Abstract
The prothrombotic state in patients with atrial fibrillation (AF) is related to endothelial injury, the activation of platelets and the coagulation cascade. We evaluated the levels of platelet- (CD42b) and endothelial-derived (CD144) microparticles in the plasma patients with non-valvular AF treated with dabigatran at the time of expected minimum and maximum drug plasma concentrations. Following that, we determined the peak dabigatran plasma concentration (cpeak ). CD42b increased after taking dabigatran (median [IQR] 36.7 [29.4-53.3] vs. 45.6 [32.3-59.5] cells/µL; p = 0.025). The concentration of dabigatran correlated negatively with the post-dabigatran change in CD42b (ΔCD42b, r = -0.47, p = 0.021). In the multivariate model, the independent predictors of ΔCD42b were: cpeak (HR -0.55; with a 95% confidence interval, CI [-0.93, -0.16]; p = 0.007), coronary artery disease (CAD) (HR -0.41; 95% CI [-0.79, -0.02]; p = 0.037) and peripheral artery disease (PAD) (HR 0.42; 95% CI [0.07, 0.74]; p = 0.019). CD144 did not increase after dabigatran administration. These data suggest that low concentrations of dabigatran may be associated with platelet activation. PAD and CAD have distinct effects on CD42b levels during dabigatran treatment.
Keywords
Introduction
Atrial fibrillation (AF) remains the most common cardiac arrhythmias in clinical practice, which is associated with an increased risk of stroke and systemic embolism. AF itself enhances endothelial dysfunction, platelet aggregation and coagulation. 1,2 Dabigatran, a non-vitamin K oral anticoagulant (NOAC) specifically and reversibly blocks the activity of thrombin, exerting direct antithrombotic and indirect anti-platelet action. 3,4 Effective anticoagulant treatment with NOACs such as dabigatran is an essential process for improving the prognosis of patients with AF. 5,6
Circulating microparticles (MPs) are membrane vesicles that are <2 µm in size, which are released into the plasma from all morphotic blood elements and the vascular endothelium through exotic budding of the plasma membrane in response to cellular apoptosis or activation. 7 MPs have surface antigens characteristic of the cells from which they were made. MPs play a crucial role in haemostasis and thrombosis. 8,9 MPs derived from platelets (PMPs) and endothelial-derived MPs (EMPs) participate in coagulation and haemostasis by inducing platelet aggregation or developing platelet and fibrin rich thrombi. 7 Circulating procoagulant MPs might reflect a hypercoagulable state that contributes to arterial thromboembolism. 10,11 Circulating EMPs have been linked to the severity, lesion volume, and outcome of acute ischemic stroke. 12 Some medications, such as antihypertensives, lipid-lowering agents and antioxidant drugs modulate PMP and EMP levels. 13 –16
Recent clinical findings suggest that MPs may play a role in the hypercoagulable state in AF. 10,17 –19 Previous studies 13 have showed increased levels of PMPs and EMPs in patients with AF, indicating elevated endothelial damage and platelet activation. Moreover, PMPs have been reported to be used for monitoring antithrombotic therapy in patients with type 2 diabetes. 20,21
The aim of this study was to evaluate the levels of PMPs and EMPs, namely platelet glycoprotein Ib (GPIb, cluster of differentiation 42b, CD42b) and vascular endothelial cadherin (VE-cadherin, CD144), respectively, in patients with non-valvular AF treated with dabigatran.
Material and Methods
Design
In this prospective observational study, we evaluated patients with non-valvular AF treated with dabigatran etexilate (dabigatran). According to the protocol of the study, we have determined in-plasma peak dabigatran and microparticle (CD42b and CD144) concentrations at the time of expected minimum and maximum dabigatran concentrations. The trough sample was obtained at 12 hours after taking the drug. The post-dose samples were collected 1 to 3 hours after drug ingestion.
