Abstract
Two concepts relating to venous thromboembolism (VTE) prevention have recently emerged—“appropriate” prophylaxis and “preventable” VTE. We evaluated whether a human alert, as part of a pharmacy intervention program, can increase appropriate prophylaxis and decrease preventable symptomatic VTE in hospitalized patients. This prospective study with retrospective data collection was conducted utilizing data from 1879 patients in 2006 as a control cohort. The intervention cohort data were from 1646 patients during 2007, after program implementation. The rate of appropriate prophylaxis increased from 23.8% in 2006 to 37.9% in 2007 (odds ratio 1.8; 95% confidence interval [CI] = 1.6-2.1; P < .0001). Preventable VTE incidence was reduced by 74% (95% CI = 44%-88%) from 18.6 to 4.9 per 1000 patient discharges in 2006 and 2007, respectively (P = .0006). In conclusion, a pharmacy-led multifaceted intervention can significantly increase the rates of appropriate prophylaxis and significantly reduce the incidence of preventable VTE in hospitalized patients.
Introduction
Venous thromboembolism (VTE) is the most preventable cause of death for hospitalized patients and the number one strategy to improve patient safety in US hospitals. 1 Despite the fact that published guidelines have been in existence for >20 years, extensive gaps in VTE prevention in hospitals continue to exist globally. 2 –4
Efforts to improve in-hospital VTE prophylaxis would thus help to decrease the overall burden of hospital-acquired VTE. As such, the concept of “appropriate prophylaxis” (AP) has recently emerged in the medical literature and denotes appropriate type, dose, and duration of thromboprophylaxis. 5 –8 Several recent studies have demonstrated AP rates of between 13.3% and 33.9%, in US hospitals. 6 –9 In addition, potentially preventable VTE is defined by the National Quality Forum (NQF) as the number of patients diagnosed with confirmed VTE during hospitalization (not present at arrival), who did not receive VTE prophylaxis between hospital admission and the day before the VTE diagnostic order date. 10 Both the NQF and the Joint Commission (JC) have recently given final endorsement to their VTE measures, with the only outcome measure being the incidence of “preventable” VTE. 10
Hospital-based studies have demonstrated improved prophylaxis rates with either computer alerts or human alert programs, 11 –13 but only one randomized study has demonstrated a significant reduction in hospital VTE rates. 14 “Active” interventions as compared to “passive” interventions (eg, guideline dissemination) have demonstrated to be more efficacious in preventing VTE and increasing prophylaxis rates. 15 To date, no studies have used, measured, or demonstrated an improvement in AP rates and “preventable” VTE with specific hospital-based interventions. We undertook a study to evaluate whether a “human alert,” provided by a pharmacy intervention management program, would have an effect on both increasing AP and subsequently decreasing preventable VTE events in the setting of an acute-care hospital.
Methods and Materials
A prospective, before-after intervention cohort study with retrospective data collection was conducted using 2006 data as a control cohort and 2007 patients as the intervention cohort. Design and inception for the study began in early 2005. A multifaceted, physician-championed, pharmacy-led program to increase AP rates and prevent VTE was implemented in January 2007 at Lovelace Medical Center, a 254-bed, for-profit community hospital located in Albuquerque, New Mexico. Ethics approval was granted by the Institutional Review Board and Medical Executive Committee. Education, audit, and feedback were provided by 2 study investigators (C.E.M. and A.C.S.) to individual physicians, medical sections, and the general medical staff as part of the intervention throughout 2007. Any transferred data were de-identified in accordance with the Health Insurance Portability and Accountability Act (HIPAA) of 1996. The primary end point was AP and the secondary end point was “preventable” VTE events. Overall VTE events were also included in the final analysis.
Patients were placed in 1 of 3 mutually exclusive subgroups based on hospital discharge summary and billing records: critical care, surgical, or medical. Surgical and medical patients were identified using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis and procedure codes indicative of the surgery and condition of interest. A critical care patient was one defined as having stayed ≥1 day in an intensive care unit. Patients were placed in the most severe subgroup for which they qualified, with critical care being the highest in severity and medical conditions being the lowest. Patients were further classified into 7 mutually exclusive diagnostic groups adapted from the Seventh Edition of the American College of Chest Physicians (ACCP) guidelines (Table 1). 16 Surgical procedures were classified as major or nonmajor. Major operations were any intra-abdominal operation and any other operations lasting ≥45 minutes. Surgical VTE risk was defined based on the Seventh ACCP guidelines. Final determination of at-risk status and the recommended thromboprophylaxis prescribed were based on the assessments of “additional risk factors” as delineated in the Seventh ACCP guidelines. 16 Risk factors were identified using relevant ICD-9-CM diagnoses and procedure codes (Appendix A).
Classifications of At-Risk Diagnostic Groups a

a Modified from Geerts WH et al.16
For AP, the data source included discharge-summary records, which contained demographic data (age, gender, and race), admission and discharge dates, referral source, and type of insurance. Primary and secondary codes for diagnoses and procedures in ICD-9-CM format and specialty of the attending or admitting physician were also evaluated. Billing records provided daily information on inpatient services provided, and pharmacy data describing medication type, quantity, and dose. For VTE outcomes, all medical records were reviewed by 2 study investigators (C.E.M. and A.C.S.).
Inclusion and Exclusion Criteria
Patients aged ≥18 years at admission, who had an inpatient stay of ≥2 days during the study period, were eligible for inclusion. In addition, at-risk patients with a previous index stay who were readmitted for treatment of a “hospital-acquired VTE” within 90 days were also included. Patients with ≥1 of the following criteria were excluded from the analyses: transferred from another acute-care facility; pregnancy-related discharge diagnosis owing to precautions for anticoagulant use in pregnant women; other conditions where pharmacological thromboprophylaxis could be contraindicated, including active bleeding or indicating a potentially high risk of bleeding due to certain liver diseases, malignant hypertension, certain blood diseases, active peptic ulcer, and renal dysfunction; patients with a community-acquired VTE on admission; and patients admitted on therapeutic dose warfarin or low-molecular-weight heparin for other medical reasons (Appendix A).
Pharmacy Intervention Management Program: Identification of At-Risk Patients
Pharmacists used an individualized risk-assessment form ([RAF]; Appendix B) based on the Seventh ACCP guidelines, 16 in order to determine contraindications to pharmacologic and mechanical prophylaxis, as well as VTE risk factors for new admissions and any patients transferred to a critical care unit. Pharmacists contacted the attending physician for at-risk patients not currently prescribed AP when no contraindications were present, actively reminding them to prophylax at-risk patients. The contact was in person or via telephone communication. Venous thromboembolism risk factors and overall VTE risk level, as well as contraindications if present, were communicated to the attending physician. In addition, the recommended thromboprophylactic strategy was communicated to the attending physician during this “human alert” communication. For patients with contraindications to pharmacoprophylaxis and with no contraindications to mechanical prophylaxis, mechanical prophylaxis was recommended (Appendix B). For rare patients that had both contraindications to mechanical and pharmacologic prophylaxis, no recommendation for thromboprophylaxis was made. In addition, the RAF was completed and signed with the VTE risk factors and recommendation for prophylaxis, if applicable, in the patient medical record within the progress notes section (Appendix B).
The primary pharmacologic agent recommended was the low-molecular-weight heparin enoxaparin. If patients had severe renal insufficiency, unfractionated heparin 5000 units twice daily was recommended. For those patients with contraindications to anticoagulation, sequential compression devices were the primary recommendation.
If a pharmacist recommendation was not accepted for an at-risk patient with no contraindication, the attending physician received a direct call from the lead pharmacist. If the recommendation was still not accepted, the issue was elevated to the physician champion who subsequently contacted the responsible attending physician. This design created a multilevel hierarchy of possibly 1, 2, or 3 active reminders if physicians chose not to prophylax an at-risk patient.
In units where interdisciplinary rounds occurred, VTE prophylaxis was also addressed. Hospital staff and medical staff education were provided on a perpetual basis postintervention. Audit and feedback of performance was provided to each medical group/team and individual providers, approximately monthly, postintervention by the lead pharmacist and physician champion. Preventable VTE events were reviewed and discussed with the responsible provider.
