with that of the anti-Xa level, DASH (D-dimer, Age, Sex, Hormonal therapy) and VTE-BLEED (actiVe cancer, male with uncontrolled hyperTension at baseline, anaEmia, history of BLeeding, agE > _60 years, rEnal Dysfunction) scores in guidelines. 5 The parameters collected at the initiation of DOACs treatment were adopted for the INR level, anti-Xa level, DASH score, and VTE-BLEED score.
The definition of VTE recurrence was as follows: upon the completion of 3 or 6 months of standard anticoagulation, there are presence of new defect on ventilation/perfusion (V/Q) scan, new thrombi in the uninvolved pulmonary arteries on previous computed tomography pulmonary angiography (CTPA), new thrombi in the previously uninvolved venous segments or an extension of venous diameter more than 4 mm on the prior thrombi of previous compression ultrasonography (CUS). 17 The definition of bleeding conformed to the International Society on Thrombosis and Haemostasis (ISTH) criteria. Clinically relevant bleeding was defined as the composite of major bleeding and clinically relevant non-major bleeding.18,19 Major bleeding was defined as fatal bleeding, symptomatic bleeding in critical areas or organs, surgical site bleeding causing second intervention and/or hemodynamic instability, hemarthrosis resulting in immobilization, prolonged hospitalization, delayed wound healing, or deep wound infection, and bleeding causing a fall in hemoglobin level of 20 g/L or more, or a transfusion of 2 or more units of whole blood or red cells with temporal association within 24 to 48 h to the bleeding.18,19 Clinically relevant non-major bleeding was defined as any sign or symptom of hemorrhage that does not meet the definition of major bleeding20,21 but warrants medical intervention, increased level of care, hospitalization, or onsite evaluation. 22 VTE recurrence and bleeding events were adjudicated by investigators who were unaware of the INR, anti-Xa, DASH, and VTE-BLEED scores of patients. Provoked VTE was defined as VTE occurring after immobilization for at least 3 days or major surgery within 4 weeks prior to the diagnosis, in patients with active cancer, after recent long flights (>4 h), during pregnancy or peripartum period, and in patients receiving oral contraceptives or hormone replacement treatment. Unprovoked VTE was defined as VTE occurring without any of the abovementioned provoking factors. 23
For the retrospective cohort, the INR level at the initiation were compared among 3 groups including the recurrence group which contained patients with VTE recurrence, the bleeding group which contained patients with clinically relevant bleeding, and the non-recurrent VTE and bleeding (non-R&B) group which contained patients with neither VTE recurrence nor clinically relevant bleeding. The number summation of patients in all 3 groups may exceed the actual total number of all patients since there could be patients with concurrent VTE recurrence and clinically relevant bleeding. Correlation between INR and VTE recurrence as well as clinically relevant bleeding were analyzed. Efficiency of INR in the prediction of VTE recurrence and clinically relevant bleeding were analyzed. Cumulative time-dependent incidence of clinically relevant bleeding among patients with different strata of INR were compared. For the bleeding group, the correlation were analyzed between INR and first-time bleeding which was represented by the time interval between the initiation of anticoagulation and the moment of first clinically relevant bleeding event, as well as the times of clinically relevant bleeding events. By using the acquired cutoffs range of INR from the retrospective cohort, the predictive efficiency of the baseline INR for VTE recurrence and clinically relevant bleeding was validated and synchronously compared with that of mean level of peak and trough anti-Xa in the prospective cohort. In addition, the predictive efficiency of the baseline INR for VTE recurrence was compared with that of DASH score 24 in which post-VKA D-dimer was replaced by initial D-dimer, whereas that for clinically relevant bleeding was compared with VTE-BLEED score. 25
All the measurements were performed in the laboratory department of each participating hospital, by similar protocols. Pre-analytical handling of coagulation tests conformed to the Clinical and Laboratory Standards Institute (CLSI) H21-A5. 26 With respect to PT/INR, the instruments used were Sysmex automatic blood coagulometers, whereas the reagent kits used were ThrombopLastin-DS (Fisher Diagnostics, a division of Fisher Scientific, Co.L.L.C.). Drug calibrated anti-Xa was used in the current study. Anti-Xa was calibrated with rivaroxaban or edoxaban.27,28 D-dimer measurement conformed to the ISTH standardization. 29
Data for analyses were retrieved from electronic medical record systems of each participating hospital. The reporting of this study basically conformed to the STROBE statement. 30 All the patients in the prospective cohort signed informed consent. The study protocol was approved by the institutional review board of Shanghai Xinhua Hospital (approval number: XHEC-SHHDC-2020-002).
Study Population
As per the inclusion and exclusion criteria, eligible patients were incorporated into the current study. The inclusion criteria comprised: (1) All eligible patients were 18 years old or older. (2) All eligible patients were the hospitalized ones due to an established diagnosis of acute VTE after undergoing CTPA, CUS, and/or V/Q scan. (3) All eligible patients had no contraindication to DOACs and underwent DOACs anticoagulation for 3 or 6 months after the diagnosis of the acute VTE. In the case of clinically relevant bleeding, DOACs anticoagulation was transiently suspended till the recovery of bleeding, and reduced in dosage if bleeding happened again. (4) All eligible patients underwent CTPA, CUS, and/or V/Q scan to detect the absence or presence of recurrent VTE upon the completion of 3 or 6 months of DOACs anticoagulation, irrespective of the symptoms of VTE recurrence or not. The exclusion criteria comprised: (1) Patients who had an objective history of chronic thromboembolic disease (CTED). 5 (2) Patients who underwent thrombolysis due to hemodynamic instability, percutaneous catheter-directed treatment, surgical embolectomy, or implantation of inferior vena cava filter (IVCF), due to contraindication of anticoagulation. (3) Patients whose DOACs anticoagulation was prematurely (less than 3 months) terminated or suspended more than 1 month in the case of fatal major bleeding. (4) Patients who underwent concurrent antiplatelet agents therapy during the 3 or 6 months of DOACs treatment.