Study Patients
After giving their informed consent, a total of 39 patients with documented AF treated with dabigatran for 3 months or more who were screened in our department between August 2015 to August 2017, were enrolled in the study. The exclusion criteria were unstable cardiovascular diseases (unstable coronary artery disease [CAD], exacerbation of chronic heart failure, uncontrolled hypertension), recent (<3 months) thromboembolic events (stroke/TIA or systemic embolism), connective tissue disease, uncontrolled thyroid disease and chronic kidney disease stage 4 or more. 22
The CHA2DS2-VASc score was used to evaluate the risk of stroke or systemic embolism. 23 The patients’ bleeding risk was estimated using the HAS-BLED score. 24
Patients were evaluated at enrollment for dabigatran dosage based on current guidelines. 5 We used a standardized questionnaire to collect the patients’ demographics data, information about cardiovascular risk factors and current treatment. 22 Patients were monitored for 12 months for cases of stroke and systemic embolism, as well as major bleeding. The comorbidities were defined in the Online supplemental material.
Blood Collection and Laboratory Analysis
Dabigatran plasma measurement
Plasma samples were collected in Monovette ® vacuum tubes (S-Monovette, Sarstedt), containing 3.2% trisodium citrate (10:1). Plasma concentrations of dabigatran were determined with the BCS XP Automated Blood Coagulation Analyzer System (Siemens Healthcare Diagnostics Products GmbH, Marburg, Germany), using Hyphen BioMed products (Neuville Sur Oise, France), as per the manufacturer’s instructions. 25
Reference Ranges
The therapeutic ranges have not been clearly defined for NOACs. Using the Randomized Evaluation of Long Term Anticoagulant Therapy With Dabigatran Etexilate (RE-LY) study 26 , we defined the “peak range” in the post-dose samples for dabigatran at a given dose. The estimated total dabigatran plasma concentrations after oral administration of 110mg or 150 mg b.i.d. in the RELY study were 52-275 ng/ml and 74.3-383 ng/ml, respectively. 26
Platelet-Derived and Endothelial-Derived Microparticles Determination
PMP and EMP levels were measured through the direct fluorescence method using flow cytometry assay, as previously described. 27,28 Venous blood was drawn into Vacutainer™ CTAD vacuum glass tubes (Becton Dickinson), containing 0.11 M buffered trisodium citrate solution, 15 M theophylline, 3.7 M adenosine and 0.198 M dipyridamole. The blood samples were centrifuged at room temperature at 1550G for 20 minutes in order to separate larger morphotic elements. The obtained supernatant was transferred into Eppendorf tubes, leaving about 200 µl of fluid above the cell line, and the tubes were frozen at -80C. After thawing, the material was centrifuged on a MiniSpin Plus centrifuge (Eppendorf, Germany) at room temperature at 13000G, for 2 minutes. From the prepared sample, 100µl of the supernatant was moved to TruCount tubes (Becton Dickinson, USA). Characteristic cell markers were used for each type of MP to evaluate these different types. The sample was labelled with monoclonal antibodies against CD42b and CD144 (5µl of each). After thorough mixing of the sample, the material was incubated for 20 minutes at room temperature in the dark. 100µl of the diluted (1:10) buffer “Annexin V Binding Buffer” (the diluted buffer was filtered using a 0.2 µm Whatman filter) and 5 µl Annexin V FITC were then added to the sample. The sample was then mixed again and incubated for 10 minutes at room temperature in the dark. After incubation, 300µl of diluted (1:10) Annexin V Binding Buffer was added. Finally, the prepared samples were analyzed using the FACSCalibur flow cytometer (Becton Dickinson, USA). 29 The change in PMPs and EMPs (ΔCD42b and ΔC144, respectively) were defined as MP concentrations after the administration of dabigatran minus the baseline. The technicians performing these measurements were blinded to the treatment groups.
