Abstract
Introduction
Coronary artery disease (CAD) is a complex, chronic, and multifactorial disorder and has become a major source of morbidity and mortality in different parts of the world.1–4 Myocardial infarction (MI), an extreme manifestation of CAD, usually occurs following the development of an acute occlusive thrombus due to a ruptured coronary artery atheromatous plaque. Much of the risk of MI is explained by conventional risk factors such as age, male gender, smoking, hypertension, hypercholesterolemia, diabetes mellitus, obesity, and sedentary lifestyle.5–7 But the exact underlying mechanisms of MI are not fully understood, which may be due to an interaction between the individual’s genetic makeup and various environmental factors.8,9 In recent years, most studies have looked at associations between polymorphic variants in candidate genes and the risk of developing MI.10–12 A potential candidate gene system is the matrix metalloproteinase (MMP) family.
The MMPs are a large family of zinc-dependent enzymes that are capable of degrading most components of the extracellular matrix (ECM) and contribute to both normal and pathological tissue remodeling. 13 Matrix metalloproteinase 9, also known as gelatinase B or 92-kDa type IV collagenase, is one of the MMPs found to be highly expressed in the vulnerable regions of atherosclerotic plaques, and for this reason it has been suggested MMP-9 plays a pivotal role in vascular remodeling and development of atherosclerotic lesion, and it also has an important role in arterial plaque rupture.14,15 Epidemiological studies have shown that MMP-9 levels are increased in the circulation of patients with MI.16,17 Recent research suggested that genetic variation that affects the expression of MMP-9 may contribute to the occurrence of cardiovascular disease. 18
Smoking is widely acknowledged as a major common environmental factor associated with the risk of CAD.19,20 Smoking is associated with a higher serum level of cholesterol, coronary vasomotor activity, platelet aggregation, and prothombotic state. 21 The smoking-associated risk of CAD has been reported to be greater in some genetic variants.22,23 Knowing that in most cases MI has a multifactorial basis, involving a number of genes and environmental factors interacting to determine whether the disease will develop, we also tried to look at the association and linkage between the MMP-9 gene polymorphisms and MI in a Uighur population. In addition, the interaction of the MMP-9 gene polymorphisms with smoking was assessed.
Materials and Methods
Participants
The study was designed as a case–control study. A total of 835 Uighur participants were recruited from January 2006 to March 2010 at the First Affiliated Hospital of Xinjiang Medical University, Urumqi, China. The case group consisted of 384 patients with an acute MI (AMI) or non-ST segment elevation myocardial infarction (NSTEMI) evidenced by coronary arteriography. Diagnosis of AMI and NSTEMI was made by characteristic symptoms (chest pain), electrocardiographic changes (new pathologic Q waves, at least 1 mm ST elevation in any 2 or more contiguous limb leads or a new left bundle branch block, or new persistent ST-T wave changes diagnostic of a non-Q wave MI), and serum creatinine kinase-MB isoenzyme (CK-MB) elevations (more than 3-fold higher than the upper reference limit) according to the guidelines. 24 Those with the presence of cardiogenic shock, with known liver, thyroid, or renal diseases; malignancy or pregnancy; or with acute viral infection within the previous 4 weeks were excluded. The control group consisted of 451 participants without anomaly in coronary arteriography and no history of CAD, no symptoms suggestive of CAD, no electrocardiographic signs of CAD, no regional wall motion abnormalities, and no relevant valvular abnormalities in echocardiograms, to exclude the presence of CAD. 25 Control participants with any history of heart disease, neoplasm, cardiomyopathy, or severe illness limiting life expectancy or refusing consent were excluded. Hypertension was defined as having a blood pressure above 140 mm Hg systolic and or 90 mm Hg diastolic at 3 consecutive measurements divided by 15 minutes intervals or any antihypertensive agent use. 26 Hypercholesterolemia was defined as serum total cholesterol (TC) levels ≥6.2 mmol/L (240 mg/dL) or current use of antihyperlipidemic medication. Diabetes was defined as fasting plasma glucose levels ≥7.0 mmol/L (126 mg/dL) or 2 hours after 75 g of oral glucose loading glucose levels ≥11.1 mmol/L (200 mg/dL), or current use of antidiabetic medication. 27 Participants were considered smokers when consuming more than 5 cigarettes per day or nonsmokers when never smoked or had stopped smoking at least 1 year before sample collection. Persons who drank 100 g or more alcohol per day in the previous 6 months were considered alcohol users. Weight and height measurements were performed with a standardized scale with light clothes and naked feet. Body mass index (BMI) was calculated as weight (in kilograms) divided by height (in meters) squared.