Statistical Analyses
Comparison of measurement data was performed by using T-test or ANOVA. Between-group comparison of rates was performed by Chi-square test. Correlation between INR and VTE recurrence as well as clinically relevant bleeding were analyzed by using multivariable Cox proportional hazard model, with the results expressed as hazard ratios and their 95% confidence intervals. Receiver operator characteristic (ROC) curve was used to analyze the predictive efficiency for VTE recurrence and clinically relevant bleeding by INR in the retrospective cohort and that by anti-Xa in the prospective cohort. Comparison of cumulative time-dependent incidence of clinically relevant bleeding among patients with different strata of INR was performed by using Kaplan-Meier curve analysis. For patients with clinically relevant bleeding in the retrospective cohort, the correlation between INR and first-time bleeding as well as times of bleeding events were performed by using Pearson correlation analysis.
For the prospective cohort, based on the population proportion of hospitalized acute VTE incidence which was maximum 2.69 per 1000 individuals according to a previous study, 31 a confidence level being 99%, and a margin of error being 1%, a total of 179 or more patients were needed to be measured. In consideration of a possibly unavoidable rate of lost to follow-up which was controlled to be below 10% as far as possible, a total of 199 or more patients were demanded. Pairwise comparison of diagnostic consistency and dominance tests between INR and anti-Xa in the prospective cohort were performed by using Cohen's Kappa coefficient analysis and McNemar's test, respectively. Statistical analyses were performed by using SPSS 26. A P-value being less than .05 denoted statistical significance.
Results
Characteristics of Patients in Retrospective Cohort
A total of 1253 hospitalized patients who underwent VTE reexamination following the completion of standard and effective anticoagulation for 3 or 6 months after the diagnosis of acute VTE between 2012 and 2021 from the participating hospitals were incorporated into the current study as per the inclusion criteria. According to the exclusion criteria, 17 patients with CTED, 36 patients who underwent thrombolysis, percutaneous catheter-directed treatment, or implantation of IVCF, 48 patients whose DOACs anticoagulation was prematurely terminated or suspended more than 1 month, and 69 patients who underwent concurrent antiplatelet agents therapy with DOACs treatment were excluded. A total of 1083 patients in the retrospective cohort entered into the analytical procedures.
The mean age of all patients in the retrospective cohort was 68.6 years old. The number of female and male patients were 519 (47.9%) and 564 (52.1%), respectively. Among all 1083 patients with VTE, PE, DVT, and PE with DVT were 329 (30.4%), 301 (27.8%), and 453 (41.8%), respectively. A total of 727 patients underwent 3 months of DOACs treatment, whereas 356 ones underwent 6 months of DOACs treatment. After the VTE reexamination following the completion of 3 or 6 months of standard DOACs anticoagulation, the number of patients in the recurrence, bleeding, and non R&B groups were 31, 150, and 908, respectively. There were 6 patients who developed both VTE recurrence and clinically relevant bleeding (Table 1).
Demographics and Clinical Characteristics of all Patients at Baseline.
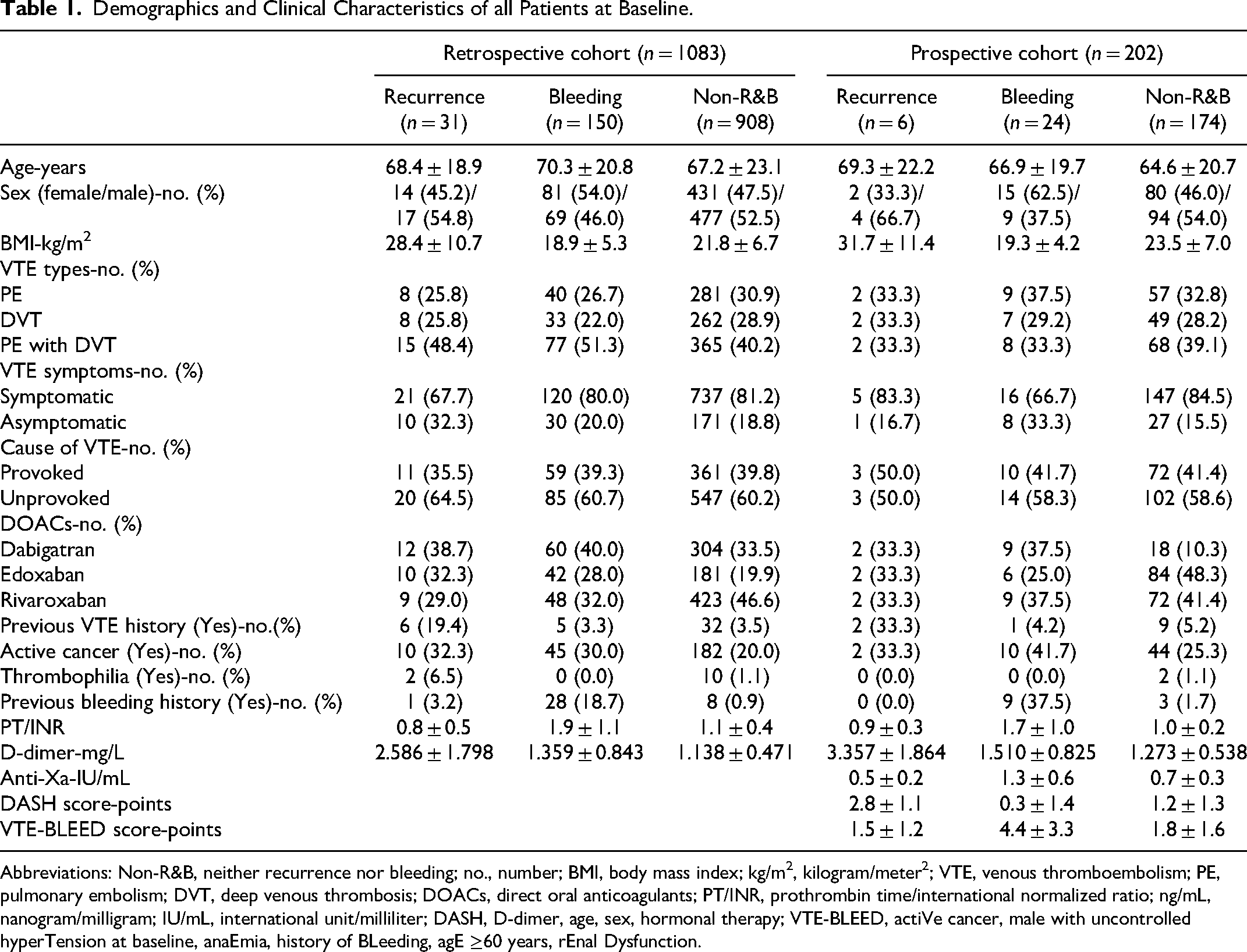
Abbreviations: Non-R&B, neither recurrence nor bleeding; no., number; BMI, body mass index; kg/m2, kilogram/meter2; VTE, venous thromboembolism; PE, pulmonary embolism; DVT, deep venous thrombosis; DOACs, direct oral anticoagulants; PT/INR, prothrombin time/international normalized ratio; ng/mL, nanogram/milligram; IU/mL, international unit/milliliter; DASH, D-dimer, age, sex, hormonal therapy; VTE-BLEED, actiVe cancer, male with uncontrolled hyperTension at baseline, anaEmia, history of BLeeding, agE ≥60 years, rEnal Dysfunction.