Statistical Analysis
The study was powered to have a 90% chance of detecting a 10% difference in MPs between groups at p = 0.05, based on a previous study. 13 13 patients were required in each group in order to demonstrate such a difference in MPs. Quantitative variables were reported as the mean (±standard deviation) or median (interquartile range [IQR], where appropriate. The Shapiro-Wilk test was used to check the normality of distribution. The quantitative variables were evaluated between groups using the t-Student test or the Mann-Whitney U test. The Wilcoxon-Sign test was used to evaluate MPs between and after dabigatran administration. Moreover, a linear dependence between variables was tested using Pearson’s correlation or Spearman’s rank test, as appropriate. In order to identify independent factors affecting the level of MPs, all clinical and laboratory variables that showed the association with MPs in the univariate model (P < 0.15) were included in the multivariate analysis. The models were adjusted for age and sex. P values <0.05 were considered statistically significant. Data were analyzed using STATISTICA 13.0 (Statsoft Inc, Tulsa, OK).
Ethics Approval and Consent to Participate
The study was approved by the Jagiellonian University Ethics Committee (Trial registration No. 122.6120.97.2015; registered May 28, 2015, http://kbet.cm.uj.edu.pl/), Cracow, Poland. All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and national research committee.
Results
Patient Characteristics
The study group was comprised of 39 patients with AF (paroxysmal n = 8 [20.5%]; persistent n = 31 [79.5%]) treated with dabigatran. This includes 28 patients taking a standard dose of 150 mg twice daily (b.i.d.) and 11 patients receiving a reduced dose of 110 mg b.i.d. Table 1 shows the prevalence of cardiovascular risk factors. Most patients (n = 30; 76.9%) were at a high risk for stroke (CHA2DS2-VASc score ≥2). Two patients (5.1%) had a low stroke risk (score of 0; These patients received anticoagulation before undergoing cardioversion). One-fifth of patients (n = 8; 20.5%) had a high risk of bleeding (HAS-BLED score ≥3). Patients who were treated with 150mg of dabigatran were younger, had a lower CHA2DS2-VASc score and a higher creatinine clearance compared to those treated with a dose of 110 mg b.i.d. (Table 1, Suppl. Table 1).
Baseline Characteristic of Patients.

Values are presented as, n (%) or mean ± standard deviation.
Abbreviations: BMI, body mass index; CHA2DS2-VASc score, Congestive heart failure, Hypertension, Age ≥75 years, Diabetes, Prior stroke/transient ischemic attack (TIA)/thromboembolism, Vascular disease, Age 65–74 years, Sex category (female); HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio (INR), Elderly, Drugs/Alcohol concomitantly; ARB, angiotensin II receptor blockers; Hb, haemoglobin; PLT, platelet level; ClCr, creatinine clearance according to the Cockcroft-Gault equation.
Plasma Concentrations of Anticoagulants
The post-dose plasma concentration of dabigatran was determined in 24 patients. Seventeen patients (70.8%) had a plasma drug concentration at the peak range that showed no difference between 150 mg b.i.d. and 110 mg b.i.d. (70.6% [n = 12] vs. 57.1% [n = 5], respectively, p = 0.53).
Microparticle Plasma Concentrations and Their Determinants
CD42b
We compared plasma concentrations of PMPs before and after taking dabigatran.
Patients with PAD had higher levels of CD42b before and after the administration of dabigatran. A subgroup with a history of myocardial infarction had higher CD42b levels before administration of dabigatran, whereas patients treated with angiotensin II receptor blockers, dihydropyridines and metformin had higher CD42b levels after anticoagulant administration (Table 2).
PMP in AF Patients Treated With Dabigatran Before and After Taking an Anticoagulant.