Controls and patients with MI patients were matched by gender and age ± 5 years. All participants were genetically unrelated ethnic Uighur population from Xinjiang Uighur Autonomous Region of China. Each participant was interviewed after informed consent was obtained. The study was approved by the ethics committee of the First Affiliated Hospital of Xinjiang Medical University.
Biochemical Analysis
Five milliliter venous blood samples were obtained from all participants after 12-hour fasting and placed in EDTA tubes and stored at −80°C until the time of assay. The serum concentrations of triglyceride (TG), TC, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and blood glucose were measured by the standard methods in the Central laboratory of the First Affiliated Hospital of Xinjiang Medical University.
Matrix Metalloproteinase 9 Genotyping
Genomic DNA was extracted from peripheral blood leukocytes by standard methods (Boiteke Corporation, Beijing, China). Genotyping was performed with the method of polymerase chain reaction and restriction fragment length polymorphism (PCR-RFLP). The primers of two sites were designed by the Primer Premier 5.0 software (PREMIER Biosoft International, Vancover, Canada) and syntheses were performed by the Shanghai Biological Engineering Company Limited (Shanghai, China). The PCR product of −1562C>T and R279Q polymorphism sites were then digested with the restriction enzyme SphI and SmaI (Fermentas Corporation, Lithuania) at 37°C for 16 hours and separated by electrophoresis on a 2% agarose gel and visualized by ethidium bromide. Detailed descriptions of the methods used are summarized in Tables 1 and 2.
PCR Primer Sets and Conditions for Matrix Metalloproteinase (MMP- 9) Gene
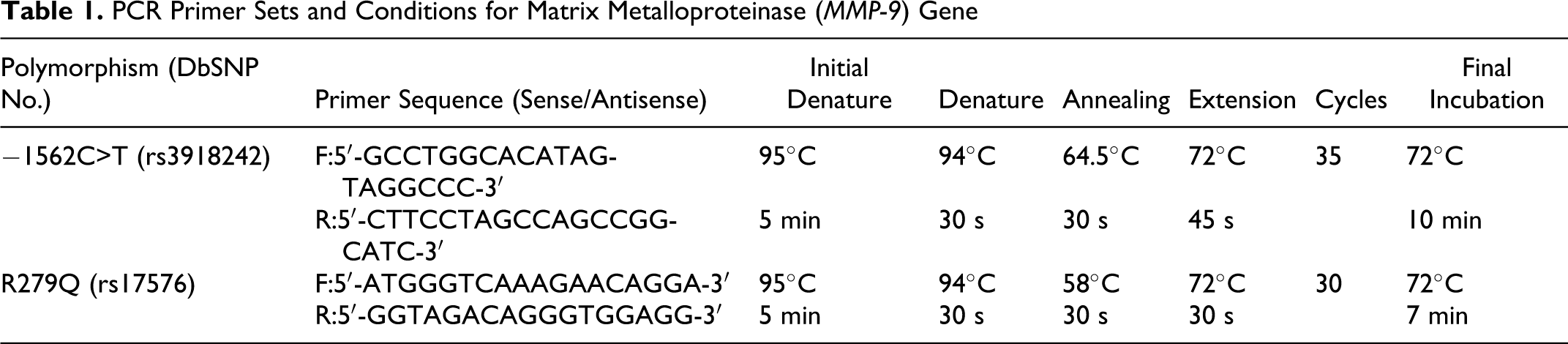
Restriction Enzymes, Conditions and Product Lengths for Analysis of MMP-9 Gene
Abbreviations: MMP-9, matrix metalloproteinase; PCR, polymerase chain reaction.
