Abstract
Induction chemotherapy is associated with increased thrombosis risk in children with acute lymphoblastic leukemia (ALL). In this prospective study, we explored the effects of ALL and induction chemotherapy on the procoagulant, anticoagulant, and fibrinolytic systems in 20 children with ALL. The levels of
Introduction
Thrombosis is a serious complication of childhood acute lymphoblastic leukemia (ALL) occurring in at least 5% of patients.
1
The pathogenesis of increased thrombosis risk is multifactorial and may involve hypercoagulability secondary to the disease or its treatment, central venous catheters (CVCs), and inherited thrombophilia.
2
–4 Because thromboembolic complications occur almost exclusively during induction and consolidation chemotherapy of ALL, many investigators have studied hemostatic changes during these intensive chemotherapy periods.
5
Although abnormalities in coagulation parameters vary with chemotherapy protocol and timing of investigation, almost all studies have demonstrated that antithrombin III (AT) level decreases during treatment with
Patients
Twenty children (11 boys and 9 girls) with newly diagnosed ALL, consecutively admitted to our hospital were enrolled for the study. Their age ranged between 3 and 15 years with a median value of 5.5 years. The diagnosis was established according to the bone marrow morphology, blast immunophenotype, and cytogenetic and/or molecular genetic findings. Fifteen patients had pre-B ALL (PB-ALL) and 5 had T-ALL. Blast counts in peripheral blood were calculated by a hematologist. All patients were treated through a CVC, according to Berlin-Frankfurt-Munster (BFM) ALL-95 Protocol, as described elsewhere. 16 Briefly, induction therapy consisted of 60 mg/m2 oral prednisolone (PRD) for 4 weeks, 1.5 mg/m2 intravenous (iv) VCR and 30 mg/m2 iv DNR on days 8, 15, 22, and 29, and 5.000 iu/m2 iv ASP Medac on days 12, 15, 18, 21, 24, 27, 30, and 33.
Complete blood counts, coagulation screening, and blood chemistry were performed at least twice weekly. All patients were negative both for inherited thrombophilia risk factors, which included factorVG1691A, prothrombin G20210A, and MTHFR C677 T mutations; levels of protein C (PC), protein S (PS), AT, lipoprotein (a) and homocysteine, and for personal and family histories of thrombosis. Control group consisted of 10 age-matched healthy children. Informed consent were obtained from parents and also from the children themselves if he or she is >12 years of age. The study was approved by the Institutional Review Board of Gazi University Medical School.
Methods
Markers of procoagulant, anticoagulant, and fibrinolytic systems were studied in patients at 4 different time points: at diagnosis, before institution of chemotherapy protocol (T0), at the end of PRD monotherapy on day 8 (T1), 3 days after the first dose of VNC and DNR on day 11 (T2), and 2 days after the first dose of ASP on day 14 (T3). None of the patients received anticoagulant therapy, fresh frozen plasma, or AT concentrate during the study period.
Venous blood samples from patients and controls were drawn in vacutainer tubes (Becton Dickinson, UK) buffered with 3.2% sodium citrate, spun at 1500 rpm for 10 minutes and the plasma samples were aliquoted into plastic tubes and kept at −80°C until assayed. Before analysis, the samples were quick thawed in a 37°C water bath for 5 minutes. The levels of prothrombine time (Thromborel-S), activated partial thromboplastin time (Pathromtin), fibrinogen (Multifibren U),
The plasma concentration of thrombin-activated fibrinolysis inhibitor (TAFI) antigen (Asserachrom TAFI) and total tissue factor pathway inhibitor (TFPI) antigen were measured using kits from Diagnostica Stago (Asniers, France). The antigen level of PAI-1 was determined using ELISA kit from TECHNOZYM (Vienna, Austria).The level of C-reactive protein (CRP) was measured by rate nephelometry (Beckman Instruments, Inc, Brea, California).
Statistical Analysis
Statistical analysis were performed using SPSS 11.5 (SPSS Inc, Chicago, Illinois), and P values of <.05 were considered statistically significant. Correlation was calculated by Spearman correlation test. The Mann-Whitney U test was used to compare data from the 2 study groups. Wilcoxon test was used to detect changes with time within each group.
Results
All of the study patients were steroid responsive (day 8 blast count <1000/µL) and completed the induction chemotherapy uneventfully. At diagnosis, the median white blood cell (WBC) and blast counts were 8550/µL (range: 1460-110 000 µL) and 4540/µL (range 880-105 000/µL), respectively. The median blast count was significantly higher in T-ALL (median: 33.876; range: 1380-90 080/µL) than in PB-ALL (median: 4752; range: 880-105 430/µL). Two (10%) of the 20 patients developed CVC-related deep venous thrombosis (DVT) in upper extremity, one at the fourth and the other at the fifth week of induction therapy. The levels of hemostatic parameters in patients at diagnosis and at 3 different time points after the start of chemotherapy are shown in Table 1.