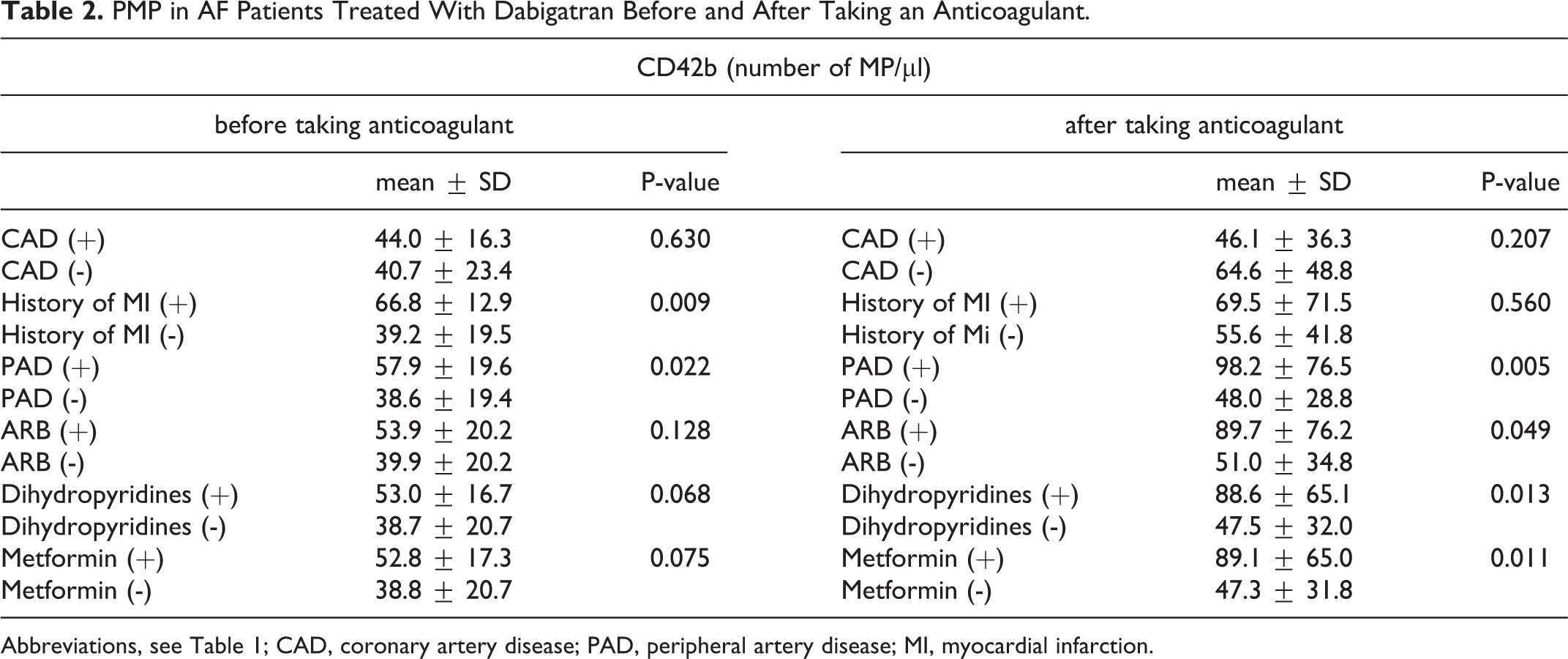
Abbreviations, see Table 1; CAD, coronary artery disease; PAD, peripheral artery disease; MI, myocardial infarction.
CD42b levels before taking anticoagulants positively correlated with the number of pack-years (r = 0.69, p = 0.013). CD42b levels after taking anticoagulants correlated positively with the CHA2DS2-VASc score (r = 0.33, p = 0.040) and was borderline negatively correlated with dabigatran concentrations (r = -0.39, p = 0.060).
After dabigatran administration CD42b increased (median [IQR] 36.7 [29.4-53.3] vs. 45.6 [32.3-59.5] cells/µL; p = 0.025) (Figure 1). In patients with coronary artery disease (CAD) after taking dabigatran we observed a smaller increase in CD42b compared to patients without CAD (102.4% [60.5-130.1] vs. 144.6% [101.5-245.4], p = 0.038). Among patients with peripheral artery disease (PAD), there was a trend toward higher post-dabigatran CD42b compared to the remainder of the cohort (179.1% [52.2-272.3] vs. 120.4% [92.2-152.1], p = 0.158). The concentration of dabigatran correlated negatively with ΔCD42b (r = -0.47, p = 0.021) (Figure 1).