Correlation Between Risk Factors and VTE Recurrence as Well as Bleeding
Among the variables related to VTE recurrence and bleeding, which comprised age, sex, body mass index (BMI), active cancer, recent surgery, unprovoked VTE, symptoms of VTE, previous VTE history, immobilization, pregnancy, autoimmune diseases, thrombophilia, previous bleeding history, hepatic dysfunction, renal dysfunction, uncontrolled hypertension, INR, and D-dimer, a sequential univariable and multivariable Cox regression analysis showed that sex, BMI, cause of VTE, previous VTE history, active cancer, thrombophilia, previous bleeding history, INR, and D-dimer were correlated with VTE recurrence or bleeding. Male sex (1.433 [1.104-1.828], P = .001), BMI ≥ 30 kg/m2 (1.622[1.234-2.010], P = .001), unprovoked VTE (1.858[1.267-2.449], P < .001), previous VTE history (2.025 [1.380-2.670], P < .001), active cancer (1.488 [1.127-2.030], P = .001), thrombophilia (1.731 [1.359-2.103], P = .001), INR < 1.0 (1.505 [1.137-1.873], P = .001), and D-dimer ≥ 500 ng/mL (1.453 [1.132-1.774], P = .005) were in favor of VTE recurrence, whereas female sex (0.713 [0.516-0.910], P = .008), previous bleeding history (0.618 [0.374-0.862], P < .001), and INR ≥ 1.0 (0.701 [0.476-0.936], P = .001) were in favor of bleeding. The rest risk factors were neither correlated with VTE recurrence nor bleeding. The INR value was an independent predictor for VTE recurrence and clinically relevant bleeding (Figure 1).
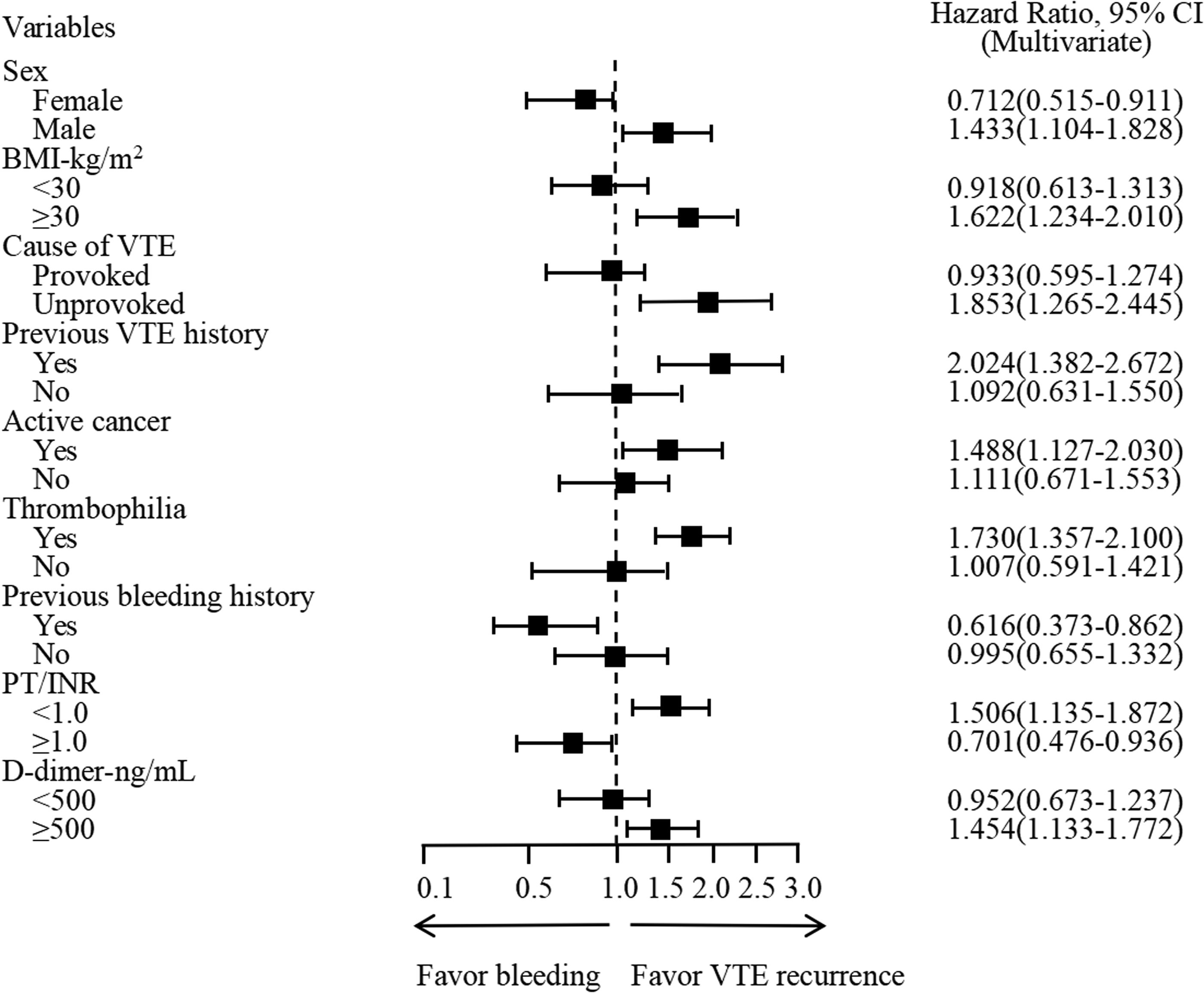
Correlation between risk factors and VTE recurrence as well as bleeding.