Hemostatic Parameters Before and During Chemotherapy in Children With Acute Lymphoblastic Leukemia.a
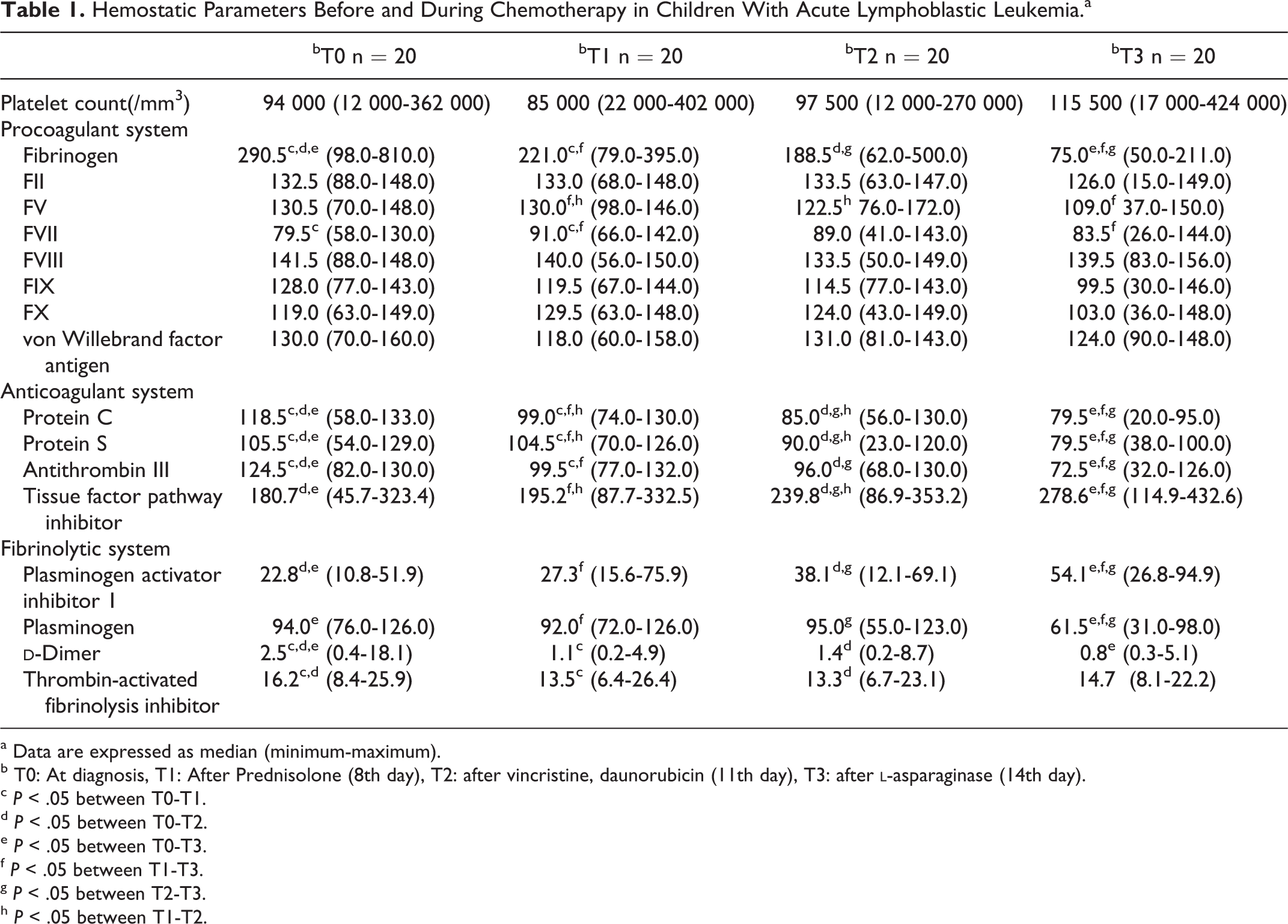
a Data are expressed as median (minimum-maximum).
b T0: At diagnosis, T1: After Prednisolone (8th day), T2: after vincristine, daunorubicin (11th day), T3: after
c P < .05 between T0-T1.
d P < .05 between T0-T2.
e P < .05 between T0-T3.
f P < .05 between T1-T3.
g P < .05 between T2-T3.
h P < .05 between T1-T2.