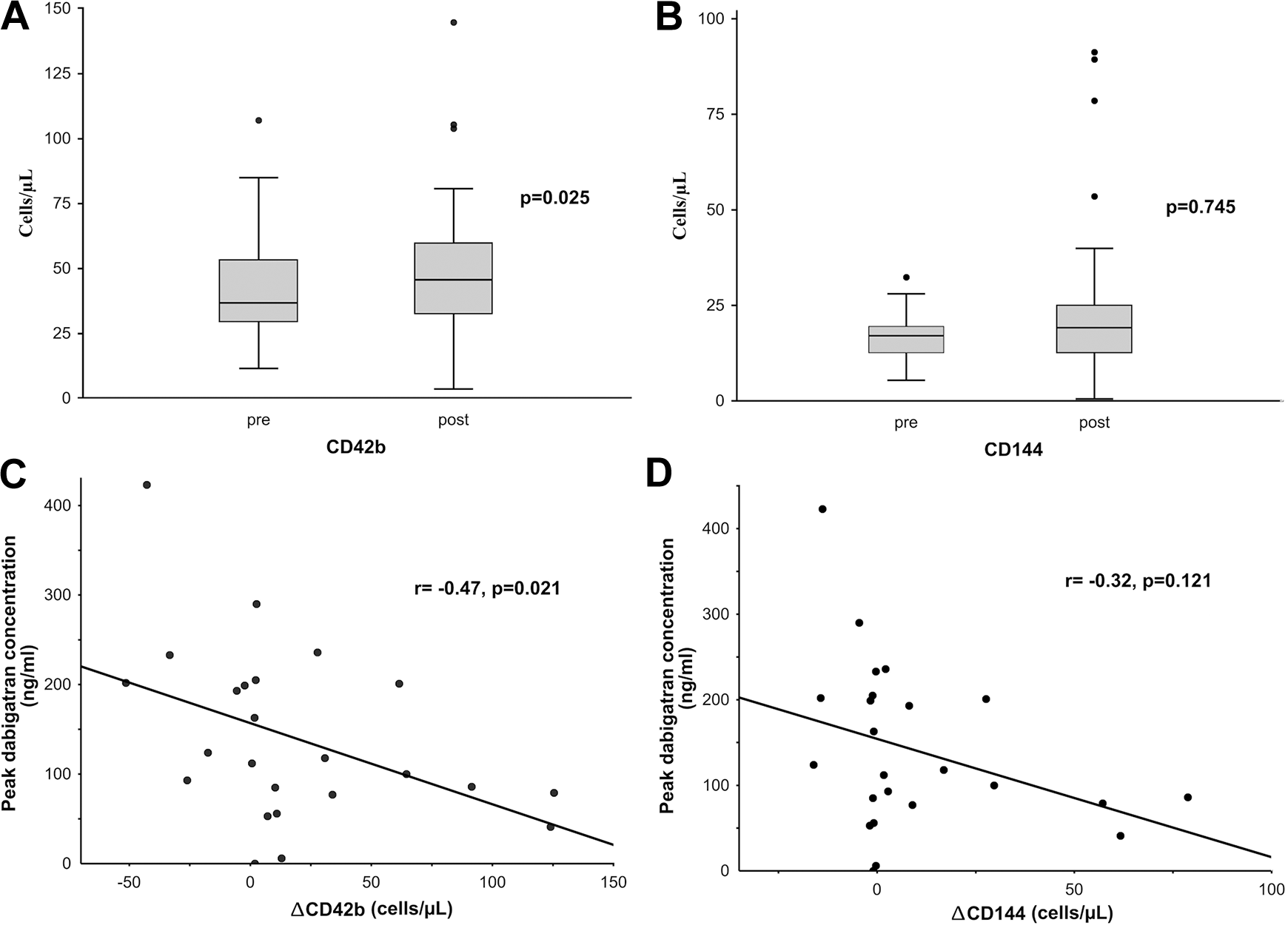
Associations between CD42b and CD144 and administration of dabigatran (pre, post, Panels A and B) in patients with atrial fibrillation (AF). Correlations of ΔCD42b (Panel C), ΔCD144 (Panel D) with peak dabigatran concentration in subjects with AF. Values are presented as median (interquartile range), and black points indicate an outlier.