Appropriate Prophylaxis
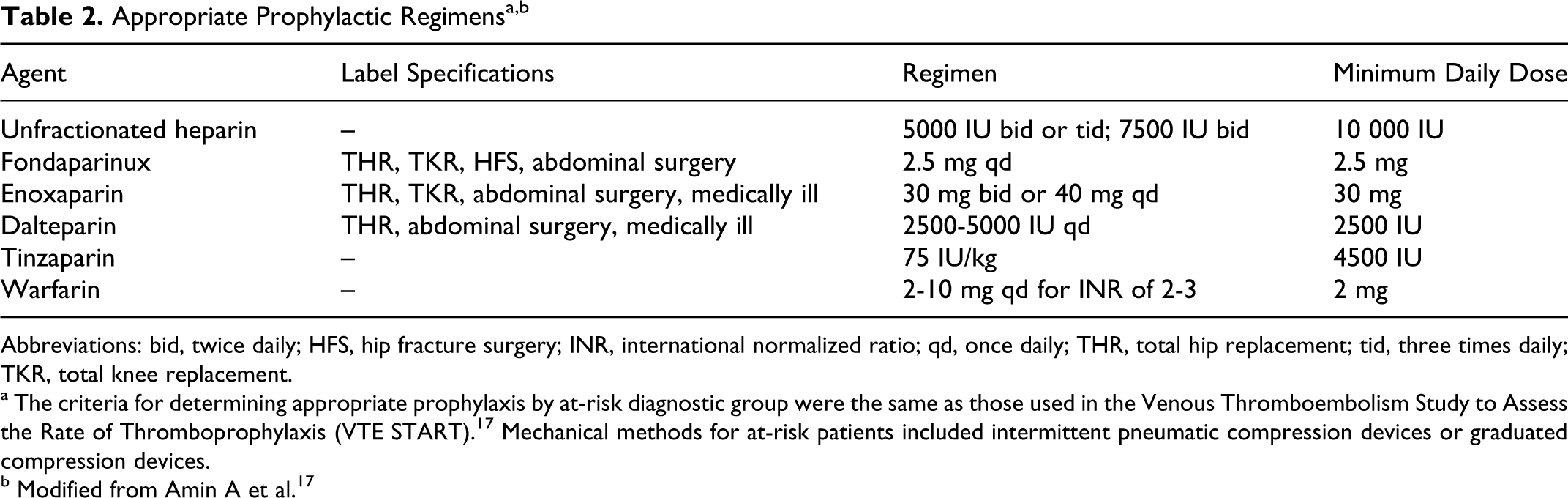
Appropriate prophylaxis was defined as appropriate type, dose, and duration. Appropriate prophylaxis rates were determined using 4 criteria: (a) whether the patient received any thromboprophylaxis; (b) whether an appropriate type of thromboprophylaxis (mechanical or pharmacologic) was used; (c) whether the pharmacologic regimen (if any) was given at a dose greater or equal to the minimum recommended daily dose; and (d) whether the regimen was administered for greater or equal to the recommended number of days. The criteria for determining AP by at-risk diagnostic group were the same as those used in the Venous Thromboembolism Study to Assess the Rate of Thromboprophylaxis (VTE START). 17 In summary, appropriate types of prophylaxis (ie, mechanical and pharmacologic) were defined for the at-risk patient diagnostic groups using the Seventh ACCP guideline recommendations. 16 Appropriate dose was defined by the package insert information from the various drugs when such information existed or by typical doses recommended in various guidelines (Table 2). 17 Due to the typical delay in the initiation of thromboprophylaxis, thromboprophylaxis not being used the day of surgery, and partial days of stay, appropriate duration was defined as length of stay minus 2 days for surgical patients and length of stay minus 1 day for medical patients. The duration of pharmacologic prophylaxis was calculated by summing the number of days for which a relevant billing code was recorded. Similarly, for mechanical prophylaxis, the duration was calculated as the total number of days between the first billing date and the discharge date for the applicable billing code.
Abbreviations: bid, twice daily; HFS, hip fracture surgery; INR, international normalized ratio; qd, once daily; THR, total hip replacement; tid, three times daily; TKR, total knee replacement.
a The criteria for determining appropriate prophylaxis by at-risk diagnostic group were the same as those used in the Venous Thromboembolism Study to Assess the Rate of Thromboprophylaxis (VTE START).17 Mechanical methods for at-risk patients included intermittent pneumatic compression devices or graduated compression devices.
b Modified from Amin A et al.17
Venous Thromboembolism and Safety Outcomes
Venous thromboembolism events within 90 days of hospital admission were identified and calculated by patient subgroup. An ICD-9-CM report was run on a monthly basis to identify patients admitted with, or discharged with, any diagnosis of VTE, including thrombophlebitis. Patient medical records were reviewed to determine whether the VTE event was hospital-acquired or community-acquired and if it was “preventable” or “nonpreventable.” Community-acquired VTE (ie, present on admission) events were excluded from the outcomes. Only hospital-acquired VTE events, “preventable” or “nonpreventable,” were included in the overall VTE outcomes, and “preventable” VTE was also tracked separately. “Preventable” VTE was defined as VTE that occurred if a patient did not receive AP prior to the event or during the prior index stay. Upper-extremity (UE) deep-vein thrombosis (DVT) was included in overall VTE events but was considered “nonpreventable” VTE; thrombophlebitis was excluded as a VTE event.
All VTE events were symptomatic and verified by objective methods. For DVT, ultrasonographic evidence of the loss of vein compressibility or evidence of a filling defect on conventional contrast venography 18 were used to verify the event. For pulmonary embolism (PE), high-probability ventilation–perfusion scanning 19 and filling defects on computed tomography of the chest with contrast medium 20 or conventional pulmonary angiography 21 were used to verify the event. For patients with >1 VTE event, only the first VTE event was counted. Suspected but not confirmed VTE events were not included in the outcomes. Major and minor bleeding events were not tracked.
Statistical Methods
Measures of central tendency (mean, median, and mode) and measures of dispersion (range and standard deviation) were calculated and reported for continuous variables (eg, age, length of stay) for each cohort. Appropriate prophylaxis calculation was performed by dividing the number of appropriately treated patients by the number of patients at risk. Categorical baseline demographics and risk factors were compared using chi-square statistics, interval variables such as length of stay were compared using Wilcoxon test. Rates of “preventable” and overall VTE events were calculated and compared using Poisson regression and log rank survival analysis. Relative change in the rate of AP was compared using multivariable logistic regression. For all statistical models, the level of significance was set at .05. Results from the Poisson regression were summarized using the rate ratio with the corresponding 95% confidence interval (CI). Results from the logistic regression were summarized and reported in terms of odds ratio (OR) with the corresponding 95% CI.
Results
Patient Characteristics
A total of 1879 discharges were included in 2006 and 1646 in 2007. Patient exclusions are detailed in Figure 1 . Most patient characteristics were similar between the 2 cohorts (Table 3). There was a decrease in the number of surgical patients and an increase in medical patients; a decrease in central venous catheterization placement, but an increase in the number of patients with obesity and cancer therapy; and a decrease in the number of patients with no risk factors, but an increase in the number of patients with ≥2 risk factors from 2006 to 2007.