Effects of Disease
All patients were examined at diagnosis before the start of chemotherapy (T0) to assess the effects of disease on hemostasis. The mean level of vWf:Ag, FVIII,
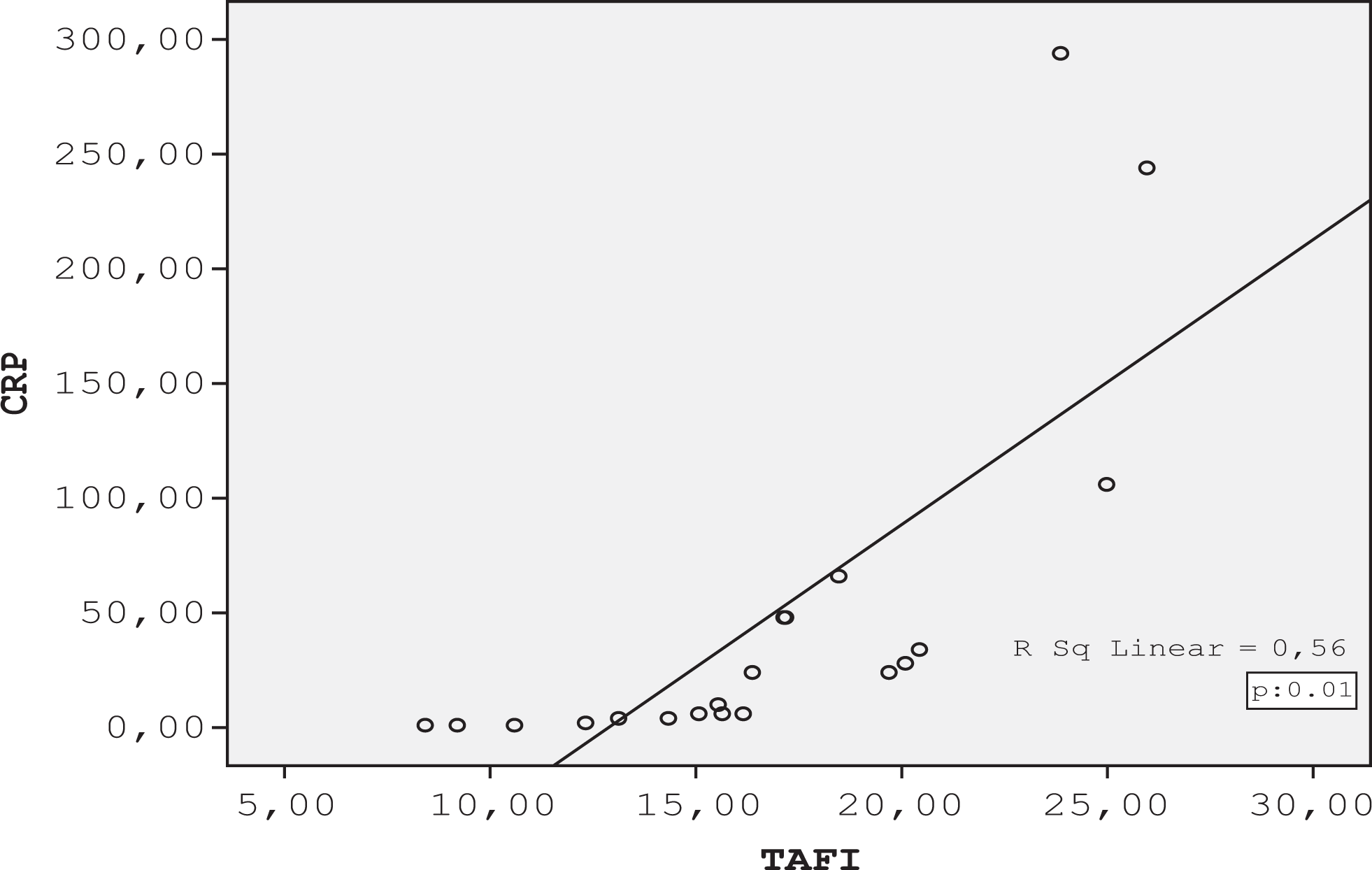
Correlation between C-reactive protein (CRP) and thrombin activatable fibrinolysis inhibitor (TAFI) level in children with acute lymphoblastic leukemia.
Effects of Age
Patient age showed statistically significant correlation only with TAFI level (Figure 2). Patients >9 years of age had significantly higher