Abstract
Deep venous thrombosis (DVT) is a complication of stroke. Our aim was to determine the frequency of DVT in patients with acute stroke, risk factors for its development, and its influence on the 3-month outcome. A total of 323 consecutive patients with acute stroke were enrolled. We performed ultrasound imaging within 7 days after stroke. Deep venous thrombosis was found in 8.7% of patients, only in those with ischemic stroke. Patients with DVT were more frequently female (71.4% vs 49.5%), had prestroke Modified Rankin scale (mRS) 3 to 5 (42.9% vs 15.3%), elevated C-reactive protein (CRP) serum level (65.4% vs 32.5%), and a trend toward elevated serum fibrinogen level (85.7% vs 70.1%; P = .08). In a multivariate analysis, elevated CRP (odds ratio [OR] 3.15) and prestroke disability (OR 2.89) were independent risk factors for DVT. Deep venous thrombosis occurs in <10% of patients with acute stroke and does not significantly affect the 3-month outcome. Prestroke dependency and elevated CRP level at baseline are independent risk factors for DVT.
Introduction
Venous thromboembolism (VTE), including deep venous thrombosis (DVT) and pulmonary embolism (PE), is a potentially life-threatening complication in surgical and nonsurgical patients. Deep venous thrombosis has been reported to develop in 10% to 75% of immobilized poststroke patients. Such a wide range of results may be attributed to methodological differences between particular studies (eg, DVT diagnostic method, its timing, postmortem verification, symptomatic and asymptomatic DVT, immobilized or mobile patients, and stroke severity) and is a good rationale for further observational studies. 1–5 Pulmonary embolism, which may be DVT’s sequel, is considered the most common cause of autopsy-verified death between the second and fourth week after stroke. 6 It affects up to 20% of patients and leads to 1% to 2% of ischemic stroke-related deaths in hemiplegic patients not treated with prophylactic doses of heparin. 7
Previous studies suggest that the risk of DVT in patients with ischemic stroke is associated with older age, 8 atrial fibrillation, 9 and a higher degree of limb paresis. 10
Most studies addressing the issue of DVT and stroke tend to focus on patients with lower limb paresis. However, for epidemiological purposes, it seems more reasonable to include all patients with stroke, irrespective of the level of lower limb paresis.
Our aim was to determine the frequency of DVT in a real-world population of patients with acute stroke, identify risk factors for its development, and the influence of DVT on a 3-month stroke outcome.
Material and Methods
We enrolled consecutive acute stroke patients admitted to our Stroke Unit in 2 periods (December 2007 to May 2008 and August 2008 to May 2009). The diagnosis of stroke was based on clinical signs, symptoms, and brain computed tomography (CT) imaging at admission. Patients with subarachnoid hemorrhage (SAH) were excluded from the study.
Information about preexisting comorbidities and receiving oral anticoagulants or heparins were obtained from medical records and patients (or their proxies if necessary). We also collected data on their serum inflammatory markers (ie, routine C-reactive protein [CRP] and fibrinogen) within the first 24 hours of hospital stay. Normal range of CRP level in our laboratory was <10 and <4 mg/dL for fibrinogen.
Prestroke disability and neurological deficit at admission were measured with Modified Rankin scale (mRS) and National Institute of Health Stroke scale (NIHSS), respectively. Stroke severity was defined as follows: mild NIHSS ≤7 points (pts), moderate NIHSS 8 to 14 pts, and severe NIHSS > 14 points. Rankin score was evaluated by a physician blinded to ultrasonographic (USG) result.
Stroke outcomes were overall 3-month mortality and combined death or dependency (3-6 points in mRS). The follow-up examination was done during a routine visit in the outpatient clinic 3 months after stroke onset by an experienced physician blinded to the patient’s DVT status. In case of missed appointment, we interviewed the patient or a proxy by phone.