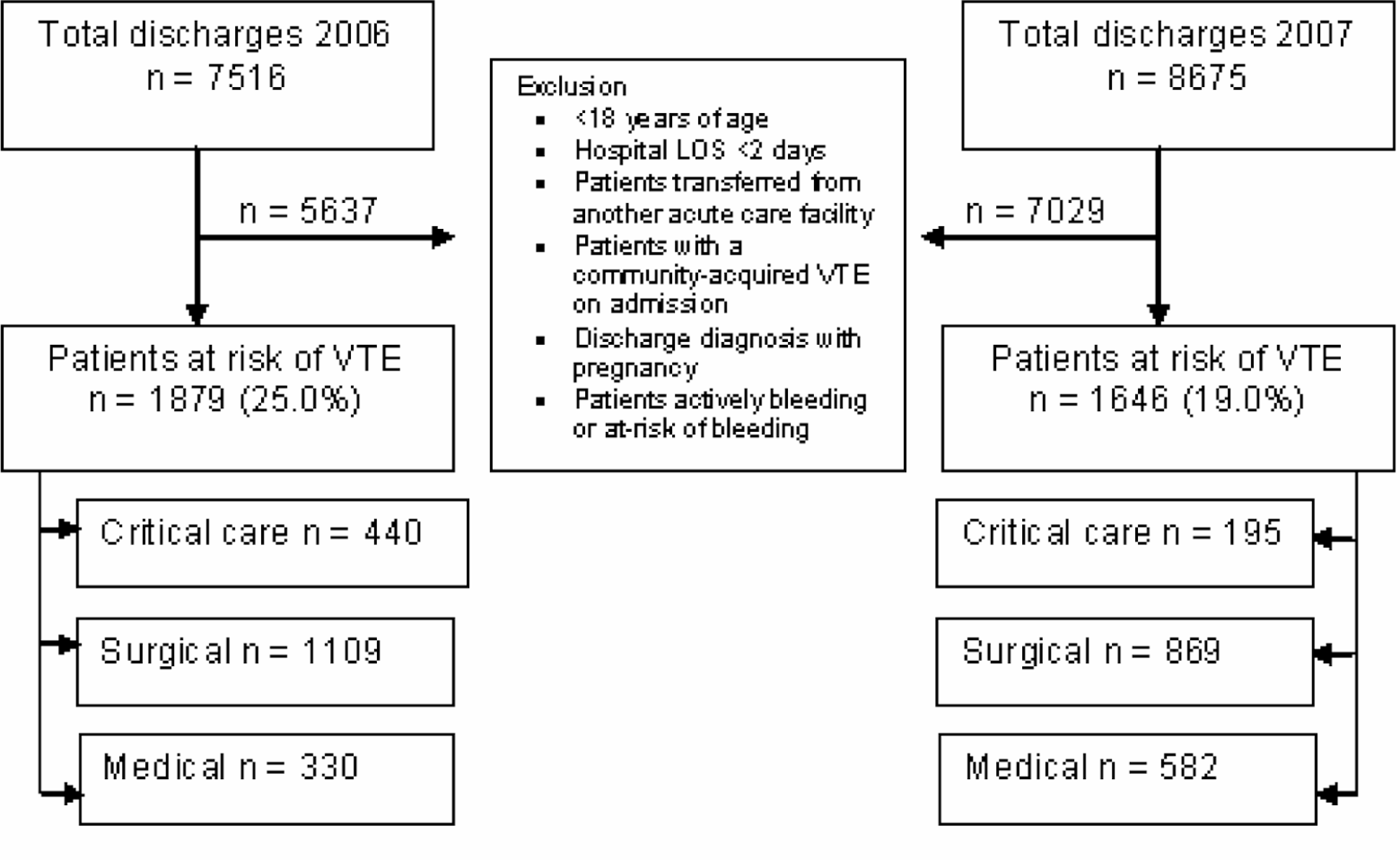
Flowchart of patients included and excluded from the study. LOS indicates length of stay; VTE, venous thromboembolism.
Patient Characteristics
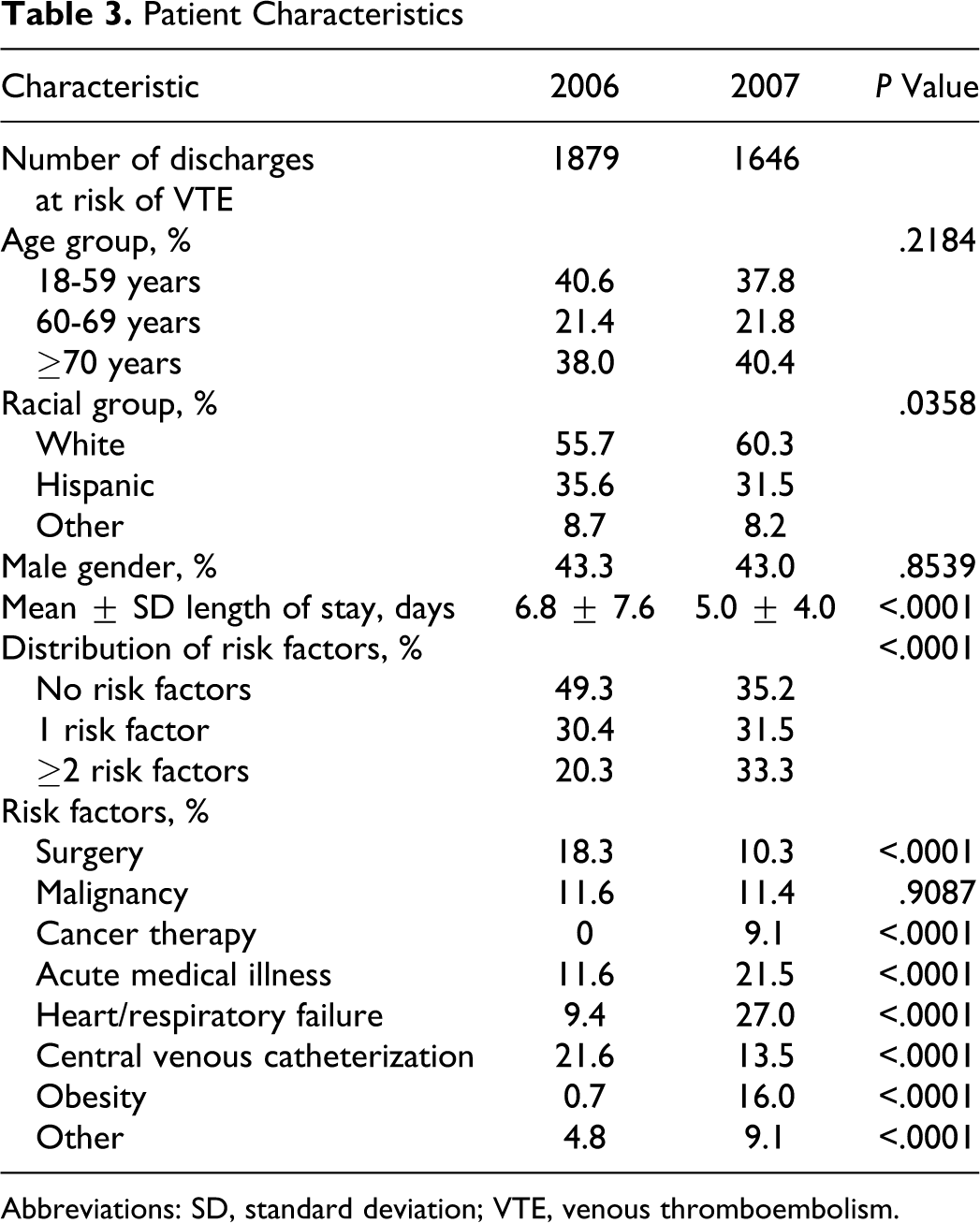
Abbreviations: SD, standard deviation; VTE, venous thromboembolism.
Outcomes: VTE Events and AP Rates
The overall ORs for increased AP comparing 2007 with 2006 were 2.5, 1.6, 2.1, and 1.8 for critical care, surgical, medical, and overall discharges, respectively (all Ps < .0001) (Table 4). For the overall population, rates of “any” prophylaxis and AP were 62.8% and 23.8% in 2006 versus 81.5% and 37.9% in 2007, respectively (P < .0001 for both). In critical care patients, rates of “any” prophylaxis and of AP were 66.3% and 14.6% in 2006 versus 83.0% and 34.1% in 2007, respectively (P < .0001). Similarly, in medical patients, rates of “any” prophylaxis and AP were 38.5% and 22.3% in 2006 versus 76.7% and 38.2% in 2007, respectively (P < .0001). In the surgical patients, rates of “any” prophylaxis and AP were 68.2% and 27.9% in 2006 versus 84.3% and 38.5% in 2007, respectively (P < .0001).