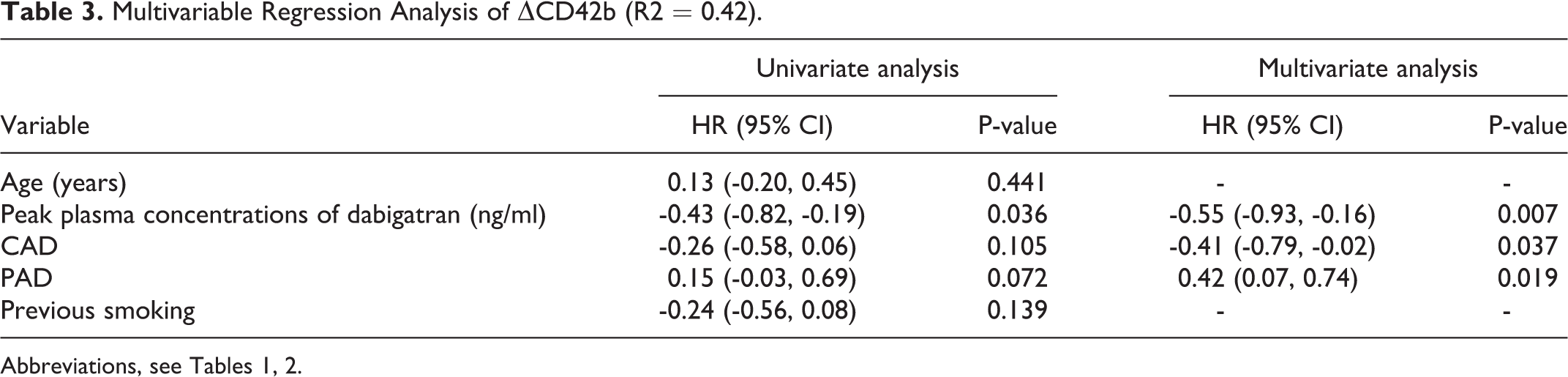
In the multivariable regression analysis, the independent predictors of ΔCD42b were the peak plasma concentration of dabigatran (HR -0.55; 95% CI [-0.93, -0.16]), CAD (HR -0.41; 95% CI [-0.79, -0.02]; and PAD (HR 0.42; 95% CI [0.07, 0.74]), (R2 = 0.42, Table 3).
Multivariable Regression Analysis of ΔCD42b (R2 = 0.42).
CD144
Patients treated with dihydropyridines and metformin had a higher CD144 count both before and after dabigatran administration. Those treated with a statin had a higher CD144 count before dabigatran administration, whereas patients with PAD and history of stroke/TIA had higher CD144 after anticoagulant administration (Suppl. Table 2). CD144 levels after taking anticoagulant correlated positively with the CHA2DS2-VASc score (r = 0.32, p = 0.044).
CD144 did not increase after taking anticoagulants (17.1 [12.6-19.5] vs. 19.2 [12.5-25.1] cells/µL, p = 0.745) (Figure 1). In a subgroup analysis, a post-dabigatran increase in CD144 levels was observed in patients with PAD (149.8% [102.9-367.7] vs. 96.7% [85.7-156.1], remainders, p = 0.023). The concentration of dabigatran did not correlate with ΔCD144 (r = -0.32, p = 0.121) (Figure 1).