None of DVT prevention methods were used routinely. As there are no direct guidelines regarding stroke patients, prophylactic doses of low-molecular-weight heparins (LMWHs) were administered at physican’s discretions in cases clinically judged to be at increased risk of developing DVT. Patients diagnosed with DVT were treated with full doses of LMWH. Thromboembolic deterrent stockings (TEDSs) were not used in our center.
Deep Venous Thrombosis Detection Methods
Ultrasonographic (USG) examination was performed within the first 7 days after admission by an experienced trained physician (J.P.B.) blinded to patients’ baseline factors. We used Vivid 7 Dimension (Georgia) with the 7- to 10-MHz linear probe. The diagnosis of DVT was made if a noncompressible segment was present (compression ultrasound [CUS] test) or flow impairment was identified on color Doppler imaging. This technique is recognized as sufficiently sensitive and specific, especially in the detection of proximal DVT. Its sensitivity in detection of DVT in the lower limbs is estimated between 97% and 100% and specificity between 98% and 99%. 11,12 Patients were examined for both proximal (localized in the popliteal, femoral, and common femoral vein) and distal (peroneal and tibial veins) DVT. The type of DVT (ie, symptomatic or asymptomatic) was determined by the treating physician and stated in the medical records.
Ethics Committee Approval
The study was approved by the local Ethics committee. As DVT screening with Doppler USG is noninvasive and safe, we relied on patient’s or proxy verbal informed consent. Outcome evaluation at 3 months after stroke was implemented in our department’s clinical routine prior to the study, so separate informed consent was not necessary.
Statistical Methods
Categorical variables were presented as a ratio with number of valid observations and continuous variables as median with quartiles (Q1 and Q3). By calculating the proportions, we excluded unknown values from the denominator. In basic comparative statistics, we applied chi-square test or 2-sided Fisher exact test, and Mann-Whitney U test, as appropriate.
Multivariate logistic regression was adjusted for major independent outcome predictors. To avoid variable selection caused by spurious correlations, only variables showing a relationship to the outcome (defined P < .1 in the univariate model) were included as potential predictors. Subsequently, we used an interactive backward stepwise approach with the P < .05 of the likelihood ratio test for exclusion of excess factors to identify the final multivariate model for each major outcome. We considered P value <.05 statistically significant. For statistical analyses Statistica 8.0 (StatSoft, Inc, 2008) was used.
Results
In the recruitment period of 16 months, a total of 425 acute stroke patients were admitted to our stroke unit. We excluded 102 patients who did not undergo USG evaluation within 7 days after admission due to death, early discharge, or unstable general condition not allowing proper evaluation in the USG laboratory. Those patients had more severe neurologic deficit at baseline (median NIHSS score 8.5 vs 5.0 pts; P < .001), more frequently decreased level of consciousness (39.8% vs 15.8%; P < .001), and higher ratio of prestroke disability (mRS score 2-5 pts 35.3% vs 26.6%; P = .09). Thirty-nine (38%) not included patients died during the hospital stay at a median of 2 days (interquartile range [IQR]: 2-4). Detailed description of both groups is presented in Table 1 .