TAFI level than in those <9 years of age. Other hemostatic parameters did not differ significantly between these age groups (Tables 2 and 3).
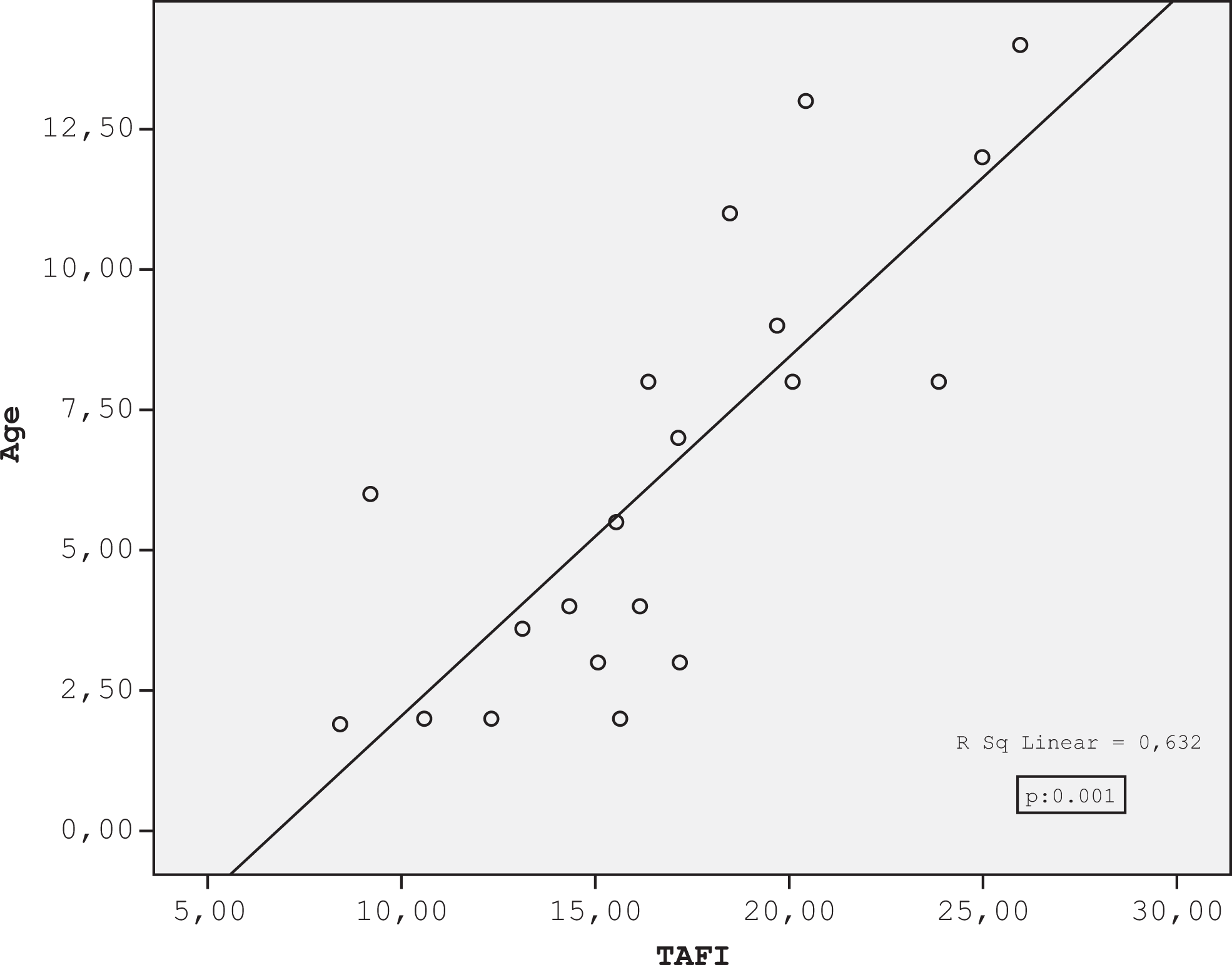
Correlation between age (year) and thrombin activatable fibrinolysis inhibitor (TAFI) level in children with acute lymphoblastic leukemia.
Platelet Count and Procoagulant System According to the Age in Children With Acute Lymphoblastic Leukemia.a
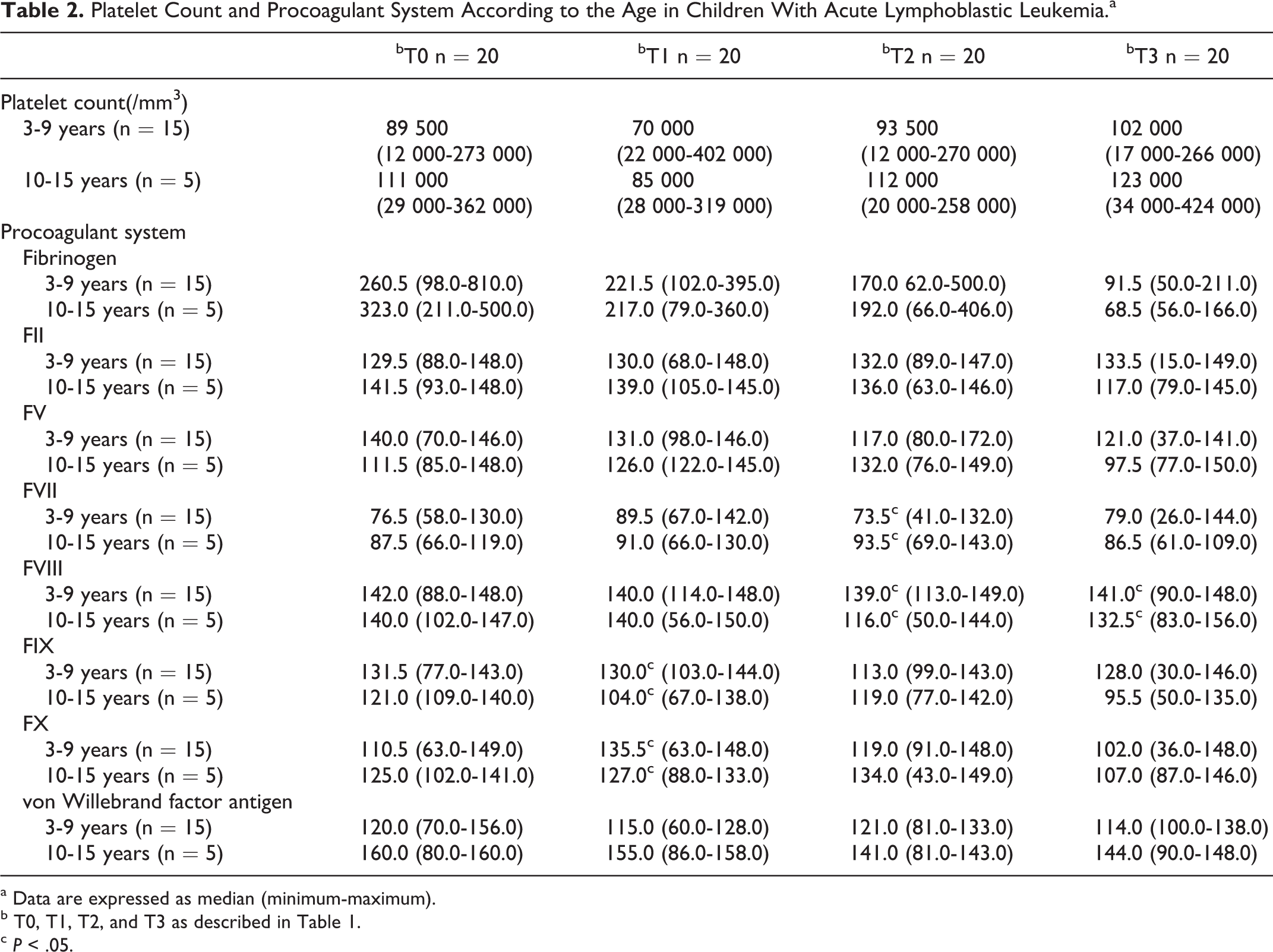
a Data are expressed as median (minimum-maximum).