Statistical Analysis
All data were analyzed using SPSS for Windows 13.0 (SPSS Inc, Chicago, Illinois). All polymorphisms were tested for confirmation with Hardy-Weinberg expectations in both groups separately. Measurement data are shown as means ± standard deviation (SD), and the differences between the case and control groups were evaluated using the chi-square test (for categorical variables) and independent sample t test (for continuous variables). Differences in the frequencies of the alleles and genotypes were compared between patient and control groups by the chi-square test when appropriate. We estimated odds ratios (ORs) and 95% confidence intervals (CIs) for the effect of MMP-9 polymorphisms on MI risk using logistic regression analyses adjusted for the effects of other cardiovascular risk factors. Logistic regression analysis was also used to test for interaction between MMP-9 polymorphisms and smoking. A 2-tailed P < .05 was considered statistically significant.
Results
Clinical Characteristics
The distributions of clinical characteristics between MI group and control group are shown in Table 3. No significant differences were seen between the 2 groups with regard to gender distribution, age, triglyceride, HDL-C, apolipoprotein A I, apolipoprotein B, blood urea nitrogen, serum creatinine, and the drinking status (all P > .05). However, TC, LDL-Co, systolic blood pressure, diastolic blood pressure, and BMI were significantly higher in the case group than in the control group (all P < .05). There were significantly higher percentages of smokers and patients with diabetes mellitus or hypertension in the case group (all P < .05).
Clinical Characteristics of the Study Population
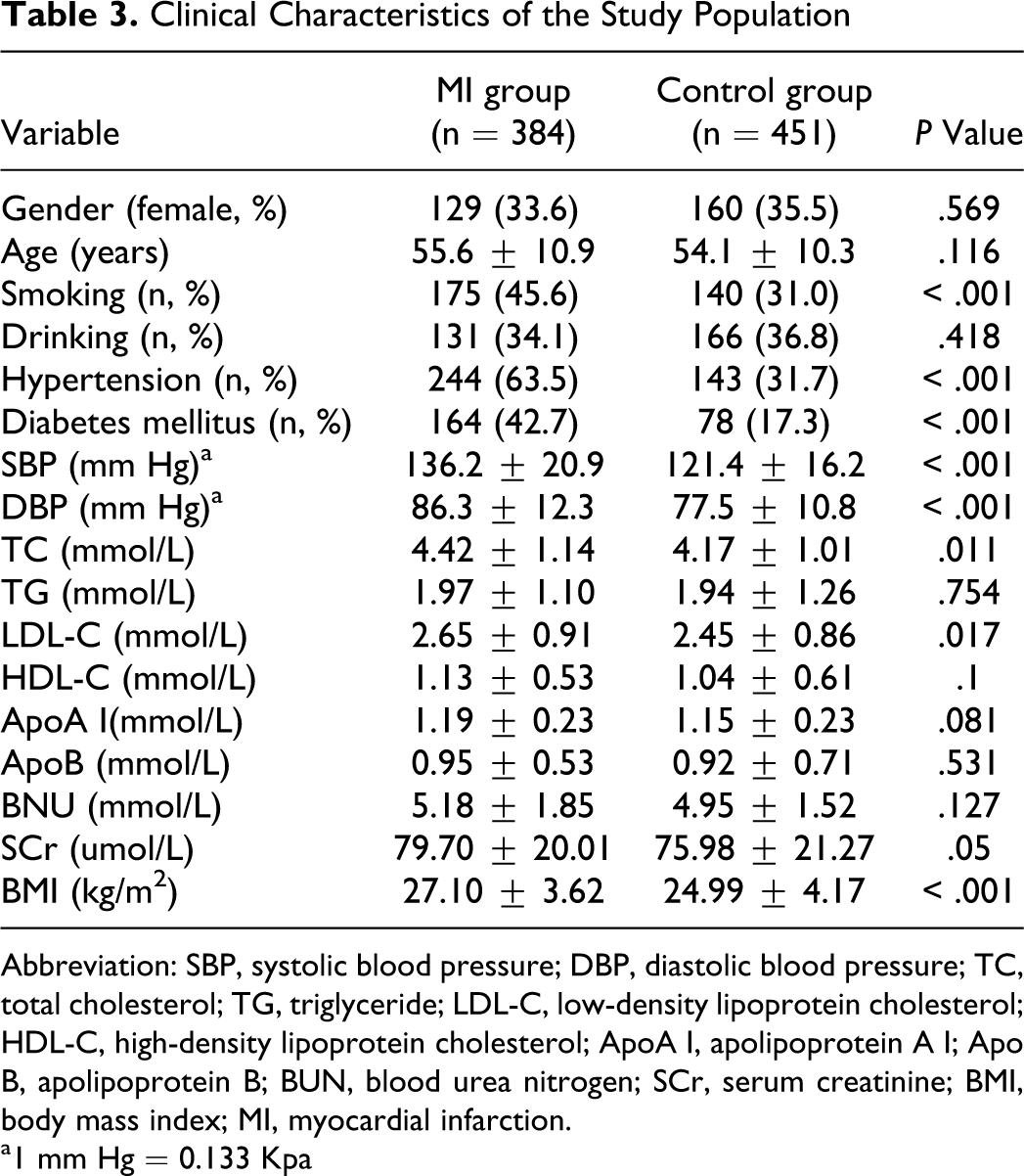
Abbreviation: SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoA I, apolipoprotein A I; Apo B, apolipoprotein B; BUN, blood urea nitrogen; SCr, serum creatinine; BMI, body mass index; MI, myocardial infarction.