Comparative Characteristic of the Included and not Included Patients a
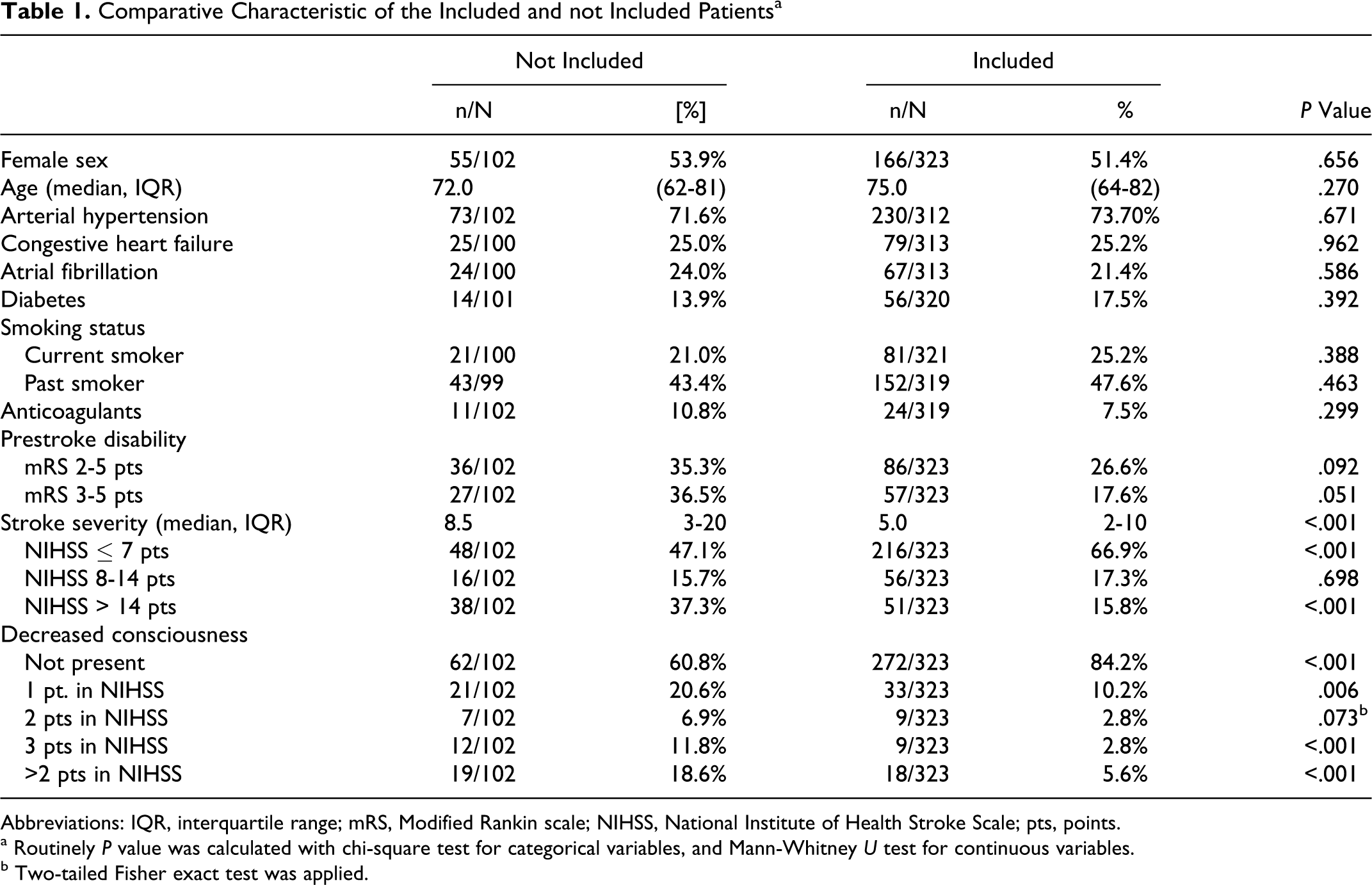
Abbreviations: IQR, interquartile range; mRS, Modified Rankin scale; NIHSS, National Institute of Health Stroke Scale; pts, points.
a Routinely P value was calculated with chi-square test for categorical variables, and Mann-Whitney U test for continuous variables.
b Two-tailed Fisher exact test was applied.
The final analysis involved 323 Caucasian patients admitted with acute ischemic (92.9%) and hemorrhagic (7.1%) stroke. Detailed baseline characteristic and outcomes are presented in Table 2 . Ultrasound examination was performed with median delay of 3 days from stroke onset (IQR: 2-5 days).