Prediction of VTE Recurrence and Bleeding by INR
In a ROC curve analysis by using baseline INR value to predict VTE recurrence, the sensitivity and specificity were 79.4% and 92.8%, respectively. The area under the curve (AUC) was 0.881 (0.803-0.960) (P = .025). The cutoff value of INR was 0.9. An INR being lower than 0.9 implied a higher risk of VTE recurrence than an INR being higher than 0.9. With respect to the prediction of clinically relevant bleeding, the ROC curve analysis revealed that the sensitivity and specificity were 85.7% and 77.9%, respectively, by using the baseline INR value. The AUC was 0.876 (0.786-0.967) (P < .001). The cutoff value of INR was 2.1. An INR being higher than 2.1 implied a higher risk of bleeding than an INR being lower than 2.1 (Figure 2).
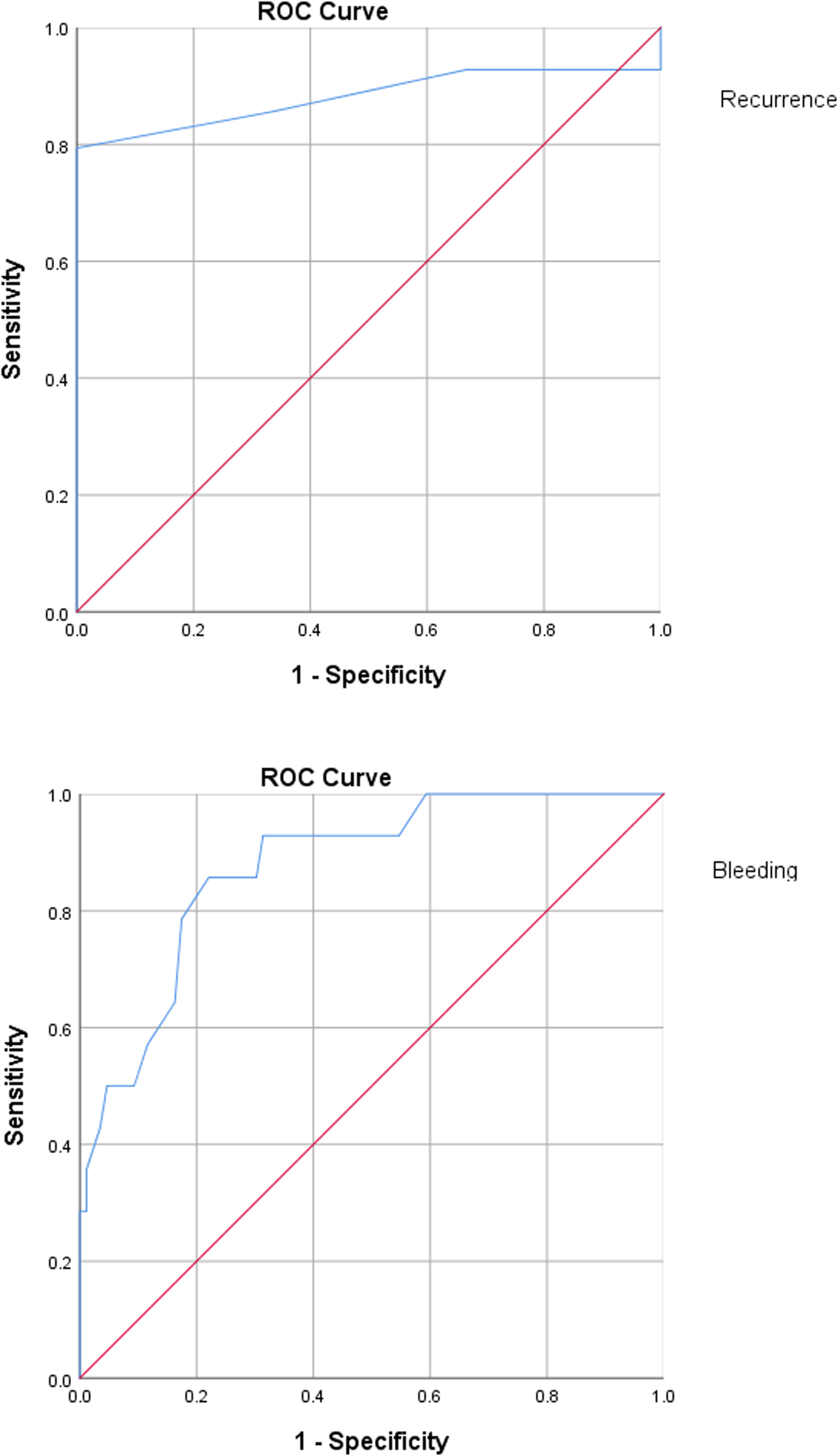
Prediction of VTE recurrence and bleeding by INR.