12-month follow-up
Neither ischemic stroke nor systemic thromboembolism were observed during the 12-month follow-up period. A new thrombus in the left atrial appendage was revealed in 2 patients with the dabigatran plasma concentration “in the peak range.” There were 2 major bleedings, including 1 hemorrhagic stroke among patients receiving a standard dose of dabigatran.
Discussion
This prospective study shows that in AF patients, dabigatran administration is associated with the release of PMPs (CD42b), a marker of platelet activation. A negative correlation between the concentration of dabigatran and ΔCD42b suggests a harmful effect associated with low doses of this anticoagulant. Moreover, comorbidities such as PAD and CAD may exert an antagonistic effect of on the change in CD42b.
Formation of MPs is an essential part of the physiological coagulation process. Aminophospholipids on the surface of PMPs and EMPs have a large number of binding sites for plasma coagulation factors (IXa, VIII, Va and IIa). Therefore, activation of blood coagulation proteins may occur both on whole plaques and on MPs. 30 Glycoprotein Ib (GPIb, CD42b) is a component of the GPIb-V-IX complex on platelets, which allows platelet adhesion and platelet plug formation at sites of vascular injury. Vascular endothelial cadherin (VE-cadherin, CD144) is an endothelial-specific adhesion molecule located at the junctions between endothelial cells. These molecules maintain and control endothelial cell contacts, and play an important role in controlling vascular permeability and leukocyte extravasation. 31 The mechanism of hemostasis is shown in Figure 2.

Components of hemostasis. Coagulation factors in the bright blue, dark blue and green frames belong to the intrinsic, extrinsic and common blood coagulation pathways. Black horizontal arrows indicate transformation from inactive to active forms. Red bar-headed lines show the inhibition of active protease forms and targets of anticoagulants. Black dashed arrows indicate an impact on the process. Abbreviations: APC+PS, activated protein C + protein S, AT, antithrombin, Ca2+-calcium, F, factor, GPIb, glycoprotein Ib, HMWK, high molecular weight kininogen, PK, plasma prekallikrein, PL, phospholipids, TF, tissue factor, TFPI, tissue factor protease inhibitor, tPA, tissue plasminogen activator, uPA, urokinase plasminogen activator, VE-cadherin, vascular endothelial cadherin [based on 32 ].
The lack of a need for routine laboratory monitoring is the primary advantage of NOACs over Vitamin K antagonists. Dabigatran is one of the most commonly used NOACs in the antithrombotic management of patients with AF. 33 Dabigatran has a unique mechanism through which it might function as an anticoagulant. In addition to blocking the catalytic activity of thrombin, it may displace thrombin from fibrin or platelets, 34 and inhibit GPIbα-mediated platelet aggregation. 35 Despite this, there are reports 36,37 where dabigatran therapy has been associated with an increase in acute coronary events when compared with warfarin. The benefits of dabigatran over warfarin seem to outweigh the small increase in the risk of MI. 38 In addition to its antithrombotic effect, dabigatran has been suggested 39 to exert some pro-thrombotic effect due to its fostering the ligation of thrombin to its high-affinity platelet receptor (GPIbα) in patients with AF. Petzold et al. 39 described increased platelet aggregation in patients with AF treated with dabigatran compared with those receiving VKA. Siwaponanan et al. reported an increase in total circulating MP levels, as well as evidence of elevated endothelial damage and platelet activation, as demonstrated by the increased PMPs and EMPs levels in AF patients. 13 The use of dabigatran causes suppression of fibrin formation and thus the formation of thrombi. However, this drug may lead to platelet activation as seen in the increase in PMPs, which are markers of platelet activation.