Baseline Characteristics of the Study Population and Outcomes
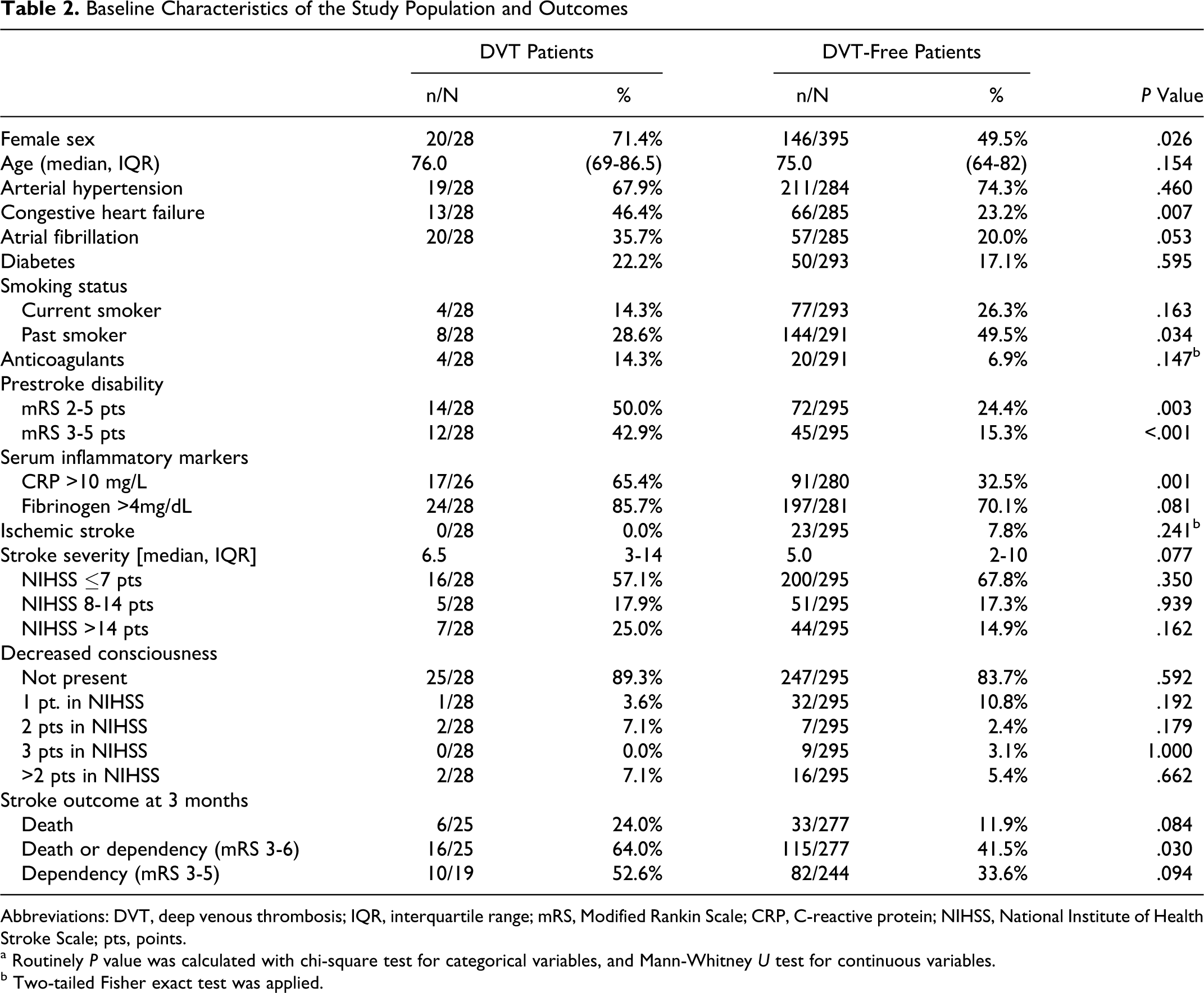
Abbreviations: DVT, deep venous thrombosis; IQR, interquartile range; mRS, Modified Rankin Scale; CRP, C-reactive protein; NIHSS, National Institute of Health Stroke Scale; pts, points.
a Routinely P value was calculated with chi-square test for categorical variables, and Mann-Whitney U test for continuous variables.
b Two-tailed Fisher exact test was applied.