Cumulative Time-Dependent Bleeding Incidence in Patients With Different INR
With the acquired cutoffs of baseline INR value which ranged from 0.9 to 2.1 in the retrospective cohort, all the patients were classified into the low INR subgroup (INR < 0.9) (n = 346), intermediate INR subgroup (0.9 ≤ INR ≤ 2.1) (n = 534) and high INR subgroup (INR > 2.1) (n = 203). The cumulative incidence of bleeding in the low, intermediate, and high INR subgroups were 36 (10.4%), 72 (13.5%), and 42 (20.7%), respectively (P < .001). Clinically relevant bleeding events occurred more and sooner among the patients in the high INR subgroup than the low INR subgroup, whereas the bleeding events in the intermediate INR subgroup fell in between (Figure 3).
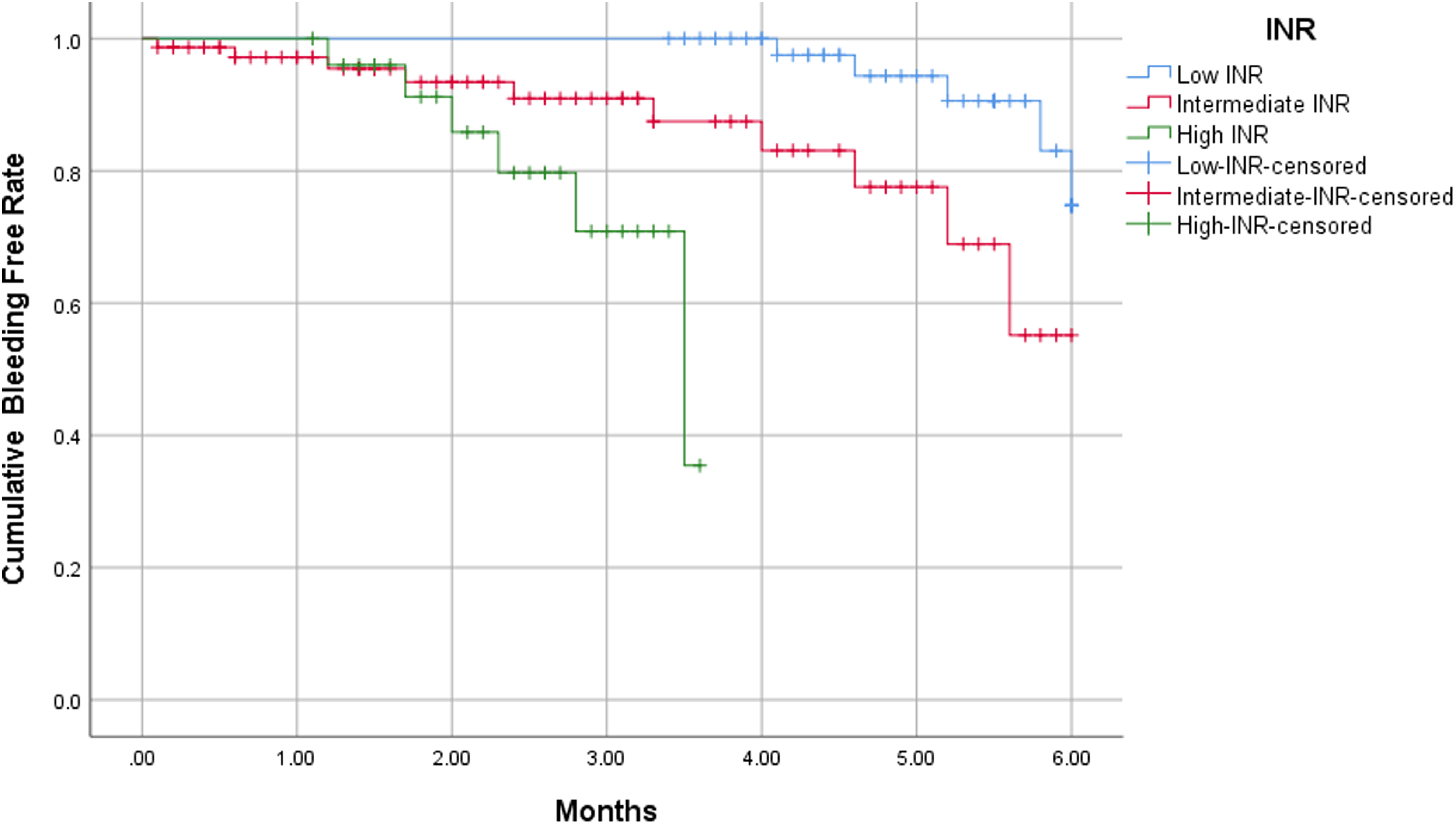
Cumulative time-dependent bleeding incidence in patients with different INR.
Correlation Between INR and First-Time Bleeding as Well as Times of Bleeding
In a Pearson correlation analysis, it was revealed that the baseline INR value was correlated with the first-time bleeding, among the patients with clinically relevant bleeding (R = 0.687, P < .001). The higher the baseline INR value, the sooner the first-time clinically relevant bleeding event, and vice versa. The Pearson correlation analysis also indicated that the baseline INR value was correlated with the times of clinically relevant bleeding events during the 3 or 6 months of DOACs treatment (R = 0.780, P < .001). The higher the baseline INR value, the more the clinically relevant bleeding events (Figure 4).