a1 mm Hg = 0.133 Kpa
Matrix Metalloproteinase 9 − 1562 C>T and R 279 Q Genotype and Allele Frequency Distribution
Genotype and allele frequencies of MMP-9 (−1562C>T and R279Q) in controls and patients with MI are shown in Table 4. The distribution of genotypes were not significantly deviated from the Hardy-Weinberg equilibrium in both MI and control groups (all P > .05). The distribution of −1562C>T genotype was significantly different between the MI and control group ( χ 2 = 9.054, P = .011), and the −1562 T allele frequency was significantly higher in MI group than that in the control group (14.19% vs 9.31%; P = .002). The frequency of variant genotypes (CT/TT) for −1562C>T in the patients with MI (25.52%) was significantly higher than that in the controls (17.29%; P = .004). By further analyses we found that an individual who had T allele (TT or CT genotype) was at an increased crude risk of MI (OR = 1.64, P = .004). There were no significant differences in both genotypes and allele distribution of the R279Q polymorphism between 2 groups (all P > .05).
Genotype Distribution and Allele Frequencies of MMP-9 Polymorphisms Between 2 Groups
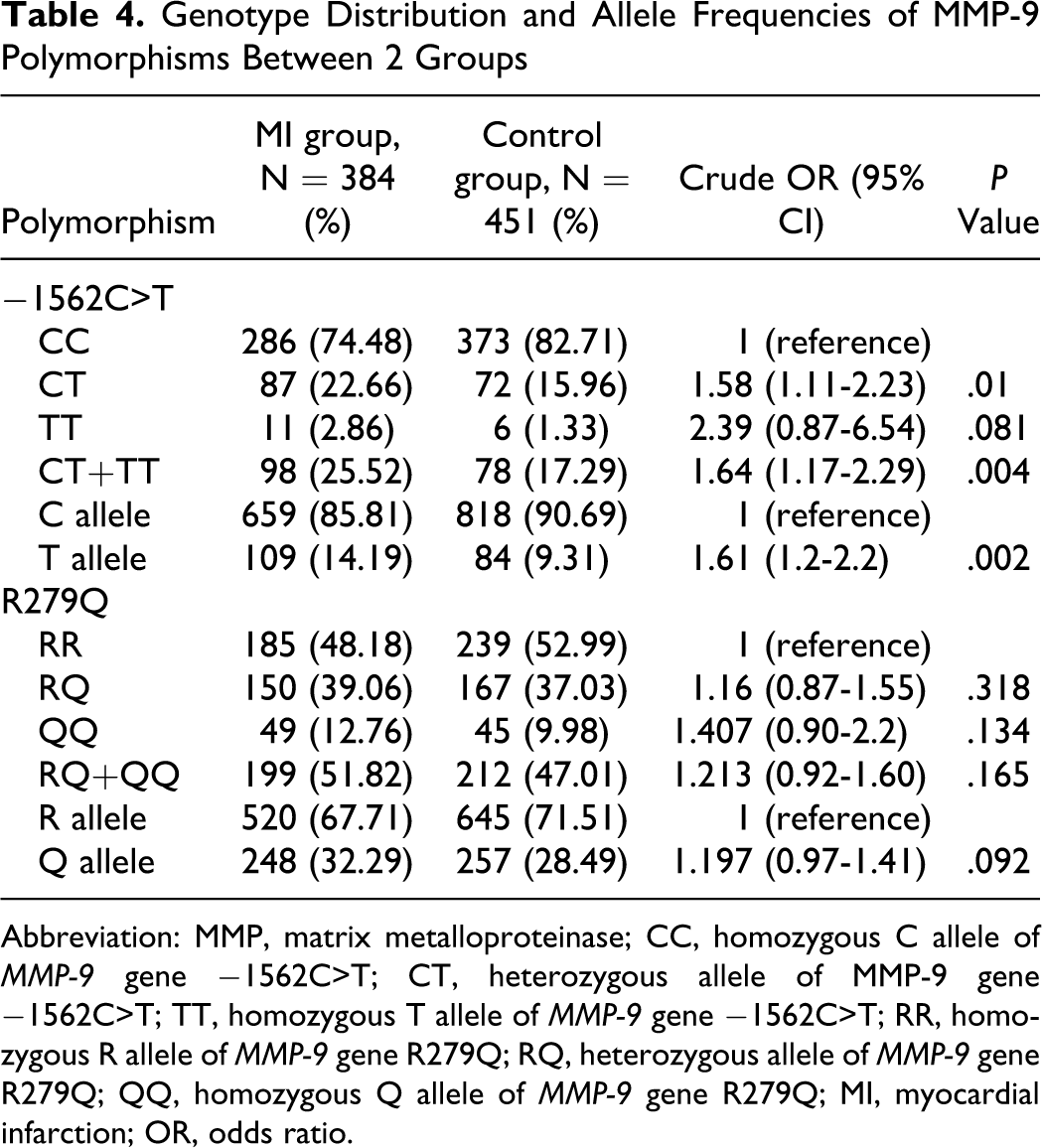