b T0, T1, T2, and T3 as described in Table 1.
c P < .05.
Anticoagulant and Fibrinolytic Systems According to the Age in Children With Acute Lymphoblastic Leukemia.a
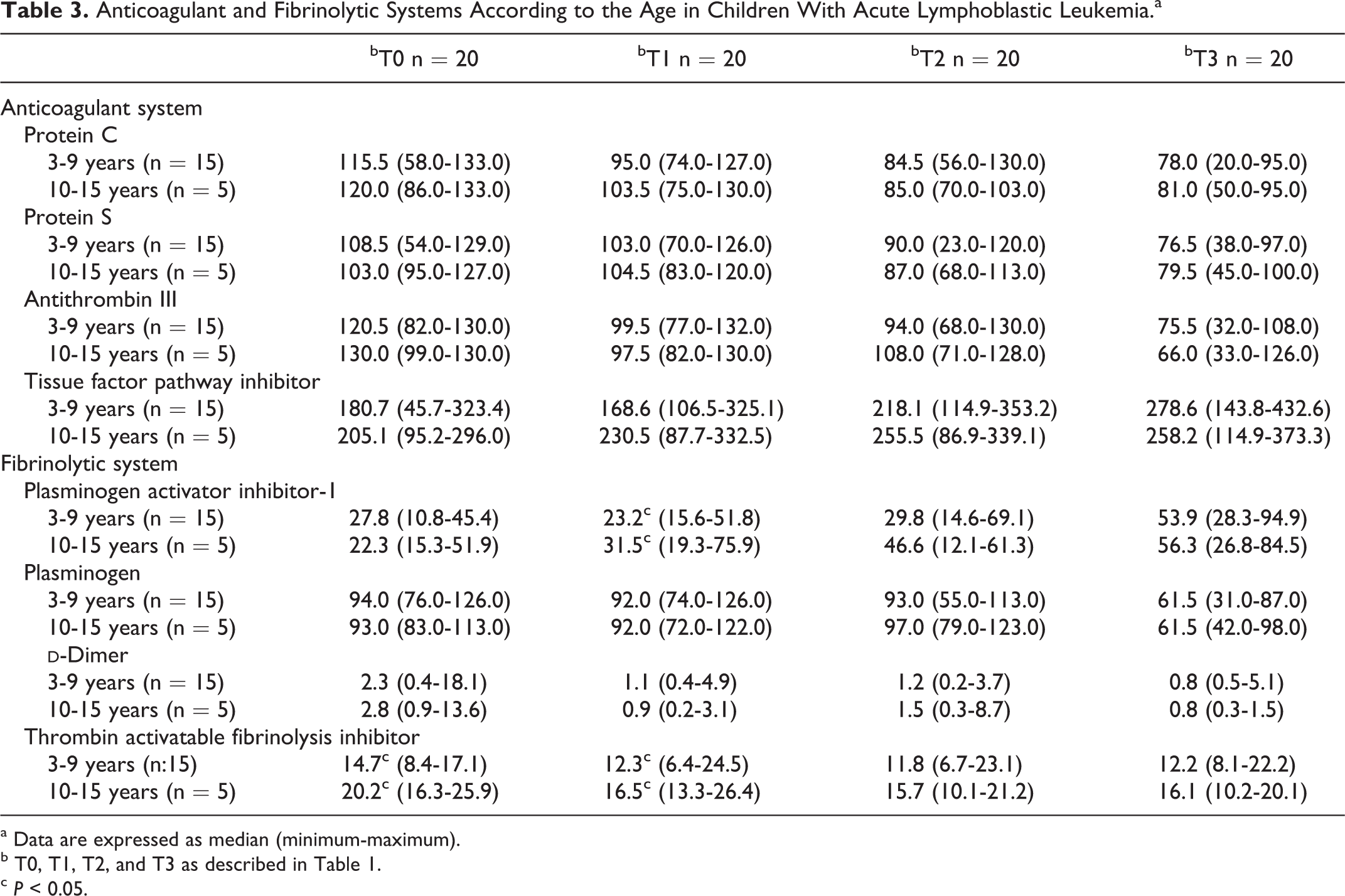
a Data are expressed as median (minimum-maximum).
b T0, T1, T2, and T3 as described in Table 1.
c P < 0.05.