Deep venous thrombosis was detected in 28 (8.7%) patients. It was predominantly distal (n = 22; 7.4%), and in 1 patient it was bilateral. Only 6 of 28 patients with DVT had associated clinical symptoms. It was edema or/and pain of the lower extremity. Patients with DVT were more frequently female (71.4% vs 49.5%; P < .05), dependent prior to stroke (42.9% vs 15.3%; P < .001), and had elevated CRP serum levels (65.4% vs 32.5%; P < .001). There was also a tendency toward elevated fibrinogen serum level (85.7% vs 70.1%; P = .08) and higher NIHSS score at baseline (median 6.5 vs 5.0; P = .08). Detailed comparative statistics are presented in Table 2.
We found no association between the development of DVT and the level of lower limb paresis (Table 3 ).
Association Between the Development of DVT and the Level of Lower Limb Paresis
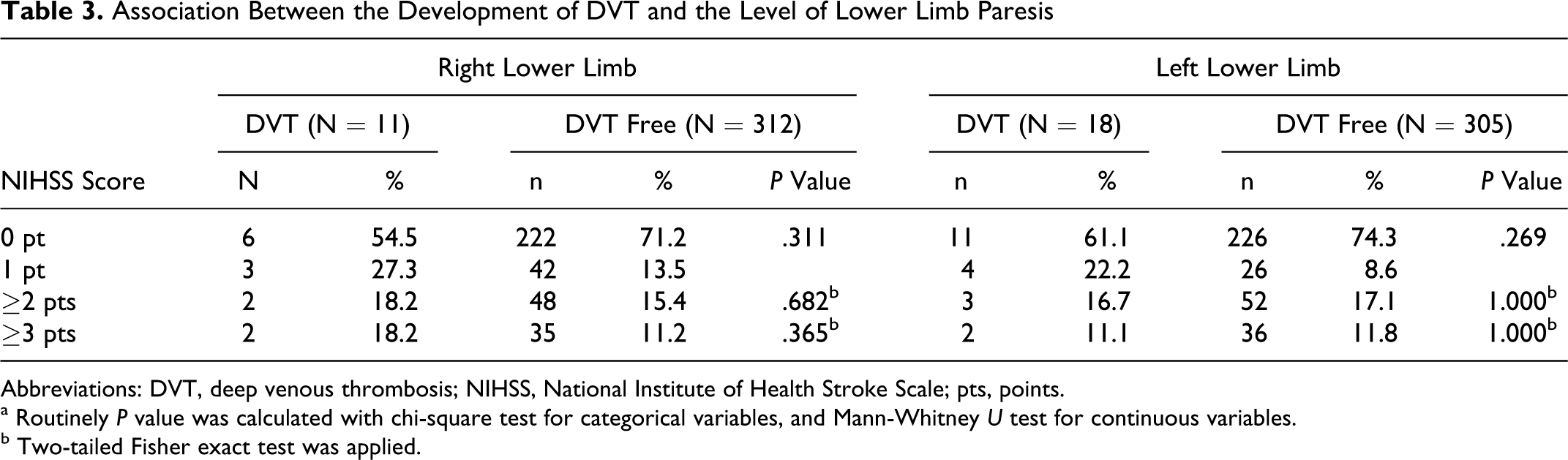
Abbreviations: DVT, deep venous thrombosis; NIHSS, National Institute of Health Stroke Scale; pts, points.
a Routinely P value was calculated with chi-square test for categorical variables, and Mann-Whitney U test for continuous variables.
b Two-tailed Fisher exact test was applied.