Abbreviation: MMP, matrix metalloproteinase; CC, homozygous C allele of MMP-9 gene −1562C>T; CT, heterozygous allele of MMP-9 gene −1562C>T; TT, homozygous T allele of MMP-9 gene −1562C>T; RR, homozygous R allele of MMP-9 gene R279Q; RQ, heterozygous allele of MMP-9 gene R279Q; QQ, homozygous Q allele of MMP-9 gene R279Q; MI, myocardial infarction; OR, odds ratio.
Interaction Between Smoking and MMP-9 Polymorphisms for MI
The whole sample was further divided according to the current smoking status, and the differences by −1562C>T or R279Q polymorphisms (homozygous or heterozygous carriers) were evaluated. At −1562C>T locus, compared with the CC genotype and nonsmokers, smokers carrying mutant T allele (CT or TT genotype) showed significantly higher risk (OR = 3.64, 95% CI: 2.04-6.49). Among individual who carry CC genotype, smoking history had 1.8-fold risk of MI (OR = 1.81, 95% CI: 1.32-2.49). In participants who did not smoke, the −1562 T allele (CT or TT genotype) was associated with an increase risk of MI (OR = 1.56, 95% CI: 1.03-2.37). At R279Q polymorphism locus, compared with 279 RR genotype carriers who did not smoke, the smoker with 279 RQ or QQ genotype had high risk of MI (OR = 2.44, 95% CI: 1.62-3.69). Among individual who had RR genotype, smoking history was associated with a 1.56-fold risk of MI (OR = 1.56, 95% CI: 1.05-2.31; Table 5).