Effects of PRD Monotherapy
On day 8 of PRD monotherapy (T1), the levels of FVII, PC, PS, and AT increased and the levels of FBG,
Effects of PRD, VCR, and DNR
Patients were re-examined on day 11 of chemotherapy (T2), 48 hours after the administration of VCR and DNR. The levels of PC, PS, and FV decreased but TFPI increased significantly as compared to T1; when compared with baseline, the levels of FBG, PC, PS, AT,
Effects of PRD, VCR, DNR, and ASP
There was a statistically significant decrease in FBG, PC, PS, AT, and PLG and increase in PAI-1 and TFPI at T3 as compared to T2; whereas FBG, PC, PS, AT, PLG, and
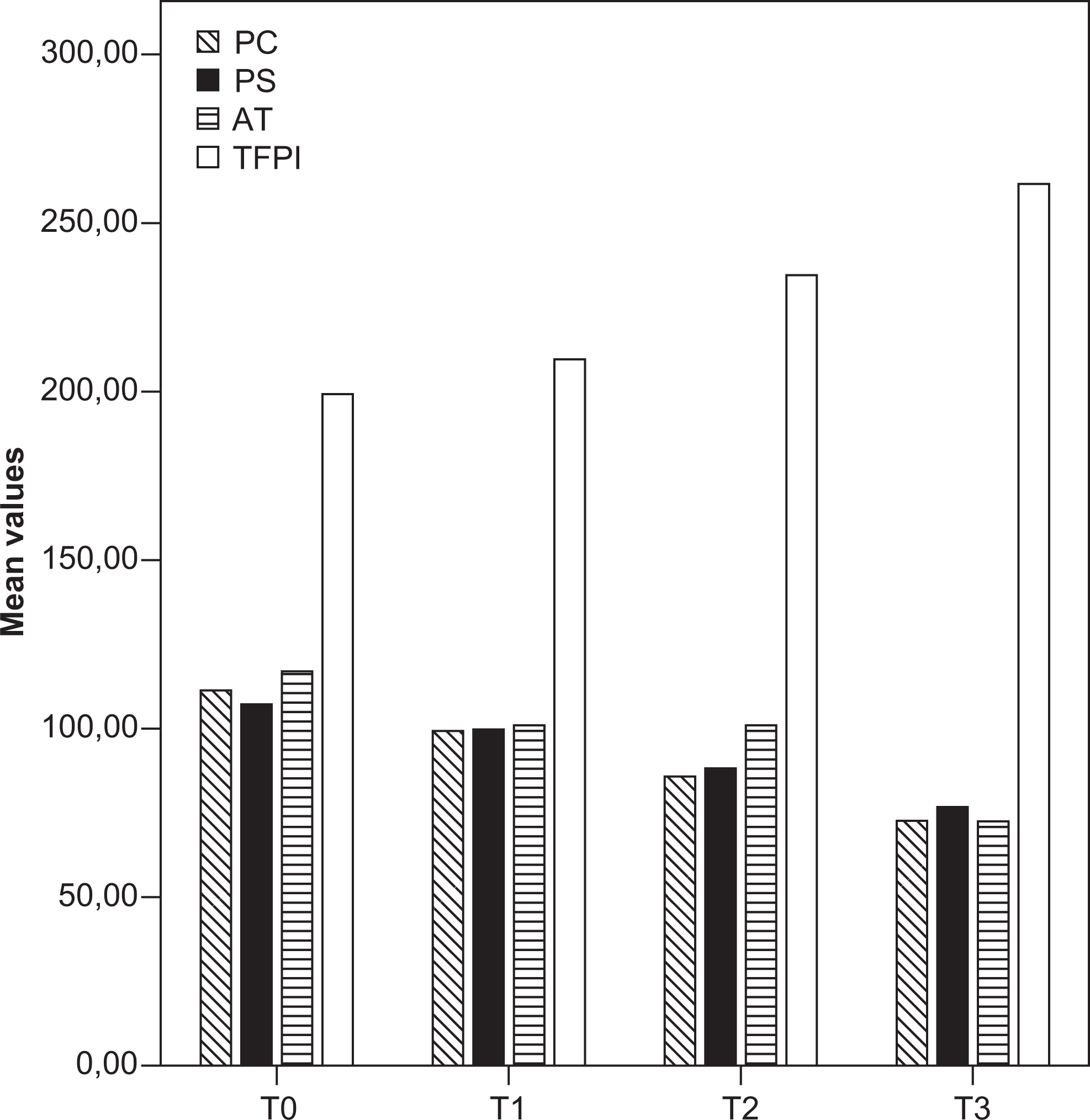
Mean values of protein C (PC), protein S (PS), antithrombin III (AT), and tissue factor pathway inhibitor (TFPI) in children with acute lymphoblastic leukemia (T0, T1, T2, and T3, at diagnosis and on the 8th, 11th, and 14th day of chemotherapy, respectively).