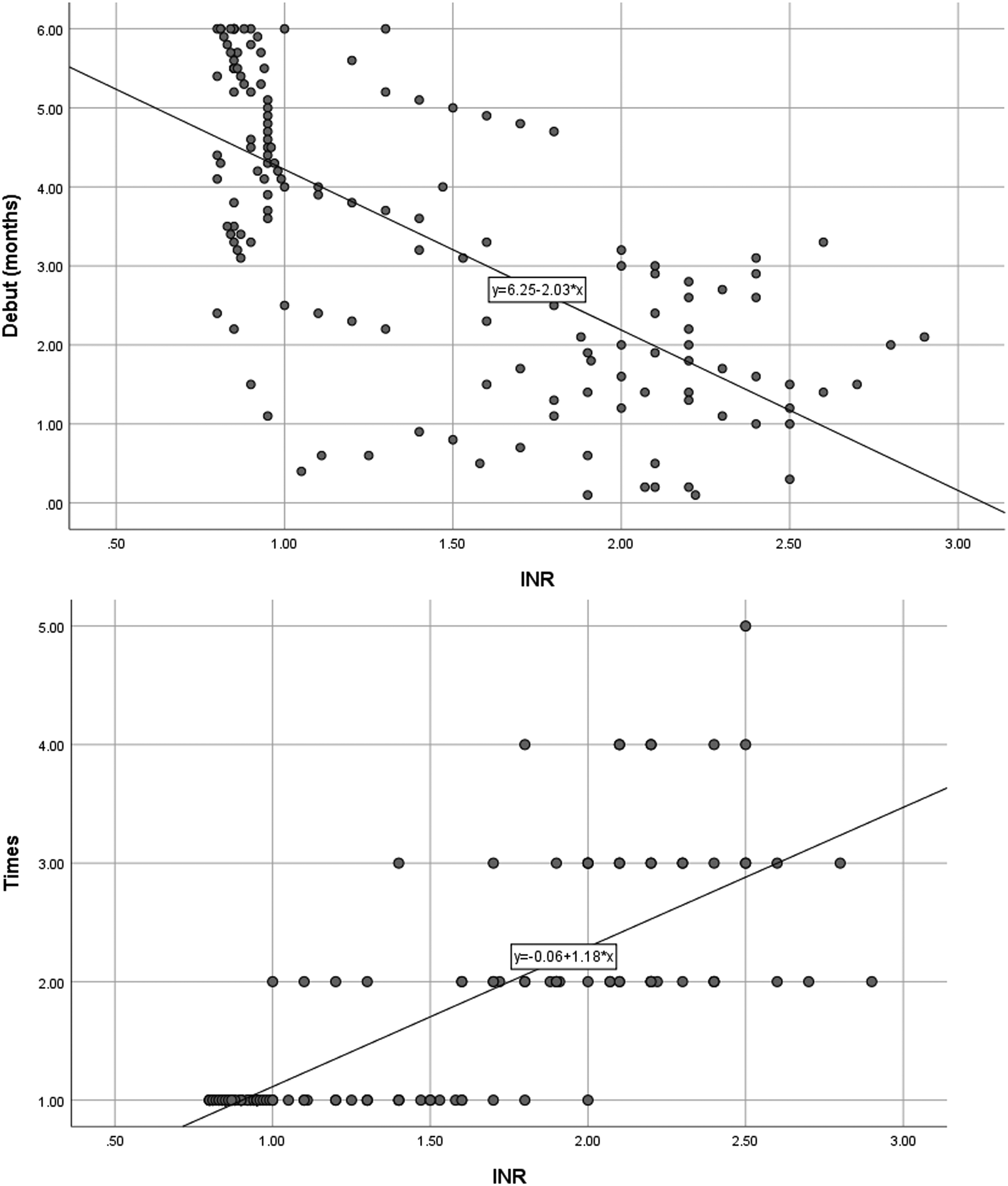
Correlation between INR and first-time bleeding as well as times of bleeding.
Validation of Predictive Efficiency of INR for Outcomes
From Jan, 2021 through Apr, 2022, a total of 239 VTE patients from the participating hospitals were consecutively incorporated into the prospective cohort based on the inclusion criteria. According to the exclusion criteria, 2 patients with CTED, 5 patients who underwent thrombolysis or implantation of IVCF, 4 patients whose DOACs anticoagulation was prematurely terminated or suspended more than 1 month, and 16 patients who underwent concurrent antiplatelet agents therapy with DOACs treatment were excluded. With 10 patients lost to follow-up, a total of 202 patients completed the whole study (Table 1).
According to the acquired cutoff range of INR and the baseline INR value of the patients in the prospective cohort, they were stratified into the low (INR < 0.9) (n = 8), intermediate (0.9 ≤ INR ≤ 2.1)( n = 160), and high (INR > 2.1) (n = 34) INR groups, which denoted possible recurrence, non-R&B, and bleeding, respectively. After the 3 or 6 months of DOACs therapy, the calibration demonstrated that there were actually 4 (50%) patients with VTE recurrence, 156 (97.5%) patients with non-R&B, and 20 (58.8%) patients with bleeding in the low, intermediate, and high INR groups, respectively.
As a control, anti-Xa level was adopted as a potential biomarker for the prediction of VTE recurrence and bleeding for the prospective cohort. In a ROC curve analysis, the sensitivity and specificity of baseline anti-Xa level were 83.2% and 69.4% for the prediction of VTE recurrence, respectively. The AUC was 0.711 (0.542-0.880) (P = .037). The cutoff value of anti-Xa was 0.6 IU/mL. An anti-Xa level being lower than 0.6 IU/mL implied a higher risk of VTE recurrence than an anti-Xa level being higher than 0.6 IU/mL. Meanwhile, the sensitivity and specificity of baseline anti-Xa level were 74.8% and 65.7% for the prediction of bleeding, respectively. The AUC was 0.683 (0.535-0.831) (P = .029). The cutoff value of anti-Xa was 1.4 IU/mL. An anti-Xa level being higher than 1.4 IU/mL implied a higher risk of bleeding than an anti-Xa level being lower than 1.4 IU/mL. Based on the acquired cutoff range of anti-Xa and the baseline anti-Xa level of the patients in the prospective cohort, they were stratified into the low (n = 9), intermediate (n = 155), and high (n = 38) anti-Xa groups, which denoted possible recurrence, non-R&B, and bleeding, respectively. In a calibration, there were 3 (33.3%) patients with VTE recurrence, 148 (95.5%) patients with non-R&B, and 16 (42.1%) patients with bleeding in the low, intermediate, and high anti-Xa groups, respectively. In the comparison of diagnostic consistency between INR and anti-Xa, the Kappa coefficient was 0.375 (P < .001). For the comparison of dominance between INR and anti-Xa, the P-value of McNemar's test was .008 (Table 2).
Calibration of Predictive Efficiency of INR and Anti-Xa in Prospective Cohort.