Compared with 2006, preventable VTE was reduced by 74% (18.6 per 1000 patient discharges in 2006 versus 4.9 per 1000 patient discharges in 2007; P = .0006; Table 4). Similar results were obtained using log rank survival analysis (P = .0003; Figure 2 ).
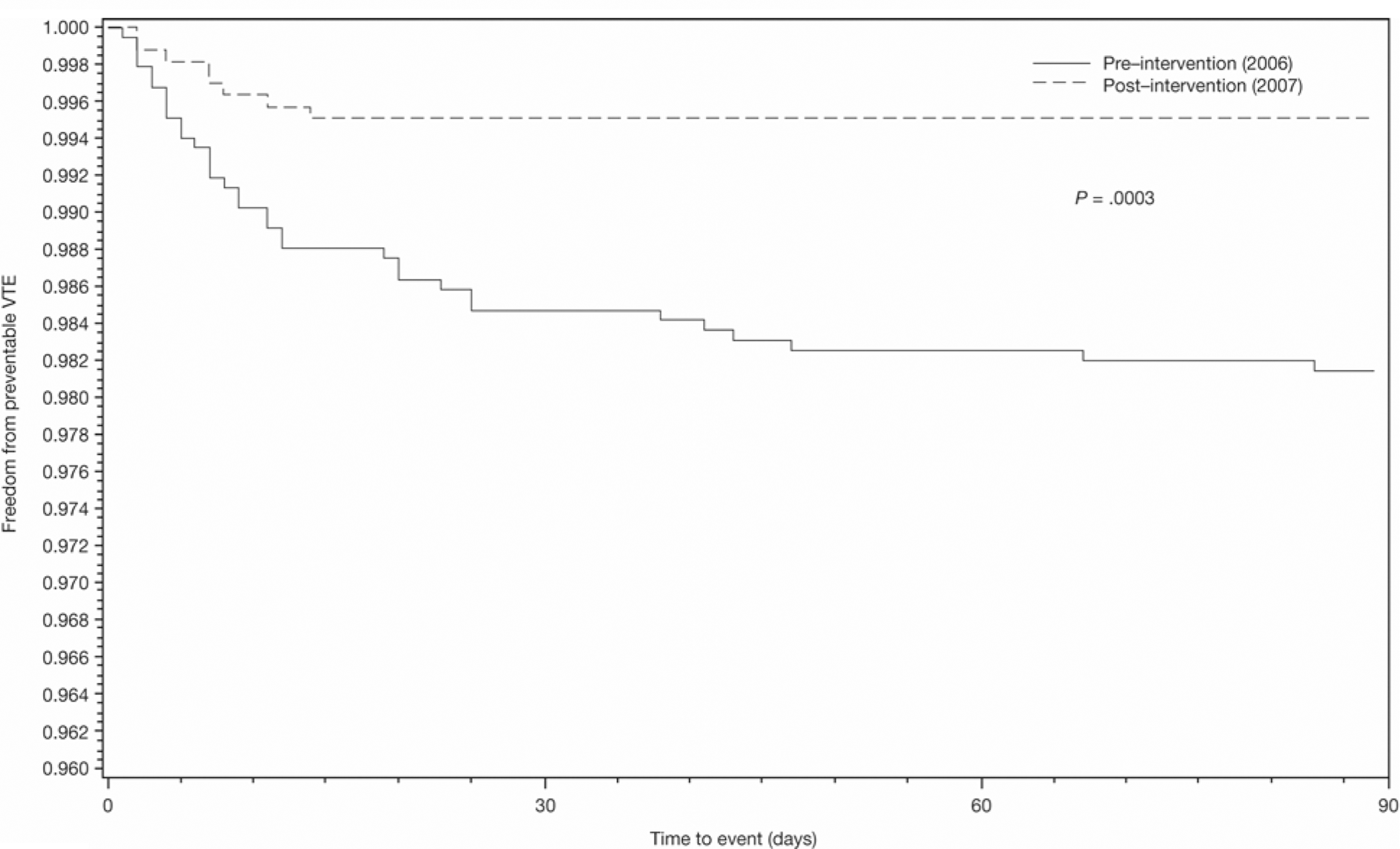
Freedom from preventable venous thromboembolism (VTE) at 90 days.
Appropriate Prophylaxis and Preventable Venous Thromboembolism (VTE) Outcomes
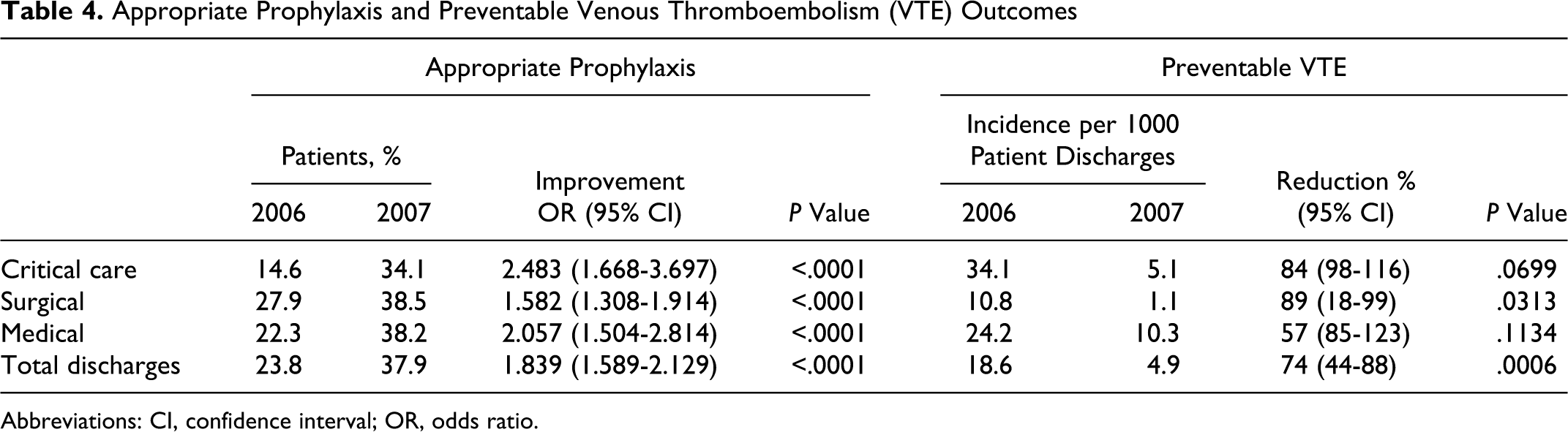
Abbreviations: CI, confidence interval; OR, odds ratio.
A nonsignificant trend for a reduction in overall VTE events by 44% (37.2 vs 25.5 per 1000 patient discharges) approached statistical significance (P = .0624). Overall VTE events included UE DVT, central venous catheter-associated UE DVT, lower-extremity DVT, and PE (the number of overall and preventable VTE events are reported in Table 5 ). It should be noted that 17 UE DVT events, occurring spontaneously or associated with a central venous catheter, were observed in 2007 and 13 in 2006. Preventable PE events were 5.3 per 1000 patient discharges in 2006 and 2.4 per 1000 patient discharges in 2007; however, the numbers were too small to determine statistical significance.
Overall and Preventable Venous Thromboembolism Events in the Control and Intervention Patient Cohorts

Abbreviations: DVT, deep-vein thrombosis; PE, pulmonary embolism.
Discussion
The results of this study demonstrate that interdisciplinary collaboration between pharmacists and physicians, with an active human alert in the hospital setting, can significantly increase rates of “appropriate” prophylaxis and significantly decrease rates of clinically apparent “preventable” VTE (by 74%), while also increasing the rates of “any” prophylaxis.
Only one previous study has demonstrated an increase in prophylaxis rates and a decrease in hospital-acquired VTE using computerized electronic alerts. 14 A study using a physician-alert system has demonstrated an increase in hospital-based prophylaxis rates but no decrease in overall VTE rates. 22> Furthermore, other studies using hospital-based computerized reminder systems or pharmacist interventions have demonstrated improved prophylaxis rates without a corresponding decrease in VTE rates, 11,12 or with VTE event rates that were too small to draw reasonable conclusions. 13 The hierarchical design of having a physician champion and dual reminder system to reduce hospital-acquired VTE may warrant further studies in the future.