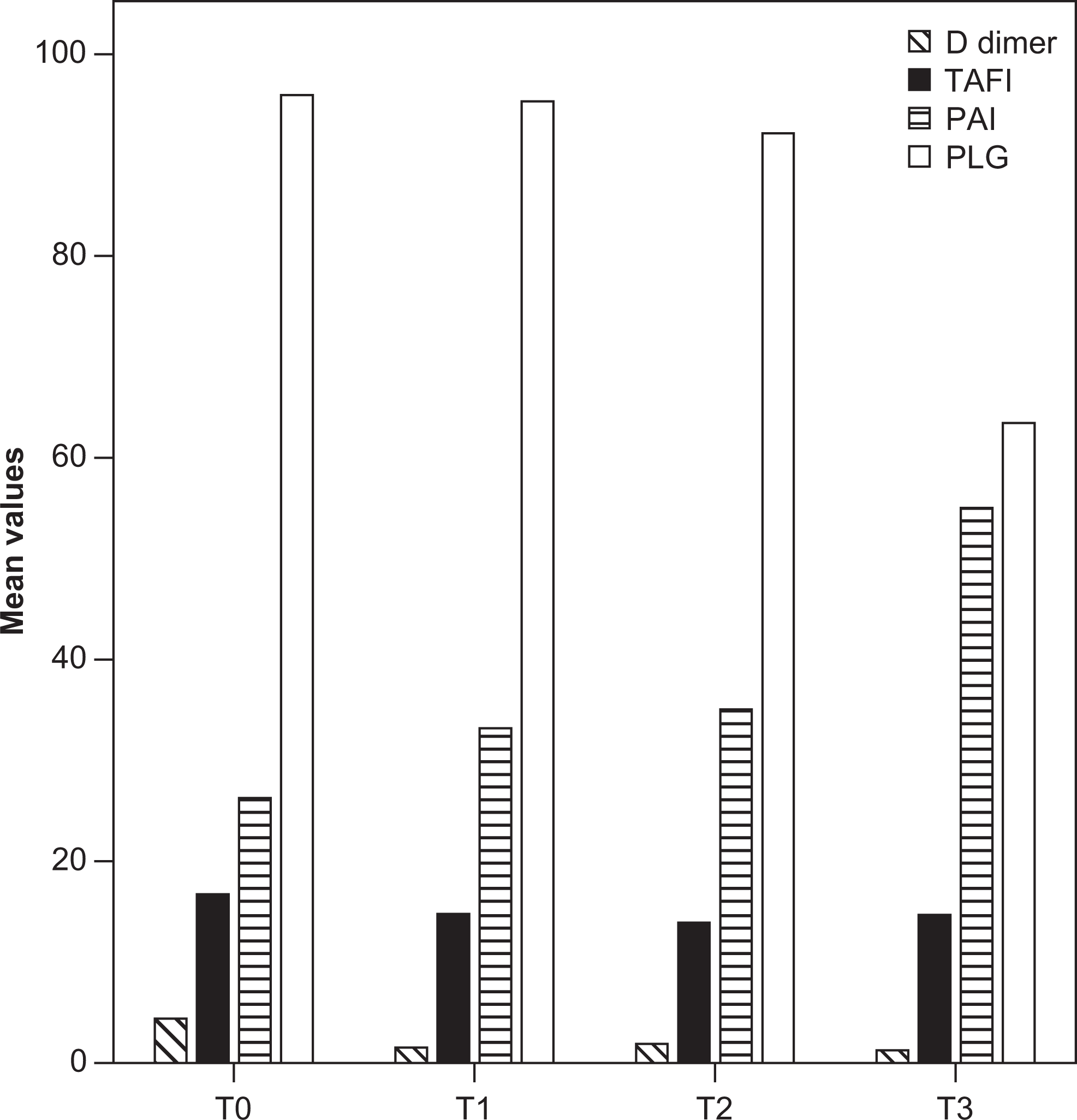
Mean values of D-dimer, thrombin activatable fibrinolysis inhibitor (TAFI), plasminogen activator inhibitor 1 (PAI-1), and plasminogen (PLG) in children with acute lymphoblastic leukemia (T0, T1, T2, and T3, at diagnosis, and on the 8th, 11th, and 14th day of chemotherapy, respectively).
Discussion
Thrombosis results from imbalance between procoagulant, anticoagulant, and fibrinolytic systems. A prothrombotic state can occur in patients with cancer due to the ability of cancer cells to produce and secrete procoagulant/profibrinolytic substances and inflammatory cytokines or to interact with platelets, monocytes, and endothelial cells, leading to activation of coagulation.
17,18
Studies in children with ALL have shown that markers of coagulation activation including
Data on the state of fibrinolytic system at diagnosis of childhood ALL are scarce. The level of PAI-1 was found to be normal in one and elevated in another study. 24,26 In our study, there were no significant differences in PAI-1 and PLG levels between patient and control groups. Interestingly, the level of TAFI, which is a carboxypeptidase enzyme downregulating fibrinolysis, was elevated in patients at diagnosis compared to controls. This is a novel finding not reported previously in ALL. The TAFI is activated by thrombin thrombomoduline complex and to a lesser extent by plasmin and inhibit fibrinolysis by decreasing PLG binding to fibrin, thereby representing a link between coagulation and fibrinolysis. Elevated level of TAFI has been identified as a risk factor for thrombosis in several diseases or conditions. 27
The mechanisms involved in the activation of coagulation in ALL remain to be addressed. Blasts cells from patients with ALL show weak expression of TF at their surface.