Association Between Smoking and the MMP-9 Gene Polymorphisms on MI a
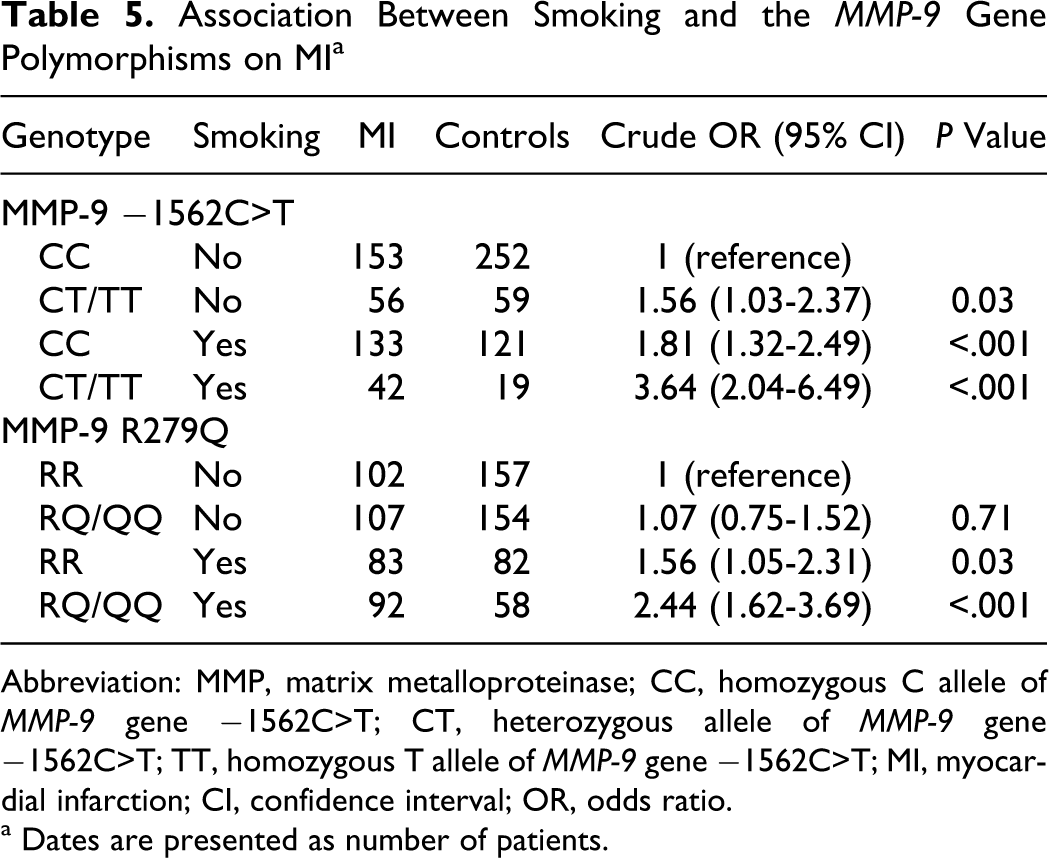
Abbreviation: MMP, matrix metalloproteinase; CC, homozygous C allele of MMP-9 gene −1562C>T; CT, heterozygous allele of MMP-9 gene −1562C>T; TT, homozygous T allele of MMP-9 gene −1562C>T; MI, myocardial infarction; CI, confidence interval; OR, odds ratio.
a Dates are presented as number of patients.
Identification of Independent Risk Factors for MI
A multiple logistic regression model showed 3 independent factors: diabetes mellitus (OR = 2.94, 95% CI: 1.46-5.91; P < .001), hypertension (OR = 2.51, 95% CI: 1.59-3.97; P < .001), and smoking (OR = 1.89, 95% CI: 1.52-2.34; P = .004). After adjusting for those conventional cardiovascular risk factors such as age, gender, hypertension, hypercholesterolemia, and diabetes mellitus, the −1562 T allele (TT or CT genotype) carriers still had an approximately 1.3-fold higher relative risk of developing MI than those with the CC homozygote (OR = 1.31, 95% CI: 1.22-1.40; P = .004). Moreover, there was a synergistic effect between smoking and −1562 T allele for the occurrence of MI (OR = 4.42, 95% CI: 2.74-7.13; P < .001). In addition, we found no association between R279Q polymorphism and MI risk in this cohort. But the interaction of R279Q polymorphism with smoking was an independent risk factor for MI (OR = 2.07, 95% CI: 1.04-4.13; P = .021), it was high than the independent effect of smoking (OR = 1.89) to MI (Table 6).