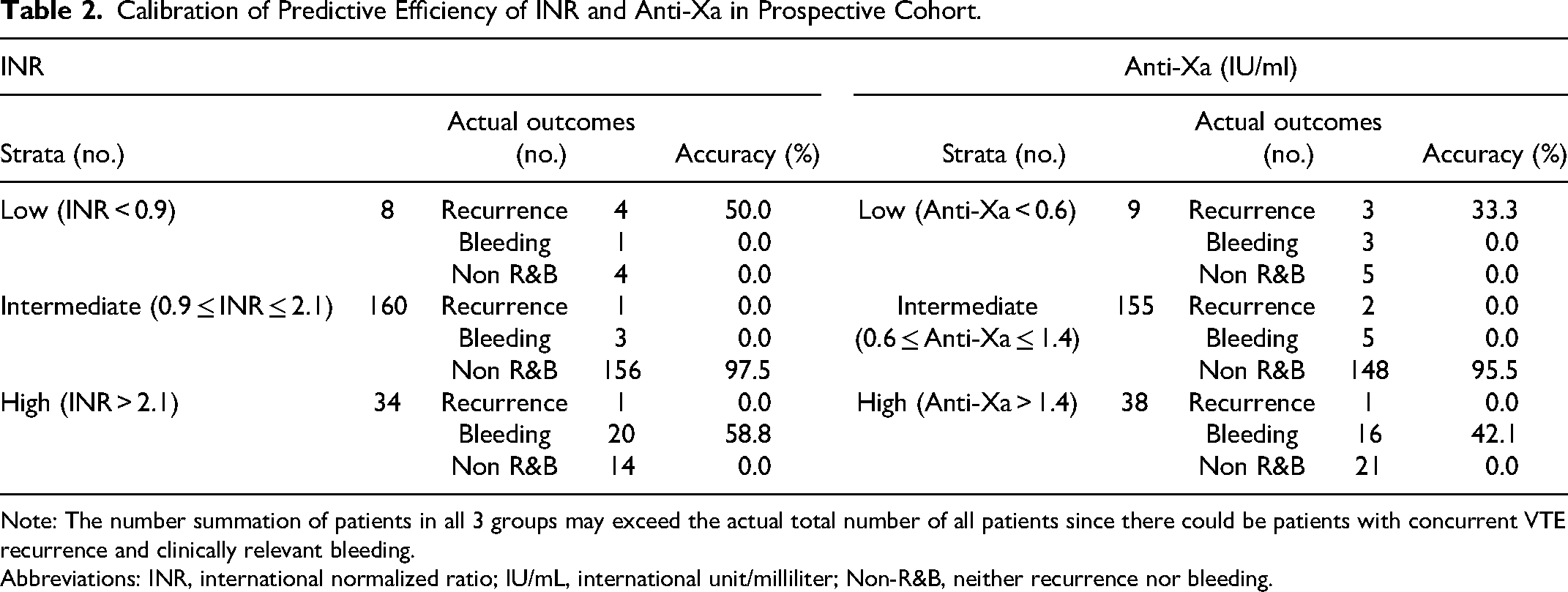
Note: The number summation of patients in all 3 groups may exceed the actual total number of all patients since there could be patients with concurrent VTE recurrence and clinically relevant bleeding.
Abbreviations: INR, international normalized ratio; IU/mL, international unit/milliliter; Non-R&B, neither recurrence nor bleeding.
According to DASH score, 12 patients were identified as “VTE recurrence likely,” in which actually 4 (33.3%) patients had recurrence, being less than the calibration rate (50%) of a low INR (<0.9) (P = .015). According to VTE-BLEED score, 35 patients were identified as “bleeding likely,” in which actually 18 (51.4%) patients had bleeding, being similar to the calibration rate (58.8%) of a high INR (>2.1) (P = .256).
A subgroup analysis with regard to the validation of predictive efficiency of INR for outcomes among patients who underwent different DOACs types was conducted for the prospective cohort. For patients who underwent dabigatran (n = 29), the accuracy for predicting VTE recurrence by the low INR was 50% (1/2), for predicting non-R&B by the intermediate INR was 95.8% (23/24), for predicting bleeding by the high INR was 33.3% (1/3). For patients who underwent edoxaban (n = 92), the accuracy for predicting VTE recurrence by the low INR was 40% (2/5), for predicting non-R&B by the intermediate INR was 98.6% (68/69), for predicting bleeding by the high INR was 55.6% (10/18). For patients who underwent rivaroxaban (n = 83), the accuracy for predicting VTE recurrence by the low INR was 50% (1/2), for predicting non-R&B by the intermediate INR was 97.0% (65/67), for predicting bleeding by the high INR was 64.3% (9/14). The results of subgroup analysis were similar to the overall results, except the accuracy for predicting bleeding by the high INR (33.3%) of dabigatran was lower than those of the other 2 DOACs (P < .001).
Discussion
The findings interpreted from the current results were as follows: (1) The PT/INR value at the initiation of DOACs treatment was an independent predictor for VTE recurrence and bleeding in patients with acute VTE who underwent 3 or 6 months of oral DOACs treatment. (2) The baseline INR value could be used to predict VTE recurrence and clinically relevant bleeding in a short-term which was 3 or 6 months. Its predictive efficiency outperformed that of anti-Xa level and DASH score.
Although there were no previous studies comparable to this one, the current findings are basically consistent with those in 2 relevant previous studies.13,14 In the study of Kodato et al, a lower INR level was associated with a higher risk of DVT occurrence in patients receiving edoxaban thromboprophylaxis, 13 being similar with our study except ours targeted VTE recurrence. In the study of Ofek et al, there was a strong correlation between INR level and anti-Xa concentrations which is used to monitor DOACs anticuagulation, in patients receiving rivaroxaban for the prevention and treatment of VTE. Although there is no direct evidence, the study of Ofek et al indirectly suggests that INR may be predictive of the occurrence or recurrence of VTE in patients receiving DOACs, being in favor of the current study. Nevertheless, both of the 2 studies did not concern the role of INR in bleeding for patients receiving DOACs, which was clarified in the current study.