28,29 Plasma levels of TF and procoagulant phospholipids have been shown to be elevated at presentation in children with ALL.
23
However, evidence linking blast cells to the coagulation abnormalities remains controversial. Athale et al reported that the levels of vWf:Ag,
Inflammatory mediators such as interlekin 6 (IL-6), tumor necrosis factor-α (TNF-α), and CRP are involved in the activation of coagulation.
32
Because they increase, independently of infections at diagnosis of ALL,
33
we investigated the effect of CRP, a surrogate marker for inflammatory cytokines on hemostasis in our study group. Fifty-five percent (11 of 20) of patients had elevated CRP level at presentation. These patients had significantly higher level of FBG, AT, and TAFI as compared to those with normal CRP levels, whereas FVIII, vWf:Ag, and
Chemotherapy is a well-established risk factor for thrombosis. Glucocorticoid which is an essential element of induction chemotherapy in childhood ALL has been implicated in promoting thrombosis. They have different effects on hemostatic system depending on the context in which they are given; the levels of FVII, FVIII, and FXI increase in healthy individuals, whereas vWf:Ag, FBG, and PAI-1 levels decrease in active inflammation.
34
Although studies of hemostatic systems during GC monotherapy in children with ALL have given variable and often discrepant results, most of them showed increase in vWf:Ag, FVIII, and PAI-1 levels and decrease in FBG,
Data on the effects of VCR and DNM in coagulation system in childhood ALL are scarce. In vitro studies have shown that VCR and DNM reduce the activation of PC and induces large increases in thrombin generation at the surface of cancer cells.
35 Furthermore, presence of antracyclin in induction protocol increases the risk of thrombosis in children with ALL.
2
In the present study, the levels of FBG, PC, PS, and AT decreased, whereas TFPI level significantly increased 3 days after the administration of VCR and DNM as compared to their previous (T1) values. A slight but not statistically significant increase in
The ASP exerts its antileukemic activity through intracellular asparagine depletion, leading to the impairment of protein synthesis/secretion and apoptotic cell death. 36 The effects of ASP on coagulation system in children with ALL vary with the type of ASP preparation, dosages, timing of blood sampling, and other cytostatic agents in chemotherapy regimen. 2 , 4 - 13 Abnormalities in coagulation is more severe after Escherichia coli ASP as compared to Erwinia ASP. 37 In accord with previous studies, we found significant decrease in FBG, PC, PS, AT, and PLG levels and increase in PAI-1 levels after a single injection of E coli ASP (T3) as compared to their pre-ASP (T2) and baseline values (T0). The level of TFPI markedly increased after ASP, indicating extrinsic pathway activation of coagulation. The progressive increase in TFPI level in our study is in line with the study of Schneider et al who showed that free TFPI, TF activity, vWf:Ag, and thrombomoduline levels increased during induction therapy with PRD, VCR, DNM, and ASP. 23 Thus, the procoagulant state induced by extrinsic coagulation activation, and high FVIII level which persisted during the entire study period, impaired anticoagulant capacity and decreased fibrin lysis due to low PLG and high PAI-1 and TAFI levels produce a favorable environment for thrombosis formation. Replacement therapy with fresh frozen plasma or cryoprecipitate for hypofibrinogenemia may augment this hypercoagulable state and further increase the risk of thrombosis.
Two patients in our study group developed DVT. In consistent with previous observations, thrombosis was CVC related and developed in the fourth and fifth week of induction therapy, when platelet counts and fibrinogen levels recovered (data not shown). Infections secondary to chemotherapy-induced neutropenia are also more common during this period and may contribute to the development of thrombosis by increasing thrombin generation. 38 Interestingly, our patients with DVT were adolescents older than 12 years of age. Silverman et al reported that children with ALL between the age of 9 and 18 years had a higher incidence of thrombosis than younger children. 39 In our study, patients older than 9 years of age had higher TAFI level than in younger age group, while other parameters were not significantly different. Appel et al observed more profound decrease in procoagulant and anticoagulant factors in children with ALL between 11 and 18 years of age than in younger age group during ASP monotherapy phase, following 4-week induction chemotherapy with GC, VCR, and DNM. 11 It appears that hemostatic abnormalities vary not only with composition but also with the duration of induction chemotherapy as well as patient age.
In conclusion, our results showed that coagulation is activated and fibrinolysis is impaired at diagnosis, whereas induction chemotherapy leads to reactivation of coagulation and progressive impairment in fibrinolytic and anticoagulant capacities in childhood ALL. Our study is the first to show that TAFI is elevated and may contribute to increased thrombosis risk in children with ALL. Inflammatory response induced by the disease or infections may contribute to activation of coagulation and inhibition of fibrinolysis by affecting the levels of fibrinolytic proteins, thereby the development of thrombosis in ALL. Our study is limited by the small number of patients and lack of functional studies of all hemostatic proteins. Disease and treatment-related factors involved in the dynamic changes in hemostatic system need to be investigated in a larger group of children with ALL having different clinical and biological characteristics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