Previous findings have suggested that MPs can play both a procoagulant role, as well as having a role in anticoagulation. Tans et al. 40 demonstrated that PMPs also possess potent anticoagulant functionality. MPs with phosphatidylserine on their surface are involved in the formation of thrombin, which activates the C protein involved in the degradation of coagulation factors Va and VIIIa. The fibrinolytic activity of EMPs is mainly based on the activity of tissue plasminogen activator (tPA). The presence of tPA on the surface of MPs increases plasma plasmin activity. 41,42
Choudhury et al. showed evidence of platelet activation (i.e., high PMPs and sP-selectin levels) in AF patients, but they suggest that this is likely due to underlying cardiovascular diseases rather than the arrhythmia per se. 19,43 In our study, both PMPs and EMPs correlated positively with the CHA2DS2-VASc score after taking an anticoagulant, suggesting an increased thromboembolic risk.
In our study, we observed that the presence of CAD was associated with a lower increase in CD42b. This may be related to a higher concentration of CD42b at the baseline in CAD patients, especially in patients with a history of MI. However, we also observed a greater increase in CD144 levels in the subgroup of patients with PAD, which may be associated with greater endothelial activation after taking dabigatran.
The limitations of our study include the sample size. However, the number of subjects was sufficient to detect the differences between the groups, given the results of the power calculation. Due to a high thromboembolic risk in the real-life AF population, 44 we did not enroll patients with AF and without an indication for anticoagulant treatment. Despite the particularly carefully selected study group (numerous exclusion criteria for inclusion in the study), we cannot rule out the impact of comorbidities on EMPs and PMPs in patients with AF. In our analyses, we did not take into account the assessment of the effect of microparticle concentration on adverse events because of too small the 12 month follow-up event rate. Finally, the mechanisms underlying thrombogenesis in AF are clearly complex and remain only partially understood, and therefore further studies are required to evaluate the role of PMPs and EMPs in the pathophysiology of AF.
Conclusions
In patients with AF, the administration of dabigatran is associated with an increase in CD42b levels. The release of platelet microparticles strongly correlates with the concentration of dabigatran in the blood, suggesting that lower dabigatran concentrations are not able to diminish post-drug platelet activation. This mechanism may potentially lead to paradoxical thrombosis in AF patients treated with dabigatran. We believe that larger studies are necessary to evaluate the relationship between MPs and dabigatran in AF.
Supplemental Material
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620972467 - Flow Cytometric Assessment of Endothelial and Platelet Microparticles in Patients With Atrial Fibrillation Treated With Dabigatran
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620972467 for Flow Cytometric Assessment of Endothelial and Platelet Microparticles in Patients With Atrial Fibrillation Treated With Dabigatran by Aleksandra Lenart-Migdalska, Leszek Drabik, Magdalena Kaźnica-Wiatr, Lidia Tomkiewicz-Pająk, Piotr Podolec and Maria Olszowska in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
Ethical approval to report this case series was obtained from Jagiellonian University Ethics Committee (Trial registration No. 122.6120.97.2015), Cracow, Poland. Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Acknowledgments
The study was supported by a grant from Jagiellonian University Medical College. The authors gratefully acknowledge Prof. Ewa Wypasek from the Laboratory of Molecular Biology in John Paul II Hospital and both Prof. Piotr Marek Radziwon and Mateusz Dziemiańczuk from the Regional Center for Blood Donation and Blood Treatment in Bialystok for their support in determining microparticles using a flow cytometer.
Contribution Statement
The concept and design of the study: ALM, MO, data collection: ALM, MKW, LTP data analysis: ALM, LD, MKW, LTP, PP, MO, draft manuscript preparation: ALM, LD, MO, final manuscript approval: ALM, LD, MKW, LTP, PP, MO.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author (s) disclosed receipt of the following financial support for the research, authorship, and / or publication of this article: The study was supported by a grant from Jagiellonian University Medical College. This article was supported by the science fund of the John Paul II Hospital, Cracow, Poland (no. FN10 / 2020 to ALM).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