Elevated serum CRP level, with odds ratio (OR) 3.15 (95% confidence interval [CI]: 1.31-7.60), and prestroke dependency (mRS score of 3-5 points), with OR 2.89 (95%CI: 1.19-7.05), proved to be independent risk factors for DVT in multivariate logistic regression. There was also a strong tendency for congestive heart failure diagnosed before stroke onset (OR 2.23; 95% CI: 0.94-5.27). Positive history of smoking showed a tendency toward decreased risk of DVT (OR 0.44; 95% CI: 0.17-1.14). Detailed ORs for DVT are presented below (Figure 1 )
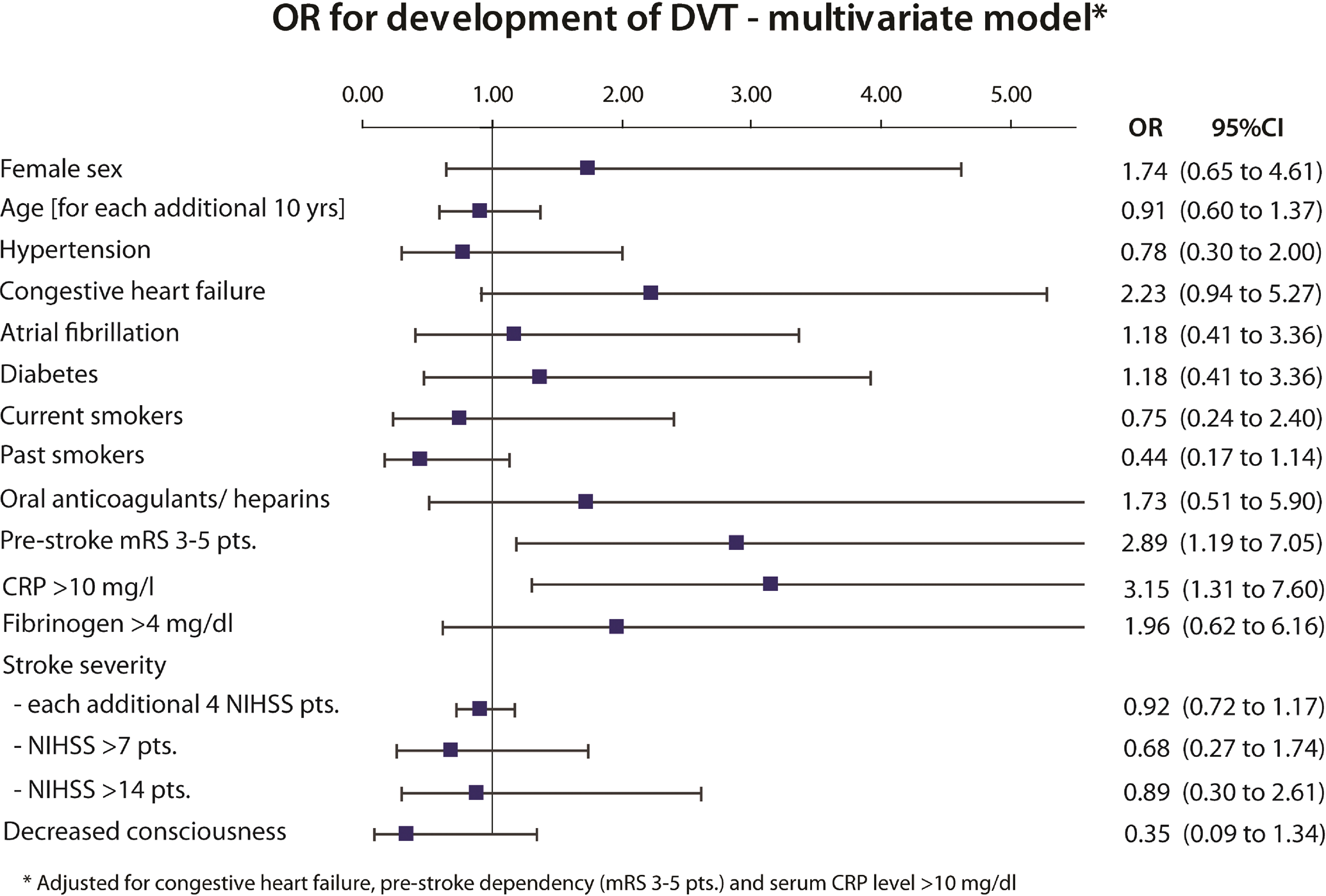
Risk factors for the development of deep venous thrombosis (DVT) in acute stroke patients.