Logistic Regression Analysis of Association Between the MMP-9 Genotypes and Risk of MI
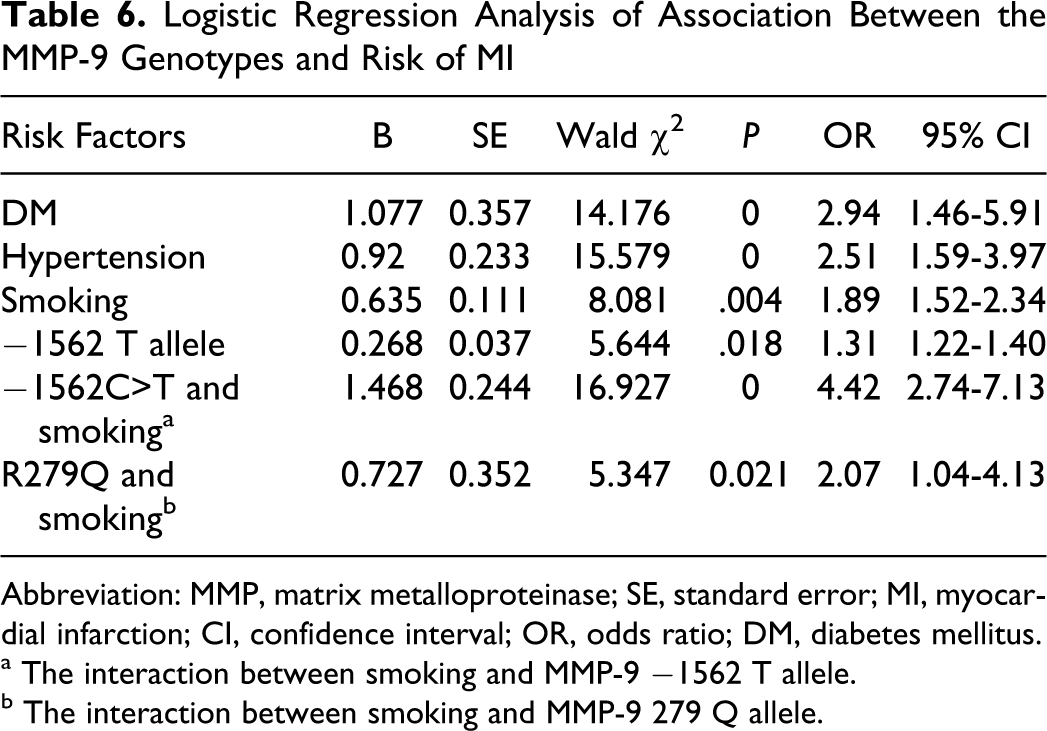
Abbreviation: MMP, matrix metalloproteinase; SE, standard error; MI, myocardial infarction; CI, confidence interval; OR, odds ratio; DM, diabetes mellitus.
a The interaction between smoking and MMP-9 −1562 T allele.
b The interaction between smoking and MMP-9 279 Q allele.
Discussion
To the best of our knowledge, this is the first study that has investigated the gene–environment interaction between smoking and 2 potentially functional polymorphisms of the MMP-9 gene in relation to MI susceptibility in Chinese Uighur population. Our data demonstrated that the −1562 T allele of MMP-9 gene is significantly associated with an increased risk of MI. However, we found no association between R279Q polymorphism and MI risk in this cohort. In addition, there was significant synergism between 2 locus polymorphisms and smoking to MI in this population.
The human MMP-9 gene is located on chromosome 20q12.2-13.1, and a number of SNPs in the promoter, coding, and untranslated regions have been reported. 28 Of these, 2 polymorphisms are of special interest: promoter −1562C>T polymorphism and codon 279 polymorphism (R279Q). In the former polymorphism, functional studies indicate that the −1562C>T polymorphism has an allele-specific effect on MMP-9 transcription. DNA-protein interaction assays have revealed that the sequence between nucleotide position −1567 and −1559 relative to the transcription start site of the MMP-9 gene, which encompasses the −1562 polymorphic site, can interact with a nuclear protein whose entity is still unknown. 29 And the latter is located in the gelatinase-specific fibronectin type II domain, which leads to an amino acid exchange (arginine [R] to glutamine [Q]) in the catalytic domain which presumably enhances substrate binding.30,31 It is therefore possible that the amino acid conversion associated with this polymorphism affects the activity of this enzyme.