Many of the studies attempting to demonstrate improvements in VTE events with prophylaxis did not use a clear definition of AP, including type, dose, and duration. 11 –14,22,23 “Appropriate” prophylaxis provides a more meaningful and definitive measure than in previous trials of various interventions. In prior studies, AP rates have been significantly lower than “any” prophylaxis rates. 8,9 Our study suggests that AP may be a more sensitive marker of “preventable” VTE than “any” prophylaxis, as rates of thromboprophylaxis may have been overestimated using the latter approach. As a hospital patient’s clinical status is constantly changing, and patients are often transferred within the hospital with orders being rewritten or reissued, it is important to look at appropriate type, dose, and duration as compared with the hospital length of stay because many errors of omission can potentially occur at the time of transfer. For these reasons, as the present study highlights, future studies should incorporate and use the more stringent definition of “appropriate” prophylaxis
Second, although overall VTE is important, no trials of pharmacologic or mechanical methods attempting to reduce VTE have demonstrated a reduction in UE DVT events, 24 as these events have a different pathogenesis and natural course compared with lower-extremity DVT. 25 As such, a strength of our study, and a limitation of previous studies, was the explicit exclusion of UE DVT as preventable VTE. It is also important to distinguish between “preventable” and “nonpreventable” VTE in future studies, especially in light of “preventable” VTE being the outcome measure assessed by the NQF and the JC. 10
Third, it should be noted that this intervention was performed in an intervention-naive hospital and that improvements in VTE prophylaxis and VTE outcomes are more likely when being studied in this setting as compared to a center or centers that already have high rates of prophylaxis and low rates of VTE. 14,15
The present study validates previous pharmacy studies which have demonstrated that interdisciplinary approaches can improve prophylaxis rates. 12,23 It also provides evidence for more operational methods to initiate a thromboprophylaxis program to improve quality, as compared with elaborate computerized alert programs, which are currently unavailable in many hospital systems. With the continued focus in the United States on quality and outcome measures of thromboprophylaxis and minimizing rehospitalizations, similar active and interdisciplinary approaches may help to improve the quality of care within hospitals. In addition, the study also validates that an individualized RAF, completed by trained professionals, can improve preventable VTE outcomes within hospitals, and that similar team-based programs may be generalizable to other professionals and for other quality focuses.
A limitation of this study is that it was not designed as a randomized, blinded, controlled trial. However, because the standard of care in US hospitals based upon national quality initiatives is to maximize AP for at-risk patients, such a trial may no longer be of a reasonable design to undertake within this setting. A second limitation is that the comparative frequencies of specific types of prophylaxis, including combination prophylaxis, were not tracked for the cohorts. A third limitation is that major and minor bleeding were not assessed between cohorts. Of note, patients who were considered at-risk of bleeding were excluded from the study. Differences in patient characteristics between the 2006 and 2007 cohorts may be seen as a potential study limitation, as well as performance of this study at a single site. These patient differences are likely to be a reflection of changes at the hospital site, including an increase in the volume of medical patients, an effort to minimize unnecessary central venous catheterization, and physician education about assessment of obesity. Specifically, the shift toward more medical patients, and fewer surgical patients, in the 2007 cohort may be perceived as a potential limitation because prophylaxis and VTE rates are typically lower in medical patients compared with surgical patients. Confounding from this shift would likely reduce AP and VTE rates so this may be a perceived limitation. However, the magnitude of effect in reduction of preventable symptomatic VTE events may have been underestimated, as the patients in the 2007 cohort had more VTE risk factors overall than the comparator cohort in 2006. Accordingly, it is possible that the 2007 population was healthier despite more VTE risk factors, and this effect contributed to the reduction in preventable VTE. Finally, the RAF is complex with a large number of data points that require trained personnel to complete (Appendix B). Some hospitals may not have the personnel or resources to execute a complex RAF and this may be perceived as a potential limitation.