In patients with DVT, there was a tendency toward higher 3-month mortality (24.0% vs 11.9%; P = .084). However, it was not confirmed in the multivariate model adjusted for all independent outcome predictors. In our heart failure study, severity of neurological deficit and decreased level of consciousness proved to be independent factors influencing mortality.
Unfavorable outcome (mRS 3-6 pts) occurred significantly more frequently in the DVT group (64.0% vs 41.5%; P = .030). However, it was also not confirmed in the multivariate model. In our study population, the independent risk factors for death or dependency after 3 months were older age, prestroke mRS of 2 to 5 pts, and the severity of neurological deficit.
Discussion
We found DVT in 8.7% of patients with acute stroke admitted to our center. In the literature, DVT within 14 days after stroke has been reported in 10% to 75% of patients, depending on the method used for DVT detection. 1–5 Clinically manifested DVT occurs much less frequently and ranges 13–17 from 1.7% to 5.0%, which is in concordance with our results, as only 6 of 28 DVTs were symptomatic. Asian population is probably less prone to the DVT development than other races. According to Tan et al, it develops in 2.4% within the first week, reaching 4.8% within the first month poststroke. 18 However, their study excluded patients with prestroke immobility and clinical suspicion of DVT, which may explain the small number of detected DVTs. The opposite results are presented by De Silva et al who assessed the incidence of DVT in 105 Asian patients with acute ischemic stroke using USG. In the initial evaluation performed 7 to 10 days poststroke, they found DVT in 30% of patients, which was significantly more frequently distal (32/105 vs 8/105). In the follow-up evaluation (days 25-30 poststroke), DVT was detected in 45% of patients. 19
In a recent large trial evaluating the effectiveness of thigh-length graduated compression stockings (GCSs), in which 2518 patients were randomly allocated either to GCS (n = 1256) or to routine care (n = 1262), DVT was detected with a compression Doppler ultrasound of both legs at 7 to 10 days after stroke and when practical, again at 25 to 30 days after enrollment. Symptomatic or asymptomatic DVT was found in 126 (10.0%) patients allocated to thigh-length GCS and in 133 (10.5%) allocated to avoid GCS, 20 which is in concordance with our results.
In our study, DVT was predominantly distal (7.4% vs 2.2%), which is consistent with the literature. 7,19,21–23 We decided not to concentrate solely on proximal DVT because distal venous thrombosis also poses an indirect but significant threat. It is due to the risk of proximal extension and possible PE. 7 The thrombi propagate above the knee in 20% of patients. 24 Some complications of distal DVT (eg, postphlebitic syndrome) may evolve over 3 months poststroke. 25 In our study, only 6 of 28 DVT patients developed clinically symptomatic DVT. According to an autopsy-based study by Viitanen et al, PE tend to develop 2 to 4 weeks poststroke and has been reported to be the most common cause of death at that time. 6 This may explain why none of our patients developed PE during hospital stay.
Although partial or total immobilization due to lower limb paresis in poststroke patients is considered the major risk factor for VTE, 5,8,10 it was not confirmed by our findings. Limb paresis and immobilization are supposed to reduce muscle pumping action in the legs, which results in venous blood flow reduction. 5 According to a recent meta-analysis of epidemiological studies, the risk of VTE in immobilized patients, not necessarily due to stroke, is doubled. 26 In our study group, limb paresis was present in 49.2% of patients, but only in 28.8% of them it was of moderate or severe degree (NIHSS ≥2 points). This may account for the relatively low occurrence of DVT. The study is also not powered enough to draw negative conclusions about coexistence of DVT and limb paresis.
Higher occurrence of DVT in patients with hemorrhagic stroke was reported in a few published articles. 27–29 Based on data derived from the National Hospital Discharge Survey, including 14 109 000 ischemic and 1 606 000 hemorrhagic stroke hospitalizations from 1979 to 2003, Skaf et al reported that DVT occurred in 0.74% and 1.37% of patients, respectively. 27 In our study, none of the 23 (7.1%) patients with hemorrhagic stroke developed DVT, which may be explained by their low absolute number.