Since Zhang et al 29 reported that the −1562 T allele of MMP-9 had a higher promoter activity and T allele carriers tended to have a higher risk of severe CAD, other studies have also confirmed this association with CAD.32,33 But the results were still not consistent, because 3 published studies on 3266 German and 268 Turks with angiographically documented CAD failed to confirm an association with the T allele.34,35 Recently, Zhi et al 36 found that MMP-9 −1562 CT/TT genotypes may contribute to CAD in diabetics and MI in CAD patients in a Chinese population. Fallah et al 37 reported that the −1562C>T polymorphism in the MMP-9 gene potentially play a role in the manifestation of coronary atherosclerosis but did not have any effect on the number of diseased vessels in Iranians. Genetic polymorphisms may vary among different ethnic groups. In this study, we found that −1562 CT/TT genotypes were associated with a significantly increased risk of MI. This is consistent with the studies from evidence that genetic epidemiology indicates that −1562 T allele carriers are predisposed to the development of coronary atherosclerosis that results in significant coronary stenosis. 32 At R279Q locus, in 1 study, 38 the 279Q allele was associated with increased MMP-9 levels and the combined end point of cardiovascular death and nonfatal MI. Whereas in the other 3 studies, R279Q polymorphism was not found to be associated with the risk of CAD and stable angina.32,39,40 Similar to the findings of the above-mentioned 3 studies, we did not find significant main effects of R279Q on MI risk.
Smoking doubles the risk of MI and ischemic stroke, 41 but the mechanisms by which smoking increases the risk of these atherothrombotic events are not fully understood. Studies indicated that smoking enhances systemic coagulability, as evidenced by increased circulating thrombin generation and activity, 42 fibrinogen levels,42,43 platelet activation, 43 and enhanced plaque tissue factor content and procoagulant activity, 44 by which smoking may predispose to acute arterial thrombosis.
It has been reported that MMP-9 brings about de-stabilizing structural changes in vulnerable atherosclerotic plaques.14,45 Matrix metalloproteinase 9 also potentiates the chemokine interleukin (IL)-8 and modifies the local chemokine profile, 46 and hence may promote cellular infiltration of the plaque, weakening the fiber cap of the atherosclerotic plaque, and increasing the size of the lipid core.47,48 These processes render the plaque susceptible to rupture due to reduced mechanical strength and hence increase the probability of atherothrombotic ischemia. As we know, thrombosis is generally accepted as the most common pathogenetic pathway of MI. Thus, we speculated that there should be a synergistic interaction between the effect of smoking and the MMP-9 genetic variation for MI.
In our study population, compared with the nonsmoker with −1562 CC genotype, the smoker with −1562 CT or TT genotype was associated with a 1.31-fold risk of MI. Multiple logistic regression analysis showed that the interaction between smoking and −1562 T allele significantly increase a 4.42-fold risk of MI. Although R279Q locus did not show significant differences between patients and controls, but the smoker with 279Q allele (RQ or QQ genotype) was significant risk factor for MI (OR = 2.07, 95% CI: 1.04-4.13; P = .001). The interaction between R279Q polymorphism and smoking was high than the independent effect of smoking (OR = 1.89). However, our relatively small study may influence the results. Thus, the results need to be replicated in other populations from different parts of China, and further investigations should be performed to explore the mechanisms of this gene–environment or other possible pharmacokinetic interactions.
Limitations of This Study
There are some limitations in the present study, the most obvious limitation of the present study is its sample size, though it had enough power to detect a moderate level of ORs, but it may have led to weak statistical significance and wide CIs when estimating ORs. In addition, the sample was well characterized and was from a homogenous Uighur population, which reduces the risks of population stratification and false associations.
Conclusions
This study suggested that MMP-9 −1562C>T polymorphism could be associated with the susceptibility to MI in Chinese Uighur population, and the −1562 T allele carriers might be at high risk of the development of MI. The interaction between −1562C>T polymorphism and smoking was a significantly independent risk factor for MI. The R279Q polymorphism was not associated with MI, but the interaction between it and smoking had significantly increased risk of MI.
Footnotes
The authors declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Great Technology Special Item Foundation of Xinjiang, China (200733146-3).