Despite INR has not been considered as a predictor for VTE recurrence according to the guidelines,5,32 it was revealed that, an INR value <1.5 during the first 3 months of treatment was a significant risk factor for a late VTE recurrence during a 21-month follow-up, in a prospective study including 297 patients with acute unprovoked VTE who underwent oral anticoagulation therapy (warfarin as the first-choice agent). 33 On the other hand, INR is also regarded as a predictor for bleeding. The “L” in the “HAS-BLED” that stands for “Labile INR” which is defined as a therapeutic time in range < 60% is adopted as one of the items in the HAS-BLED prediction model for quantifying bleeding risk.5,34 According to a network meta-analysis, HAS-BLED score is a balanced bleeding risk assessment tool in terms of sensitivity and specificity. 35 In addition, in another bleeding risk assessment model (RAM) which is termed IMPROVE bleeding score, hepatic failure which is defined as INR >1.5 is adopted as one of the items of the RAM. 36
Anti-Xa level which is high associated with DOACs is also strongly correlated with INR. 14 The ISTH guidelines favor PT as the test for screening and anti-Xa assay as the test for the quantification of rivaroxaban. The CHEST guidelines also favor anti-Xa assay for the quantification of rivaroxaban. Nevertheless, neither guidelines ascertain the assays specific for edoxaban. In addition, with respect to dabigatran, the ISTH guidelines recommend activated partial thromboplastin time as the test for screening and dilute thrombin time (TT) as the test for quantification, whereas the CHEST guidelines recommend the TT and ecarin clotting time (ECT), respectively, instead of anti-Xa. PT/INR is also recommended for the detection of factor Xa inhibitors in the absence of anti-Xa assay, and for the detection of dabigatran in the absence of TT or ECT.37,38,39
It is worth noting that the BMI was relatively high in the recurrence group, whereas relatively low in the bleeding group, in both retrospective and prospective cohorts of the current study. In the correlation analysis, BMI ≥30 kg/m2 was associated with VTE recurrence, whereas BMI <30 kg/m2 was associated with neither VTE recurrence nor bleeding. This is relatively close to a retrospective study, in which obese patients >120 kg receiving DOACs or VKA were associated with increased VTE recurrence, without significant differences in bleeding, compared to those ≤120 kg. 40 Nevertheless, recent retrospective studies suggested that the use of DOACs at conventional doses in obese patients with VTE yielded similar rates of VTE recurrences or major bleeding to patients with normal weight.41,42 Evidence from large-scale randomized controlled trial regarding this issue may be needed in the future.
Certain clinical implications may derive from the current study. First, clinicians may need to pay attention to the PT/INR value at the initiation of DOACs anticoagulation, so as to assess the risk of VTE recurrence or bleeding in the future. Accordingly, for patients with either risk of VTE recurrence or bleeding, caveat should be given to alert them for the possible recurrence or bleeding. Second, in case of anti-Xa assay is unavailable, INR could be an alternative after the lead-in of LMWH to evaluate the efficiency and safety of the pending DOACs use. Third, INR may be more widely applicable than anti-Xa is. With respect to patients who receive dabigatran, INR may be the only option between itself and anti-Xa, for the prognostic prediction after use. Last, the current findings did not suggest that DOACs should be used in the same way that VKA is used by adjusting dose with INR, since there was no evidence supporting that.
Limitations
Limitations should be acknowledged for the current study. First, the first part of the study was of retrospective nature, whereas the second prospective part of the study contained an inconsiderable sample size albeit it has met the statistical requirements. Relevant large prospective studies are warranted in the future. Second, the endpoint INR and its dynamic changes during the period of anticoagulation were unclear. The role of interim INR or the trend of INR change in the prediction of VTE recurrence and bleeding remains undefined. Nevertheless, this does not necessarily influence the predictive performance of baseline INR. Third, although anti-platelet therapy concurrent with DOACs was excluded, there still could be difference in initial risk of VTE recurrence or bleeding and in factors affecting initial INR, which may affect the outcomes of patients. Nevertheless, it is unnecessary to equalize all the predisposing factors related to VTE recurrence or bleeding, or factors affecting initial INR among all patients, for their existence do not directly concern the performance of INR in predicting the outcomes of patients. In addition, the multivariable Cox regression analysis in this study demonstrated that INR was an independent predictor of VTE recurrence or bleeding, regardless of the difference in initial risks associated with outcomes among patients. Fourth, the current study can merely indicate that INR predicts VTE recurrence and clinically relevant bleeding in the first 3 or 6 months of anticoagulation period. In the cases beyond that period, it remains unclear that whether INR still works. Last, all the patients being studied underwent a lead-in LMWH anticuagulation for 5 to 10 days prior to DOACs treatment. Accordingly, the current results may not be applicable for VTE patients who undergo DOACs anticoagulation without a LMWH lead-in.
Conclusions
In conclusion, baseline PT/INR value at the initiation of DOACs treatment is an independent predictor for VTE recurrence upon the completion of 3 or 6 months of anticoagulation, and clinically relevant bleeding during the first 3 or 6 months of anticoagulation. The current findings may contribute a fresh and accessible prognostic predictor to patients with acute VTE who receive DOACs anticoagulation.
Footnotes
Author Contributions
Concept and design: WX, YZ. Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: All authors. Critical revision of the manuscript for important intellectual content: All authors. Statistical analysis: WX, YZ, YC, MX. Administrative, technical, or material support: WX, YZ. Supervision: WX, YZ. Guarantor: WX, YZ.
Data Sharing Statements
Data are available upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study protocol was approved by the institutional review boards of Shanghai Xinhua Hospital (approval number: XHEC-SHHDC-2020-002).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the key sub-specialty construction funding of the Pudong Health System (PWZy2020-15), the construction funding of clinical characteristic discipline of Pudong Health System (PWYts2021-04), and the international talent training program of Shanghai Xinhua Hospital (2023YGJRC04).