In a recent article, Ogata et al shows that the risk of DVT is increased in patients with hemorrhagic stroke, as well as in patients with higher degree of neurological deficit and more severe consciousness disturbances (measured in NIHSS) 29 . Our findings revealed a strong trend toward higher NIHSS score in the patients with DVT (median 6.5 vs 5.0; P = .077). However, it was not confirmed in the multivariate model.
A number of studies provide evidence that the development of DVT is associated with elevation of systemic inflammation markers, 30,31 which is fully consistent with our findings. Cause-and-results relationship of increased inflammatory markers and DVT is currently discussed. Some authors suggest that increased serum inflammatory markers, including CRP, are the results and not the reason for DVT development. 32
Although increased level of
According to our data, prestroke dependency and possibly preexisting congestive heart failure appear to be the most important clinical predictors of DVT. Accounting for unconfirmed influence of stroke severity and the level of limb paresis, it may be assumed that relatively high proportion of DVT diagnosed in our patients was already present at admission. It is also possible that prestroke dependent patients are simply at increased risk of developing new DVT. Either way, it does not change the fact that stroke-related immobilization may accelerate clot formation.
Study Limitations
Our study has certain limitations, which may have resulted in the understating of the incidence of DVT and its complications. The number of DVT-positive patients (n = 28) was suboptimal for conducting extensive multivariate analysis on DTV predictors. The final model involved 4 variables, which may be considered acceptable. However, the analysis should be regarded more exploratory than definite, especially in forming negative conclusions.
Ultrasound was proved to be highly sensitive and specific in the detection of proximal DVT in symptomatic patients, but its sensitivity for distal DVT detection is lower (62.1% vs 93.9%). 36 Venography still remains the “gold standard” of DVT detection. However, it may be complicated by allergic reactions (mild in 0.7%-12.7% and severe in 0.02%-0.04% of patients), 37 local pain and discomfort, nausea and vomiting, dizziness, superficial phlebitis, and edema. Serious complications are infrequent and include bronchospasm. 38 Therefore, we decided to rely only on ultrasound examination, which is noninvasive, more feasible, and safer than venography. In our study, it was performed by a single trained physician, whose accuracy in DVT detection was previously validated in a nonstroke setting. Such approach by avoiding interobserver variability allowed to obtain uniform results.
A total of 102 patients (24%) were not included in the final analysis due to early death, indication for early transfer to another wards, or poor general condition not allowing proper USG evaluation within the first 7 days. Therefore, we may assume that our study population consisted of patients with stroke eligible for full-time treatment in the neurological ward, with exclusion of very severe outliners. We did not perform autopsy in patients who died during the first 7 days of hospital stay (n = 22) on a routine basis, including 4 patients diagnosed with DVT. Therefore, we cannot exclude that their death was caused by PE. We also assessed patients very early after stroke, so we were not able to directly differentiate between newly developed and preexisting DVT. On the other hand, the incidence of DVT may be lower than in other studies, where screening was performed later after the stroke. Finally, the physicians assessing the 3-month outcomes were not blinded to patients’ baseline neurological status.
The 2-month gap in recruitment was caused by work arrangements of J.P.B., not relevant to the study.
Conclusions
In conclusion, our study shows that DVT in everyday clinical practice occurs in <10% of patients with acute mild-to-severe stroke. It appears not to affect significantly their 3-month outcomes. Patients with prestroke dependency and elevated serum CRP level are at increased risk of DVT independently of stroke severity. Therefore, it may be reasonable to provide such patients with additional attention and proper DVT prophylaxis in order to avoid thrombotic complications of life-threatening potential.
Footnotes
Acknowledgments
The authors would like to thank Professor Martin Dennis (University of Edinburgh, Scotland) for his valuable advice while preparing the manuscript.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