Strengths of this study include stringent inclusion and exclusion criteria across well-defined, large, hospital-based study populations, precise measures of thromboprophylaxis, and objectively verified outcomes. Other strengths include the use of a standardized, evidence-based risk-assessment scheme in addition to a precise time point during which the pharmacy intervention management program was initiated, as part of an explicit hospital-quality intervention addressing VTE prevention. Another study strength is the use of the more recent definitions of “appropriate” prophylaxis and “preventable” VTE and demonstrating improvements in both. Although there has been no formal postintervention cohort for analysis, our records show continued and sustained effect through 2010, with only 3 preventable VTE cases in the first 9 months of 2010, despite increased growth and volumes.
Conclusion
A pharmacy-led multifaceted intervention can significantly increase rates of AP and significantly reduce preventable VTE in hospitalized patients. An “individualized” risk assessment method, used by trained professionals with an active human alert, is an acceptable method for identifying patients at risk of VTE and reducing VTE outcomes.
Footnotes
Appendix A
International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) Codes for Exclusion Criteria and Venous Thromboembolism (VTE) Risk Factors as Defined in the Study Protocol
| ICD-9-CM diagnosis codes, ICD-9-CM procedure codes, and therapy names | |
|---|---|
|
|
|
| Patients <18 years of age | |
| Patients with a hospital length of stay <2 days | |
| Patients transferred from another acute care facility | |
| Community-acquired VTE on admission | ‘415.11’−‘415.19’, ‘451.11’−‘451.19’, ‘451.2’, ‘451.81’, ‘451.83’, ‘451.84’, ‘451.89’, ‘453.2’, ‘453.40’−‘453.42’, ‘453.8’, ‘453.9’ |
| Pregnant on admission | ‘630’−‘633.9’, ‘634.0’−‘639.9’, ‘640.0’−‘648.94’, ‘650’−‘659.93’, ‘660.00’−‘669.93’, ‘670’−‘677’, ‘72−75’ |
| Patients at risk of bleeding | |
| Liver diseases | ‘006.3’, ‘070.00’−‘070.93’, ‘091.62’, ‘095.3’, ‘121.0’, ‘121.1’, ‘121.3’, ‘122.0’, ‘122.5’, ‘122.8’, ‘155.0’−‘155.2’, ‘197.7’, ‘211.5’, ‘230.8’, ‘235.3’, ‘271.0’, ‘275.0’, ‘277.3’, ‘286.7’, ‘570’, ‘571.0’−‘571.9’, ‘572.0’−‘572.8’, ‘573.0’−‘573.9’, ‘782.4’, ‘789.1’, ‘794.8’, ‘864.0’−‘864.19’, ‘996.82’, ‘997.4’, ‘V10.07’ |
| Malignant hypertension | ‘401.0’, ‘402.0’, ‘403.0’, ‘404.0’, ‘405.0’ |
| Blood disease | ‘280.0’−‘280.9’, ‘281.0’−‘281.9’, ‘282.0’−‘282.3’, ‘283.41’−‘282.49’, ‘282.5’, ‘282.60’−‘282.69’, ‘282.7’−‘282.9’, ‘283.0’, ‘283.10’−‘283.19’, ‘283.2’, ‘283.9’, ‘284.0’−‘284.9’, ‘285.0’, ‘285.1’, ‘285.21’−‘285.29’, ‘285.8’, ‘285.9’, ‘286.0’−‘286.9’, ‘287.0’−‘287.2’, ‘287.30’−‘287.39’, ‘287.4’−‘287.9’, ‘288.x’, ‘289.4’, ‘289.6’, ‘289.7’, ‘289.81’−‘289.89’, ‘289.9’ |
| Active peptic ulcer | ‘531.00’−‘531.3’, ‘532.00’−‘532.3’, ‘533.00’−‘533.3’, ‘534.00’−‘534.3’, ‘530.2x’ |
| Antiplatelet agents | Clopidogrel, ticlopidine, cilostazol |
| NSAIDs and >65 years of age | Celecoxib, choline magnesium trisalicylate, diclofenac, diflunisal, etodolac, fenoprofen, flurbiprofen, ibuprofen, indomethacin, ketoprofen, ketorolac, meclofenamate, mefenamic acid, meloxicam, nabumetone, naproxen, oxaprozin, piroxicam, salicylates, salsalate, sulindac, tolmetin |
| Renal dysfunction | ‘593.9’, ‘584.0’−‘584.9’, ‘585.0’−‘585.9’, ‘586’, ‘403’, ‘639.3’, ‘669.3’, ‘958.5’ |
| Patients actively bleeding | ‘252.8’, ‘255.4’, ‘362.81’, ‘363.61’, ‘363.62’, ‘364.41’, ‘374.81’, ‘376.32’, ‘377.42’, ‘430’, ‘431’, ‘432.0’−‘432.9’, ‘446.21’, ‘448.9’, ‘455.2’, ‘455.3’, ‘455.8’, ‘456.0’, ‘456.20’, ‘459’, ‘519.09’, ‘530.7’, ‘530.82’, ‘531.00’, ‘531.01’, ‘531.20’, ‘531.21’, ‘531.40’, ‘531.41’, ‘531.60’, ‘531.61’, ‘532.00’, ‘532.01’, ‘532.20’, ‘532.21’, ‘532.40’, ‘532.41’, ‘532.60’, ‘532.61’, ‘533.00’, ‘533.01’, ‘533.20’, ‘533.21’, ‘533.40’, ‘533.41’, ‘533.60’, ‘533.61’, ‘534.00’, ‘534.01’, ‘534.20’, ‘534.21’, ‘534.40’, ‘534.41’, ‘534.60’, ‘534.61’, ‘535.01’, ‘535.11’, ‘535.21’, ‘535.31’, ‘535.41’, ‘535.51’, ‘535.61’, ‘537.83’, ‘537.84’, ‘562.12’, ‘562.13’, ‘569.3’, ‘578.0’−‘578.9’, ‘593.81’, ‘596.7’, ‘596.8’, ‘602.1’, ‘607.82’, ‘620.1’, ‘622.8’, ‘623.8’, ‘626.6’, ‘627’, ‘627.1’, ‘784.7’, ‘784.8’, ‘800.2x’, ‘800.3x’, ‘800.7x’, ‘800.8x’, ‘801.2x’, ‘801.3x’, ‘801.7x’, ‘801.8x’, ‘803.2x’, ‘803.3x’, ‘803.7x’, ‘803.8x’, ‘804.2x’, ‘804.3x’, ‘804.7x’, ‘804.8x’, ‘852.0x’−‘852.5x’, ‘853.0x’−‘853.5x’, ‘958.2’, ‘996.7x’, ‘997.02’, ‘998.11’, ‘E870.x’, ‘6.02’, ‘21.00’−‘21.09’, ‘28.7’, ‘39.41’, ‘39.98’, ‘42.33’, ‘44.40’−‘44.49’, ‘45.43’, ‘49.95’, ‘54.12’, ‘57.93’, ‘60.94’ |
|
|
|
| Surgery | |
|
General, vascular, gynecologic, and urologic surgery
General surgery Transplant |
Diagnosis: ‘996.80’−‘996.89’, ‘V42.0’−‘V42.7’, ‘V42.81’−‘V42.89’, ‘V42.9’, ‘V58.44’, ‘E878.0’ Procedure: ‘11.60’−‘11.69’, ‘16.59’, ‘33.50’, ‘33.52’, ‘37.51’, ‘41.00’−‘41.09’, ‘41.94’, ‘46.97’, ‘49.74’, ‘ 50.51’, ‘50.59’, ‘52.80’−‘52.86’, ‘55.61’, ‘55.69’ |
| Other general surgery | Procedure: ‘07.02’, ‘07.12’−‘07.99’, ‘21.00’−‘21.09’, ‘21.30’−‘21.32’, ‘28.0’, ‘28.2’−‘28.7’, ‘28.91’−‘28.99’, ‘31.1’, ‘31.21’, ‘31.29’, ‘34.71’, ‘40.21’−‘40.59’, ‘41.2’, ‘41.41’−‘41.43’, ‘41.5’, ‘41.93’, ‘42.31’−‘42.39’, ‘42.82’, ‘42.91’, ‘43.0’, ‘43.11’−‘44.03’, ‘44.21’−‘44.32’, ‘44.39’−‘44.42’, ‘44.49’−‘44.66’, ‘44.69’−‘44.91’, ‘45.00’−‘45.03’, ‘45.15’, ‘45.26’, ‘45.27’, ‘45.30’−‘45.95’ ‘46.01’−‘46.79’, ‘46.91’−‘46.94’, ‘47.09’, ‘47.19’−‘47.99’, ‘48.25’, ‘48.26’, ‘48.0’, ‘48.1’, ‘48.31’−‘48.99’, ‘49.01’−‘49.12’, ‘49.31’−‘49.39’, ‘49.43’−‘49.73’, ‘49.75’−‘49.93’, ‘49.95’, ‘50.0’, ‘50.12’, ‘50.21’−‘50.29’, ‘50.3’, ‘50.4’, ‘50.61’, ‘50.69’, ‘51.02’−‘51.04’, ‘51.13’, ‘51.21’,‘51.22’, ‘51.31’−‘51.87’, ‘51.89’−’51.99’, ‘52.01’−‘52.09’, ‘52.21’−‘52.7, ‘52.92’, ‘52.95’, ‘52.96’, ‘52.99’, ‘54.0’, ‘54.11’−‘54.19’, ‘54.23’, ‘54.3’, ‘54.4’, ‘54.52’−’54.75’, ‘54.92’−‘54.95’, ‘85.0’, ‘85.12’, ‘85.20’, ‘85.41’−‘85.48’, ‘85.81’, ‘86.03’−‘86.05’, ‘86.11’−‘86.23’, ‘86.27’,‘86.28’, ‘86.4’, ‘86.59’ |
| Major vascular surgery | ‘36.03’−‘36.99’, ‘38.00’−‘38.89’, ‘38.92’−‘38.99’, ‘39.1’, ‘39.22’−‘39.26’, ‘39.28’−‘39.41’, ‘39.44’−‘39.79’, ‘39.8’, ‘39.91’−‘39.94’, ‘39.96’−‘39.99’ |
| Major gynecologic surgery | ‘65.01’, ‘65.09’, ‘65.12’−‘65.89’, ‘65.92’−‘65.92’, ‘66.02’, ‘66.19’−‘66.79’, ‘66.92’−‘66.94’, ‘66.97’−‘66.99’, ‘67.2’, ‘67.31’−‘67.69’, ‘68.0’, ‘68.13’, ‘68.14’, ‘68.19’−‘68.59’, ‘68.6’−‘68.9’, ‘69.01’−‘69.49’, ‘69.95’, ‘69.98’, ‘69.99’, ‘70.11’−‘70.14’, ‘70.31’−‘70.92’, ‘71.09’, ‘71.22’−‘71.79’, ‘71.8’, ‘71.9’ |
| Major urologic surgery | ‘55.01’−‘55.12’, ‘55.24’−‘55.59’, ‘55.7’, ‘55.81’−‘55.99’, ‘56.1’, ‘56.2’, ‘56.34’, ‘56.39’−‘56.99’, ‘57.12’−‘57.22’, ‘57.34’, ‘57.39’, ‘57.51’−‘57.92’, ‘57.96’−‘57.99’, ‘58.0’, ‘58.1’, ‘58.31’−‘58.49’, ‘58.5’, ‘58.91’−‘58.99’, ‘60.0’, ‘60.11’−‘60.19’, ‘60.3’−‘60.5’, ‘60.61’−‘60.82’, ‘60.93’, ‘60.94’, ‘60.99’, ‘62.0’, ‘62.12’−‘62.99’, ‘63.01’−‘63.99’, ‘64.2’, ‘64.3’ |
| Laparoscopic surgery | ‘44.38’, ‘44.67’, ‘44.68’, ‘44.95’−‘44.98’, ‘47.01’, ‘47.11’, ‘50.19’, ‘51.11’, ‘51.23’, ‘51.24’, ‘54.21’, ‘54.51’, ‘59.03’, ‘59.12’ |
| Orthopedic surgery | |
| Elective hip arthroplasty | ‘81.51’−‘81.53’ |
| Elective knee arthroplasty | ‘81.54’, ‘81.55’ |
| Knee arthroscopy | ‘80.26’ |
| Hip fracture surgery | ‘79.05’, ‘79.15’, ‘79.25’, ‘79.35’ |
| Elective spine surgery | ‘80.19’, ‘80.29’, ‘80.39’, ‘80.50’−‘80.59’, ‘80.79’, ‘80.89’, ‘80.99’, ‘81.00−‘81.08’, ‘81.30’−‘81.39’, ‘81.62−‘81.66’, ‘84.51’, ‘84.58’, ‘84.59’ |
| Neurosurgery | ‘01.11’−‘01.59’, ‘01.6’, ‘02.01’−‘02.99’, ‘03.01’−‘03.29’, ‘03.32’−‘03.79’, ‘03.93’−‘03.99’, ‘04.01’−‘04.07’, ‘04.12’−‘04.79’, ‘04.91’−‘04.99’, ‘05.0’, ‘05.11’−‘05.89’, ‘05.9’ |
| Trauma, spinal cord injury, burns | |
| Trauma | ‘800.00’−‘804.99’, ‘805.00’−‘805.98’, ‘807.00’−‘825.39’, ‘826.0’−‘829.1’, ‘850.0’, ‘850.10’−‘850.12’, ‘850.2’−‘850.9’, ‘851.00’−‘854.19’, ‘860.0’−‘860.5’, ‘861.00’−‘868.19’, ‘869.0’−‘869.1’, ‘870.0’−‘871.9’, ‘872.00’−‘881.22’, ‘882.0’−‘897.7’, ‘900.00’−‘904.54’, ‘904.6’−‘904.9’, ‘925.1’−‘926.0’, ‘926.11’−‘926.19’, ‘926.8’−‘926.9’, ‘927.00’−‘927.21’, ‘927.3’−‘927.9’, ‘928.00’−‘928.21’, ‘928.3’−‘929.9’, ‘950.0’−‘957.9’, ‘958.4’,‘958.5’, ‘958.8’, ‘959.01’−‘959.19’, ‘959.2’−‘959.9’ |
| Acute spinal cord injuries | ‘806.00’−‘806.79’, ‘806.8’, ‘806.9’, ‘907.2’, ‘952.00’−‘952.19’, ‘952.2’−‘952.9’ |
| Burns | ‘940.0’−‘940.9’, ‘941.30’−‘941.59’, ‘942.30’−‘942.59’, ‘943.30’−‘943.59’, ‘944.30’−‘944.58’, ‘945.30’−‘945.59’, ‘946.3’−‘946.5’, ‘947.0’−‘947.9’, ‘948.00’−‘948.99’, ‘949.0’−‘949.5’ |
| Malignancy | ‘140.0’−‘149.9’, ‘150.0’−‘159.9’, ‘160.0’−‘165.9’, ‘170.0’−‘176.9’, ‘179’, ‘180.0’−‘189.9’, ‘190.0’−‘198.7’, ‘198.81’−‘198.89’, ‘199.0’, ‘199.1’, ‘200.00’−‘202.99’, ‘203.00’−‘208.91’, ‘230.0’−‘234.9’ |
| Cancer therapy | Abarelix, aldesleukin, alemtuzumab, alitretinoin, altretamine, amifostine, anastrozole, arsenic trioxide, asparaginase, azacitidine, bevacizumab, bexarotene capsules, bexarotene gel, bleomycin, bortezomib, busulfan intravenous, busulfan oral, calusterone, capecitabine, carboplatin, carmustine, carmustine with Polifeprosan 20 Implant, cetuximab, chlorambucil, cisplatin, cladribine, clofarabine, cyclophosphamide, cytarabine, cytarabine liposomal, dacarbazine, dactinomycin, actinomycin D, dasatinib, daunomycin, daunorubicin, daunorubicin liposomal, decitabine, denileukin, dexrazoxane, docetaxel, doxorubicin, doxorubicin liposomal, epirubicin, erlotinib, estramustine, etoposide phosphate, etoposide, VP-16, exemestane, floxuridine (intraarterial), fludarabine, fluorouracil, 5-FU, fulvestrant, gefitinib, gemcitabine, gemtuzumab ozogamicin, goserelin acetate, histrelin acetate, hydroxyurea, ibritumomab tiuxetan, idarubicin, ifosfamide, imatinib mesylate, irinotecan, lenalidomide, letrozole, leuprolide acetate, levamisole, lomustine, CCNU, meclorethamine, nitrogen mustard, megestrol acetate, melphalan, Procedure codes for radiation therapy: ‘92.20’−‘92.29’, ‘92.30’−‘92.39’ |
| Use of SERMs | Tamoxifen, raloxifene, toremifene, clomiphene |
| Acute medical illness Severe respiratory disease | ‘136.3’, ‘466.0’, ‘466.11’, ‘466.19’, ‘480.0’−‘482.2’, ‘482.30’−‘482.89’, ‘482.9’−‘484.8’, ‘485’,‘486’, ‘487.0’−‘487.8’, ‘490’, ‘491.0’, ‘491.1’, ‘491.20’−‘491.22’, ‘491.8’, ‘491.9’, ‘492.0’, ‘492.8’, ‘493.00’−‘493.92’, ‘494.0’−‘495.9’, ‘496’, ‘500’−‘505’, ‘506.0’−‘508.9’, ‘510.0’, ‘510.9’, ‘511.0’−‘511.9’, ‘512.0’, ‘512.8’, ‘513.0’, ‘513.1’, ‘514’, ‘515’, ‘516.0’−‘516.9’, ‘517.1’−‘517.8’, ‘518.0’, ‘518.2’−‘518.6’, ‘519.00’−‘519.09’, ‘519.1’ |
| AMI | ‘410.x0’, ‘410.x1’ |
| Stroke | ‘433.01’, ‘433.11’, ‘433.21’, ‘433.31’, ‘433.81’, ‘433.91’, ‘434.01’, ‘434.11’, ‘434.91’, ‘436’, ‘997.02’ |
| Severe infectious disease | ‘001.x’−‘009.x’, ‘010.x’−‘018.x’, 020.x’−027.x’, ‘030.x’−041.x’, ‘042.x’, 045.x−‘049.x’, ‘050.x’−‘057.x’, ‘060.x’−‘066.x’, ‘070.x’−‘079.x’, ‘080.x’−‘088.x’, ‘090.x’−‘099.x’, ‘100.x’−‘104.x’, ‘110.x’−‘118.x’, ‘120.x’−‘129.x’, ‘130.x’−‘136.x’, ‘137.x’−‘139.x’, ‘320.x’, ‘321.x’, ‘322.x’, ‘323.x’, ‘324.x’, ‘326.x’, ‘480.x’, ‘481.x’, ‘482.x’, ‘483.x’, ‘484.x’, ‘485.x’, ‘486.x’, ‘487.x’, ‘540.0’, ‘540.1’, ‘540.9’, ‘550.0’, ‘551.x’, ‘567.x’, ‘569.5’, ‘572.0’, ‘590.x’, ‘599.0’, ‘595.x’, ‘601.x’, ‘611.0’, ‘614.x’, ‘615.x’, ‘616.x’, ‘680.x’, ‘681.x’, ‘682.x’, ‘683.x’, ‘684.x’, ‘685.x’, ‘686.x’, ‘711.x’, ‘730.x’, ‘785.52’, ‘995.91’, ‘995.92’, ‘999.3’ |
| Heart or respiratory failure | |
| Heart failure | ‘398.91’, ‘402.01’, ‘402.11’, ‘402.91’, ‘404.01’, ‘404.03’, ‘404.11’, ‘404.13’, ‘404.91’, ‘404.93’, ‘428.0’, ‘428.1’, ‘428.20’−‘428.43’, ‘428.9’ |
| Respiratory failure | ‘786.01’−‘786.09’ |
| IBD | ‘564.1’, ‘564.5’, ‘555.0’−‘555.9’, ‘556.0’−‘556.9’ |
| Nephrotic syndrome | ‘581.0’−‘581.3’, ‘581.81’, ‘581.89’, ‘581.9’ |
| Myeloproliferative disorders | ‘205.x0’, ‘206.x0’, ‘207.x0’, ‘238.4’, ‘238.7’, ‘289.0’, ‘289.6’, ‘289.89’ |
| Paroxysmal nocturnal hemoglobinuria | ‘283.2’ |
| Central venous catheterization | ‘38.93’−‘38.94’ |
| Inherited or acquired thrombophilia | ‘286.0’−‘286.9’ |
| Obesity | ‘278.00’, ‘278.01’, ‘V77.8’ |
| Varicose veins | ‘454.0’−‘454.9’ |
| Pregnancy and postpartum | (EXCLUDED FROM THE STUDY) |
| Previous VTE | ‘V12.51’ |
| Estrogen-containing oral contraception or hormone replacement therapy Estrogen-containing oral contraception | Alesse, Apri, Aviane, Brevicon®, Demulen®, Desogen®, Estrostep®, Estrostep® Fe, Genora®, Jenest®, Levlen®, Levlite®, Levora®, Lo/Ovral®, Loestrin®, Loestrin® Fe, Low-Ogestrel®, Microgestin®, Microgestin® Fe, Mircette®, Modicon®, Necon®, Norinyl®, Nordette®, Nortrel®, Ogestrel®, Ortho-Cept®, Ortho-Cyclen®, Ortho-Novum®, Ortho Tri-Cyclen®, Ovcon®, Ovral®, Seasonale®, Tri-Levlen®, Tri-Norinyl®, Triphasil®, Trivora®, Yasmin®, Zovia® |
| Estrogen-containing hormone replacement therapy | Activella, femhrt, Ortho-Prefest, Premphase, Prempro, Estrasorb, EstroGel, Alora, Climara, Estraderm, Menostar, Vivelle, Vivelle-Dot, Estrace, Estring, Femring, Premarin, Vagifem, Delestrogen, DEPO-Estradiol, Premarin Intravenous, Cenestin, Enjuvia, Femtrace, Gynodiol, Menest, Ogen |


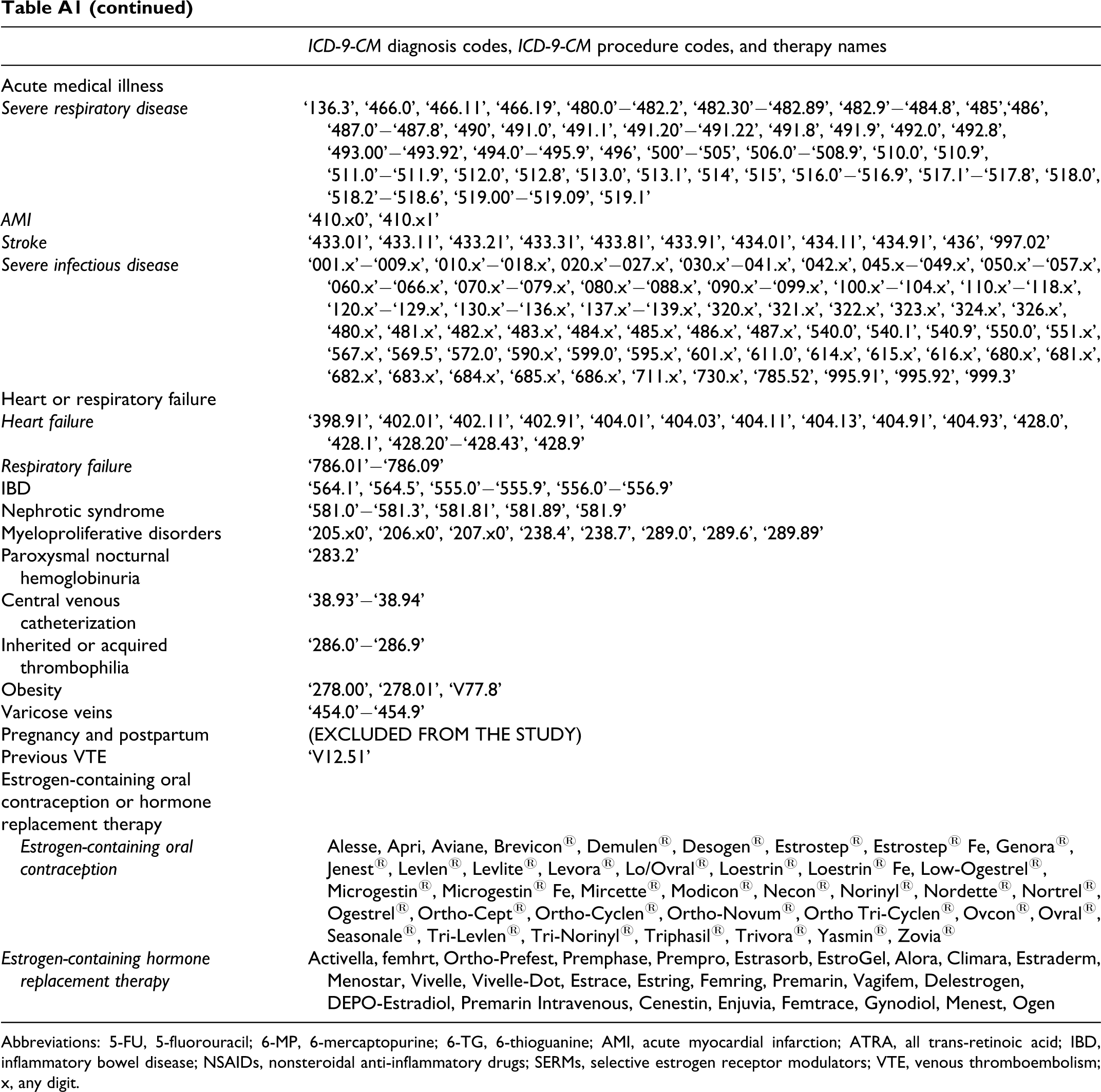
Abbreviations: 5-FU, 5-fluorouracil; 6-MP, 6-mercaptopurine; 6-TG, 6-thioguanine; AMI, acute myocardial infarction; ATRA, all trans-retinoic acid; IBD, inflammatory bowel disease; NSAIDs, nonsteroidal anti-inflammatory drugs; SERMs, selective estrogen receptor modulators; VTE, venous thromboembolism; x, any digit.
Appendix B Venous Thromboembolism Risk-Assessment Form
Acknowledgments
Dr Mahan would like to express his thanks to the clinical pharmacists, administration, and medical staff at Lovelace Medical Center for aiding in this quality improvement initiative. The study was funded by sanofi-aventis. The authors received editorial support in the preparation of this manuscript, which was funded by sanofi-aventis, Bridgewater, NJ, USA. Hester van Lier, PhD, provided the editorial support. The sponsor played no role in study design or methodology.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Dr. Mahan is an unpaid consultant for Johnson and Johnson Pharmaceutical Research & Development and Ortho-McNeil Janssen Scientific Affairs. Dr. Mahan has received travelling fellowship funds from the North American Thrombosis Forum and honoraria as a consultant and speaker from Boehringer Ingelheim and sanofi-aventis pharmaceuticals and as a consultant from Polymedix and Leo Pharmaceuticals.
Dr. Spyropoulous has received honoraria as a consultant for Bayer, Eisai, Boehringer-Ingelheim, Bristol-Myers Squibb, Pfizer, Johnson & Johnson, and sanofi-aventis.
Dr Hussein is consultant for Lovelace as related to this study, and has received research support and is a consultant for Schering Plough, Pfizer, Amylin, Merck, and AstraZeneca.
Dr Amin has received research support from sanofi-aventis, Otsuka and Boehringer- Ingelheim, and is on the speakers bureau for sanofi-aventis, Otsuka and Boehringer-Ingelheim.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by sanofi-aventis.